
FULL PRESCRIBING INFORMATION: CONTENTS *
WARNING: SEVERE ACUTE EXACERBATIONS OF HEPATITIS B, PATIENTS
CO-INFECTED WITH HIV AND HBV, AND LACTIC ACIDOSIS AND
HEPATOMEGALY
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Timing of Administration
2.2 Recommended Dosage in Adults
2.3 Recommended Dosage in Pediatric Patients
2.4 Renal Impairment
2.5 Hepatic Impairment
2.6 Duration of Therapy
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Severe Acute Exacerbations of Hepatitis B
5.2 Patients Co-infected with HIV and HBV
5.3 Lactic Acidosis and Severe Hepatomegaly with Steatosis
6 ADVERSE REACTIONS
6.1 Clinical Trial Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Racial/Ethnic Groups
8.7 Renal Impairment
8.8 Liver Transplant Recipients
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Outcomes in Adults
14.2 Outcomes in Pediatric Subjects
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
* Sections or subsections omitted from the full prescribing information are not listed.
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use BARACLUDE
safely and effectively. See full prescribing information for BARACLUDE.
BARACLUDE
®
(entecavir) tablets, for oral use
BARACLUDE
®
(entecavir) oral solution
Initial U.S. Approval: 2005
WARNING: SEVERE ACUTE EXACERBATIONS OF
HEPATITIS B, PATIENTS CO-INFECTED WITH HIV AND HBV,
and LACTIC ACIDOSIS AND HEPATOMEGALY
See full prescribing information for complete boxed warning.
• Severe acute exacerbations of hepatitis B have been reported in patients
who have discontinued anti-hepatitis B therapy, including entecavir.
Hepatic function should be monitored closely for at least several
months after discontinuation. Initiation of anti-hepatitis B therapy may
be warranted. (5.1)
• BARACLUDE is not recommended for patients co-infected with human
immunodeficiency virus (HIV) and hepatitis B virus (HBV) who are not
also receiving highly active antiretroviral therapy (HAART), because of
the potential for the development of resistance to HIV nucleoside reverse
transcriptase inhibitors. (5.2)
• Lactic acidosis and severe hepatomegaly with steatosis, including
fatal cases, have been reported with the use of nucleoside analogue
inhibitors. (5.3)
------------------------------ INDICATIONS AND USAGE ------------------------------
BARACLUDE is a hepatitis B virus nucleoside analogue reverse transcriptase inhibitor
indicated for the treatment of chronic hepatitis B virus infection in adults and children
at least 2 years of age with evidence of active viral replication and either evidence of
persistent elevations in serum aminotransferases (ALT or AST) or histologically active
disease. (1)
---------------------------DOSAGE AND ADMINISTRATION ---------------------------
• Nucleoside-inhibitor-treatment-naïve with compensated liver disease (greater
than or equal to 16 years old): 0.5 mg once daily. (2.2)
• Nucleoside-inhibitor-treatment-naïve and lamivudine-experienced pediatric
patients at least 2 years of age and weighing at least 10 kg: dosing is based on
weight. (2.3)
• Lamivudine-refractory or known lamivudine or telbivudine resistance substitutions
(greater than or equal to 16 years old): 1 mg once daily. (2.2)
• Decompensated liver disease (adults): 1 mg once daily. (2.2)
• Renal impairment: Dosage adjustment is recommended if creatinine clearance
is less than 50 mL/min. (2.4)
• BARACLUDE (entecavir) should be administered on an empty stomach. (2.1)
------------------------- DOSAGE FORMS AND STRENGTHS -------------------------
• Tablets: 0.5 mg and 1 mg (3, 16)
• Oral solution: 0.05 mg/mL (3, 16)
-------------------------------- CONTRAINDICATIONS --------------------------------
• None. (4)
--------------------------- WARNINGS AND PRECAUTIONS ---------------------------
• Severe acute exacerbations of hepatitis B virus infection after discontinuation:
Monitor hepatic function closely for at least several months. (5.1, 6.1)
• Co-infection with HIV: BARACLUDE is not recommended unless the patient is also
receiving HAART. (5.2)
• Lactic acidosis and severe hepatomegaly with steatosis: If suspected, treatment
should be suspended. (5.3)
-------------------------------- ADVERSE REACTIONS --------------------------------
• In adults, the most common adverse reactions (≥3%, all severity grades) are
headache, fatigue, dizziness, and nausea. The adverse reactions observed in
pediatric patients were consistent with those observed in adults. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Bristol-Myers Squibb at
1-800-721-5072 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
---------------------------USE IN SPECIFIC POPULATIONS ---------------------------
• Liver transplant recipients: Limited data on safety and efficacy are available. (8.8)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 11/2019

BARACLUDE
®
(entecavir)
FULL PRESCRIBING INFORMATION
1
WARNING: SEVERE ACUTE EXACERBATIONS OF HEPATITIS B, PATIENTS
CO-INFECTED WITH HIV AND HBV, and LACTIC ACIDOSIS AND HEPATOMEGALY
Severe acute exacerbations of hepatitis B have been reported in patients
who have discontinued anti-hepatitis B therapy, including entecavir. Hepatic
function should be monitored closely with both clinical and laboratory
follow-up for at least several months in patients who discontinue
anti-hepatitis B therapy. If appropriate, initiation of anti-hepatitis B therapy
may be warranted [see Warnings and Precautions (5.1)].
Limited clinical experience suggests there is a potential for the development
of resistance to HIV (human immunodeficiency virus) nucleoside reverse
transcriptase inhibitors if BARACLUDE is used to treat chronic hepatitis B
virus (HBV) infection in patients with HIV infection that is not being treated.
Therapy with BARACLUDE is not recommended for HIV/HBV co-infected
patients who are not also receiving highly active antiretroviral therapy
(HAART) [see Warnings and Precautions (5.2)].
Lactic acidosis and severe hepatomegaly with steatosis, including
fatal cases, have been reported with the use of nucleoside analogue
inhibitors alone or in combination with antiretrovirals [see Warnings and
Precautions (5.3)].
INDICATIONS AND USAGE
BARACLUDE
®
(entecavir) is indicated for the treatment of chronic hepatitis B virus
infection in adults and pediatric patients 2 years of age and older with evidence of active
viral replication and either evidence of persistent elevations in serum aminotransferases
(ALT or AST) or histologically active disease.
2 DOSAGE AND ADMINISTRATION
2.1 Timing of Administration
BARACLUDE should be administered on an empty stomach (at least 2 hours after a meal
and 2 hours before the next meal).
2.2 Recommended Dosage in Adults
Compensated Liver Disease
The recommended dose of BARACLUDE for chronic hepatitis B virus infection in
nucleoside-inhibitor-treatment-naïve adults and adolescents 16 years of age and older
is 0.5 mg once daily.
The recommended dose of BARACLUDE in adults and adolescents (at least 16 years
of age) with a history of hepatitis B viremia while receiving lamivudine or known
lamivudine or telbivudine resistance substitutions rtM204I/V with or without rtL180M,
rtL80I/V, or rtV173L is 1 mg once daily.
Decompensated Liver Disease
The recommended dose of BARACLUDE for chronic hepatitis B virus infection in adults
with decompensated liver disease is 1 mg once daily.
2.3 Recommended Dosage in Pediatric Patients
Table 1 describes the recommended dose of BARACLUDE for pediatric patients 2 years
of age or older and weighing at least 10 kg. The oral solution should be used for patients
with body weight up to 30 kg.
Table 1: Dosing Schedule for Pediatric Patients
Recommended Once-Daily Dose of Oral Solution (mL)
Body Weight (kg)
Treatment-Naïve
Patients
a
Lamivudine-Experienced
Patients
b
10 to 11 3 6
greater than 11 to 14 4 8
greater than 14 to 17 5 10
greater than 17 to 20 6 12
greater than 20 to 23 7 14
greater than 23 to 26 8 16
greater than 26 to 30 9 18
greater than 30 10 20
a
Children with body weight greater than 30 kg should receive 10 mL (0.5 mg) of oral
solution or one 0.5 mg tablet once daily.
b
Children with body weight greater than 30 kg should receive 20 mL (1 mg) of oral
solution or one 1 mg tablet once daily.
2.4 Renal Impairment
In adult subjects with renal impairment, the apparent oral clearance of entecavir
decreased as creatinine clearance decreased [see Clinical Pharmacology (12.3)]. Dosage
adjustment is recommended for patients with creatinine clearance less than 50 mL/min,
including patients on hemodialysis or continuous ambulatory peritoneal dialysis (CAPD),
as shown in Table 2. The once-daily dosing regimens are preferred.
Table 2: Recommended Dosage of BARACLUDE in Adult Patients with Renal
Impairment
Creatinine Clearance
(mL/min)
Usual Dose
(0.5 mg)
Lamivudine-Refractory
or Decompensated
Liver Disease (1 mg)
50 or greater 0.5 mg once daily 1 mg once daily
30 to less than 50
0.25 mg once daily
a
OR
0.5 mg every 48 hours
0.5 mg once daily
OR
1 mg every 48 hours
10 to less than 30
0.15 mg once daily
a
OR
0.5 mg every 72 hours
0.3 mg once daily
a
OR
1 mg every 72 hours
Less than 10
Hemodialysis
b
or CAPD
0.05 mg once daily
a
OR
0.5 mg every 7 days
0.1 mg once daily
a
OR
1 mg every 7 days
a
For doses less than 0.5 mg, BARACLUDE Oral Solution is recommended.
b
If administered on a hemodialysis day, administer BARACLUDE after the hemodialysis
session.
Although there are insufficient data to recommend a specific dose adjustment of
BARACLUDE in pediatric patients with renal impairment, a reduction in the dose or an
increase in the dosing interval similar to adjustments for adults should be considered.
2.5 Hepatic Impairment
No dosage adjustment is necessary for patients with hepatic impairment.
2.6 Duration of Therapy
The optimal duration of treatment with BARACLUDE for patients with chronic hepatitis B
virus infection and the relationship between treatment and long-term outcomes such as
cirrhosis and hepatocellular carcinoma are unknown.
3 DOSAGE FORMS AND STRENGTHS
• BARACLUDE 0.5 mg film-coated tablets are white to off-white, triangular-shaped,
and debossed with “BMS” on one side and “1611” on the other side.
• BARACLUDE 1 mg film-coated tablets are pink, triangular-shaped, and debossed
with “BMS” on one side and “1612” on the other side.
• BARACLUDE oral solution, 0.05 mg/mL, is a ready-to-use, orange-flavored, clear,
colorless to pale yellow, aqueous solution. Ten milliliters of the oral solution provides
a 0.5 mg dose and 20 mL provides a 1 mg dose of entecavir.
4 CONTRAINDICATIONS
None.
5 WARNINGS AND PRECAUTIONS
5.1 Severe Acute Exacerbations of Hepatitis B
Severe acute exacerbations of hepatitis B have been reported in patients who have
discontinued anti-hepatitis B therapy, including entecavir [see Adverse Reactions
(6.1)]. Hepatic function should be monitored closely with both clinical and laboratory
follow-up for at least several months in patients who discontinue anti-hepatitis B therapy.
If appropriate, initiation of anti-hepatitis B therapy may be warranted.
5.2 Patients Co-infected with HIV and HBV
BARACLUDE has not been evaluated in HIV/HBV co-infected patients who were not
simultaneously receiving effective HIV treatment. Limited clinical experience suggests
there is a potential for the development of resistance to HIV nucleoside reverse
transcriptase inhibitors if BARACLUDE is used to treat chronic hepatitis B virus infection
in patients with HIV infection that is not being treated [see Microbiology (12.4)]. Therefore,
therapy with BARACLUDE is not recommended for HIV/HBV co-infected patients who are
not also receiving HAART. Before initiating BARACLUDE therapy, HIV antibody testing
should be offered to all patients. BARACLUDE has not been studied as a treatment for
HIV infection and is not recommended for this use.
5.3 Lactic Acidosis and Severe Hepatomegaly with Steatosis
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been
reported with the use of nucleoside analogue inhibitors, including BARACLUDE, alone
or in combination with antiretrovirals. A majority of these cases have been in women.
Obesity and prolonged nucleoside inhibitor exposure may be risk factors. Particular
caution should be exercised when administering nucleoside analogue inhibitors to any
patient with known risk factors for liver disease; however, cases have also been reported
in patients with no known risk factors.
Lactic acidosis with BARACLUDE use has been reported, often in association with
hepatic decompensation, other serious medical conditions, or drug exposures.
Patients with decompensated liver disease may be at higher risk for lactic acidosis.

BARACLUDE
®
(entecavir) BARACLUDE
®
(entecavir)
Laboratory Abnormalities
Frequencies of selected treatment-emergent laboratory abnormalities reported during
therapy in four clinical trials of BARACLUDE compared with lamivudine are listed in
Table 4.
Table 4: Selected Treatment-Emergent
a
Laboratory Abnormalities Reported in
Four Entecavir Clinical Trials Through 2 Years
Nucleoside-Inhibitor-Naïve
b
Lamivudine-Refractory
c
Test
BARACLUDE
0.5 mg
n=679
Lamivudine
100 mg
n=668
BARACLUDE
1 mg
n=183
Lamivudine
100 mg
n=190
Any Grade 3–4
laboratory abnormality
d
35% 36% 37% 45%
ALT >10 × ULN and
>2 × baseline
2% 4% 2% 11%
ALT >5 × ULN 11% 16% 12% 24%
Albumin <2.5 g/dL <1% <1% 0 2%
Total bilirubin >2.5 × ULN 2% 2% 3% 2%
Lipase ≥2.1 × ULN
7% 6% 7% 7%
Creatinine >3 × ULN 0 0 0 0
Confirmed creatinine
increase ≥0.5 mg/dL
1% 1% 2% 1%
Hyperglycemia, fasting
>250 mg/dL
2% 1% 3% 1%
Glycosuria
e
4% 3% 4% 6%
Hematuria
f
9% 10% 9% 6%
Platelets <50,000/mm
3
<1% <1% <1% <1%
a
On-treatment value worsened from baseline to Grade 3 or Grade 4 for all parameters
except albumin (any on-treatment value <2.5 g/dL), confirmed creatinine increase
≥0.5 mg/dL, and ALT >10 × ULN and >2 × baseline.
b
Studies AI463022 and AI463027.
c
Includes Study AI463026 and the BARACLUDE 1 mg and lamivudine treatment arms
of Study AI463014, a Phase 2 multinational, randomized, double-blind study of three
doses of BARACLUDE (0.1, 0.5, and 1 mg) once daily versus continued lamivudine
100 mg once daily for up to 52 weeks in subjects who experienced recurrent viremia
on lamivudine therapy.
d
Includes hematology, routine chemistries, renal and liver function tests, pancreatic
enzymes, and urinalysis.
e
Grade 3 = 3+, large, ≥500 mg/dL; Grade 4 = 4+, marked, severe.
f
Grade 3 = 3+, large; Grade 4 = ≥4+, marked, severe, many.
ULN=upper limit of normal.
Among BARACLUDE-treated subjects in these studies, on-treatment ALT elevations
greater than 10 times the upper limit of normal (ULN) and greater than 2 times baseline
generally resolved with continued treatment. A majority of these exacerbations were
associated with a ≥2 log
10
/mL reduction in viral load that preceded or coincided with the
ALT elevation. Periodic monitoring of hepatic function is recommended during treatment.
Exacerbations of Hepatitis After Discontinuation of Treatment
An exacerbation of hepatitis or ALT flare was defined as ALT greater than 10 times
ULN and greater than 2 times the subject’s reference level (minimum of the baseline
or last measurement at end of dosing). For all subjects who discontinued treatment
(regardless of reason), Table 5 presents the proportion of subjects in each study who
experienced post-treatment ALT flares. In these studies, a subset of subjects was allowed
to discontinue treatment at or after 52 weeks if they achieved a protocol-defined response
to therapy. If BARACLUDE is discontinued without regard to treatment response, the rate
of post-treatment flares could be higher. [See Warnings and Precautions (5.1).]
Table 5: Exacerbations of Hepatitis During Off-Treatment Follow-up, Subjects in
Studies AI463022, AI463027, and AI463026
Subjects with ALT Elevations >10 × ULN and >2 × Reference
a
BARACLUDE Lamivudine
Nucleoside-inhibitor-naïve
HBeAg-positive 4/174 (2%) 13/147 (9%)
HBeAg-negative 24/302 (8%) 30/270 (11%)
Lamivudine-refractory 6/52 (12%) 0/16
a
Reference is the minimum of the baseline or last measurement at end of dosing.
Median time to off-treatment exacerbation was 23 weeks for BARACLUDE-treated
subjects and 10 weeks for lamivudine-treated subjects.
Treatment with BARACLUDE should be suspended in any patient who develops clinical
or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity (which
may include hepatomegaly and steatosis even in the absence of marked transaminase
elevations).
6 ADVERSE REACTIONS
The following adverse reactions are discussed in other sections of the labeling:
• Exacerbations of hepatitis after discontinuation of treatment [see Boxed Warning,
Warnings and Precautions (5.1)].
• Lactic acidosis and severe hepatomegaly with steatosis [see Boxed Warning,
Warnings and Precautions (5.3)].
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction
rates observed in the clinical trials of a drug cannot be directly compared to rates in the
clinical trials of another drug and may not reflect the rates observed in practice.
Clinical Trial Experience in Adults
Compensated Liver Disease
Assessment of adverse reactions is based on four studies (AI463014, AI463022,
AI463026, and AI463027) in which 1720 subjects with chronic hepatitis B virus infection
and compensated liver disease received double-blind treatment with BARACLUDE
0.5 mg/day (n=679), BARACLUDE 1 mg/day (n=183), or lamivudine (n=858) for up to
2 years. Median duration of therapy was 69 weeks for BARACLUDE-treated subjects
and 63 weeks for lamivudine-treated subjects in Studies AI463022 and AI463027 and
73 weeks for BARACLUDE-treated subjects and 51 weeks for lamivudine-treated subjects
in Studies AI463026 and AI463014. The safety profiles of BARACLUDE and lamivudine
were comparable in these studies.
The most common adverse reactions of any severity (≥3%) with at least a possible
relation to study drug for BARACLUDE-treated subjects were headache, fatigue, dizziness,
and nausea. The most common adverse reactions among lamivudine-treated subjects
were headache, fatigue, and dizziness. One percent of BARACLUDE-treated subjects in
these four studies compared with 4% of lamivudine-treated subjects discontinued for
adverse events or abnormal laboratory test results.
Clinical adverse reactions of moderate-severe intensity and considered at least possibly
related to treatment occurring during therapy in four clinical studies in which BARACLUDE
was compared with lamivudine are presented in Table 3.
Table 3: Clinical Adverse Reactions
a
of Moderate-Severe Intensity (Grades 2–4)
Reported in Four Entecavir Clinical Trials Through 2 Years
Nucleoside-Inhibitor-Naïve
b
Lamivudine-Refractory
c
Body System/
Adverse Reaction
BARACLUDE
0.5 mg
n=679
Lamivudine
100 mg
n=668
BARACLUDE
1 mg
n=183
Lamivudine
100 mg
n=190
Any Grade 2–4
adverse reaction
a
15% 18% 22% 23%
Gastrointestinal
Diarrhea <1% 0 1% 0
Dyspepsia <1% <1% 1% 0
Nausea <1% <1% <1% 2%
Vomiting <1% <1% <1% 0
General
Fatigue 1% 1% 3% 3%
Nervous System
Headache 2% 2% 4% 1%
Dizziness <1% <1% 0 1%
Somnolence <1% <1% 0 0
Psychiatric
Insomnia <1% <1% 0 <1%
a
Includes events of possible, probable, certain, or unknown relationship to treatment
regimen.
b
Studies AI463022 and AI463027.
c
Includes Study AI463026 and the BARACLUDE 1 mg and lamivudine treatment arms
of Study AI463014, a Phase 2 multinational, randomized, double-blind study of three
doses of BARACLUDE (0.1, 0.5, and 1 mg) once daily versus continued lamivudine
100 mg once daily for up to 52 weeks in subjects who experienced recurrent viremia
on lamivudine therapy.
BARACLUDE
®
(entecavir) BARACLUDE
®
(entecavir)
Table 6: Principal Analyses of Time to Adjudicated Events - Randomized
Treated Subjects
Number of Subjects with Events
Endpoint
c
BARACLUDE
N=6,216
Non-ETV
N=6,162
Hazard Ratio
[BARACLUDE:Non-ETV]
(CI
a
)
Primary Endpoints
Overall malignant
neoplasm
331 337 0.93 (0.800, 1.084)
Liver-related HBV
disease progression
350 375 0.89 (0.769, 1.030)
Death 238 264 0.85 (0.713, 1.012)
Secondary Endpoints
Non-HCC malignant
neoplasm
95 81 1.10 (0.817, 1.478)
HCC 240
b
263 0.87 (0.727, 1.032)
Analyses were stratified by geographic region and prior HBV nucleos(t)ide experience.
a
95.03% CI for overall malignant neoplasm, death, and liver-related HBV disease
progression; 95% CI for non-HCC malignant neoplasm and HCC.
b
One subject had a pre-treatment HCC event and was excluded from the analysis.
c
Overall malignant neoplasm is a composite event of HCC or non-HCC malignant
neoplasm. Liver-related HBV disease progression is a composite event of liver-related
death, HCC, or non-HCC HBV disease progression.
CI = confidence interval; N = total number of subjects.
Limitations of the study included population changes over the long-term follow-up
period and more frequent post-randomization treatment changes in the non-ETV group.
In addition, the study was underpowered to demonstrate a difference in the non-HCC
malignancy rate because of the lower than expected background rate.
Adverse Reactions from Postmarketing Spontaneous Reports
The following adverse reactions have been reported during postmarketing use of
BARACLUDE. Because these reactions were reported voluntarily from a population of
unknown size, it is not possible to reliably estimate their frequency or establish a causal
relationship to BARACLUDE exposure.
Immune system disorders: Anaphylactoid reaction.
Metabolism and nutrition disorders: Lactic acidosis.
Hepatobiliary disorders: Increased transaminases.
Skin and subcutaneous tissue disorders: Alopecia, rash.
7 DRUG INTERACTIONS
Since entecavir is primarily eliminated by the kidneys [see Clinical Pharmacology (12.3)],
coadministration of BARACLUDE with drugs that reduce renal function or compete for
active tubular secretion may increase serum concentrations of either entecavir or the
coadministered drug. Coadministration of entecavir with lamivudine, adefovir dipivoxil, or
tenofovir disoproxil fumarate did not result in significant drug interactions. The effects of
coadministration of BARACLUDE with other drugs that are renally eliminated or are known
to affect renal function have not been evaluated, and patients should be monitored closely
for adverse events when BARACLUDE is coadministered with such drugs.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women
exposed to BARACLUDE during pregnancy. Healthcare providers are encouraged to
register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
Risk Summary
Prospective pregnancy data from the APR are not sufficient to adequately assess the risk
of birth defects, miscarriage or adverse maternal or fetal outcomes. Entecavir use during
pregnancy has been evaluated in a limited number of individuals reported to the APR and
the number of exposures to entecavir is insufficient to make a risk assessment compared
to a reference population. The estimated background rate for major birth defects is 2.7%
in the U.S. reference population of the Metropolitan Atlanta Congenital Defects Program
(MACDP). The rate of miscarriage is not reported in the APR. All pregnancies have a
background risk of birth defect, loss, or other adverse outcomes. In the U.S. general
population, the estimated background risk of miscarriage in clinically recognized
pregnancies is 15–20%.
In animal reproduction studies, no adverse developmental effects were observed with
entecavir at clinically relevant exposures. No developmental toxicities were observed at
systemic exposures (AUC) approximately 25 (rats) and 200 (rabbits) times the exposure at
the maximum recommended human dose (MRHD) of 1 mg/day (see Data).
Decompensated Liver Disease
Study AI463048 was a randomized, open-label study of BARACLUDE 1 mg once daily
versus adefovir dipivoxil 10 mg once daily given for up to 48 weeks in adult subjects
with chronic HBV infection and evidence of hepatic decompensation, defined as a
Child-Turcotte-Pugh (CTP) score of 7 or higher [see Clinical Studies (14.1)]. Among the
102 subjects receiving BARACLUDE, the most common treatment-emergent adverse
events of any severity, regardless of causality, occurring through Week 48 were peripheral
edema (16%), ascites (15%), pyrexia (14%), hepatic encephalopathy (10%), and
upper respiratory infection (10%). Clinical adverse reactions not listed in Table 3 that
were observed through Week 48 include blood bicarbonate decreased (2%) and
renal failure (<1%).
Eighteen of 102 (18%) subjects treated with BARACLUDE and 18/89 (20%) subjects
treated with adefovir dipivoxil died during the first 48 weeks of therapy. The majority
of deaths (11 in the BARACLUDE group and 16 in the adefovir dipivoxil group) were
due to liver-related causes such as hepatic failure, hepatic encephalopathy, hepatorenal
syndrome, and upper gastrointestinal hemorrhage. The rate of hepatocellular carcinoma
(HCC) through Week 48 was 6% (6/102) for subjects treated with BARACLUDE and
8% (7/89) for subjects treated with adefovir dipivoxil. Five percent of subjects in either
treatment arm discontinued therapy due to an adverse event through Week 48.
No subject in either treatment arm experienced an on-treatment hepatic flare
(ALT >2 × baseline and >10 × ULN) through Week 48. Eleven of 102 (11%) subjects
treated with BARACLUDE and 11/89 (13%) subjects treated with adefovir dipivoxil had a
confirmed increase in serum creatinine of 0.5 mg/dL through Week 48.
HIV/HBV Co-infected
The safety profile of BARACLUDE 1 mg (n=51) in HIV/HBV co-infected subjects enrolled
in Study AI463038 was similar to that of placebo (n=17) through 24 weeks of blinded
treatment and similar to that seen in non-HIV infected subjects [see Warnings and
Precautions (5.2)].
Liver Transplant Recipients
Among 65 subjects receiving BARACLUDE in an open-label, post-liver transplant trial
[see Use in Specific Populations (8.8)], the frequency and nature of adverse events were
consistent with those expected in patients who have received a liver transplant and the
known safety profile of BARACLUDE.
Clinical Trial Experience in Pediatric Subjects
The safety of BARACLUDE in pediatric subjects 2 to less than 18 years of age is based
on two clinical trials in subjects with chronic HBV infection (one Phase 2 pharmacokinetic
trial [AI463028] and one Phase 3 trial [AI463189]). These trials provided experience in
168 HBeAg-positive subjects treated with BARACLUDE for a median duration of 72 weeks.
The adverse reactions observed in pediatric subjects who received treatment with
BARACLUDE were consistent with those observed in clinical trials of BARACLUDE in
adults. Adverse drug reactions reported in greater than 1% of pediatric subjects included
abdominal pain, rash events, poor palatability (“product taste abnormal”), nausea,
diarrhea, and vomiting.
6.2 Postmarketing Experience
Data from Long-Term Observational Study
Study AI463080 was a randomized, global, observational, open-label Phase 4 study to
assess long-term risks and benefits of BARACLUDE (0.5 mg/day or 1 mg/day) treatment
as compared to other standard-of-care HBV nucleos(t)ide analogues in subjects with
chronic HBV infection.
A total of 12,378 patients were treated with BARACLUDE (n=6,216) or other HBV
nucleos(t)ide treatment [non-entecavir (ETV)] (n=6,162). Patients were evaluated at
baseline and subsequently every 6 months for up to 10 years. The principal clinical
outcome events assessed during the study were overall malignant neoplasms,
liver-related HBV disease progression, HCC, non-HCC malignant neoplasms, and death.
The study showed that BARACLUDE was not significantly associated with an increased
risk of malignant neoplasms compared to other standard-of-care HBV nucleos(t)ides,
as assessed by either the composite endpoint of overall malignant neoplasms or the
individual endpoint of non-HCC malignant neoplasms. The most commonly reported
malignancy in both the BARACLUDE and non-ETV groups was HCC followed by
gastrointestinal malignancies. The data also showed that long-term BARACLUDE use was
not associated with a lower occurrence of HBV disease progression or a lower rate of
death overall compared to other HBV nucleos(t)ides. The principal clinical outcome event
assessments are shown in Table 6.
BARACLUDE
®
(entecavir) BARACLUDE
®
(entecavir)
Data
Animal Data
Entecavir was administered orally to pregnant rats (at 2, 20, and 200 mg per kg per
day) and rabbits (at 1, 4, and 16 mg per kg per day) during organogenesis (on gestation
Days 6 through 15 [rat] and 6 through 18 [rabbit]). In rats, embryofetal toxicity including
post-implantation loss, resorptions, tail and vertebral malformations, skeletal variations
including reduced ossification (vertebrate, sternebrae, and phalanges) and extra lumbar
vertebrae and ribs, and lower fetal body weights were observed at systemic exposures
(AUC) 3,100 times those in humans at the MRHD. Maternal toxicity was also observed at
this dose level. In rabbits, embryofetal toxicity including post-implantation loss, resorptions
and skeletal variations, including reduced ossification (hyoid) and increased incidence of
13
th
rib, were observed at systemic exposures (AUC) 883 times those in humans at the
MRHD. There were no signs of embryofetal toxicity when pregnant animals received oral
entecavir at 28 (rat) and 212 (rabbit) times the human exposure (AUC) at the MRHD. In a
pre/postnatal development study, entecavir was administered orally to pregnant rats at
0.3, 3, and 30 mg per kg per day from gestation day 6 to lactation/post-partum day 20.
No adverse effects on the offspring occurred at up to the highest dose evaluated, resulting
in exposures (AUC) greater than 94 times those in humans at the MRHD.
8.2 Lactation
Risk Summary
It is not known whether BARACLUDE is present in human breast milk, affects human
milk production, or has effects on the breastfed infant. When administered to lactating
rats, entecavir was present in milk (see Data). The developmental and health benefits of
breastfeeding should be considered along with the mother’s clinical need for BARACLUDE
and any potential adverse effects on the breastfed infant from BARACLUDE or from the
underlying maternal condition.
Data
Entecavir was excreted into the milk of lactating rats following a single oral dose of 10 mg
per kg on lactation day 7. Entecavir in milk was approximately 25% that in maternal
plasma (based on AUC).
8.4 Pediatric Use
BARACLUDE was evaluated in two clinical trials of pediatric subjects 2 years of age
and older with HBeAg-positive chronic HBV infection and compensated liver disease.
The exposure of BARACLUDE in nucleoside-inhibitor-treatment-naïve and lamivudine-
experienced pediatric subjects 2 years of age and older with HBeAg-positive chronic
HBV infection and compensated liver disease receiving 0.015 mg/kg (up to 0.5 mg
once daily) or 0.03 mg/kg (up to 1 mg once daily), respectively, was evaluated in Study
AI463028. Safety and efficacy of the selected dose in treatment-naïve pediatric subjects
were confirmed in Study AI463189, a randomized, placebo-controlled treatment trial
[see Indications and Usage (1), Dosage and Administration (2.3), Adverse Reactions (6.1),
Clinical Pharmacology (12.3), and Clinical Studies (14.2)].
There are limited data available on the use of BARACLUDE in lamivudine-experienced
pediatric patients; BARACLUDE should be used in these patients only if the potential
benefit justifies the potential risk to the child. Since some pediatric patients may require
long-term or even lifetime management of chronic active hepatitis B, consideration should
be given to the impact of BARACLUDE on future treatment options [see Microbiology (12.4)].
The efficacy and safety of BARACLUDE have not been established in patients less than
2 years of age. Use of BARACLUDE in this age group has not been evaluated because
treatment of HBV in this age group is rarely required.
8.5 Geriatric Use
Clinical studies of BARACLUDE did not include sufficient numbers of subjects aged
65 years and over to determine whether they respond differently from younger subjects.
Entecavir is substantially excreted by the kidney, and the risk of toxic reactions to this
drug may be greater in patients with impaired renal function. Because elderly patients
are more likely to have decreased renal function, care should be taken in dose selection,
and it may be useful to monitor renal function [see Dosage and Administration (2.4)].
8.6 Racial/Ethnic Groups
There are no significant racial differences in entecavir pharmacokinetics. The safety and
efficacy of BARACLUDE 0.5 mg once daily were assessed in a single-arm, open-label
trial of HBeAg-positive or -negative, nucleoside-inhibitor-naïve, Black/African American
(n=40) and Hispanic (n=6) subjects with chronic HBV infection. In this trial, 76% of
subjects were male, the mean age was 42 years, 57% were HBeAg-positive, the mean
baseline HBV DNA was 7.0 log
10
IU/mL, and the mean baseline ALT was 162 U/L. At
Week 48 of treatment, 32 of 46 (70%) subjects had HBV DNA <50 IU/mL (approximately
300 copies/mL), 31 of 46 (67%) subjects had ALT normalization (≤1 × ULN), and
12 of 26 (46%) HBeAg-positive subjects had HBe seroconversion. Safety data were similar
to those observed in the larger controlled clinical trials.
Because of low enrollment, safety and efficacy have not been established in the US
Hispanic population.
8.7 Renal Impairment
Dosage adjustment of BARACLUDE is recommended for patients with creatinine clearance
less than 50 mL/min, including patients on hemodialysis or CAPD [see Dosage and
Administration (2.4) and Clinical Pharmacology (12.3)].
8.8 Liver Transplant Recipients
The safety and efficacy of BARACLUDE were assessed in a single-arm, open-label trial
in 65 subjects who received a liver transplant for complications of chronic HBV infection.
Eligible subjects who had HBV DNA less than 172 IU/mL (approximately 1000 copies/mL)
at the time of transplant were treated with BARACLUDE 1 mg once daily in addition to
usual post-transplantation management, including hepatitis B immune globulin. The trial
population was 82% male, 39% Caucasian, and 37% Asian, with a mean age of 49 years;
89% of subjects had HBeAg-negative disease at the time of transplant.
Four of the 65 subjects received 4 weeks or less of BARACLUDE (2 deaths,
1 re-transplantation, and 1 protocol violation) and were not considered evaluable. Of the
61 subjects who received more than 4 weeks of BARACLUDE, 60 received hepatitis B
immune globulin post-transplant. Fifty-three subjects (82% of all 65 subjects treated)
completed the trial and had HBV DNA measurements at or after 72 weeks treatment
post-transplant. All 53 subjects had HBV DNA <50 IU/mL (approximately 300 copies/mL).
Eight evaluable subjects did not have HBV DNA data available at 72 weeks, including
3 subjects who died prior to study completion. No subjects had HBV DNA values
≥50 IU/mL while receiving BARACLUDE (plus hepatitis B immune globulin). All 61
evaluable subjects lost HBsAg post-transplant; 2 of these subjects experienced recurrence
of measurable HBsAg without recurrence of HBV viremia. This trial was not designed to
determine whether addition of BARACLUDE to hepatitis B immune globulin decreased the
proportion of subjects with measurable HBV DNA post-transplant compared to hepatitis B
immune globulin alone.
If BARACLUDE treatment is determined to be necessary for a liver transplant recipient
who has received or is receiving an immunosuppressant that may affect renal function,
such as cyclosporine or tacrolimus, renal function must be carefully monitored both before
and during treatment with BARACLUDE [see Dosage and Administration (2.4) and Clinical
Pharmacology (12.3)].
10 OVERDOSAGE
There is limited experience of entecavir overdosage reported in patients. Healthy subjects
who received single entecavir doses up to 40 mg or multiple doses up to 20 mg/day for
up to 14 days had no increase in or unexpected adverse events. If overdose occurs, the
patient must be monitored for evidence of toxicity, and standard supportive treatment
applied as necessary.
Following a single 1 mg dose of entecavir, a 4-hour hemodialysis session removed
approximately 13% of the entecavir dose.
11 DESCRIPTION
BARACLUDE
®
is the tradename for entecavir, a guanosine nucleoside analogue
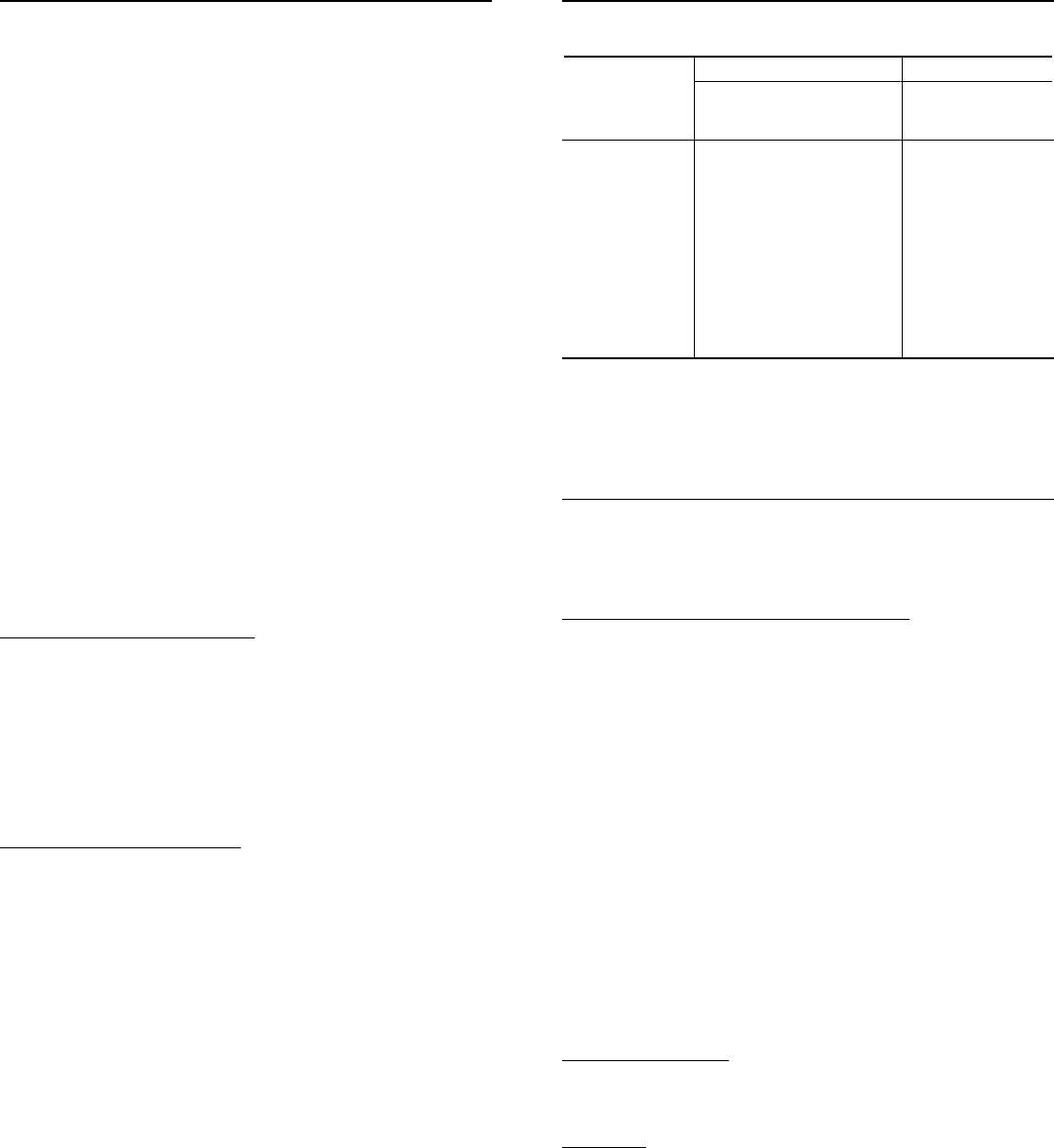
with selective activity against HBV. The chemical name for entecavir is 2-amino-1,
9-dihydro-9-[(1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylenecyclopentyl]-
6H-purin-6-one, monohydrate. Its molecular formula is C
12
H
15
N
5
O
3
•H
2
O, which
corresponds to a molecular weight of 295.3. Entecavir has the following structural
formula:
Entecavir is a white to off-white powder. It is slightly soluble in water (2.4 mg/mL),
and the pH of the saturated solution in water is 7.9 at 25° C ± 0.5° C.
BARACLUDE film-coated tablets are available for oral administration in strengths of
0.5 mg and 1 mg of entecavir. BARACLUDE 0.5 mg and 1 mg film-coated tablets contain
the following inactive ingredients: lactose monohydrate, microcrystalline cellulose,
crospovidone, povidone, and magnesium stearate. The tablet coating contains titanium
dioxide, hypromellose, polyethylene glycol 400, polysorbate 80 (0.5 mg tablet only),
and iron oxide red (1 mg tablet only). BARACLUDE Oral Solution is available for oral
administration as a ready-to-use solution containing 0.05 mg of entecavir per milliliter.
BARACLUDE Oral Solution contains the following inactive ingredients: maltitol, sodium
citrate, citric acid, methylparaben, propylparaben, and orange flavor.

BARACLUDE
®
(entecavir) BARACLUDE
®
(entecavir)
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Entecavir is an antiviral drug against hepatitis B virus [see Microbiology (12.4)].
12.3 Pharmacokinetics
The single- and multiple-dose pharmacokinetics of entecavir were evaluated in healthy
subjects and subjects with chronic hepatitis B virus infection.
Absorption
Following oral administration in healthy subjects, entecavir peak plasma concentrations
occurred between 0.5 and 1.5 hours. Following multiple daily doses ranging from
0.1 to 1 mg, C
max
and area under the concentration-time curve (AUC) at steady state
increased in proportion to dose. Steady state was achieved after 6 to 10 days of
once-daily administration with approximately 2-fold accumulation. For a 0.5 mg oral
dose, C
max
at steady state was 4.2 ng/mL and trough plasma concentration (C
trough
) was
0.3 ng/mL. For a 1 mg oral dose, C
max
was 8.2 ng/mL and C
trough
was 0.5 ng/mL.
In healthy subjects, the bioavailability of the tablet was 100% relative to the oral solution.
The oral solution and tablet may be used interchangeably.
Effects of food on oral absorption: Oral administration of 0.5 mg of entecavir with a
standard high-fat meal (945 kcal, 54.6 g fat) or a light meal (379 kcal, 8.2 g fat) resulted
in a delay in absorption (1.0–1.5 hours fed vs. 0.75 hours fasted), a decrease in C
max
of
44%–46%, and a decrease in AUC of 18%–20% [see Dosage and Administration (2)].
Distribution
Based on the pharmacokinetic profile of entecavir after oral dosing, the estimated
apparent volume of distribution is in excess of total body water, suggesting that entecavir
is extensively distributed into tissues.
Binding of entecavir to human serum proteins in vitro was approximately 13%.
Metabolism and Elimination
Following administration of
14
C-entecavir in humans and rats, no oxidative or acetylated
metabolites were observed. Minor amounts of phase II metabolites (glucuronide and
sulfate conjugates) were observed. Entecavir is not a substrate, inhibitor, or inducer of
the cytochrome P450 (CYP450) enzyme system. See Drug Interactions, below.
After reaching peak concentration, entecavir plasma concentrations decreased
in a bi-exponential manner with a terminal elimination half-life of approximately
128–149 hours. The observed drug accumulation index is approximately 2-fold with once-
daily dosing, suggesting an effective accumulation half-life of approximately 24 hours.
Entecavir is predominantly eliminated by the kidney with urinary recovery of unchanged
drug at steady state ranging from 62% to 73% of the administered dose. Renal clearance
is independent of dose and ranges from 360 to 471 mL/min suggesting that entecavir
undergoes both glomerular filtration and net tubular secretion [see Drug Interactions (7)].
Special Populations
Gender: There are no significant gender differences in entecavir pharmacokinetics.
Race: There are no significant racial differences in entecavir pharmacokinetics.
Elderly: The effect of age on the pharmacokinetics of entecavir was evaluated following
administration of a single 1 mg oral dose in healthy young and elderly volunteers.
Entecavir AUC was 29.3% greater in elderly subjects compared to young subjects. The
disparity in exposure between elderly and young subjects was most likely attributable
to differences in renal function. Dosage adjustment of BARACLUDE should be based on
the renal function of the patient, rather than age [see Dosage and Administration (2.4)].
Pediatrics: The steady-state pharmacokinetics of entecavir were evaluated in nucleoside-
inhibitor-naïve and lamivudine-experienced HBeAg-positive pediatric subjects 2 to less
than 18 years of age with compensated liver disease. Results are shown in Table 7.
Entecavir exposure among nucleoside-inhibitor-naïve subjects was similar to the exposure
achieved in adults receiving once-daily doses of 0.5 mg. Entecavir exposure among
lamivudine-experienced subjects was similar to the exposure achieved in adults receiving
once-daily doses of 1 mg.
Table 7:
Pharmacokinetic Parameters in Pediatric Subjects
Nucleoside-Inhibitor-Naïve
a
n=24
Lamivudine-Experienced
b
n=19
C
max
(ng/mL)
(CV%)
6.31
(30)
14.48
(31)
AUC
(0–24)
(ng•h/mL)
(CV%)
18.33
(27)
38.58
(26)
C
min
(ng/mL)
(CV%)
0.28
(22)
0.47
(23)
a
Subjects received once-daily doses of 0.015 mg/kg up to a maximum of 0.5 mg.
b
Subjects received once-daily doses of 0.030 mg/kg up to a maximum of 1 mg.
Renal impairment: The pharmacokinetics of entecavir following a single 1 mg dose were
studied in subjects (without chronic hepatitis B virus infection) with selected degrees
of renal impairment, including subjects whose renal impairment was managed by
hemodialysis or continuous ambulatory peritoneal dialysis (CAPD). Results are shown in
Table 8 [see Dosage and Administration (2.4)].
Table 8: Pharmacokinetic Parameters in Subjects with Selected Degrees of
Renal Function
Renal Function Group
Baseline Creatinine Clearance (mL/min)
Unimpaired
>80
n=6
Mild
>50–≤80
n=6
Moderate
30–50
n=6
Severe
<30
n=6
Severe
Managed with
Hemodialysis
a
n=6
Severe
Managed
with CAPD
n=4
C
max
(ng/mL)
(CV%)
8.1
(30.7)
10.4
(37.2)
10.5
(22.7)
15.3
(33.8)
15.4
(56.4)
16.6
(29.7)
AUC
(0-T)
(ng•h/mL)
(CV)
27.9
(25.6)
51.5
(22.8)
69.5
(22.7)
145.7
(31.5)
233.9
(28.4)
221.8
(11.6)
CLR (mL/min)
(SD)
383.2
(101.8)
197.9
(78.1)
135.6
(31.6)
40.3
(10.1)
NA
NA
CLT/F (mL/min)
(SD)
588.1
(153.7)
309.2
(62.6)
226.3
(60.1)
100.6
(29.1)
50.6
(16.5)
35.7
(19.6)
a
Dosed immediately following hemodialysis.
CLR = renal clearance; CLT/F = apparent oral clearance.
Following a single 1 mg dose of entecavir administered 2 hours before the hemodialysis
session, hemodialysis removed approximately 13% of the entecavir dose over
4 hours. CAPD removed approximately 0.3% of the dose over 7 days [see Dosage and
Administration (2.4)].
Hepatic impairment: The pharmacokinetics of entecavir following a single 1 mg dose
were studied in adult subjects (without chronic hepatitis B virus infection) with moderate
or severe hepatic impairment (Child-Turcotte-Pugh Class B or C). The pharmacokinetics
of entecavir were similar between hepatically impaired and healthy control subjects;
therefore, no dosage adjustment of BARACLUDE is recommended for patients with
hepatic impairment. The pharmacokinetics of entecavir have not been studied in pediatric
subjects with hepatic impairment.
Post-liver transplant: Limited data are available on the safety and efficacy of BARACLUDE
in liver transplant recipients. In a small pilot study of entecavir use in HBV-infected
liver transplant recipients on a stable dose of cyclosporine A (n=5) or tacrolimus (n=4),
entecavir exposure was approximately 2-fold the exposure in healthy subjects with
normal renal function. Altered renal function contributed to the increase in entecavir
exposure in these subjects. The potential for pharmacokinetic interactions between
entecavir and cyclosporine A or tacrolimus was not formally evaluated [see Use in Specific
Populations (8.8)].
Drug Interactions
The metabolism of entecavir was evaluated in in vitro and in vivo studies. Entecavir is
not a substrate, inhibitor, or inducer of the cytochrome P450 (CYP450) enzyme system.
At concentrations up to approximately 10,000-fold higher than those obtained in
humans, entecavir inhibited none of the major human CYP450 enzymes 1A2, 2C9, 2C19,
2D6, 3A4, 2B6, and 2E1. At concentrations up to approximately 340-fold higher than
those observed in humans, entecavir did not induce the human CYP450 enzymes 1A2,
2C9, 2C19, 3A4, 3A5, and 2B6. The pharmacokinetics of entecavir are unlikely to be
affected by coadministration with agents that are either metabolized by, inhibit, or induce
the CYP450 system. Likewise, the pharmacokinetics of known CYP substrates are unlikely
to be affected by coadministration of entecavir.
The steady-state pharmacokinetics of entecavir and coadministered drug were not
altered in interaction studies of entecavir with lamivudine, adefovir dipivoxil, and tenofovir
disoproxil fumarate [see Drug Interactions (7)].
12.4 Microbiology
Mechanism of Action
Entecavir, a deoxyguanosine nucleoside analogue with activity against HBV reverse
transcriptase (rt), is efficiently phosphorylated to the active triphosphate form, which
has an intracellular half-life of 15 hours. By competing with the natural substrate
deoxyguanosine triphosphate, entecavir triphosphate functionally inhibits all three
activities of the HBV reverse transcriptase: (1) base priming, (2) reverse transcription
of the negative strand from the pregenomic messenger RNA, and (3) synthesis of the
positive strand of HBV DNA. Entecavir triphosphate is a weak inhibitor of cellular DNA
polymerases α, β, and δ and mitochondrial DNA polymerase γ with K
i
values ranging
from 18 to >160 μM.
Antiviral Activity
Entecavir inhibited HBV DNA synthesis (50% reduction, EC
50
) at a concentration of
0.004 µM in human HepG2 cells transfected with wild-type HBV. The median EC
50
value
for entecavir against lamivudine-resistant HBV (rtL180M, rtM204V) was 0.026 µM (range
0.010–0.059 μM).
The coadministration of HIV nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs)
with BARACLUDE is unlikely to reduce the antiviral efficacy of BARACLUDE against HBV
or of any of these agents against HIV. In HBV combination assays in cell culture, abacavir,
didanosine, lamivudine, stavudine, tenofovir, or zidovudine were not antagonistic to the
anti-HBV activity of entecavir over a wide range of concentrations. In HIV antiviral assays,

BARACLUDE
®
(entecavir) BARACLUDE
®
(entecavir)
entecavir was not antagonistic to the cell culture anti-HIV activity of these six NRTIs or
emtricitabine at concentrations greater than 100 times the C
max
of entecavir using the
1 mg dose.
Antiviral Activity Against HIV
A comprehensive analysis of the inhibitory activity of entecavir against a panel of laboratory
and clinical HIV type 1 (HIV-1) isolates using a variety of cells and assay conditions yielded
EC
50
values ranging from 0.026 to >10 µM; the lower EC
50
values were observed when
decreased levels of virus were used in the assay. In cell culture, entecavir selected for an
M184I substitution in HIV reverse transcriptase at micromolar concentrations, confirming
inhibitory pressure at high entecavir concentrations. HIV variants containing the M184V
substitution showed loss of susceptibility to entecavir.
Resistance
In Cell Culture
In cell-based assays, 8- to 30-fold reductions in entecavir phenotypic susceptibility were
observed for lamivudine-resistant strains. Further reductions (>70-fold) in entecavir
phenotypic susceptibility required the presence of amino acid substitutions rtM204I/V
with or without rtL180M along with additional substitutions at residues rtT184, rtS202,
or rtM250, or a combination of these substitutions with or without an rtI169 substitution
in the HBV reverse transcriptase. Lamivudine-resistant strains harboring rtL180M
plus rtM204V in combination with the amino acid substitution rtA181C conferred
16- to 122-fold reductions in entecavir phenotypic susceptibility.
Clinical Studies
Nucleoside-inhibitor-naïve subjects: Genotypic evaluations were performed on evaluable
samples (>300 copies/mL serum HBV DNA) from 562 subjects who were treated
with BARACLUDE for up to 96 weeks in nucleoside-inhibitor-naïve studies (AI463022,
AI463027, and rollover study AI463901). By Week 96, evidence of emerging amino acid
substitution rtS202G with rtL180M and rtM204V substitutions was detected in the HBV of
2 subjects (2/562=<1%), and 1 of them experienced virologic rebound (≥1 log
10
increase
above nadir). In addition, emerging amino acid substitutions at rtM204I/V and rtL80I,
rtV173L, or rtL180M, which conferred decreased phenotypic susceptibility to entecavir in
the absence of rtT184, rtS202, or rtM250 changes, were detected in the HBV of 3 subjects
(3/562=<1%) who experienced virologic rebound. For subjects who continued treatment
beyond 48 weeks, 75% (202/269) had HBV DNA <300 copies/mL at end of dosing
(up to 96 weeks).
HBeAg-positive (n=243) and -negative (n=39) treatment-naïve subjects who failed to
achieve the study-defined complete response by 96 weeks were offered continued
entecavir treatment in a rollover study. Complete response for HBeAg-positive was
<0.7 MEq/mL (approximately 7 × 10
5
copies/mL) serum HBV DNA and HBeAg loss
and, for HBeAg-negative was <0.7 MEq/mL HBV DNA and ALT normalization. Subjects
received 1 mg entecavir once daily for up to an additional 144 weeks. Of these
282 subjects, 141 HBeAg-positive and 8 HBeAg-negative subjects entered the long-term
follow-up rollover study and were evaluated for entecavir resistance. Of the 149 subjects
entering the rollover study, 88% (131/149), 92% (137/149), and 92% (137/149) attained
serum HBV DNA <300 copies/mL by Weeks 144, 192, and 240 (including end of dosing),
respectively. No novel entecavir resistance-associated substitutions were identified in a
comparison of the genotypes of evaluable isolates with their respective baseline isolates.
The cumulative probability of developing rtT184, rtS202, or rtM250 entecavir resistance-
associated substitutions (in the presence of rtL180M and rtM204V substitutions) at
Weeks 48, 96, 144, 192, and 240 was 0.2%, 0.5%, 1.2%, 1.2%, and 1.2%, respectively.
Lamivudine-refractory subjects: Genotypic evaluations were performed on evaluable
samples from 190 subjects treated with BARACLUDE for up to 96 weeks in studies of
lamivudine-refractory HBV (AI463026, AI463014, AI463015, and rollover study AI463901).
By Week 96, resistance-associated amino acid substitutions at rtT184, rtS202, or
rtM250, with or without rtI169 changes, in the presence of amino acid substitutions
rtM204I/V with or without rtL80V, rtV173L/M, or rtL180M emerged in the HBV from
22 subjects (22/190=12%), 16 of whom experienced virologic rebound (≥1 log
10
increase
above nadir) and 4 of whom were never suppressed <300 copies/mL. The HBV from 4
of these subjects had entecavir resistance substitutions at baseline and acquired further
changes on entecavir treatment. In addition to the 22 subjects, 3 subjects experienced
virologic rebound with the emergence of rtM204I/V and rtL80V, rtV173L/M, or rtL180M.
For isolates from subjects who experienced virologic rebound with the emergence
of resistance-associated substitutions (n=19), the median fold-change in entecavir
EC
50
values from reference was 19-fold at baseline and 106-fold at the time of virologic
rebound. For subjects who continued treatment beyond 48 weeks, 40% (31/77) had
HBV DNA <300 copies/mL at end of dosing (up to 96 weeks).
Lamivudine-refractory subjects (n=157) who failed to achieve the study-defined complete
response by Week 96 were offered continued entecavir treatment. Subjects received 1 mg
entecavir once daily for up to an additional 144 weeks. Of these subjects, 80 subjects
entered the long-term follow-up study and were evaluated for entecavir resistance.
By Weeks 144, 192, and 240 (including end of dosing), 34% (27/80), 35% (28/80), and
36% (29/80), respectively, attained HBV DNA <300 copies/mL. The cumulative probability
of developing rtT184, rtS202, or rtM250 entecavir resistance-associated substitutions (in
the presence of rtM204I/V with or without rtL180M substitutions) at Weeks 48, 96, 144,
192, and 240 was 6.2%, 15%, 36.3%, 46.6%, and 51.5%, respectively. The HBV of 6
subjects developed rtA181C/G/S/T amino acid substitutions while receiving entecavir, and
of these, 4 developed entecavir resistance-associated substitutions at rtT184, rtS202, or
rtM250 and 1 had an rtT184S substitution at baseline. Of 7 subjects whose HBV had an
rtA181 substitution at baseline, 2 also had substitutions at rtT184, rtS202, or rtM250 at
baseline and another 2 developed them while on treatment with entecavir.
In a post-approval integrated analysis of entecavir resistance data from 17 Phase 2
and 3 clinical trials, an emergent entecavir resistance-associated substitution rtA181C
was detected in 5 out of 1461 (0.3%) subjects during treatment with entecavir. This
substitution was detected only in the presence of lamivudine resistance-associated
substitutions rtL180M plus rtM204V.
Cross-resistance
Cross-resistance has been observed among HBV nucleoside analogue inhibitors.
In cell-based assays, entecavir had 8- to 30-fold less inhibition of HBV DNA synthesis
for HBV containing lamivudine and telbivudine resistance-associated substitutions
rtM204I/V with or without rtL180M than for wild-type HBV. Substitutions rtM204I/V with
or without rtL80I/V, rtV173L, or rtL180M, which are associated with lamivudine and
telbivudine resistance, also confer decreased phenotypic susceptibility to entecavir. The
efficacy of entecavir against HBV harboring adefovir resistance-associated substitutions
has not been established in clinical trials. HBV isolates from lamivudine-refractory subjects
failing entecavir therapy were susceptible in cell culture to adefovir but remained resistant
to lamivudine. Recombinant HBV genomes encoding adefovir resistance-associated
substitutions at either rtA181V or rtN236T had 1.1- or 0.3-fold shifts in susceptibility to
entecavir in cell culture, respectively.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term oral carcinogenicity studies of entecavir in mice and rats were carried out
at exposures up to approximately 42 times (mice) and 35 times (rats) those observed
in humans at the highest recommended dose of 1 mg/day. In mouse and rat studies,
entecavir was positive for carcinogenic findings. It is not known how predictive the results
of rodent carcinogenicity studies may be for humans [see Adverse Reactions (6.2)].
In mice, lung adenomas were increased in males and females at exposures 3 and
40 times those in humans. Lung carcinomas in both male and female mice were
increased at exposures 40 times those in humans. Combined lung adenomas and
carcinomas were increased in male mice at exposures 3 times and in female mice at
exposures 40 times those in humans. Tumor development was preceded by pneumocyte
proliferation in the lung, which was not observed in rats, dogs, or monkeys administered
entecavir, supporting the conclusion that lung tumors in mice may be a species-specific
event. Hepatocellular carcinomas were increased in males and combined liver adenomas
and carcinomas were also increased at exposures 42 times those in humans. Vascular
tumors in female mice (hemangiomas of ovaries and uterus and hemangiosarcomas of
spleen) were increased at exposures 40 times those in humans. In rats, hepatocellular
adenomas were increased in females at exposures 24 times those in humans; combined
adenomas and carcinomas were also increased in females at exposures 24 times those
in humans. Brain gliomas were induced in both males and females at exposures 35 and
24 times those in humans. Skin fibromas were induced in females at exposures 4 times
those in humans.
Mutagenesis
Entecavir was clastogenic to human lymphocyte cultures. Entecavir was not mutagenic in
the Ames bacterial reverse mutation assay using S. typhimurium and E. coli strains in the
presence or absence of metabolic activation, a mammalian-cell gene mutation assay, and
a transformation assay with Syrian hamster embryo cells. Entecavir was also negative in
an oral micronucleus study and an oral DNA repair study in rats.
Impairment of Fertility
In reproductive toxicology studies, in which animals were administered entecavir at up
to 30 mg/kg for up to 4 weeks, no evidence of impaired fertility was seen in male or
female rats at systemic exposures greater than 90 times those achieved in humans
at the highest recommended dose of 1 mg/day. In rodent and dog toxicology studies,
seminiferous tubular degeneration was observed at exposures 35 times or greater than
those achieved in humans. No testicular changes were evident in monkeys.
14 CLINICAL STUDIES
14.1 Outcomes in Adults
At 48 Weeks
The safety and efficacy of BARACLUDE in adults were evaluated in three Phase 3
active-controlled trials. These studies included 1633 subjects 16 years of age or older
with chronic hepatitis B virus infection (serum HBsAg-positive for at least 6 months)
accompanied by evidence of viral replication (detectable serum HBV DNA, as measured
by the bDNA hybridization or PCR assay). Subjects had persistently elevated ALT levels at
least 1.3 times ULN and chronic inflammation on liver biopsy compatible with a diagnosis
of chronic viral hepatitis. The safety and efficacy of BARACLUDE were also evaluated in a
study of 191 HBV-infected subjects with decompensated liver disease and in a study of
68 subjects co-infected with HBV and HIV.
Nucleoside-inhibitor-naïve Subjects with Compensated Liver Disease
HBeAg-positive: Study AI463022 was a multinational, randomized, double-blind study of
BARACLUDE 0.5 mg once daily versus lamivudine 100 mg once daily for a minimum of
52 weeks in 709 (of 715 randomized) nucleoside-inhibitor-naïve subjects with chronic
hepatitis B virus infection, compensated liver disease, and detectable HBeAg. The mean
age of subjects was 35 years, 75% were male, 57% were Asian, 40% were Caucasian,
and 13% had previously received interferon-α. At baseline, subjects had a mean

BARACLUDE
®
(entecavir) BARACLUDE
®
(entecavir)
Knodell Necroinflammatory Score of 7.8, mean serum HBV DNA as measured by
Roche COBAS Amplicor
®
PCR assay was 9.66 log
10
copies/mL, and mean serum ALT level
was 143 U/L. Paired, adequate liver biopsy samples were available for 89% of subjects.
HBeAg-negative (anti-HBe-positive/HBV DNA-positive): Study AI463027 was a
multinational, randomized, double-blind study of BARACLUDE 0.5 mg once daily versus
lamivudine 100 mg once daily for a minimum of 52 weeks in 638 (of 648 randomized)
nucleoside-inhibitor-naïve subjects with HBeAg-negative (HBeAb-positive) chronic
hepatitis B virus infection and compensated liver disease. The mean age of subjects was
44 years, 76% were male, 39% were Asian, 58% were Caucasian, and 13% had previously
received interferon-α. At baseline, subjects had a mean Knodell Necroinflammatory
Score of 7.8, mean serum HBV DNA as measured by Roche COBAS Amplicor PCR assay
was 7.58 log
10
copies/mL, and mean serum ALT level was 142 U/L. Paired, adequate
liver biopsy samples were available for 88% of subjects.
In Studies AI463022 and AI463027, BARACLUDE was superior to lamivudine on the
primary efficacy endpoint of Histologic Improvement, defined as a 2-point or greater
reduction in Knodell Necroinflammatory Score with no worsening in Knodell Fibrosis
Score at Week 48, and on the secondary efficacy measures of reduction in viral load
and ALT normalization. Histologic Improvement and change in Ishak Fibrosis Score are
shown in Table 9. Selected virologic, biochemical, and serologic outcome measures are
shown in Table 10.
Table 9: Histologic Improvement and Change in Ishak Fibrosis Score at Week 48,
Nucleoside-Inhibitor-Naïve Subjects in Studies AI463022 and AI463027
Study AI463022
(HBeAg-Positive)
Study AI463027
(HBeAg-Negative)
BARACLUDE
0.5 mg
n=314
a
Lamivudine
100 mg
n=314
a
BARACLUDE
0.5 mg
n=296
a
Lamivudine
100 mg
n=287
a
Histologic Improvement (Knodell Scores)
Improvement
b
72% 62% 70% 61%
No improvement 21% 24% 19% 26%
Ishak Fibrosis Score
Improvement
c
39% 35% 36% 38%
No change 46% 40% 41% 34%
Worsening
c
8% 10% 12% 15%
Missing Week 48 biopsy 7% 14% 10% 13%
a
Subjects with evaluable baseline histology (baseline Knodell Necroinflammatory Score ≥2).
b
≥2-point decrease in Knodell Necroinflammatory Score from baseline with no worsening of the
Knodell Fibrosis Score.
c
For Ishak Fibrosis Score, improvement = ≥1-point decrease from baseline and worsening
= ≥1-point increase from baseline.
Table 10: Selected Virologic, Biochemical, and Serologic Endpoints at Week 48,
Nucleoside-Inhibitor-Naïve Subjects in Studies AI463022 and AI463027
Study AI463022
(HBeAg-Positive)
Study AI463027
(HBeAg-Negative)
BARACLUDE
0.5 mg
n=354
Lamivudine
100 mg
n=355
BARACLUDE
0.5 mg
n=325
Lamivudine
100 mg
n=313
HBV DNA
a
Proportion undetectable
(<300 copies/mL) 67% 36% 90% 72%
Mean change from baseline
(log
10
copies/mL)
–6.86 –5.39 –5.04 –4.53
ALT normalization (≤1 × ULN) 68% 60% 78% 71%
HBeAg seroconversion 21% 18% NA NA
a
Roche COBAS Amplicor PCR assay [lower limit of quantification (LLOQ) = 300 copies/mL].
Histologic Improvement was independent of baseline levels of HBV DNA or ALT.
Lamivudine-refractory Subjects with Compensated Liver Disease
Study AI463026 was a multinational, randomized, double-blind study of BARACLUDE
in 286 (of 293 randomized) subjects with lamivudine-refractory chronic hepatitis B
virus infection and compensated liver disease. Subjects receiving lamivudine at study
entry either switched to BARACLUDE 1 mg once daily (with neither a washout nor
an overlap period) or continued on lamivudine 100 mg for a minimum of 52 weeks.
The mean age of subjects was 39 years, 76% were male, 37% were Asian, 62% were
Caucasian, and 52% had previously received interferon-α. The mean duration of prior
lamivudine therapy was 2.7 years, and 85% had lamivudine resistance substitutions
at baseline by an investigational line probe assay. At baseline, subjects had a mean
Knodell Necroinflammatory Score of 6.5, mean serum HBV DNA as measured by
Roche COBAS Amplicor PCR assay was 9.36 log
10
copies/mL, and mean serum ALT level
was 128 U/L. Paired, adequate liver biopsy samples were available for 87% of subjects.
BARACLUDE was superior to lamivudine on a primary endpoint of Histologic Improvement
(using the Knodell Score at Week 48). These results and change in Ishak Fibrosis Score are
shown in Table 11. Table 12 shows selected virologic, biochemical, and serologic endpoints.
Table 11: Histologic Improvement and Change in Ishak Fibrosis Score at
Week 48, Lamivudine-Refractory Subjects in Study AI463026
BARACLUDE
1 mg
n=124
a
Lamivudine
100 mg
n=116
a
Histologic Improvement (Knodell Scores)
Improvement
b
55% 28%
No improvement 34% 57%
Ishak Fibrosis Score
Improvement
c
34% 16%
No change 44% 42%
Worsening
c
11% 26%
Missing Week 48 biopsy 11% 16%
a
Subjects with evaluable baseline histology (baseline Knodell Necroinflammatory
Score ≥2).
b
≥2-point decrease in Knodell Necroinflammatory Score from baseline with no worsening
of the Knodell Fibrosis Score.
c
For Ishak Fibrosis Score, improvement = ≥1-point decrease from baseline and
worsening = ≥1-point increase from baseline.
Table 12: Selected Virologic, Biochemical, and Serologic Endpoints at Week 48,
Lamivudine-Refractory Subjects in Study AI463026
BARACLUDE
1 mg
n=141
Lamivudine
100 mg
n=145
HBV DNA
a
Proportion undetectable
(<300 copies/mL)
19% 1%
Mean change from baseline
(log
10
copies/mL)
–5.11 –0.48
ALT normalization (≤1 × ULN) 61% 15%
HBeAg seroconversion 8% 3%
a
Roche COBAS Amplicor PCR assay (LLOQ = 300 copies/mL).
Histologic Improvement was independent of baseline levels of HBV DNA or ALT.
Subjects with Decompensated Liver Disease
Study AI463048 was a randomized, open-label study of BARACLUDE 1 mg once daily
versus adefovir dipivoxil 10 mg once daily in 191 (of 195 randomized) adult subjects
with HBeAg-positive or -negative chronic HBV infection and evidence of hepatic
decompensation, defined as a Child-Turcotte-Pugh (CTP) score of 7 or higher. Subjects
were either HBV-treatment-naïve or previously treated, predominantly with lamivudine
or interferon-α.
In Study AI463048, 100 subjects were randomized to treatment with BARACLUDE and
91 subjects to treatment with adefovir dipivoxil. Two subjects randomized to treatment
with adefovir dipivoxil actually received treatment with BARACLUDE for the duration of
the study. The mean age of subjects was 52 years, 74% were male, 54% were Asian,
33% were Caucasian, and 5% were Black/African American. At baseline, subjects had a
mean serum HBV DNA by PCR of 7.83 log
10
copies/mL and mean ALT level of 100 U/L;
54% of subjects were HBeAg-positive; 35% had genotypic evidence of lamivudine
resistance. The baseline mean CTP score was 8.6. Results for selected study endpoints
at Week 48 are shown in Table 13.
Table 13: Selected Endpoints at Week 48, Subjects with Decompensated Liver
Disease, Study AI463048
BARACLUDE
1 mg
n=100
a
Adefovir Dipivoxil
10 mg
n=91
a
HBV DNA
b
Proportion undetectable (<300 copies/mL) 57% 20%
Stable or improved CTP score
c
61% 67%
HBsAg loss 5% 0
Normalization of ALT (≤1 × ULN)
d
49/78 (63%) 33/71 (46%)
a
Endpoints were analyzed using intention-to-treat (ITT) method, treated subjects
as randomized.
b
Roche COBAS Amplicor PCR assay (LLOQ = 300 copies/mL).
c
Defined as decrease or no change from baseline in CTP score.
d
Denominator is subjects with abnormal values at baseline.
ULN=upper limit of normal.

BARACLUDE
®
(entecavir) BARACLUDE
®
(entecavir)
Lamivudine-refractory Subjects
Among lamivudine-refractory subjects (Study AI463026), 77 (55%) BARACLUDE-
treated subjects and 3 (2%) lamivudine subjects continued blinded treatment for
up to 96 weeks. In this cohort of BARACLUDE subjects, 31 (40%) subjects achieved
HBV DNA <300 copies/mL, 62 (81%) subjects had ALT ≤1 × ULN, and 8 (10%) subjects
demonstrated HBeAg seroconversion at the end of dosing.
14.2 Outcomes in Pediatric Subjects
The pharmacokinetics, safety and antiviral activity of BARACLUDE in pediatric subjects
were initially assessed in Study AI463028. Twenty-four treatment-naïve and 19
lamivudine-experienced HBeAg-positive pediatric subjects 2 to less than 18 years of
age with compensated chronic hepatitis B virus infection and elevated ALT were treated
with BARACLUDE 0.015 mg/kg (up to 0.5 mg) or 0.03 mg/kg (up to 1 mg) once daily.
Fifty-eight percent (14/24) of treatment-naïve subjects and 47% (9/19) of lamivudine-
experienced subjects achieved HBV DNA <50 IU/mL at Week 48 and ALT normalized in
83% (20/24) of treatment-naïve and 95% (18/19) of lamivudine-experienced subjects.
Safety and antiviral efficacy were confirmed in Study AI463189, a study of BARACLUDE
among 180 nucleoside-inhibitor-treatment-naïve pediatric subjects 2 to less than 18 years
of age with HBeAg-positive chronic hepatitis B infection, compensated liver disease, and
elevated ALT. Subjects were randomized 2:1 to receive blinded treatment with BARACLUDE
0.015 mg/kg up to 0.5 mg/day (N=120) or placebo (N=60). The randomization was
stratified by age group (2 to 6 years; >6 to 12 years; and >12 to <18 years). Baseline
demographics and HBV disease characteristics were comparable between the 2 treatment
arms and across age cohorts. At study entry, the mean HBV DNA was 8.1 log
10
IU/mL
and mean ALT was 103 U/L. The primary efficacy endpoint was a composite of HBeAg
seroconversion and serum HBV DNA <50 IU/mL at Week 48 assessed in the first 123
subjects reaching 48 weeks of blinded treatment. Twenty-four percent (20/82) of subjects
in the BARACLUDE-treated group and 2% (1/41) of subjects in the placebo-treated group
met the primary endpoint. Forty-six percent (38/82) of BARACLUDE-treated subjects and
2% (1/41) of placebo-treated subjects achieved HBV DNA <50 IU/mL at Week 48. ALT
normalization occurred in 67% (55/82) of BARACLUDE-treated subjects and 22% (9/41)
of placebo-treated subjects; 24% (20/82) of BARACLUDE-treated subjects and 12% (5/41)
of placebo-treated subjects had HBeAg seroconversion.
16 HOW SUPPLIED/STORAGE AND HANDLING
BARACLUDE
®
(entecavir) Tablets and Oral Solution are available in the following strengths
and configurations of plastic bottles with child-resistant closures:
Product
Strength and
Dosage
Form
Description Quantity NDC Number
0.5 mg film-
coated tablet
White to off-white, triangular-shaped
tablet, debossed with “BMS” on
one side and “1611” on the other side.
30 tablets 0003-1611-12
1 mg film-
coated tablet
Pink, triangular-shaped tablet,
debossed with “BMS” on one side
and “1612” on the other side.
30 tablets 0003-1612-12
0.05 mg/mL
oral solution
Ready-to-use, orange-flavored,
clear, colorless to pale yellow,
aqueous solution in a 260 mL bottle.
210 mL 0003-1614-12
BARACLUDE Oral Solution is a ready-to-use product; dilution or mixing with water or
any other solvent or liquid product is not recommended. Each bottle of the oral solution
is accompanied by a dosing spoon that is calibrated in 0.5 mL increments up to 10 mL.
Storage
BARACLUDE Tablets should be stored in a tightly closed container at 25°C (77°F);
excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room
Temperature]. Store in the outer carton to protect from light.
BARACLUDE Oral Solution should be stored in the outer carton at 25°C (77°F); excursions
permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room
Temperature]. Protect from light. After opening, the oral solution can be used up to the
expiration date on the bottle. The bottle and its contents should be discarded after the
expiration date.
17 PATIENT COUNSELING INFORMATION
See FDA-approved patient labeling (Patient Information).
Severe Acute Exacerbation of Hepatitis after Discontinuation of Treatment
Inform patients that discontinuation of anti-hepatitis B therapy, including BARACLUDE,
may result in severe acute exacerbations of hepatitis B. Advise the patient to not
discontinue BARACLUDE without first informing their healthcare provider [see Warnings
and Precautions (5.1)].
Risk of Development of HIV-1 Resistance in Patients with HIV-1 Coinfection
Inform patients that if they have or develop HIV infection and are not receiving effective
HIV treatment, BARACLUDE may increase the risk of development of resistance to
HIV medication [see Warnings and Precautions (5.2)].
Subjects Co-infected with HIV and HBV
Study AI463038 was a randomized, double-blind, placebo-controlled study of BARACLUDE
versus placebo in 68 subjects co-infected with HIV and HBV who experienced recurrence
of HBV viremia while receiving a lamivudine-containing highly active antiretroviral (HAART)
regimen. Subjects continued their lamivudine-containing HAART regimen (lamivudine dose
300 mg/day) and were assigned to add either BARACLUDE 1 mg once daily (51 subjects)
or placebo (17 subjects) for 24 weeks followed by an open-label phase for an additional
24 weeks where all subjects received BARACLUDE. At baseline, subjects had a mean
serum HBV DNA level by PCR of 9.13 log
10
copies/mL. Ninety-nine percent of subjects
were HBeAg-positive at baseline, with a mean baseline ALT level of 71.5 U/L. Median
HIV RNA level remained stable at approximately 2 log
10
copies/mL through 24 weeks of
blinded therapy. Virologic and biochemical endpoints at Week 24 are shown in Table 14.
There are no data in patients with HIV/HBV co-infection who have not received prior
lamivudine therapy. BARACLUDE has not been evaluated in HIV/HBV co-infected patients
who were not simultaneously receiving effective HIV treatment [see Warnings and
Precautions (5.2)].
Table 14:
Virologic and Biochemical Endpoints at Week 24, Study AI463038
BARACLUDE 1 mg
a
n=51
Placebo
a
n=17
HBV DNA
b
Proportion undetectable (<300 copies/mL) 6% 0
Mean change from baseline (log
10
copies/mL) –3.65 +0.11
ALT normalization (≤1 × ULN) 34%
c
8%
c
a
All subjects also received a lamivudine-containing HAART regimen.
b
Roche COBAS Amplicor PCR assay (LLOQ = 300 copies/mL).
c
Percentage of subjects with abnormal ALT (>1 × ULN) at baseline who achieved ALT
normalization (n=35 for BARACLUDE and n=12 for placebo).
For subjects originally assigned to BARACLUDE, at the end of the open-label phase
(Week 48), 8% of subjects had HBV DNA <300 copies/mL by PCR, the mean change
from baseline HBV DNA by PCR was −4.20 log
10
copies/mL, and 37% of subjects with
abnormal ALT at baseline had ALT normalization (≤1 × ULN).
Beyond 48 Weeks
The optimal duration of therapy with BARACLUDE is unknown. According to protocol-
mandated criteria in the Phase 3 clinical trials, subjects discontinued BARACLUDE
or lamivudine treatment after 52 weeks according to a definition of response based
on HBV virologic suppression (<0.7 MEq/mL by bDNA assay) and loss of HBeAg (in
HBeAg-positive subjects) or ALT <1.25 × ULN (in HBeAg-negative subjects) at Week 48.
Subjects who achieved virologic suppression but did not have serologic response
(HBeAg-positive) or did not achieve ALT <1.25 × ULN (HBeAg-negative) continued
blinded dosing through 96 weeks or until the response criteria were met. These
protocol-specified subject management guidelines are not intended as guidance for
clinical practice.
Nucleoside-inhibitor-naïve Subjects
Among nucleoside-inhibitor-naïve, HBeAg-positive subjects (Study AI463022),
243 (69%) BARACLUDE-treated subjects and 164 (46%) lamivudine-treated subjects
continued blinded treatment for up to 96 weeks. Of those continuing blinded treatment
in Year 2, 180 (74%) BARACLUDE subjects and 60 (37%) lamivudine subjects achieved
HBV DNA <300 copies/mL by PCR at the end of dosing (up to 96 weeks). 193 (79%)
BARACLUDE subjects achieved ALT ≤1 × ULN compared to 112 (68%) lamivudine
subjects, and HBeAg seroconversion occurred in 26 (11%) BARACLUDE subjects and
20 (12%) lamivudine subjects.
Among nucleoside-inhibitor-naïve, HBeAg-positive subjects, 74 (21%) BARACLUDE
subjects and 67 (19%) lamivudine subjects met the definition of response at Week
48, discontinued study drugs, and were followed off treatment for 24 weeks. Among
BARACLUDE responders, 26 (35%) subjects had HBV DNA <300 copies/mL, 55 (74%)
subjects had ALT ≤1 × ULN, and 56 (76%) subjects sustained HBeAg seroconversion
at the end of follow-up. Among lamivudine responders, 20 (30%) subjects had HBV
DNA <300 copies/mL, 41 (61%) subjects had ALT ≤1 × ULN, and 47 (70%) subjects
sustained HBeAg seroconversion at the end of follow-up.
Among nucleoside-inhibitor-naïve, HBeAg-negative subjects (Study AI463027), 26 (8%)
BARACLUDE-treated subjects and 28 (9%) lamivudine-treated subjects continued
blinded treatment for up to 96 weeks. In this small cohort continuing treatment in
Year 2, 22 BARACLUDE and 16 lamivudine subjects had HBV DNA <300 copies/mL by
PCR, and 7 and 6 subjects, respectively, had ALT ≤1 × ULN at the end of dosing (up
to 96 weeks).
Among nucleoside-inhibitor-naïve, HBeAg-negative subjects, 275 (85%) BARACLUDE
subjects and 245 (78%) lamivudine subjects met the definition of response at Week
48, discontinued study drugs, and were followed off treatment for 24 weeks. In this
cohort, very few subjects in each treatment arm had HBV DNA <300 copies/mL by PCR
at the end of follow-up. At the end of follow-up, 126 (46%) BARACLUDE subjects and
84 (34%) lamivudine subjects had ALT ≤1 × ULN.

BARACLUDE
®
(entecavir) BARACLUDE
®
(entecavir)
Lactic Acidosis and Severe Hepatomegaly
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have
been reported with use of drugs similar to BARACLUDE. Advise patients to contact
their healthcare provider immediately and stop BARACLUDE if they develop clinical
symptoms suggestive of lactic acidosis or pronounced hepatotoxicity [see Warnings and
Precautions (5.3)].
Missed Dosage
Inform patients that it is important to take BARACLUDE on a regular dosing schedule on
an empty stomach (at least 2 hours after a meal and 2 hours before the next meal) and
to avoid missing doses as it can result in development of resistance [see Dosage and
Administration (2.1)].
Treatment Duration
Advise patients that in the treatment of chronic hepatitis B, the optimal duration of
treatment is unknown. The relationship between response and long-term prevention of
outcomes such as hepatocellular carcinoma is not known.
Instructions for Use
Inform patients using the oral solution to hold the dosing spoon in a vertical position
and fill it gradually to the mark corresponding to the prescribed dose. Rinsing of the
dosing spoon with water is recommended after each daily dose. Some patients may find
it difficult to accurately measure the prescribed dose using the provided dosing spoon;
therefore, patients/caregivers should refer to the steps in the Patient Information section
that demonstrate the correct technique of using the provided dosing spoon to measure
the prescribed BARACLUDE dose.
Pregnancy Registry
Advise patients that there is a pregnancy exposure registry that monitors pregnancy
outcomes in women exposed to BARACLUDE during pregnancy [see Use in Specific
Populations (8.1)].

BARACLUDE
®
(entecavir)
Patient Information
BARACLUDE
®
(BEAR ah klude)
(entecavir)
Tablets
BARACLUDE
®
(BEAR ah klude)
(entecavir)
Oral Solution
Read this Patient Information before you start taking BARACLUDE and each time you get a rell. There may be
new information. This information does not take the place of talking with your healthcare provider about your
medical condition or treatment.
What is the most important information I should know about BARACLUDE?
1. Your hepatitis B virus (HBV) infection may get worse if you stop taking BARACLUDE. This usually
happens within 6 months after stopping BARACLUDE.
• Take BARACLUDE exactly as prescribed.
• Do not run out of BARACLUDE.
• Do not stop BARACLUDE without talking to your healthcare provider.
• Your healthcare provider should monitor your health and do regular blood tests to check your liver if you
stop taking BARACLUDE.
2. If you have or get HIV that is not being treated with medicines while taking BARACLUDE,
the HIV virus may develop resistance to certain HIV medicines and become harder to treat. You
should get an HIV test before you start taking BARACLUDE and anytime after that when there is a chance
you were exposed to HIV.
BARACLUDE can cause serious side effects including:
3. Lactic acidosis (buildup of acid in the blood). Some people who have taken BARACLUDE or
medicines like BARACLUDE (a nucleoside analogue) have developed a serious condition called
lactic acidosis. Lactic acidosis is a serious medical emergency that can cause death. Lactic acidosis must
be treated in the hospital. Reports of lactic acidosis with BARACLUDE generally involved patients who were
seriously ill due to their liver disease or other medical condition.
Call your healthcare provider right away if you get any of the following signs or symptoms of lactic
acidosis:
• You feel very weak or tired.
• You have unusual (not normal) muscle pain.
• You have trouble breathing.
• You have stomach pain with nausea and vomiting.
• You feel cold, especially in your arms and legs.
• You feel dizzy or light-headed.
• You have a fast or irregular heartbeat.
4. Serious liver problems. Some people who have taken medicines like BARACLUDE have developed
serious liver problems called hepatotoxicity, with liver enlargement (hepatomegaly) and fat
in the liver (steatosis). Hepatomegaly with steatosis is a serious medical emergency that can
cause death.
Call your healthcare provider right away if you get any of the following signs or symptoms of
liver problems:
• Your skin or the white part of your eyes turns yellow (jaundice).
• Your urine turns dark.
• Your bowel movements (stools) turn light in color.

BARACLUDE
®
(entecavir)
• You don’t feel like eating food for several days or longer.
• You feel sick to your stomach (nausea).
• You have lower stomach pain.
You may be more likely to get lactic acidosis or serious liver problems if you are female, very overweight, or
have been taking nucleoside analogue medicines, like BARACLUDE, for a long time.
What is BARACLUDE?
BARACLUDE is a prescription medicine used to treat chronic hepatitis B virus (HBV) in adults and children 2 years
of age and older who have active liver disease.
• BARACLUDE will not cure HBV.
• BARACLUDE may lower the amount of HBV in the body.
• BARACLUDE may lower the ability of HBV to multiply and infect new liver cells.
• BARACLUDE may improve the condition of your liver.
• It is not known whether BARACLUDE will reduce your chances of getting liver cancer or liver damage
(cirrhosis), which may be caused by chronic HBV infection.
• It is not known if BARACLUDE is safe and effective for use in children less than 2 years of age.
What should I tell my healthcare provider before taking BARACLUDE?
Before you take BARACLUDE, tell your healthcare provider if you:
• have kidney problems. Your BARACLUDE dose or schedule may need to be changed.
• have received medicine for HBV before. Some people, especially those who have already been treated
with certain other medicines for HBV infection, may develop resistance to BARACLUDE. These people may
have less benet from treatment with BARACLUDE and may have worsening of hepatitis after resistant
virus appears. Your healthcare provider will test the level of the hepatitis B virus in your blood regularly.
• have any other medical conditions.
• are pregnant or plan to become pregnant. It is not known if BARACLUDE will harm your unborn baby. Talk
to your healthcare provider if you are pregnant or plan to become pregnant.
Antiretroviral Pregnancy Registry. If you take BARACLUDE while you are pregnant, talk to your
healthcare provider about how you can take part in the BARACLUDE Antiretroviral Pregnancy Registry.
The purpose of the pregnancy registry is to collect information about the health of you and your baby.
• are breastfeeding or plan to breastfeed. It is not known if BARACLUDE can pass into your breast milk.
You and your healthcare provider should decide if you will take BARACLUDE or breastfeed.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter
medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you have taken a medicine
to treat HBV in the past.
Know the medicines you take. Keep a list of your medicines with you to show your healthcare provider and
pharmacist when you get a new medicine.
How should I take BARACLUDE?
• Take BARACLUDE exactly as your healthcare provider tells you to.
• Your healthcare provider will tell you how much BARACLUDE to take.
• Your healthcare provider will tell you when and how often to take BARACLUDE.
• Take BARACLUDE on an empty stomach, at least 2 hours after a meal and at least 2 hours before the
next meal.
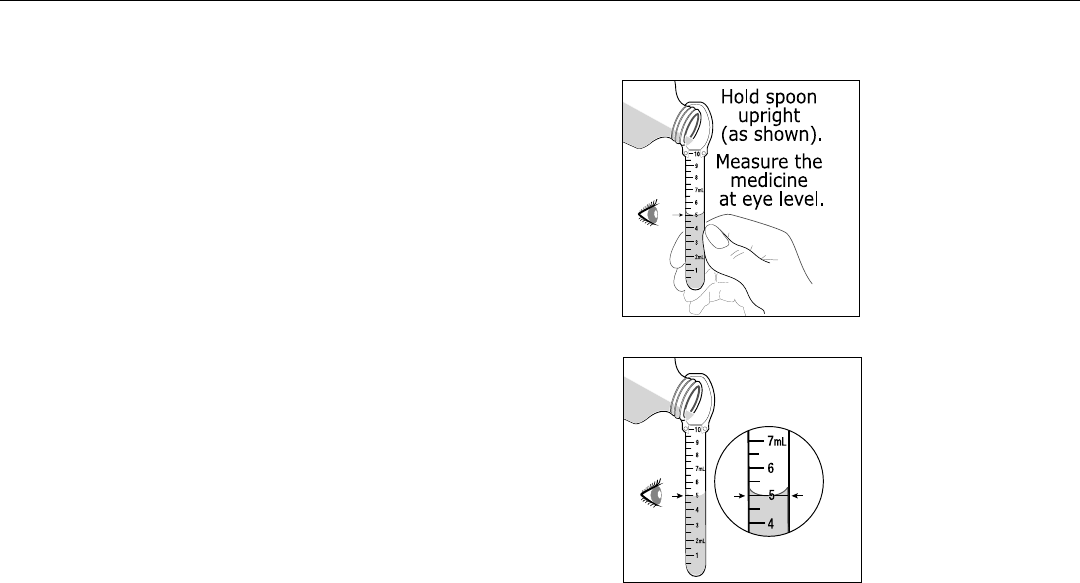
Figure 2
BARACLUDE
®
(entecavir)
• If you are taking BARACLUDE Oral Solution, or giving it to your child, carefully measure the dose with
the dosing spoon provided, as follows:
• Hold the dosing spoon in an upright
(vertical) position and slowly ll it to the
measurement line on the dosing spoon
that is the same as the prescribed dose.
Bring the dosing spoon to eye level to be
sure that the level of the BARACLUDE Oral
Solution is at the correct measurement
line (see Figure 1).
• With the dosing spoon at eye level, holding
it with the measurement lines facing
you, check that it has been lled to the
correct measurement line. The top of the
BARACLUDE Oral Solution in the dosing
spoon will look curved, not at. Measure
the dose of BARACLUDE Oral Solution at
the bottom of the curve. Your dose of
BARACLUDE Oral Solution is measured
correctly when the bottom of the curve is
lined up with the measurement line of the
prescribed dose. As an example, Figure 2
shows the right way to measure a 5 mL
dose of BARACLUDE (see Figure 2).
• BARACLUDE Oral Solution should be swallowed directly from the dosing spoon.
• BARACLUDE Oral Solution should not be mixed with water or any other liquid.
• After each use, rinse the dosing spoon with water and allow it to air dry.
• If you lose the dosing spoon, call your pharmacist or healthcare provider for instructions.
• Do not change your dose or stop taking BARACLUDE without talking to your healthcare provider.
• If you miss a dose of BARACLUDE, take it as soon as you remember and then take your next dose at
its regular time. If it is almost time for your next dose, skip the missed dose. Do not take two doses at
the same time. Call your healthcare provider or pharmacist if you are not sure what to do.
• When your supply of BARACLUDE starts to run low, call your healthcare provider or pharmacy for a rell.
Do not run out of BARACLUDE.
• If you take too much BARACLUDE, call your healthcare provider or go to the nearest emergency room
right away.
What are the possible side effects of BARACLUDE?
BARACLUDE may cause serious side effects. See “What is the most important information I should
know about BARACLUDE?”
The most common side effects of BARACLUDE include:
• headache
• tiredness
• dizziness
• nausea
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of BARACLUDE. For more information, ask your healthcare provider or
pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
Figure 1

BARACLUDE
®
(entecavir)
How should I store BARACLUDE?
• Store BARACLUDE Tablets or Oral Solution at room temperature, between 68°F and 77°F (20°C and 25°C).
• Keep BARACLUDE Tablets in a tightly closed container.
• Store BARACLUDE Tablets or BARACLUDE Oral Solution in the original carton, and keep the carton out of
the light.
• Safely throw away BARACLUDE that is out of date or no longer needed. Dispose of unused medicines
through community take-back disposal programs when available or place BARACLUDE in an unrecognizable
closed container in the household trash.
Keep BARACLUDE and all medicines out of the reach of children.
General information about the safe and effective use of BARACLUDE
BARACLUDE does not stop you from spreading the hepatitis B virus (HBV) to others by sex, sharing needles,
or being exposed to your blood. Talk with your healthcare provider about safe sexual practices that protect
your partner. Never share needles. Do not share personal items that can have blood or body uids on them,
like toothbrushes or razor blades. A shot (vaccine) is available to protect people at risk from becoming infected
with HBV.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaet. Do not
use BARACLUDE for a condition for which it was not prescribed. Do not give BARACLUDE to other people, even
if they have the same symptoms you have. It may harm them.
This Patient Information leaet summarizes the most important information about BARACLUDE. If you would
like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist
for information about BARACLUDE that is written for health professionals.
For more information, go to www.Baraclude.com or call 1-800-321-1335.
What are the ingredients in BARACLUDE?
Active ingredient: entecavir
Inactive ingredients in BARACLUDE Tablets: lactose monohydrate, microcrystalline cellulose, crospovidone,
povidone, magnesium stearate.
Tablet lm-coat: titanium dioxide, hypromellose, polyethylene glycol 400, polysorbate 80 (0.5 mg tablet only),
and iron oxide red (1 mg tablet only).
Inactive ingredients in BARACLUDE Oral Solution: maltitol, sodium citrate, citric acid, methylparaben, propylparaben,
and orange avor.
Distributed by:
Bristol-Myers Squibb Company
Princeton, NJ 08543 USA
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: August 2015
686US1903784-01-01 11/19
