
NIH, CDC, and FDA Program Descriptions and Research Topics 1
U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES PHS 2024
OMNIBUS SOLICITATION OF THE
NATIONAL INSTITUTES OF HEALTH,
CENTERS FOR DISEASE CONTROL AND PREVENTION, AND
FOOD AND DRUG ADMINISTRATION FOR
SMALL BUSINESS INNOVATION
RESEARCH (SBIR)
AND
SMALL BUSINESS TECHNOLOGY
TRANSFER (STTR)
GRANT APPLICATIONS
NIH, CDC, and FDA Program Descriptions and
Research Topics
SUBMISSION DATES
SEPTEMBER 5, 2024, JANUARY 5, 2025, AND
APRIL 5, 2025
National Institutes of Health (SBIR and STTR)
Centers for Disease Control and Prevention (SBIR)
Food and Drug Administration (SBIR)

NIH, CDC, and FDA Program Descriptions and Research Topics 2
Notices of Funding Opportunities, Application Instructions, and Appendices are contained in separate files. Follow the
links below to view these documents.
NOTICES OF FUNDING OPPORTUNITIES
REMINDER: ALL APPLICATIONS MUST BE SUBMITTED IN RESPONSE TO A NOTICE OF
FUNDING OPPORTUNITY THROUGH GRANTS.GOV
PHS 2023-02 OMNIBUS SOLICITATION OF THE NIH, CDC, AND FDA FOR SMALL
BUSINESS INNOVATION RESEARCH GRANT APPLICATIONS (PARENT SBIR
[R43/R44] CLINICAL TRIAL NOT ALLOWED)
PHS 2023-02 OMNIBUS SOLICITATION OF THE NIH FOR SMALL BUSINESS
TECHNOLOGY TRANSFER GRANT APPLICATIONS (PARENT STTR [R41/R42]
CLINICAL TRIAL NOT ALLOWED)
PHS 2023-02 OMNIBUS SOLICITATION OF THE NIH FOR SMALL BUSINESS INNOVATION
RESEARCH GRANT APPLICATIONS (PARENT SBIR [R43/R44] CLINICAL TRIAL
REQUIRED)
PHS 2023-02 OMNIBUS SOLICITATION OF THE NIH FOR SMALL BUSINESS
TECHNOLOGY TRANSFER GRANT APPLICATIONS (PARENT STTR [R41/R42]
CLINICAL TRIAL REQUIRED)
ADDITIONAL SPECIAL ANNOUNCEMENTS FOR SMALL BUSINESS RESEARCH
OPPORTUNITIES
APPLICATION INSTRUCTIONS
SF424 (R&R) APPLICATION INSTRUCTIONS AND ELECTRONIC SUBMISSION
INFORMATION

NIH, CDC, and FDA Program Descriptions and Research Topics 3
TABLE OF CONTENTS
NIH, CDC, AND FDA PROGRAM DESCRIPTIONS AND RESEARCH TOPICS
PROGRAM DESCRIPTIONS AND RESEARCH GRANT TOPICS ........................................... 5
NATIONAL INSTITUTES OF HEALTH (NIH) 5
NATIONAL INSTITUTE ON AGING (NIA) 7
NATIONAL INSTITUTE ON ALCOHOL ABUSE AND ALCOHOLISM (NIAAA) 12
NATIONAL INSTITUTE OF ALLERGY AND INFECTIOUS DISEASES (NIAID) 18
NATIONAL INSTITUTE OF ARTHRITIS AND MUSCULOSKELETAL AND SKIN DISEASES
(NIAMS) 33
NATIONAL INSTITUTE OF BIOMEDICAL IMAGING AND BIOENGINEERING (NIBIB) 36
NATIONAL CANCER INSTITUTE (NCI) 42
EUNICE KENNEDY SHRIVER NATIONAL INSTITUTE OF CHILD HEALTH AND HUMAN
DEVELOPMENT (NICHD) 47
NATIONAL INSTITUTE ON DRUG ABUSE (NIDA) 60
NATIONAL INSTITUTE ON DEAFNESS AND OTHER COMMUNICATION DISORDERS
(NIDCD) 73
NATIONAL INSTITUTE OF DENTAL AND CRANIOFACIAL RESEARCH (NIDCR) 76
NATIONAL INSTITUTE OF DIABETES AND DIGESTIVE AND KIDNEY DISEASES
(NIDDK) 84
NATIONAL INSTITUTE OF ENVIRONMENTAL HEALTH SCIENCES (NIEHS) 98
NATIONAL EYE INSTITUTE (NEI) 109
NATIONAL INSTITUTE OF GENERAL MEDICAL SCIENCES (NIGMS) 113
NATIONAL HEART, LUNG, AND BLOOD INSTITUTE (NHLBI) 117
NATIONAL HUMAN GENOME RESEARCH INSTITUTE (NHGRI) 124
NATIONAL INSTITUTE OF MENTAL HEALTH (NIMH) 128
NATIONAL INSTITUTE ON MINORITY HEALTH AND HEALTH DISPARITIES (NIMHD) 132
NATIONAL INSTITUTE OF NEUROLOGICAL DISORDERS AND STROKE (NINDS) 135
NATIONAL INSTITUTE OF NURSING RESEARCH (NINR) 140
NATIONAL CENTER FOR ADVANCING TRANSLATIONAL SCIENCES (NCATS) 143
NATIONAL CENTER FOR COMPLEMENTARY AND INTEGRATIVE HEALTH (NCCIH) 149
NATIONAL LIBRARY OF MEDICINE (NLM) 154
DIVISION OF PROGRAM COORDINATION, PLANNING, AND STRATEGIC INITIATIVES
(DPCPSI), OFFICE OF RESEARCH INFRASTRUCTURE PROGRAMS (ORIP) 156
OFFICE OF RESEARCH ON WOMEN’S HEALTH 161

NIH, CDC, and FDA Program Descriptions and Research Topics 4
CENTERS FOR DISEASE CONTROL AND PREVENTION (CDC) 164
NATIONAL CENTER ON BIRTH DEFECTS AND DEVELOPMENTAL DISABILITIES
(NCBDDD) 167
NATIONAL CENTER FOR CHRONIC DISEASE PREVENTION AND HEALTH PROMOTION
(NCCDPHP) 168
NATIONAL CENTER FOR EMERGING AND ZOONOTIC INFECTIOUS DISEASES
(NCEZID) 170
NATIONAL CENTER FOR ENVIRONMENTAL HEALTH (NCEH) 175
NATIONAL CENTER FOR HIV, VIRAL HEPATITIS, STD, AND TB PREVENTION (NCHHSTP)
177
NATIONAL CENTER FOR IMMUNIZATION AND RESPIRATORY DISEASES (NCIRD) 179
NATIONAL CENTER FOR INJURY PREVENTION AND CONTROL (NCIPC) 181
NATIONAL INSTITUTE FOR OCCUPATIONAL SAFETY AND HEALTH (NIOSH) 189
FOOD AND DRUG ADMINISTRATION (FDA) 195
CENTER FOR BIOLOGICS EVALUATION AND RESEARCH (CBER) 195
CENTER FOR DRUG EVALUATION AND RESEARCH (CDER) 196
CENTER FOR FOOD SAFETY AND APPLIED NUTRITION (CFSAN) 197
CENTER FOR DEVICES AND RADIOLOGICAL HEALTH (CDRH) 198
CENTER FOR VETERINARY MEDICINE (CVM) 199
OFFICE OF CRITICAL PATH PROGRAMS 200
OFFICE OF ORPHAN PRODUCTS DEVELOPMENT 201

NIH, CDC, and FDA Program Descriptions and Research Topics 5
PROGRAM DESCRIPTIONS AND RESEARCH GRANT TOPICS
The research topics shown in this solicitation represent program areas that may be of interest to small
businesses and fall within the missions of the NIH, CDC, and FDA. Small businesses are encouraged to
submit SBIR and STTR grant applications in these areas. Grant applications will be accepted and
considered in any area within the mission of the awarding components (i.e., Institutes and Centers (ICs))
identified in this solicitation. Information about the HHS SBIR and STTR programs for applicants and
awardees, including resources and programs available to HHS SBIR and STTR awardees, can be found
at https://seed.nih.gov/.
Applicants are strongly encouraged to subscribe to the NIH Guide for Grants and Contracts LISTSERV or
query program administrators periodically via email to learn of new or emerging scientific interests of the
NIH, CDC, and FDA awarding components.
You may also subscribe to the SBIR-STTR LISTSERV to get timely information about the NIH SBIR and
STTR Programs.
Additional information on each of the awarding components (ICs) and their research interests is available
electronically on the home pages shown throughout the “Research Topics” section of the solicitation.
NATIONAL INSTITUTES OF HEALTH (NIH)
NIH is the steward of medical and behavioral research for the Nation. Its mission is science in pursuit of
fundamental knowledge about the nature and behavior of living systems and the application of that
knowledge to enhance health, lengthen life, and reduce the burdens of illness and disability.
The goals of the agency are as follows:
1. to foster fundamental creative discoveries, innovative research strategies, and their applications
as a basis for ultimately protecting and improving health;
2. to develop, maintain, and renew scientific human and physical resources that will assure the
Nation's capability to prevent disease;
3. to expand the knowledge base in medical and associated sciences in order to enhance the
Nation's economic well-being and ensure a continued high return on the public investment in
research; and
4. to exemplify and promote the highest level of scientific integrity, public accountability, and social
responsibility in the conduct of science.
In realizing these goals, the NIH provides leadership and direction to programs designed to improve the
health of the Nation by conducting and supporting research:
in the causes, diagnosis, prevention, and cure of human diseases;
in the processes of human growth and development;
in the biological effects of environmental contaminants;
in the understanding of mental, addictive and physical disorders; and
in directing programs for the collection, dissemination, and exchange of information in medicine and
health, including the development and support of medical libraries and the training of medical
librarians and other health information specialists.
In addition, the NIH sponsors training of research personnel; career development of new and established
scientists; construction and renovation of research facilities and provision of other research resources.
Information about the NIH SBIR and STTR programs for applicants and awardees, including resources

NIH, CDC, and FDA Program Descriptions and Research Topics 6
and programs available to NIH SBIR and STTR awardees, can be found at https://seed.nih.gov/support-
for-small-businesses.
To carry out these responsibilities, the NIH is organized into awarding components (Institutes and
Centers). Those components that have an extramural element, that is, those that provide funds for
research and research training activities in organizations external to the NIH, are shown below. The NIH
makes every effort to finance worthy applications, including the co-funding of such applications by one or
more awarding components having relevance in the projects.
Total funding support (direct costs, indirect costs, fees) normally may not exceed $306,872 for Phase I
awards and $2,045,816 for the duration of the Phase II awards. However, this amount is subject to
change and the most current information can be found on the NIH SEED website. Awards exceeding
these amounts may be made at the discretion of an Institute or Center for applications within one of the
SBA-Approved Waiver Topics. Applicants considering a requested budget greater than these limits are
strongly encouraged to contact program staff before submitting an application.
Funding levels for projects are determined through the combined interaction among peer review, grants
management, program, budget, and other Institute and/or Center staff. These levels are based on
allowable costs that are consistent with the principles of sound cost management and in consideration of
Institute or Center priorities, constraints on the growth of average grant costs, and the availability of
funds.
Before considering and/or preparing an application to the SBIR and STTR programs, all applicants are
strongly encouraged to review the agencies’ and NIH Institutes’ and Centers’ websites and to contact
the SBIR and STTR program coordinators listed below. The Fogarty International Center, which provides
support only for conferences, postdoctoral fellowships for research in the United States and abroad, and
senior scientist exchanges between the United States and other countries, does not participate in the
SBIR and STTR program.
Contact Information
Questions of a general nature about the HHS SBIR and STTR program may be directed to:
SEED (Small business Education and Entrepreneurial Development)
Email: SEEDinfo@nih.gov
For Agency, Institute and Center Scientific/Research (Program) and Financial/Grants Management
contacts, please see the contact page

NIH, CDC, and FDA Program Descriptions and Research Topics
NIA 7
NATIONAL INSTITUTE ON AGING (NIA)
Mission
NIA’s mission is to:
•
Support and conduct genetic, biological, clinical, behavioral, social, and economic
research on aging.
•
Foster the development of research and clinician-scientists in aging.
•
Provide research resources.
•
Disseminate information about aging and advances in research to the public, health
care professionals, and the scientific community, among a variety of audiences.
Strategic Directions for Research
https://www.nia.nih.gov/about/aging-strategic-directions-research
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SEED website, unless the application fits an
SBA-approved NIA waiver topic. For topics listed in the SBA-Approved Waiver Topics, the NIA
generally will not fund Phase I applications to the Omnibus greater than $400,000 total costs or
project periods greater than 2 years; or Phase II applications greater than $2,250,000 total costs or
project periods greater than 3 years.
However, if your application is AD/ADRD focused, you may come in at $500,000 for Phase I and
$2,500,000 for Phase II (total costs). For budgetary, administrative, or programmatic reasons, the
NIA may not fund an application or may decrease the length of an award and/or the budget
recommended by a review committee.
Specific SBIR and STTR Program Information
The NIA SBIR-STTR Programs support research and product development focusing on aging and
aging- related conditions and diseases, as well as other problems and needs unique to older
Americans. NIA supports SBIR and STTR research and product development under four divisions:
Behavioral and Social Research, Biology of Aging (Aging Biology), Geriatrics and Clinical
Gerontology, and Neuroscience.
The NIA will consider any application relevant to the NIA’s mission, even if it does not
directly address one of the topics below. For additional information about NIA’s SBIR and
STTR programs please visit: https://www.nia.nih.gov/research/osbr
Specific Funding Opportunities and Programs
In addition to this Omnibus program announcement, the NIA releases targeted Notices of Funding
Opportunities (NOFOs) throughout the year. These NOFOs are listed to inform potential applicants
about other funding opportunities to which they can apply; applications submitted in response to
this Omnibus program announcement are not limited to research and development areas described
in the following targeted NOFOs. NOFOs may specify specific budget caps that are above the caps
listed for Omnibus applications. Applicants are encouraged to visit the following webpage for an up
to date list of NIA SBIR/STTR funding opportunities: https://www.nia.nih.gov/research/nia-small-
business-funding- opportunities.
For projects that aim to address Alzheimer’s Disease and Related Dementias, applicants are
encouraged to consider the following funding opportunities which allows Phase I budgets up to
$500,000 and Phase II budgets up to $2.5M (for topics covered by the approved waiver from SBA):
NIH, CDC, and FDA Program Descriptions and Research Topics
NIA 8
•
Advanced Research on Alzheimer's Disease (AD) and Alzheimer's-Disease-Related
Dementias (ADRD) (R43/R44 Clinical Trial Optional): Accepts Phase I, Phase II, Direct-
to-Phase II and fast- track applications. Details can be found
herehttps://grants.nih.gov/grants/guide/pa-files/pas-22- 196.html
•
Advancing Research on Alzheimer's Disease (AD) and Alzheimer's-Disease-Related
Dementias (ADRD) (R41/R42 Clinical Trial Optional): Accepts Phase I, Phase II, and fast-
track applications. Details can be found here: https://grants.nih.gov/grants/guide/pa-
files/PAS-22-197.html
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NIA welcomes submission of Phase IIB Competing Renewal grant applications from Phase II
SBIR/STTR awardees to continue the process of developing a wide range of aging-focused products,
including digital- mobile/cyber-health technology, pharmaceutical compounds, and medical devices.
The Phase IIB Competing Renewal award is intended to allow small businesses the opportunity to
realize further progress in commercialization, including stimulating interest in and investment by third
parties. NIA will generally not fund Phase IIBs greater than $3M total costs.
Prospective Phase IIB Competing Renewal applicants are strongly encouraged to contact NIA’s SBIR-
STTR program contact prior to consideration and preparation of a Phase IIB application and well in
advance of the SBIR-STTR submission due dates.
NIA also welcomes the submission of CRP applications to the 2 CRP NOFOs (and subsequent reissued
NOFOs):
• SBIR/STTR Commercialization Readiness Pilot (CRP) Program Technical Assistance and
Late Stage Development - Clinical Trial Not Allowed (PAR-23-219)
• SBIR/STTR Commercialization Readiness Pilot (CRP) Program Technical Assistance and
Late Stage Development - Clinical Trial Required (PAR-23-220)
Clinical Trials
Does NIA accept Clinical Trials through
the Omnibus/Parent Notices of
Funding Opportunities?
Yes
Does NIA accept Clinical Trials through
specific Notices of Funding Opportunities?
Yes
•
Advancing Research on Alzheimer's Disease
(AD) and Alzheimer's-Disease-Related
Dementias (ADRD) (R41/R42 Clinical Trial
Optional) (PAS-22-197) )
•
Advancing Research on Alzheimer's Disease
(AD) and Alzheimer's-Disease-Related
Dementias (ADRD) (R43/R44 Clinical Trial
Optional) (PAS-22-196) )

NIH, CDC, and FDA Program Descriptions and Research Topics
NIA 9
Does NIA support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
NOFOs include but are not limited to:
•
Alzheimer's Drug-Development Program
(U01 Clinical Trial Optional) (PAR-22-047)
•
Blueprint Neurotherapeutics Network (BPN):
Biologic-based Drug Discovery and
Development for Disorders of the Nervous
System (UG3/UH3 Clinical Trial Optional)
(PAR-21-163)
•
Early and Late Stage Clinical Trials for the
Spectrum of Alzheimer’s
Disease/Alzheimer’s Related Dementias
and Age-Related Cognitive Decline (R01
Clinical Trial Optional) (PAR-23-081)
•
Pilot Studies for the Spectrum of
Alzheimer’s Disease/Alzheimer’s Disease-
Related Dementias and Age-Related
Cognitive Decline (R61 Clinical Trial
Optional) (PAR-23-083
• Seamless Early-Stage Clinical Drug
Development (Phase 1 to 2a) for Novel
therapeutic Agents for the Spectrum of
Alzheimer's Disease (AD) and AD-related
Dementias (ADRD) (UG3/UH3 Clinical Trial
Required) (PAR 23 274)
• Alzheimer's Clinical Trials Consortium
(ACTC) Clinical Trials (R01 Clinical Trial
Required) (PAR 20 309)
Research Topics
The NIA will consider any application relevant to the NIA’s mission, even if it does not directly
address one of the topics below. The below topics provide an overview of interest areas for both
non-clinical trial and clinical trial applications.
A.
Alzheimer’s Disease (AD), AD-Related Dementias (ADRD), and Age-Related
Change in Brain Function. Research and development of novel interventions to
ameliorate AD/ADRD; improve AD/ADRD care; or further the understanding of the
etiology of AD/ADRD, neurodegeneration, brain connectivity, neuroplasticity, or
brain-– behavior relationships. This includes drug and non-drug interventions for
age-related cognitive decline, delirium, sleep disorders, or other central nervous
system dysfunctions, including dysfunctions of the motor, emotional, sensory, and
neuroimmune systems. This also includes novel biomarkers of neural stem cell
functions and new technologies or imaging devices that improve or study brain
connectivity; metabolism; sleep; or cognitive, motor, emotional, or sensory activity.
a.
For projects addressing AD/ADRD, you may want to consider applying to PAS-
22-196 (SBIR) and PAS-22-197 (STTR), which have higher budget limits.
B.
Aging in Place of Choice. Research and development of social, behavioral, and
environmental interventions that promote independence and aging in place by
addressing the unique needs of older adults, their healthcare providers, and
caregivers. This includes prosthetics, assistive devices and robotics, digital
technologies and software, and technology to mitigate age-related physical and
behavioral health challenges or toimprove healthcare delivery, care coordination,

NIH, CDC, and FDA Program Descriptions and Research Topics
NIA 10
and disease management.
C.
Age-Related Diseases and Conditions. Research and development of new
diagnostic tools and methods, biomarkers, therapeutics, imaging devices, and
technologies to monitor, diagnose, predict, prevent, treat, and further the
understanding of the molecular mechanisms of aging or age-related diseases and
conditions.
D.
Research Tools. Development and validation of innovative tools, resources, or
methodologies that promote the efficient, cost-effective, and high-quality
collection, analysis, or interpretation of aging-related quantitative or qualitative
data. This includes bioinformatics tools; screening platforms; surveying,
sampling, and behavioral/behavioral economics methods; and clinical
instruments to enhance the study of aging, cellular resiliencies, and aging-
related diseases.
Special Areas of Interest
Areas of particular interest related to aging biology, aging-related diseases and conditions,
behavioral health, and AD/ADRD include, but are not limited to the following:
A.
Companion diagnostics and other forms of personalized medicine.
B.
Bioinformatics, public health informatics, or data science
technologies/methods (e.g., machine learning, artificial intelligence) to
better understand aging biology and/or predict health outcomes.
C.
Novel cell and gene therapies, as well as other novel therapeutic approaches to AD/ADRD.
D.
Biomarkers and diagnostic tools for the early detection of disease.
E.
Prevention and therapeutics that directly target mechanisms related to aging biology.
F.
Assistive technology, devices, and mobile applications for older adults and caregivers.
G.
Tools, technologies, and analytic methods to address health disparities among
older adults and/or biological determinants of health disparities.
Contact Information
For more information on research topics and questions about potential NIA SBIR/STTR grant
applications and NIA’s participation in the Phase IIB or CRP programs, please contact:
Program Contacts, NIA Small Business R & D Programs:
Michael-David (“M-D”) A.R.R.Kerns, M.M., M.S., Ph.D.
National Institute on Aging (NIA)
Email: niasmallbusiness@mail.nih.gov
Bio:
https://www.nia.nih.gov/about/staff/kerns-michael-david
Rajesh Kumar, Ph.D.
National Institute on Aging (NIA) Email:
niasmallbusiness@mail.nih.gov
If there are specific questions pertaining to the interests or activities of the NIA scientific divisions, contact:

NIH, CDC, and FDA Program Descriptions and Research Topics
NIA 11
Division of Aging Biology:
Leonid Tsap, Ph.D.
National Institute on Aging (NIA)
Email: Leonid.Tsap@nih.gov
Bio: https://www.nia.nih.gov/about/staff/tsap-leonid
Division of Behavioral and Social Research:
Dinesh John, Ph.D.
National Institute on Aging (NIA)
Email: dinesh.john@nih.gov
Bio:
https://www.nia.nih.gov/about/staff/john-dinesh
Division of Geriatrics and Clinical Gerontology:
Lyndon Joseph, Ph.D.
National Institute on Aging (NIA)
Telephone: 301-496-6761
Email: lyndon.joseph@nih.gov
Bio:
https://www.nia.nih.gov/about/staff/joseph-lyndon
Division of Neuroscience:
Zane Martin, Ph.D.
National Institute on Aging (NIA)
Email:zane.martin@nih.gov
Bio: https://www.nia.nih.gov/about/staff/martin-jones-zane

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAAA 12
NATIONAL INSTITUTE ON ALCOHOL ABUSE AND ALCOHOLISM (NIAAA)
Mission
NIAAA supports research on the causes, prevention, control, and treatment of the major health
problems associated with alcohol misuse. NIAAA supports research on the causes, prevention,
control, and treatment of the major health problems associated with alcohol misuse. Through its
extramural research programs, NIAAA funds a wide range of basic and applied research to
develop new and/or improved technologies and approaches for increasing the effectiveness of
diagnosis, treatment, and prevention of Alcohol Use Disorder (AUD) and alcohol-related health
complications. NIAAA also desires to strengthen research dissemination, scientific
communications, public education, and data collection activities in the areas of its research
priorities.
Studies that examine racial, ethnic, and gender minorities as well as other underserved populations that
experience more negative alcohol-related consequences of illness and premature death than the general
population are highly encouraged and sought.
Budget Guidance
NIAAA will make awards compliant with all statutory guidelines as outlined above. Total funding
support (direct costs, indirect costs, fees) normally may not exceed the amounts defined by the
Small Business Administration (SBA), which can be found on the NIH SEED website. With the
exception of the topics indicated therein, NIAAA will generally not fund Phase I applications to the
Omnibus greater than $385K or Phase II awards over $3M total costs even if topics are listed
under the SBA-Approved Waiver Topics. Applicants considering a requested budget greater than
the standard limits are strongly encouraged to contact the NIAAA SBIR/STTR Program Director
before submitting an application. For budgetary, administrative, or programmatic reasons, NIAAA
may decrease the length of an award and/or the budget recommended by a review committee, or
not fund an application.
Specific Funding Opportunities and Programs
In addition to the Omnibus program announcement, NIAAA has targeted Notices of Funding
Opportunities (NOFOs). Please visit our NIAAA SBIR/STTR program webpage to view the latest
targeted NOFOs.
NIAAA Phase I grantees may consider applying for the I-Corps at NIH pilot program (PA-22-073).
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NIAAA will accept SBIR/STTR Phase IIB Competing Renewal grant applications from Phase
II SBIR/STTR awardees to continue the process of developing products that require approval
of a Federal regulatory agency (e.g., FDA, FCC). Such products include, but are not limited to,
medical implants, drugs, vaccines, biologicals, and new treatment or diagnostic tools that
require FDA approval. This renewal grant should allow small businesses to get to a stage
where interest and investment by third parties is more likely. To be eligible for Phase IIB
consideration, the project must retain high significance in the light of current market
conditions.
Prospective applicants are strongly encouraged to contact NIH staff well in advance of
submitting a Phase IIB Competing Renewal application by submitting to
niaaasbirsttr@mail.nih.gov a letter of intent that includes the following information:
•
Name, address, and telephone number of the Principal Investigator
•
Names of other key personnel
•
Participating institutions
•
Grant number and title
•
Progress of the Phase II award
•
Goals and justification for the Phase IIB request
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAAA 13
It is expected that only a portion of NIAAA SBIR/STTR Phase II awards will be eligible for a Phase IIB
Competing Renewal grant.
NIAAA will accept submission of CRP applications to the following NOFOs (and reissues):
•
SBIR/STTR Commercialization Readiness Pilot (CRP) Program Technical Assistance
and Late-Stage Development - Clinical Trial Not Allowed (PAR-23-219)
•
SBIR/STTR Commercialization Readiness Pilot (CRP) Program Technical Assistance
and Late-Stage Development - Clinical Trial Required (PAR-23-220)
Clinical Trials
Does NIAAA accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
Yes
Does NIAAA accept Clinical Trials
through specific Notices of Funding
Opportunities?
Yes
https://www.niaaa.nih.gov/research/niaaa-
sbir/funding-opportunities
Does NIAAA support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
https://niaaa.nih.gov/grant-funding/funding-
opportunities
Research Topics
The topics listed below reflect examples of NIAAA’s program priorities at the time of the NIH Omnibus
solicitation and should not be considered all-inclusive. NIAAA will consider ALL applications relevant to
NIAAA’s mission. The topics below include areas of interest for both pre- clinical and clinical research.
BASIC SCIENCE
Through basic scientific research, great strides have been made in understanding the mechanisms by
which alcohol exerts its effects on human health and behavior. New tools, techniques, paradigms, and
technology are needed to enable researchers to further understand the underlying biological and
behavioral mechanisms through which conditions associated with AUD develop.
Research Tools/Technologies/Devices
1.
Induced pluripotent stem cells (iPS), including disease specific cell lines and gene-edited
models (e.g., alcohol-related organ damage and disease with human iPS cell-derived
organoids) and from adult-derived human iPSCs cells representing genetic variations in
alcohol metabolism (e.g., alcohol dehydrogenase (ADH), aldehyde dehydrogenase (ALDH),
cytochrome P450 isozyme CYP2E1, and glutathione S-transferase (GST)) or models of
normal development and alterations by prenatal alcohol exposure, and embryonic stem cell
models of development and effects of alcohol exposure.
2.
Novel technologies to measure and interpret non-coding RNA (ncRNA) gene
expression, following alcohol exposure, in the brain at the cellular level or in
non-animal research models.
3.
Using single-cell transcriptomics and multiomics technologies and sequencing to reveal
the molecular fingerprint of cell states and their predicted signaling circuits in tissues
across development and AUD.
4.
Tools to detect dynamic and concurrent changes of neurotransmitters and neuromodulators
in the brain of behaving animals.
5.
Tools to detect the effects of alcohol on the central nervous system (CNS) structure and activity.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAAA 14
6.
Novel animal models, including transgenic animals.
7.
Hepatocyte cell line capable of maintaining viability and metabolic functions in culture systems
for an indefinite period.
8.
Experimental systems that mimic organ function.
9.
New methods of ethanol administration to animals that produce precise dose control
or that closely mimic types of alcohol exposure occurring in humans.
10.
New ligands that will enhance the potential usefulness of PET and SPECT
neuroimaging technologies for the study of the etiology of AUD and related
brain pathology.
11.
Humanized animal models to study AUD in different organ systems.
12.
Methods to detect epigenetic changes as disease drivers due to metabolic reprogramming by
alcohol.
13.
Tools to determine the prevalence of alcohol associated organ diseases:
alcoholic cardiomyopathy, sarcopenia, pancreatitis, pulmonary, immune
and bone diseases.
14.
Optoelectronics probes and devices used to manipulate nerve cell activity in awake
animals to better study nerve cell function in the body’s periphery.
15.
Generate organoids from iPSCs that specifically model sleep-regulating brain regions
affected by alcohol, providing a platform for studying alcohol's neurobiological impacts on
sleep at the organ level.
PREVENTION/TREATMENT/RECOVERY
Prevention strategies/programs and educational services, behavioral treatment programs, medications,
and digital health technologies are crucial in ameliorating the negative health effects and consequences
associated with AUD and alcohol misuse and recovery.
Medications Development
1.
Preclinical and/or clinical development of therapeutics for AUD and alcohol-related
complications (e.g., craving, sleep problems, withdrawal symptoms, and negative
affect).
2.
Early therapeutic discovery activities (e.g., target ID, lead compound target validation).
3.
Investigational New Drug (IND)-enabling studies.
4.
Extended formulations or reformulations of existing medications that improve
efficacy or compliance.
5.
Therapeutics for individuals with co-occurring health conditions, such as post-traumatic
stress disorder (PTSD), HIV, alcoholic hepatitis, liver fibrosis, cirrhosis, pancreatitis,
cardiomyopathy, or other alcohol-induced tissue damage.
6.
Development of precision medicine tools (e.g., biomarker panel) to predict
treatment outcomes among AUD patients.
Programs or Therapies to Prevent or Treat AUD and/or the Consequences of Alcohol Misuse,
Hazardous Drinking, and AUD Across the Lifespan
1.
Novel behavioral health or educational programs aimed at preventing or treating AUD
or associated consequences of AUD, alcohol misuse, or hazardous drinking across
the life span.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAAA 15
2.
Prevention or treatment programs tailored specifically to the needs of the following groups:
children of individuals with AUD, women, racial and ethnic underrepresented populations,
sexual and gender minority populations, individuals with Fetal Alcohol Spectrum Disorders
(FASD) across the lifespan, persons with disabilities, adolescents/young adults, the elderly,
individuals in rural settings, individuals with psychiatric comorbidities (e.g., PTSD, major
depressive disorder, etc.).
3.
Computerized versions of empirically supported prevention or treatment programs,
including but not limited to in languages other than English.
4.
Prevention curricula, videos, multi-media programs, and training materials for
use with adolescents and other population groups and in the NIAAA priority
areas.
5.
Therapeutic, skill-building, and educational program products that enhance
behavioral, neurocognitive, social, adaptive, and motor function to improve
the overall well-being of individuals with FASD and their families.
6.
Therapies to mitigate alcohol-associated adverse impact on the development of liver
and/or lung diseases.
7.
Strategies and methods to increase awareness and salience among high-risk groups of
the tragic consequences of driving after drinking.
8.
Therapies or programs specifically focused on sustaining mid- and long-term recovery from AUD.
Digital Health Tools (mHealth, health IT, wearable devices, telehealth, telemedicine, and
personalized medicine)
1.
Wearable Alcohol Biosensor - minimally invasive, near real-time detection, remote
monitoring, infrared or other non-sweat based technology preferred.
2.
Validation of promising technologies, biosensors, and research tools.
3.
Development of precision medicine tools to predict an individual’s risk for developing AUD
and/or quantify progression to an AUD diagnosis.
4.
Tools to improve the prevention or treatment of AUD and alcohol-related problems.
5.
Applications that facilitate long-term recovery support and improve continued
engagement in recovery support services.
6.
Tools to improve the identification and diagnosis of FASD and prenatal alcohol exposure.
7.
Applications or tools to improve medication safety (e.g., multiple medications,
interactions with alcohol).
8.
Mobile device applications or other health technologies to improve the effectiveness,
accessibility, and use of behavioral interventions for AUD and co-occurring disorders,
including HIV.
9.
Solutions or applications to improve minority health and health disparities with
capabilities of reaching persons in rural, remote, and under-resourced/under-served
communities.
10.
Virtual reality (VR) technology to create immersive environments that simulate real-
world scenarios involving alcohol consumption and its effects on behavior and sleep,
for understanding environmental and social factors in AUD.
11.
Non-invasive, wearable devices capable of monitoring physiological and biochemical
markers of alcohol intake and its impact on sleep patterns in real-time, utilizing
technologies like bioimpedance.
12.
App-based digital platforms that offer personalized cognitive-behavioral therapy for
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAAA 16
insomnia (CBT-I) and other sleep improvement techniques for individuals with AUD,
integrating wearable sleep data.
DIAGNOSTICS
Improving the current battery or developing new approaches to measurement, diagnosis, and
assessment of the severity of AUD, alcohol misuse and health consequences, FASD, and alcohol-
related organ damage.
Imaging Examination Technologies for Early and Precise Diagnosis of Alcohol-Related Organ
Damage
Biomarkers for AUD and alcohol-related health effects
1.
Detection (e.g., biochemical, unbiased assay) of alcohol intake for extended period
(e.g., 2 weeks, 2 months) after drinking episode.
2.
Signatures of alcohol-induced organ damage and familial risk.
3.
Reduction of time to results for current assays (e.g., phosphatidylethanol (Peth),
ethyl glucuronide (EtG)).
4.
Increase accuracy of alcohol intake detection by developing a novel combination
of biomarkers (e.g., PEth, EtG)).
5.
Improve assay methodologies for established biomarkers of alcohol consumption considering
cost, timeliness, and accessibility in a range of clinical settings.
6.
Point of care devices, for use in rural or remote primary care and hospital settings.
7.
Validation of promising biomarkers that can be used to improve clinical
research and practice (for example, diagnosis, prognosis, and treatment
response) for alcohol related health conditions, including AUD, FASD, and
alcohol associated organ injury.
8.
Tools or kits to measure aristolochic acid (AA)-adducts and advanced glycation end
products (AGEs) in serum, cerebral spinal fluid, and brain and other organs impacted by
AUD in animal models and pre-clinical settings including their relationship to the
biomarkers of neuro- inflammation.
9.
Tools to detect alcohol-induced damage in those patients with HIV infection or co-infection.
10.
Measurement and integration of ‘omics data for AUD and alcohol-related organ damage.
DATA SCIENCE
Software and tools can be used for discovery of new biomarkers and targets, precision medicine, and
other applications to increase the efficiency and efficacy of treating AUD and alcohol-related health
effects.
Data Science Tools
1.
Algorithms for integrative analysis incorporating multiple current NIAAA supported (current
and legacy), government, and public datasets, including machine learning, deep learning,
artificial intelligence, data mining and other model based and model-free approaches.
2.
Software applications for data interfaces for aggregation, imputation, harmonization,
or visualization of data from multiple sources, including current and future NIH data
systems.
3.
Algorithms and/or software tools for improving data collection, i.e., smart phone
apps, extraction of specific alcohol research parameters from existing large
databases and established public health studies, biological sensors or wearable

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAAA 17
devices.
4.
Computational and/or systems biology models of alcohol exposure, tolerance, and resilience.
5.
Computational, statistical or bioinformatics tools to organize and manage high
throughput data obtained by genomic, functional genomic, or other ‘omic strategies.
6.
Computational tools to combine multiple data modalities (e.g., omics, imaging).
7.
Application of machine learning and artificial intelligence, including large language
models, in alcohol research, including ethics and privacy concerns.
8.
Translation of ‘omics’ data into clinically relevant predictions and outcomes for AUD
and alcohol- related organ damage.
Contact Information
Megan Ryan, M.B.A.
NIAAA SBIR/STTR Program Director
National Institute on Alcohol Abuse and Alcoholism
6700B Rockledge Dr.
Rockville, MD 20852-1705
Phone: 301-827-6166
Email: mryan1@nih.gov
For general inquiries: NIAAASBIRSTTR@mail.nih.gov
For administrative and business management questions,
contact:
Jeff Thurston
SBIR/STTR Grants Management Lead
National Institute on Alcohol Abuse and Alcoholism
Phone: 301-443-9801
Email: jeffrey.thurston@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 18
NATIONAL INSTITUTE OF ALLERGY AND INFECTIOUS DISEASES (NIAID)
Mission
NIAID conducts and supports basic and applied research to better understand, treat, and ultimately
prevent infectious, immunologic, and allergic diseases. Read more about the NIAID Mission at our
website.
Budget Guidance
Total funding support (direct costs, indirect costs, fee) normally may not exceed the dollar amounts
specified by the SBA, which can be found on the NIH SEED website. Budget requests at or near
these hard caps should be well justified. Phase II/IIB applicants should note that NIAID will not
generally allow awards (of any duration) that exceed $1,000,000 total costs per year.
NIH has received a waiver from SBA, as authorized by statute, to exceed total award amount hard
caps for specific topics. The current list of approved NIAID topics is included in the SBA-Approved
Waiver Topics. Topics that align with NIAID’s priority research areas are listed for each Division; any
listed NIAID topic is sufficient to consider budget requests that exceed the hard caps. Budget
requests exceeding the hard caps must be very well justified in the “Budget Justification” attachment
to the Research and Related Budget form and be clearly consistent with the scope of the proposal.
For proposals that address an approved topic, NIAID will allow Phase I applications with budgets of up to
$300,000 total costs per year for up to 2 years; and Phase II or Phase IIB applications with budgets of up to
$1,000,000 total costs per year for up to 3 years. Requests for these budget levels must be very
well- justified. In all cases, applicants should propose a budget that is reasonable and appropriate for
completion of the research project.
NIAID staff cannot provide prior approval to exceed hard caps. Compliance with a pre-approved topic will
be confirmed at time of award by the applicant’s Grants Management Specialist and Program Officer.
NIAID will consider well justified Technical and Business Assistance (TABA) costs up to the limits
specified on the NIH SEED Website. These costs can be requested in addition to the Phase I and II
limits indicated above.
NIAID will generally not make SBIR or STTR awards with budgets that exceed these guidelines.
For budgetary, administrative, or programmatic reasons, NIAID may decide not to fund an
application or may decrease the length of an award and/or the budget recommended by a review
committee.
Specific SBIR and STTR Program Information
NIAID's Division of AIDS (DAIDS), Division of Allergy, Immunology, and Transplantation (DAIT), and
Division of Microbiology and Infectious Diseases (DMID) encourage SBIR/STTR applications related
to their mission and activities as described below. Questions regarding specific research areas may
be addressed to the NIAID Program Officials listed below. General questions about the NIAID SBIR
and STTR programs or administrative and business management concerns may be directed to the
NIAID Small Business Program Team.
When possible, applicants are encouraged to use email for communication.
For information about NIAID's Small Business Programs, please visit our website.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 19
Specific Funding Opportunities and Programs
Targeted Funding Opportunities and Notices of Special of Interest can be reviewed on the NIAID
website. However, NIAID welcomes all Phase I and II proposals (except clinical trials) for research
that is consistent with our Mission through the SBIR and STTR Omnibus Solicitations [Clinical Trial
Not Allowed].
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NIAID welcomes Phase IIB Competing Renewal Applications (SBIR only) for Phase II grants
and contracts via the Omnibus Solicitation for SBIR Grant Applications, and as indicated by
other NIAID Notices of Funding Opportunities (NOFOs). Standard NIAID Phase II funding policy
applies unless otherwise stated in the NOFO. STTR Phase II awardees may apply but must
switch programs to SBIR. Non-NIAID Phase II awardees must contact NIAID prior to submission
to confirm programmatic interest.
NIAID welcomes CRP applications from eligible NIAID Phase II/IB awardees through PAR-20-129 (and
subsequent reissued NOFOs) Please review this Notice of Funding Opportunity for details.
Clinical Trials
Does NIAID accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
No
Does NIAID accept Clinical Trials
through specific Notices of Funding
Opportunities?
Yes
NIAID SBIR Phase II Clinical Trial
Implementation Cooperative Agreement (U44):
https://grants.nih.gov/grants/guide/pa-files/PAR-
21-082.html (and subsequent reissued NOFOs)
Does NIAID support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
NIAID Clinical Trial Implementation Cooperative
Agreement (U01):
https://grants.nih.gov/grants/guide/pa-files/PAR-
21-083.html (and subsequent reissued NOFOs)
NIH Research Project Grant (Parent R01
Clinical Trial Required):
https://grants.nih.gov/grants/guide/pa-files/PA-
20-183.html (low risk clinical trial proposals,
only)
NIAID will generally consider clinical trial proposals consistent with the research topics listed
below. However, applicants are strongly encouraged to consult with NIAID Program Staff at least
10 weeks before the receipt date.
For further information, please consult NIAID’s Investigator-Initiated Clinical Trial Resources page:
https://www.niaid.nih.gov/grants-contracts/investigator-initiated-clinical-trial-resources
Research Topics
Division of AIDS (DAIDS)
The Division of AIDS (DAIDS) supports a global research portfolio to advance biological knowledge
of HIV/AIDS, its related co-infections, and co-morbidities. With the ultimate goal of creating an
“AIDS-Free Generation,” the division develops and supports the infrastructure and biomedical
research needed to: 1) Reduce HIV incidence through the development of effective biomedical
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 20
prevention strategies, including vaccines that are safe and desirable, 2) Develop novel approaches
for the treatment and cure of HIV infection, 3) Develop interventions to treat and/or prevent co-
infections and co-morbidities of greatest significance, and 4) Engage scientific and community
stakeholders to equitably implement effective interventions.
Basic Sciences Program
Supports basic and applied research on the causes, diagnosis, treatment, and prevention of HIV and AIDS.
A.
Epidemiology Branch. Population-based research, modeling, and comparative
effectiveness studies (not including clinical trials) that assess the natural history, biologic,
and clinical course of HIV/AIDS, and related outcomes, and could advance treatment
and prevention of HIV. Specific interests include phylodynamics and other factors related
to HIV transmission and associated biological and behavioral factors, basic research on
immunology, virology, and antiretroviral therapy, issues surrounding care for HIV and
other co-morbidities, interactions and impact on clinical outcomes. Development of novel
electronic tools, including devices and computer programs to enhance behaviors, such
as treatment adherence or uptake of treatment guidelines, is also of interest.
B.
Pathogenesis & Basic Research Branch. Innovative technologies for at-home self-
testing to directly detect HIV during the earliest stages of acute infection (before antibody
response) or to detect viral rebound following long-term suppression of viremia.
Identification and validation of new targets for discovery or design of strategies to prevent
HIV transmission, inhibit replication, control viremia in the absence of antiretroviral drugs,
or eradicate reservoirs of HIV that persist despite long-term antiretroviral therapy.
Innovative approaches for predicting post-treatment immunologic control of viral rebound
or for monitoring changes in the size of the rebound- competent HIV reservoir.
Determination of atomic structures relevant to HIV prevention, treatment, or cure.
C.
Targeted Interventions Branch. Discovery and development of small molecule inhibitors
with novel or underexplored mechanisms of action using standard and high-throughput
technologies; cell-based and gene therapies; RNA-based therapeutics; next-generation
biologics; novel targeting and delivery vehicles for agents active against HIV; therapeutic
vaccines and monoclonal antibodies; protein chemistry-based anti-HIV approaches;
assays to quantitate latent virus; animal models to facilitate evaluation of agents to treat or
cure HIV infection.
Vaccine Research Program
Supports the discovery, development and clinical evaluation of an HIV/AIDS vaccine.
A.
Vaccine Clinical Research Branch. Research areas: (1) phase I, II, and III domestic and
international clinical trials of candidate AIDS and TB vaccines and anti-HIV antibodies; (2)
evaluation and characterization of immune responses, virologic markers, and improved
diagnostic approaches in HIV-infected and uninfected immunized volunteers, and (3)
technologies and methods to improve clinical efficacy or reduce the burden of vaccine or
monoclonal antibody administration.
B.
Preclinical Research and Development Branch. Preclinical research to assess and
overcome specific biomedical obstacles in HIV vaccine discovery, especially by application of
innovative technologies, and/or by the development and supply of novel reagents/resources
useful for advancing original vaccine platforms including monoclonal antibody discovery and
development for prevention of HIV infection.
C.
Vaccine Translational Research Branch. VTRB enables research by advancing innovative
vaccine concepts and scalable unit operations into the development of cGMP manufactured
products. VTRB’s efforts to accelerate the development of preventive HIV-1 vaccines involves
identifying, supporting and advancing: (a) cell line development to increase Env expression,
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 21
production, quality, and yield; (b) evaluation of phase-appropriate upstream and downstream
manufacturing processes;(c) scalable and prototype process development and purification
platforms; (d) cGMP manufacturing of broad portfolio of vaccine products ranging from complex
HIV Env protein immunogens, nanoparticle-based vaccines, viral vectors, virus-like particles
(VLP), nucleic acid-based vaccines (DNA and mRNA), monoclonal antibodies for testing in
early phase human clinical trials; (e) manufacturing new and/or alternative adjuvant analogs
with similar agonist functions as those currently available for optimal immune response; (f)
novel and emerging nanoparticle antigen and adjuvant delivery modalities and dosage forms,
coformulation technologies and platforms for immunization; (g) antigen-adjuvant formulation
development, analytics development to support product characterization, in-process
operations, release, and stability testing; and (h) preclinical safety, immunogenicity, and
toxicology testing.
Therapeutics Research Program
Develops and oversees research and development of therapies for HIV disease, including
complications, co-infections and co-morbidities, in adults.
A. Drug Development and Preclinical Research Branch. Basic, preclinical, and translational
research for development of new therapies for HIV and HIV-associated co-infections, including
Mycobacterium tuberculosis and viral hepatitis; development of safer, more efficacious antiviral,
antimicrobial, and immune-based therapies, and combinations thereof, including long-acting/extended-
release approaches; target identification and validation for HIV-related co-infections and assay
development for screening potential therapeutics; preclinical research to elucidate the biology of HIV-
related co-infections, including pathogenesis, immune protection and control, and persistence and
latency; maintenance of a database of potential anti-infectives for HIV and HIV-related coinfections.
B. Laboratory and Clinical Sciences Branch. Research focused on biomarker discovery/validation
and assay development for diagnostics, including development and evaluation of practical and affordable
tests to measure viral load, drug toxicities, and drug resistance for clinical use; development and testing
of new or improved methodologies for diagnosing, monitoring, and following patients under treatment,
including tests to detect early infection in seropositive HIV-infected adult and pediatric individuals in poor
resource settings; clinical development of laboratory assays; clinical immunology, virology, and
pharmacology related to the design and conduct of clinical trials; management of quality assurance
contracts for oversight of the quality of clinical laboratory testing in support of clinical trials.
C. HIV Research Branch. Clinical research in adults to evaluate chemotherapeutic and immune-
based interventions to treat acute and chronic HIV infection and approaches to achieve sustained
remission or cure; strategies to augment HIV-specific immune responses and general host immunity to
control or clear HIV infection.
D. Complications & Co-Infections Research Branch. Clinical research in adults to evaluate new
or improved therapies and related strategies for the treatment and/or prevention of HIV-related co-
infections (exclusive of Mycobacterium tuberculosis) and non-infectious co-morbidities, including Immune
Reconstitution Inflammatory Syndrome (IRIS), in people living with HIV.
E. Tuberculosis Clinical Research Branch. Clinical research in adults to evaluate therapeutics,
therapeutic vaccines and strategies to prevent disease recurrence for tuberculosis in people living with
HIV, including those with additional medical conditions that may affect disease outcomes; clinical trials
with a primary objective to elucidate the pathophysiology and immunopathogenesis of HIV/TB co-infection
including the study of co-infection interactions and changes in the course, pathology, treatment
responses, and outcome of either infection.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 22
Prevention Science Program
Develops and oversees research and development of 1) non-vaccine biomedical HIV prevention
strategies in adolescents and adults, and 2) therapies for cure, management, treatment and prevention of
HIV and HIV-associated complications in pregnant women, infants, children, and adolescents, including
pediatric-friendly formulations. Supports domestic and international phase I, II, and III clinical trials to
evaluate these prevention or therapeutic strategies in relevant populations.
A.
Preclinical Microbicides and Prevention Research Branch. Development of non-
vaccine biomedical HIV prevention products including topical microbicides, pre-
exposure prophylaxis (PrEP), post-exposure prophylaxis (PEP), and multipurpose
prevention technologies (MPT). Emphasis on drug delivery systems (DDS) designed
to achieve systemic protection for ≥ 3 months. Development of shorter-duration
products (i.e., minimum of 7 days to <3 months), which address a compelling specific
public health need. Key populations are adolescents, cisgender women, men who
have sex with men (MSM), and transgender people.
B.
Clinical Prevention Research Branch. Development of safe and effective non-
vaccine biomedical and integrated HIV prevention interventions to reduce the
number of new HIV infections in adults and adolescents. Support the development
of HIV incidence assays, biomarkers of adherence, mathematical modeling, and
other tools needed to accomplish these objectives. Clinical development of topical
microbicides to prevent HIV infection with the goal to advance safe, effective, and
acceptable microbicide products toward licensure.
C.
Maternal, Adolescent and Pediatric Medicine Branch. Therapies for cure,
management, treatment and prevention of HIV and HIV-associated complications
including TB, in pregnant women, infants, children, and adolescents, including
development of pediatric-friendly formulations. Strategies to reduce transmission of
HIV and HIV co-infections from mother to child.
Division of Allergy, Immunology, and Transplantation (DAIT)
The Division of Allergy, Immunology, and Transplantation (DAIT) supports studies of the immune
system in health and the cause, pathogenesis, diagnosis, prevention, and treatment of disease
caused by immune dysfunction.
A.
Allergy, Asthma and Airway Biology Branch. Conditions of interest: asthma, food
allergy, eosinophilic esophagitis and gastroenteritis in relation to food allergy, atopic
dermatitis, urticaria, rhinitis, rhinosinusitis, drug allergy, sepsis. The Branch supports basic
and clinical studies investigating mechanisms of disease and new approaches to diagnose,
treat or prevent these conditions. Special interest for SBIR/STTR includes a) the
development of biomarkers as diagnostic markers, markers of disease severity and
predictive markers for treatment effectiveness, particularly of immunologic interventions such
as allergen immunotherapy for food and respiratory allergy; b) the development of new
forms of allergen immunotherapy aiming at increased tolerogenic immune responses and
decreased allergenicity.
B.
Basic Immunology Branch. The Branch supports basic and clinical research in the
following areas: adjuvant discovery and development; origin, maturation, and interactions of
immune cells; immune cell receptors, and ligands; cytokine biology; molecular basis of
immune activation, antigen recognition, and immune tolerance; immune response
regulation; hematopoiesis and stem cell biology; computational immunology; immunologic
mechanisms associated with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome;
assessment and analysis of vaccine effectiveness in neonates, pregnant women, and
adults, and basic immunology of vaccines and immunotherapeutics as medical

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 23
countermeasures for biodefense. Special interests for SBIR/STTRs include: adjuvant
discovery , development, production of biosimilars, and/or head-to-head comparisons;
bioinformatics tools for immune epitope predictions/visualization, and/or for the analysis of
multi-parameter or systems immunology data; development and validation of immunologic
reagents for analysis of immunity in non-mammalian (e.g., Xenopus laevis, zebrafish, C.
elegans) and under-represented mammalian (e.g., pig, ferret, cow, sheep, bat) models, and
development of novel/improved sample sparing methods to analyze human immune
responses from limited amounts of human sample (tissue, cells, serum, etc.).
C.
Autoimmunity and Mucosal Immunology Branch. Preclinical and clinical research to
develop and improve the diagnosis and treatment of autoimmune diseases and primary
immune deficiencies/inborn errors of immunity (not HIV); basic research of autoimmune
disease mechanisms and biomarkers; immunotherapy of disease processes; disorders
mediated by lymphocyte products; and discovery and/or development of reagents and other
tools for analysis of mucosal immunity.
D.
Transplantation Branch. Preclinical and clinical research in organ, vascularized composite
tissue and cellular transplantation: acute and chronic graft rejection, allogeneic and
xenogeneic transplantation, development of immunomodulatory agents to prevent and treat
graft rejection and to promote acute and long term graft acceptance and immunologic
tolerance, genomics of the alloimmune response, graft versus host disease for
hematopoietic stem cell transplantation, minor histocompatibility antigens, complications of
immunosuppression in transplantation, and major histocompatibility complex (MHC) region
genomics, technologies for MHC typing, and clinical applications of high-resolution HLA
typing.
E.
Radiation and Nuclear Countermeasures Program (RNCP). The RNCP will consider
preclinical research to support product development activities leading to interactions with the
Food and Drug Administration (FDA). Approaches could include those used to diagnose,
mitigate, and/or treat acute or delayed effects of radiation exposure resulting from a
radiological or nuclear incident. It is anticipated that in most cases, approval will occur in
accordance with the FDA Animal Rule (21 CFR 314.600 Subpart I for drug products and 21
CFR 601.90 Subpart H for biologic products).
Proposed activities could include:
•
Animal model studies or ex vivo approaches (e.g., human tissue chips) to
confirm/optimize product efficacy;
•
Mechanism of action studies needed for FDA consideration;
•
Good Laboratory Practice (GLP)/non-GLP
pharmacology/toxicology/pharmacokinetics/pha
rmacodynamics;
•
GLP pilot animal efficacy studies;
•
Good Manufacturing Practice product scale-up and stability studies;
•
Biomarker and biodosimetry assay/device development to determine
radiation dose and/or the biological impact of radiation exposure (in vivo and
ex vivo models acceptable).
Priority areas of product development include:
•
Approaches targeting organ systems/microbiota, for which no treatments
are available (e.g., gastrointestinal, lung, kidney, cardiac, vascular, and
skin);
•
Approaches to mitigate and/or treat radiation injury given 24 hours or later post-
irradiation;
•
Minimally invasive, predictive radiation markers, diagnostics and devices for
biodosimetry;
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 24
•
Radionuclide decorporation agents.
Division of Microbiology and Infectious Diseases (DMID)
The Division of Microbiology and Infectious Diseases (DMID) supports research to better understand,
treat, and ultimately prevent infectious diseases caused by virtually all infectious agents, except HIV.
DMID supports a broad spectrum of research from basic molecular structure, microbial physiology, and
pathogenesis, to the development of new and improved vaccines, therapeutics, and vector control
measures. DMID also supports medical diagnostics research, which is defined as research to improve
the quality of patient assessment and care that would result in the implementation of approp riate
therapeutic or preventive measures. In addition, DMID supports studies to better understand
mechanisms of pathogen transmission that may include environmental factors. DMID does not support
research directed at decontamination or the development of environmentally oriented detectors, whose
primary purpose is the identification of specific agents in the environment. Note that some of the
organisms and toxins listed below are considered NIAID priority pathogens or toxins for biodefense and
emerging infectious disease research.
A.
Bacteriology and Mycology Branch.
The branch oversees research and product development related to:
•
Bacterial infections with emphasis on hospital-associated pathogens, including
Acinetobacter, Klebsiella, Serratia, Legionella, Pseudomonas, Aeromonas,
Enterobacter, Proteus, non-enteric E.coli, staphylococci, enterococci, actinomycetes
among others;
•
Bacterial zoonoses, including plague, anthrax, tularemia, glanders, melioidosis, Lyme
disease, borrelial relapsing fevers, rickettsial diseases, anaplasmosis, ehrlichiosis,
bartonellosis, scrubtyphus, Q fever, and leptospirosis;
•
Fungal infections including those caused by Candida, Aspergillus, Cryptococcus,
Coccidiodes, Histoplasma, Blastomyces, Pneumocystis, Microsporidia, and other
pathogenic fungi.
Research is encouraged in the following general areas: (1) vaccines, adjuvants, therapeutics and
diagnostics (including target identification and characterization, device or apparatus development,
novel delivery, and preclinical evaluation); (2) strategies to combat antibacterial and antifungal drug
resistance; (3) applied proteomics and genomics; (4) host-pathogen interactions, including
pathogenesis and host response; (5) genetics, molecular, and cell biology; and (6) microbial
structure and function.
Research on all of the above is welcome, but the following areas are of particular interest to the
branch:
•
Vaccines, therapeutics, and medical diagnostics for hospital infections
•
Adjunctive therapies and non-traditional approaches to combat and treat antimicrobial
resistance
•
Diagnostics for invasive fungal diseases
•
Novel approaches for the diagnosis of Lyme disease
•
Vaccines against Coccidioidomycosis
B.
Enteric and Sexually Transmitted Infections Branch.
Enteric Section:
Enteric Infections Section research portfolios focus on enteric bacterial pathogens, their toxins,
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 25
and their infectious diseases; related sequela; and the gastrointestinal microbiota and
microbiome.
Special emphasis areas include but are not limited to those below:
• Development of vaccines to prevent bacterial enteric diseases, to protect against neurotoxins
and enterotoxins, and to combat enteric diseases in vulnerable populations.
• Development of therapeutics that focus on novel targets, that target toxin activities, and that
treat recurrent diseases.
• Development of live biotherapeutic products to restore colonization resistance to enteric
pathogens, to combat recurrent or chronic enteric disease, and to restore host immunity against
enteric pathogens.
• Development of adjunctive therapies and non-traditional approaches to treat resistant bacteria
and to combat further development of antibacterial resistance.
• Development of rapid diagnostics to identify multiple pathogens and their antimicrobial
resistance profiles that are appropriate for use in low-resource, outbreaks, and clinical settings,
as well as diagnostic approaches that differentiate asymptomatic colonization from infection.
Sexually Transmitted Infections Section:
Areas of emphasis include the development of medical diagnostics including better and more
rapid multiplex point of care tests, ability to rapidly determine antibiotic sensitivity, and novel
technologies enabling testing in low resource settings while maintaining high
sensitivity/specificity; development of new classes of antimicrobials and non-antimicrobial
treatment approaches, particularly those focused on reducing the development of antibiotic
resistance; novel delivery systems for multipurpose prevention technologies, vaccines and
therapeutics for Sexually Transmitted Infections (STIs) and other reproductive tract syndromes
such as bacterial vaginosis and pelvic inflammatory disease; understanding vaginal ecology and
immunology and approaches to developing synthetic microbiota for use as biotherapeutics or as
adjunct therapy to antibiotic treatment; development of epidemiologic and behavioral strategies
to reduce transmission of STIs; developing and evaluating interventions and products to better
serve adolescents, medically underserved populations, and minority groups who are
disproportionately affected by STIs; development of multipurpose prevention technologies to
prevent STIs, HIV, and unintended pregnancies; better understanding of the role of STIs in
infertility, premature birth, and adverse outcomes of pregnancy and how to improve outcomes;
and better understanding of the role of STIs in HIV transmission and the role of HIV in altering
the natural history of STIs.
C.
Respiratory Diseases Branch.
Research areas include: (1) viral respiratory diseases caused by influenza viruses, human
coronaviruses including SARS, MERS, and novel emerging coronaviruses, rhinoviruses,
respiratory syncytial virus and other related pneumoviruses and paramyxoviruses; (2)
mycobacterial diseases, including tuberculosis (TB) caused by bacteria of the Mycobacterium
tuberculosis complex, leprosy, Buruli ulcer and non- tuberculous mycobacterial (NTM)
diseases, particularly pulmonary infections in persons not afflicted with HIV/AIDS; (3) other
bacterial respiratory diseases including bacterial pneumonia primarily caused by Streptococcus
pneumoniae, Pseudomonas aeruginosa, and Haemophilus influenzae, pertussis, Group A and
B streptococcal diseases, meningitis, upper respiratory infections, acute exacerbations of
chronic obstructive pulmonary disease, and cystic fibrosis; and (4) mixed viral/bacterial
respiratory infections.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 26
Special emphasis areas include:
•
Development of new or improved antimicrobials (especially for antimicrobial-resistant
pathogens) and antivirals, including immunotherapeutics, immunomodulators, and host-
directed therapies to augment anti-infectives;
•
New or improved vaccines (with and without adjuvants);
•
Improved delivery systems and formulations for drugs/vaccines;
•
Microbial and host biomarkers and biosignatures suitable for diagnostic tests;
•
Development of novel or improved diagnostic tools for detection of infection and drug
resistance, including rapid point of care diagnostics and quantitation of pathogen in
response to therapy;
•
Diagnostics to distinguish viral from bacterial infections.
There is particular need for preventive and treatment countermeasures for influenza, including
universal vaccine platforms and broad-spectrum antivirals; for novel treatment of respiratory
syncytial virus (RSV) and related pneumovirus and paramyxovirus infections; for next
generation vaccines, therapeutics, and diagnostics for the prevention and treatment of
COVID-19, including pan-coronavirus approaches; for diagnostics including diagnostics for
pediatric populations, novel therapeutics, and vaccines (including adjuvants) against
Mycobacterium tuberculosis (TB); for relevant diagnostics, preventive and curative
interventions against non-HIV associated pulmonary Non-tuberculous mycobacteria (NTM);
and for the prevention, diagnosis, and treatment of Bordetella pertussis, Group A
streptococcus, and Streptococcus pneumoniae infections and other antibacterial resistant
infections.
D.
Parasitology and International Programs Branch.
Research areas: (1) protozoan infections, including amebiasis, cryptosporidiosis, cyclosporiasis,
giardiasis, leishmaniasis, malaria, trypanosomiasis, toxoplasmosis; helminth infections, including
cysticercosis, echinococcosis, lymphatic filariasis, schistosomiasis, onchocerciasis, others (e.g.,
roundworms, tapeworms, and flukes); invertebrate vectors/ectoparasites responsible for human
disease (.e.g., mosquitoes, black flies, sandflies, tsetse flies, ticks, triatomine bugs, fleas, lice,
mites), and selected intermediate hosts of parasites (e.g., snails); (2) parasite biology (genetics,
genomics, physiology, molecular biology, and biochemistry); (3) protective immunity,
immunopathogenesis, and evasion of host defense; (4) clinical, epidemiological, and natural
history studies of parasitic diseases;
(5) research and development of vaccines, drugs, immunotherapeutics and immunoprophylaxis,
and medical diagnostics; and (6) vector biology and management/control and mechanisms of
pathogen transmissions.
Research on the above is welcome, but research on the following is of particular interest to the branch:
•
New drug discovery or re-purposing of existing drugs to prevent infection and/or transmission,
or to treat parasitic diseases
•
Highly sensitive and specific diagnostics tools for parasitic diseases
•
Vaccines and vaccine technologies, monoclonal antibodies, and other immune-
mediated interventions applicable to prevention or elimination of parasitic
diseases
•
Technologies or approaches that address arthropod vector monitoring, management, and
control, to prevent transmission of vector-borne pathogens to humans
E.
Virology Branch.
The Virology Branch focuses on:
NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 27
a.
Acute viral infections caused by arthropod-borne (e.g., mosquito, tick-borne) and rodent-
borne viruses, including: dengue, zika, west nile, Japanese encephalitis, chikungunya,
yellow fever, hanta, crimean-congo hemorrhagic fever (CCHF), hazara, severe fever with
thrombocytopenia syndrome (SFTS), heartland, bourbon, tick-borne encephalitis (TBE),
powassan , lacrosse, cache valley, rift valley fever, punta toro, andes, sin nombre, hantaan;
viruses causing hemorrhagic fevers:
ebola, lassa, junin, venezuelan equine encephalitis (VEE), etc.; and other viruses, including
nipah, hendra, measles, polio, coxsackie, entero, pox, rabies, rubella, astro, calici, and rota;
pathogen X
b.
Persistent viral infections caused by viruses including adeno, borna, corona, herpes,
human T-lymphotrophic, human papilloma, parvo, and human polyoma (JC, BK, and
emerging);
c.
Acute infections with hepatitis viruses A, B, C, D and E (HAV, HBV, HCV, HDV,
and HEV); chronic infections with hepatitis viruses, B, C, D and E;
d.
Transmissible Spongiform Encephalopathies (TSE)
Areas of emphasis for SBIR/STTR applications include:
•
Development of vaccines and vaccine platforms;
•
Development of techniques to improve vaccine stability;
•
Approaches to identify antiviral targets and agents;
•
Chemical design and synthesis of novel antiviral agents;
•
Development of therapeutic, prophylactic, and postexposure prophylactic interventions;
•
Development and validation of point of care assays for disease diagnosis and to
measure response to therapy;
•
Development of new preclinical animal model systems that predict clinical efficacy of
vaccines, therapeutics and diagnostics.
The Virology Branch does not support applications covering environmental
detection and decontamination.
Office of Genomics and Advanced Technologies
The Office of Genomics and Advanced Technologies focuses on broad-based research that
emphasizes the development and improvement of high-throughput and large-large scale genomics
and other advanced technologies for the understanding of infectious diseases and the development
of multiplex platforms for medical diagnostics. The technological scope encompasses genomics,
genomic epidemiology, phylogenomics, functional genomics, proteomics, metabolomics, glycomics,
structural biology, systems biology, computational biology, bioinformatics, and diagnostics, usually
across multiple pathogens or pathogen groups.
The goal of our program is (1) to support large-scale experiments using omics, structural and
computational biology approaches, (2) to deepen the comprehension of pathogen-host interactions
and (3) to accelerate the discovery of innovative diagnostics, vaccines, and therapeutics for infectious
diseases. Our program supports the advancement of technologies and platforms that are pathogen-
independent or address multiple pathogens and may include sample preparation, instrumentation,
and instrument validation.
Special emphasis areas include:

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 28
• Development and advancement of genomic, phylogenomic, proteomic, metabolomic,
glycomic, structural biology and related technologies for infectious diseases, including single-
cell omics technologies and platforms;
• Development of bioinformatic and computational biology, including artificial
intelligence/machine learning methods and tools to advance infectious disease research; and
• Development of modeling and bioinformatic tools to integrate omics data that supports the
development of vaccines, therapeutics and diagnostics.
Office of Biodefense Research and Surety (OBRS)
The Office of Biodefense Research and Surety (OBRS) supports and oversees a trans-NIH
research portfolio to advance discovery and early development of medical countermeasures
(MCMs) against chemical threats. To learn more about OBRS and its leadership role in chemical
countermeasures research at the NIH, see NIH CCRP: A Collaborative Opportunity to Develop
Effective and Accessible Chemical Medical Countermeasures for the American People, published
in the Wiley journal Drug Development Research.
Biodefense Research Countermeasures Branch (BRCB)
The Chemical Countermeasures Research Program (CCRP) supports preclinical basic and
applied research towards understanding acute and long-term chronic toxicity resulting from
exposure to Department of Homeland Security-designated Chemicals of Concern (CoC) and
early development of MCMs to prevent mortality and serious morbidities. The ideal MCM should
have rapid post-exposure efficacy, is easily administered in a mass casualty situation (likely by first
responders in personal protective equipment) and is widely accessible in the community.
The specific injuries caused by toxic chemical exposure often manifest similarly to conditions
observed in conventional clinical practice, such as acute lung injury, acute respiratory distress
syndrome, coagulopathy, tissue fibrosis, keratopathy, neovascularization, seizure, and
neurodegeneration. As such, "treat the symptom” projects aiming to repurpose already FDA-
approved products or those in late-stage development for a conventional clinical indication are
highly encouraged.
Areas of Emphasis include but not limited to:
•
Pulmonary Agents: Development of MCMs to prevent and treat acute and/or chronic lung
injury (including edema, capillary leak, and fibrosis) resulting from exposure to agents such
as sulfur mustard, chlorine, acrolein, and phosgene.
•
Ultra-Potent Synthetic (UPS) Opioids: Development of MCMs to treat life-threatening
respiratory depression caused by acute intoxication. Treatments should be fast-acting and
effective against a variety of synthetic UPS opioids such as fentanyl, carfentanil, and
related analogs, and have a mechanism of action different from existing opioid receptor
antagonists.
•
Vesicants: Development of MCMs that mitigates dermal, ocular, and/or systemic
(including myelosuppression) toxicities after exposure to chemicals such as sulfur mustard,
nitrogen mustard, Lewisite, phosgene oxime. Candidate MCM(s) with the potential to
prevent or ameliorate chronic effects such as keratopathy is encouraged.
•
Blood/Cellular Respiration Agents: Development of MCMs to treat metabolic dysfunction
and/or coagulopathy resulting from exposure to agents such cyanide, hydrogen sulfide, and
brodifacoum. Candidate cyanide and hydrogen sulfide MCM(s) should also be effective
against smoke inhalation-related exposure.
•
Nerve Agents and Organophosphorus (OP) Pesticides: Development of MCMs to treat

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 29
acute muscarinic and nicotinic toxicities, including benzodiazepine refractory seizures,
after exposure to agents such as sarin, soman, and VX.
OBRS does not support research directed at diagnostic device development, decontamination, or the
development of environmentally oriented detectors, whose primary purpose is the identification of
specific chemicals in the environment.
Contact Information
For more information on NIAID’s SBIR/STTR research topics, program policy or to identify NIAID
Subject Matter Experts for a specific topic, please contact:
Dr. Natalia Kruchinin
SBIR/STTR Program
Coordinator Division of
Extramural Activities
National Institute of Allergy and Infectious
Diseases Phone: 240-669-2919
Email: kruchininn@niaid.nih.gov
Division of AIDS
Director: Dr. Carl Dieffenbach
Telephone: 301-496-0545
Email: CDieffenba@niaid.nih.gov
Division Small Business Representative:
Dr. Brigitte Sanders
Telephone: 240-627-3209
E-mail: sandersbe@niaid.nih.gov
Basic Sciences Program (BSP)
Director: Dr. Diana Finzi
Telephone: 301-451-2598
Email: Dfinzi@niaid.nih.gov
Epidemiology Branch (EB)
Contact: Lori Zimand
Telephone: 240-627 3212
Email: lzimand@niaid.nih.gov
Pathogenesis and Basic Research Branch (PBRB)
Contact: Dr. Karl Salzwedel
Telephone: 301-496-5332
Email: salzwedelkd@niaid.nih.gov
Targeted Interventions Branch (TIB)
Contact: Dr. Brigitte Sanders
Telephone: 240-627-3209
E-mail: sandersbe@niaid.nih.gov
Vaccine Research Program (VRP)
Director: Dr. James Lane (acting)
Telephone: 301-435-3727

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 30
Email: laneji@mail.nih.gov
Vaccine Clinical Research Branch (VCRB)
Contact: Dr. Patricia D’Souza
Telephone: 240-627-3037
Email: pdsouza@niaid.nih.gov
Preclinical Research and Development Branch (PRDB)
Contact: Dr. Anjali Singh
Telephone: 240-627-3030
Email: anjalisingh@niaid.nih.gov
Vaccine Translational Research Branch (VTRB)
Contact: Dr. Michael Pensiero
Telephone: 301-435-3749
Email: mpensiero@niaid.nih.gov
Therapeutics Research Program (TRP)
Director: Dr. Peter Kim
Telephone: 301-451-2761
Email: peter.kim2@nih.gov
Drug Development and Preclinical Research Branch (DDPRB)
Contact: Roger Ptak
Telephone: 301-761-7424
Email: roger.pt[email protected]ov
Laboratory and Clinical Sciences Branch (LCSB)
Contact: Dr. Fatima Jones
Telephone: 240-550-7863
Email: maria.jo[email protected]ov
HIV Research Branch (HIVRB)
Contact: Tia Morton, RN
Telephone: 240-627-3073
Email: frazierti@niaid.nih.gov
Complications and Co-infections Research Branch (CCRB)
Contact: Dr. Beverly Alston
Telephone: 301-435-3773
Email: balston@niaid.nih.gov
TB Clinical Research Branch (TCRB)
Contact: Dr. Andrew Vernon
Telephone: 301-761-5130
Email:
andy.vernon@nih.gov
Prevention Sciences Program (PSP)
Director: Dr. Sheryl Zwerski
Telephone: 301-402-4032
Email: szwerski@niaid.nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 31
Preclinical Microbicide & Prevention Research Branch (PMPRB)
Contact: Dr. James Cummins
Telephone: 240-292-4800
Email: cumminsje@niaid.nih.gov
Clinical Prevention Research Branch (CPRB)
Contact: Dr. Lydia Soto-Torres
Telephone: 240-292-4807
Email: lsoto-torres@niaid.nih.gov
Maternal, Adolescent and Pediatric Research Branch (MAPRB)
Contact: Judi Miller
Telephone: 240-292-4801
Email: jmillera@niaid.nih.gov
Division of Allergy, Immunology and Transplantation (DAIT)
Director: Daniel Rotrosen, M.D.
Telephone: 301-496-1886
Email: drotrosen@niaid.nih.gov
Division Small Business Representative:
Dr. Michael Minnicozzi
Telephone: 240-627-3532
E-mail: minnicozzim@niaid.nih.gov
Allergy, Asthma, and Airway Biology
Branch Chief: Alkis Togias, M.D.
Telephone: 301-496-8973
Email: togiasa@niaid.nih.gov
Basic Immunology Branch
Chief: Dr. Alison Deckhut-Augustine
Telephone: 301-496-7551, Fax: 301-480-2381
Email: augustine@niaid.nih.gov
Autoimmunity and Mucosal Immunology Branch
Chief: Dr. Ellen Goldmuntz
Telephone: 240-627 3502
Fax: 301-480-1450
Email: egoldmuntz@niaid.nih.gov
Transplantation Branch Chief:
To be announced
Telephone: TBD
Email: TBD
Radiation and Nuclear Countermeasures
Program Director: Andrea L. DiCarlo, PhD
Telephone: 240-627-3492
Email: cohena@niaid.nih.gov
RNCP SBIR Contact:
Carmen I. Rios, PhD

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAID 32
Telephone: 240-627-3553
Email: carmen.rios@nih.gov
Division of Microbiology and Infectious Diseases (DMID)
Director: Emily Erbelding, M.D., M.P.H.
Telephone: 301-435-3384
Email: emily.erbelding@nih.gov
Division Small Business Representative:
Dr. Barbara Mulach
Telephone: 240-627-3322
Email: bmulach@niaid.nih.gov
Division Small Business Representative:
Dr. Alyssa Werner
Telephone: 301-761-7525
Email: alyssa.werner@nih.gov
Bacteriology and Mycology Branch (BMB)
Contact: Dr. Alec Ritchie
Telephone: 240-627-3356
Email: aritchie@niaid.nih.gov
Enteric and Sexually Transmitted Infections Branch (ESTIB)
Contact: Dr. Tom Hiltke
Telephone: 240-627-3275
Email: thiltke@niaid.nih.gov
Parasitology and International Programs Branch (PIPB)
Contact: Dr. Annie Mo
Telephone: 240-627-3320
Email: moa@niaid.nih.gov
Respiratory Diseases Branch (RDB)
Contact: Dr. Xin-Xing Gu
Telephone: 240-627-3265
Email: guxx@niaid.nih.gov
Virology Branch (VB)
Contact: Dr. Julie Dyall
Telephone: 301-631-7296
Email: Julie.dyall@niaid.nih.gov
Office of Genomics and Advanced Technologies (OGAT)
Contact: Dr. Inka Sastalla
Telephone: 301-761-6431
Email: sastallai@mail.nih.gov
Office of Biodefense Research and Surety (OBRS)
Biodefense Research Countermeasures Branch (BRCB)
Contact: Dr. Dave Yeung
Telephone: 301-761-7237 Email: dy[email protected]ov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAMS 33
NATIONAL INSTITUTE OF ARTHRITIS AND MUSCULOSKELETAL AND SKIN
DISEASES (NIAMS)
Mission
The mission of the National Institute of Arthritis and Musculoskeletal and Skin Diseases is to
support research into the causes, treatment, and prevention of arthritis and musculoskeletal and
skin diseases, the training of basic and clinical scientists to carry out this research, and the
dissemination of information on research progress in these diseases.
For additional information about areas of interest to NIAMS, please visit the NIAMS Strategic
Plan at https://www.niams.nih.gov/about-niams/strategic-plan-fiscal-years-2020-2024.
Budget Guidance
For budgetary, administrative, or programmatic reasons, NIAMS may decide not to fund an
application or may decrease the length of an award and/or the budget recommended by a review
committee. Total funding support (direct costs, indirect costs, fees) normally may not exceed the
amounts defined by the SBA, which can be found on the NIH SEED website.
For topics listed in the SBA-Approved Waiver Topics, the NIAMS does not apply these topics to the
Omnibus Program Announcements. The NIAMS only applies the waiver topics to special Notices of
Funding Opportunities that specifically allow higher budgets than those in the Omnibus Program
Announcements. When the waiver topics are applied, NIAMS generally will not fund Phase I
applications greater than $350,000 total costs or project periods greater than 2 years; or Phase II
applications greater than $2,300,000 total costs or project periods greater than 3 years. Applicants
considering a requested budget greater than these limits are strongly encouraged to contact
program staff before submitting an application.
NIAMS provides Technical and Business Assistance (TABA) Funding. Small Businesses may
request up to $6,500 per year for a Phase I and up to $50,000 per Phase II project (across all
years) to support subcontracts or consultants above the budget cap. Small businesses should
include this budget request as part of the application and provide a detailed budget justification.
Specific SBIR and STTR Program Information
NIAMS does not participate in the SBIR/STTR clinical trial funding opportunities. NIAMS NON-
SBIR/STTR clinical trial funding opportunities support all research within the NIAMS mission areas.
It is not the intent of NIAMS to support clinical trials through the SBIR/STTR mechanism.
Applicants who wish to submit clinical trials applications to the NIAMS are encouraged to utilize
one of the NIAMS NOFOs listed at https://www.niams.nih.gov/grants-funding/conducting-clinical-
research/investigator- clinical-trial- policies.
Specific Funding Opportunities and Programs
NIAMS has published the SBIR funding opportunity PAR-23-032 promote the
translation of academic/non-profit lab research results to marketplace.
In addition, the NIAMS participates in funding opportunities for the SBIR/STTR HEAL
initiative, the Administrative Supplements to Promote Diversity in Research and
Development, and the Small Business

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAMS 34
Initiatives for Innovative Diagnostic Technology for Improving Outcomes for Maternal Health. For
currently active Notices of Funding Opportunities, please contact the program staff.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NIAMS does not accept Phase IIB renewal applications. NIAMS participates in the CRP program
and sets its own budget limits for the CRP applications in the program announcements. NIAMS
does not support clinical trials through the CRP program.
Clinical Trials
Does NIAMS accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
No
Does NIAMS accept Clinical Trials
through specific Notices of Funding
Opportunities?
No
Does NIAMS support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
https://www.niams.nih.gov/grants-
funding/conducting-clinical-
research/investigator-clinical-trial-policies
Research Topics
The NIAMS small business program supports research and development of products and
services for prevention, diagnosis and treatment of rheumatic, musculoskeletal and skin
diseases. The research topics include, but are not limited to, the following:
A.
Rheumatic Diseases. The NIAMS supports research on rheumatic and related
diseases including rheumatoid arthritis (RA), juvenile idiopathic arthritis (JIA), Lyme
arthritis, viral arthritis, gout, calcium pyrophosphate deposition disease (CPDD),
spondyloarthropathies, and systemic autoimmune diseases such as systemic lupus
erythematosus (SLE), systemic scleroderma (SSc), and autoimmune myositis.
B.
Musculoskeletal Diseases. The musculoskeletal system is composed of the skeleton,
the muscles, and connective tissues such as cartilage, tendon, and ligament. The
NIAMS supports research aimed at improving the diagnosis, treatment, and prevention
of diseases and injuries of the musculoskeletal system and its component tissues. The
topics in this area include research on musculoskeletal diseases such as osteoporosis,
osteoarthritis, muscular dystrophy, and osteogenesis imperfecta, tissue engineered
products, orthopedic devices and implants, and sports medicine and fitness.
C.
Skin Diseases. The NIAMS supports research on a wide range of skin diseases and
conditions including chronic inflammatory skin diseases such as psoriasis, rosacea,
acne vulgaris, and atopic dermatitis and autoimmune diseases such as pemphigus,
vitiligo, and alopecia areata. The NIAMS also supports research on skin repair and
regeneration in treatment of chronic wounds and reducing scar formation. Skin cancer is
an area of overlap with the National Cancer Institute (NCI), with the NIAMS focus on the
response of keratinocytes to UV light and early stages in the development of non-
melanoma skin cancer and products for prevention of melanocyte tumorigenesis.
This is not an inclusive list of all research topics covered by the NIAMS. To learn more, please
visit the NIAMS supported scientific areas at https://www.niams.nih.gov/grants-
funding/funding- opportunities/supported-scientific-areas

NIH, CDC, and FDA Program Descriptions and Research Topics
NIAMS 35
Research Topics of High Program Priority
NIAMS supports all Research and Development activities within its mission. Particular areas of
programmatic interest relative to small business initiatives include, but are not limited to:
A.
Innovative research on women’s health in the areas of musculoskeletal,
rheumatic and skin diseases
B.
Innovative research on health disparity in the areas of musculoskeletal,
rheumatic and skin diseases
C.
Innovative diagnostic technology for improving outcomes for maternal health
in NIAMS mission areas
D.
Innovative research on rare musculoskeletal, rheumatic and skin diseases
E.
Multiplex assay development for arthritis and musculoskeletal and skin diseases
F.
Lab to marketplace: translation of scientific discoveries in NIAMS mission
areas from labs into products on the market
G.
Test and/or validation of novel, state-of-the-art candidate biomarker platforms
for predicting the onset and progression of inflammatory diseases of interest to
the NIAMS and for determining the pharmacodynamics, safety and/or efficacy
of therapeutic agents targeting those diseases.
Research Topics of Lower Program Priority
The general purpose of the SBIR/STTR program is to stimulate technological innovation and increase
private sector commercialization of Innovations. Due to budget constraints, NIAMS will consider the
following research topics a lower program priority:
•
Research on a product or a technology to show equivalence to existing products
•
A product or a technology has been well funded for more than 10 years, but has not
shown any progress towards clinical testing
•
A research topic on which multiple similar technologies have been funded and
have shown scientific success
Contact Information
For general SBIR/STTR program information, contact: Dr.
Xibin Wang, NIAMS SBIR/STTR Coordinator Telephone:
301-451-3884
Fax: 301-480-1284
Email: wangx1@mail.nih.gov
For administrative and business management questions, contact:
Ms. Victoria Matthews
Telephone: 301-594-3968
Fax: 301-480-5450
Email: victoria.matthews@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIBIB 36
NATIONAL INSTITUTE OF BIOMEDICAL IMAGING AND BIOENGINEERING
(NIBIB)
Mission
The mission of the National Institute of Biomedical Imaging and Bioengineering (NIBIB) is to transform
through technology development, our understanding of disease and its prevention, detection,
diagnosis, and treatment. NIBIB supports new tools and technologies to improve human health within
its internal laboratories and through grants, collaborations, and training.
Budget Guidance
• Normally, total funding support (direct costs, indirect costs, fees) may not exceed the amounts
defined by the Small Business Administration (SBA), which can be found on the NIH SEED
website.
• Applicants considering a requested budget greater than the current SBA guidelines should ensure
that the project falls within a topic listed in the SBA-Approved Waiver Topics.
• Generally, NIBIB will not support Phase I (R41/R43) project periods greater than 1 year, or Phase
II (R42/R44) project periods greater than 2 years.
• For budgetary, administrative, or programmatic reasons, NIBIB may decide not to fund an
application or may decrease the length of an award and/or the budget recommended by a review
committee.
Specific SBIR and STTR Program Information
•
NIBIB does not typically support Phase II STTRs. Applicants interested in submitting a Phase II
application should consider the SBIR program and contact program staff at least one month before
the application deadline.
•
NIBIB does not accept Phase IIB renewal applications through the omnibus solicitations.
•
NIBIB will accept applications for support of early-stage clinical trials (see NOT-21-005) in any of
the scientific program areas within NIBIB’s mission.
Applicants are strongly encouraged to contact program staff at NIBIB-SBIR@mail.nih.gov at least
one month before submitting an application.
Specific Funding Opportunities and Programs
•
NIBIB Concept to Clinic: Commercializing Innovation (C3i) Program:
The C3i Program is designed to provide medical device innovators with specialized business
frameworks and essential tools for successful translation of biomedical technologies from the lab
(concept) to the market (clinic). Through this program, the NIH fosters the development and
commercialization of early-stage biomedical technologies by engaging investigators who are
interested in better understanding the value of their innovation in addressing an unmet market
need. The curriculum and customized mentoring provided by the C3i Program are intended to
guide investigators as they assess the commercial viability and potential business opportunity for
their innovation.
•
Targeted Funding Opportunities and Notices of Special of Interest can be reviewed on the NIBIB
website.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
•
NIBIB does not accept Phase IIB renewal application through the omnibus solicitations.
•
NIBIB participates in the Commercialization Readiness Pilot (CRP) program for
Phase II applications only.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIBIB 37
Clinical Trials
Does NIBIB accept Clinical Trials through
the Omnibus/Parent Notices of Funding
Opportunities?
Yes
See NOT-21-005 for NIBIB Clinical Trial Funding
Information
Does NIBIB accept Clinical Trials through
specific Notices of Funding Opportunities?
Yes
See NOT-21-005 for NIBIB Clinical Trial Funding
Information
Does NIBIB support Clinical Trials
through Non-SBIR/STTR Notices of
Funding Opportunities?
Yes
See NOT-21-005 for NIBIB Clinical Trial Funding
Information
Research Topics
A.
Bio-Electromagnetic Technologies. Development of technologies that use static or dynamic
electromagnetic fields for sensing, imaging, or therapeutic effects. The emphasis is on increasing
the sensitivity, spatial/temporal resolution, efficacy, or safety of bioelectromagnetic devices
through the development of novel hardware, method of operation, or pre-/post-processing
techniques for single modalities or the combination of multiple modalities. This program may
support the development of magnetic particle imaging, electrical impedance tomography,
electroencephalography, magnetoencephalography, electromagnetic-field-induced
hyperthermia/ablation, and microwave/terahertz imaging, for example.
B.
Bioanalytical Sensors. Development of sensor technologies for the detection and quantitation of
clinically relevant analytes in complex matrices for use in biomedical applications. Emphasis is on
engineering the components and functionality of bioanalytical sensors. Detection could be based on
optical, chemical, electrochemical, and/or physical (such as mechanical, gravimetric, thermal)
perturbation of a sample, for example. Examples of technologies of interest include, but are not
limited to, nano-textured substrates for analyte detection, DNA sensors for liquid biopsy, and small
molecule detectors for diagnosing infectious diseases.
C.
Image-Guided Interventions. Development of novel image-directed technologies for guidance,
navigation, tissue differentiation, and disease identification for reaching specified targets during
therapeutic procedures, which may range along the continuum from non-invasive to minimally
invasive to open surgical interventions. These technologies may range from molecular to macroscopic
scale levels. Overall emphasis is on the engineering of novel image-guided interventions to
improve outcomes of interventional procedures. In addition, emphasis includes technologies that
expand needed procedural access for individuals otherwise excluded by disease characteristics,
co-morbidities, and other parameters. Areas of priority include development of real-time or near
real-time novel image-guided technologies, with robust procedural direction or a robust receiver
operating characteristic curve. In addition, cost-efficient technologies, appropriate for low resource
settings, and/ or applicable to multiple types of interventions are strongly encouraged.
D.
Magnetic Resonance Imaging. Development of in vivo MR imaging and MR spectroscopy, for both
animal and human research and potential clinical applications. The emphasis is on the

NIH, CDC, and FDA Program Descriptions and Research Topics
NIBIB 38
development of MRI hardware and methodologies, including image acquisition and reconstruction
techniques, that would improve the speed, spatial resolution, information content, efficiency,
robustness, quality, patient experience, and safety. The emphasis should be on technological
development rather than detailed applications to specific diseases or organs.
E.
Molecular Probes and Imaging Agents. Development and biomedical application of molecular
probes and imaging agents across all imaging modalities for the visualization, characterization and
quantification of normal biological and pathophysiological processes and anatomy in living organisms at
the molecular, cellular and organ levels. The emphasis is on engineering of targeting and responsive
molecular probes of high sensitivity and specificity for PET and SPECT (radiotracers), MR (T1, T2,
CEST, hyperpolarized agents), EPR, CT, optical (fluorescent and bioluminescent probes), ultrasound
(microbubbles) and photoacoustic imaging. The imaging agents may be based on nano- and micro-
particles, liposomes, dendrimers, proteins, small organic and inorganic molecules etc., and
detectable by one or more imaging modalities. Imaging agent development through methodologies
such as chemical synthesis, biological mutagenesis, microfabrication, etc., may be pursued with an
intent of leading to in vivo biomedical application.
F.
Nuclear Medicine. Research and development of technologies and techniques that create images
out of the gamma- ray (SPECT) or positron (PET) emissions from radioactive agents that are
injected, inhaled, or ingested into the body. The emphasis is on simulation and development of
new detectors, collimators, and readout methods that enhance the signal quality of detecting
isotope emissions; designs of novel camera geometries; and correction methods that compensate
for the radiation physics properties to improve the clinical reliability of the image. Of interest are
improvements and corrections for interaction events in PET detectors and enhancement to time of
flight (TOF) image generation methods (reconstructions algorithms); as well as new collimator and
camera designs for SPECT.
G.
Optical Imaging and Spectroscopy. Development and application of optical imaging, microscopy,
and spectroscopy techniques for improving disease prevention, diagnosis, and treatment in the
medical office, at the bedside, or in the operating room. Examples of research areas include
fluorescence imaging, bioluminescence imaging, OCT, SHG, IR imaging, diffuse optical
tomography, optical microscopy and spectroscopy, confocal microscopy, and multiphoton
microscopy. The emphasis is on development of cost effective, portable, safe, and non-invasive or
minimally invasive devices, systems, and technologies for early detection, diagnosis, and treatment
for a range of diseases and health conditions.
H.
Ultrasound: Diagnostic and Interventional. Development and improvement of technologies for
diagnostic or therapeutic uses of ultrasound. The diagnostic ultrasound program includes, but is not
limited to the design, development and construction of transducers, transducer arrays, and transducer
materials, innovative image acquisition and display methods, innovative signal processing methods
and devices, and optoacoustic and thermoacoustic technology. It also includes the development of
image-enhancement devices and methods, such as contrast agents, image and data presentation and
mapping methods, such as functional imaging and image fusion. The therapeutic ultrasound program
includes, but is not limited to the design, development, and construction of transducers, transducer
arrays, interventional technologies, adjunct enhancement of non-ultrasound therapy applications,
high-intensity focused ultrasound (HIFU), or hyperthermia applications. It also includes non-
invasive or minimally invasive interventional surgical or therapy tools, ultrasound contrast agents
for therapy, targeted drug delivery, neuromodulation, and biopsy.
I.
X-ray, Electron, and Ion Beam. Research and development of technologies and techniques that
create images of internal structures, contrast agents, or molecular probes using x-rays
transmitted through the body (CT, mammography) or x-ray stimulation of secondary emissions
(x-ray fluorescence tomography). Emphasis is on simulation, design and development of new
detector systems; new readout methods that enhance the signal quality for x-ray image
generation; designs of novel imaging geometries; algorithms that compensate for the physical
properties of the detection system to improve the clinical reliability of the image (reconstruction

NIH, CDC, and FDA Program Descriptions and Research Topics
NIBIB 39
algorithms); and approaches to radiation dose reduction, especially in CT. Of interest are
diagnostic image enhancements via photon counting, dual energy, and new applications of cone-
beam tomography.
J.
Biomolecular Technologies. Development and demonstration of broadly applicable biomolecular
technologies to enable new paradigms of human health. The emphasis is on the development of
biomolecular technologies and associated computational models for biomedical intervention. NIBIB
interests include but are not limited to: molecular switches for synthetic genetic circuits; nucleases
and genome editors for DNA manipulation and regulation; engineered viruses and extracellular
vesicles for therapeutic agent delivery; transmembrane CARs for extracellular sensing; photoactive
molecular complexes for optogenetics.
K.
Bionics. Development and demonstration of broadly applicable bionic systems to enable new
paradigms of human health. The emphasis is on the development of bionic systems hardware,
software, and methodologies to improve patient health. NIBIB interests include but are not limited
to: artificial organs to replace function; electrodes and 3D printed tactile sensors for prosthetics;
implantable bioelectronic sensors and actuators for real-time, closed-loop control of tissues and
organs.
L.
Cellular and Multicellular Technologies. Development and demonstration of broadly applicable
cellular and multicellular technologies to enable new paradigms of human health. The emphasis is
on the development of cellular and multicellular technologies and associated computational models
for biomedical intervention. NIBIB interests include but are not limited to: synthetic genetic circuits
for cellular control and decision-making; engineered bacteria for microbiome regulation;
engineered T-cells for immune regulation and cancer therapy; organoids and scaffold-free tissue
assemblies for replacing organ function.
M.
Living Materials. Development and demonstration of broadly applicable living materials to enable
new paradigms of human health. The emphasis is on the development of living materials and
associated computational models for biomedical intervention. NIBIB interests include but are not
limited to: bacteria-laden hydrogels to deliver therapeutics; co-designed stem cells and scaffolds to
grow implantable tissues.
N.
Manufacturing and Biomanufacturing Tools. Development and demonstration of broadly applicable
manufacturing and biomanufacturing tools to enable the translation of new paradigms of human
health. The emphasis is on the development of manufacturing and biomanufacturing tools and
associated computational models to enable biomedical interventions. NIBIB interests include but
are not limited to: bioinks and bioprinters for 3D tissue construction; continuous production
methods for scalable manufacturing of drug delivery vehicles; inline sensors for non-destructive
evaluation of manufactured therapeutic cells; bioreactors for organoid manufacturing.
O.
Medical Devices. Development and demonstration of broadly applicable biomedical devices to
enable new paradigms of human health. The emphasis is on the development of medical device
hardware, software, and models to improve patient health. NIBIB interests include but are not
limited to: implantable bioelectronic stimulators and sensors for monitoring and modulating human
physiology; wearable sensors for monitoring health vitals; micro devices and injection systems for
therapeutic delivery; anti-bacterial and anti-coagulating coatings for implantable devices; biohybrid
devices for replacing organ function.
P.
Medical Simulators. Development and demonstration of broadly applicable medical simulators to
enable new paradigms of human health. The emphasis is on the development of medical simulator
hardware, software, and methodologies, primarily to improve patient outcomes, especially through
the reduction of medical errors. NIBIB interests include but are not limited to: virtual coaches
incorporating artificial intelligence for performance training in medical procedures and workflows;
simulation interfaces to facilitate dissemination and use of virtual environments; realistic
representations of anatomy, tissue, instrument, tactile feedback, and collision dynamics; simulator

NIH, CDC, and FDA Program Descriptions and Research Topics
NIBIB 40
designs that focus on complicated or rare procedures, including rare adverse events; simulators
that replicate realistic workflows, including planning, warm-up exercises, and rehearsal leading up
to the actual procedure; portable, easy-to-use simulators for skilled practitioners in rural and low-
resource settings.
Q.
Molecular Materials. Development and demonstration of broadly applicable molecular materials to
enable new paradigms of human health. The emphasis is on the development of molecular
materials and associated computational models for biomedical intervention. NIBIB interests include
but are not limited to: lipid nanoparticle coatings for evading the immune system; supramolecular
polymers for targeted protein degradation; drug conjugates for targeted drug delivery.
R.
Nanomaterials. Development and demonstration of broadly applicable nanomaterials to enable
new paradigms of human health. The emphasis is on the development of nanomaterials and
associated computational models for biomedical intervention. NIBIB interests include but are not
limited to: magnetic and acoustic nanoparticles for ablating cells and tissues; plasmonic nanorods
for tissue suturing; functionalized nanocarriers for drug delivery and immunotherapy.
S.
Physiomimetic Materials. Development and demonstration of broadly applicable physiomimetic
materials to enable new paradigms of human health. The emphasis is on the development of
physiomimetic materials and associated computational models for biomedical intervention. Projects
might focus on: elucidating important engineering design rules or key foundational principles
underlying future engineering, including the use of computational methods; prototyping or
redesigning platform technologies; characterizing (in vitro, ex vivo, or in vivo) broadly applicable
technologies and prototypes. NIBIB interests include but are not limited to: electrically conductive
and mechano-sensitive scaffolds for repairing tissue; photoactive adhesives for surgical sealants;
biomimetic matrices for T cell activation; artificial cells for therapeutic agent delivery.
T.
Robotics. Development and demonstration of broadly applicable robotic systems to enable new
paradigms of human health. The emphasis is on the development of robotic systems hardware,
software, and methodologies to improve patient health. NIBIB interests include but are not limited
to: robots for minimally invasive surgeries; microgrippers and drills for surgical robots; robotic
nurses for isolated patient care; soft robotic exoskeletons to replace lost capabilities; soft
elastomeric actuators for assistive robotics.
U.
Screening and High-Throughput Tools. The emphasis is on the development of screening and
high-throughput tools and associated computational models to enable biomedical
interventions. Projects might focus on: elucidating important engineering design rules or key
foundational principles underlying future engineering, including the use of computational methods;
prototyping or redesigning platform technologies and approaches; characterizing (in vitro, ex vivo,
or in vivo) broadly applicable technologies, prototypes, and lead candidate products. NIBIB
interests include but are not limited to: evolution methods for identifying therapeutic protein targets;
organs-on-chips for drug screening; microfluidic systems for high-throughput screening of
extracellular vesicles.
V.
Artificial Intelligence, Machine Learning, and Deep Learning. Design and development of artificial
intelligence, machine learning, and deep learning to enhance analysis of complex medical images
and data. The emphasis is on development of transformative machine intelligence-based systems,
emerging tools, and modern technologies for diagnosing and recommending treatments for a range
of diseases and health conditions. Unsupervised and semi-supervised techniques and
methodologies are of particular interest.
W.
Biomedical Informatics. Development of structures and algorithms to improve the collection,
annotation, aggregation, anonymization, classification, retrieval, integration, analysis, and
dissemination of quantitative and qualitative biomedical data. The emphasis is on using biomedical
information to achieve better health outcomes and smarter health care. Examples of technical
development areas in this program include but are not limited to informatics tools and resources

NIH, CDC, and FDA Program Descriptions and Research Topics
NIBIB 41
such as: databases, standards for enhanced interoperability, collaborative analysis environments,
data modeling and representation, and techniques for the integration of heterogeneous data, rational
data-driven design of experiments, visualization of data, and digital representation of rich qualitative
data. This program is intended to support NIBIB’s other program areas in biomedical imaging and
bioengineering research.
X.
Digital Health-Mobile Health and Telehealth. Development of enabling technologies that emphasize the
integration of wireless technologies with human and biological interfaces. This program includes the
development of software and hardware for telehealth and mobile health studies. This program includes
the development of software and hardware for telehealth and mobile health studies and the input
and delivery of healthcare information digitally for the analysis or monitoring of health or disease
status. The emphasis is on developing mobile health technologies driven by clinical needs and
integrating these technologies in healthcare delivery, wellness, and daily living.
Y.
Point of Care Technologies-Diagnostics. Development of rapid in-vitro diagnostic technologies and
monitoring platforms that provide real time medical evaluation and analysis of the disease status or
condition at the time and place of patient care. The program includes the delivery of healthcare that
is safe, effective, timely, patient-centered, efficient, and available in centralized and decentralized
locations. The emphasis is on developing technologies driven by clinical needs. Examples of
technology development areas in this program include but are not limited to disposable lateral flow
assays, nucleic acid testing platforms, glucose monitoring devices, etc.
Z.
Image Processing, Visual Perception, and Display. Design and development of algorithms for post-
acquisition image processing and analysis, the development of theoretical models and analysis tools
to evaluate and improve the perception of medical images, and the development of visualization
tools for improved detection. The emphasis is on using image data to achieve better health
outcomes and smarter health care. Examples of technology development areas in this program
include but are not limited to models, algorithms, software, methodologies, and other tools that will:
facilitate medical imaging research; support clinical detection, diagnosis and therapy; and improve
patient healthcare.
Contact Information
For additional information on research topics, contact:
Dr. Kari Ashmont
National Institute of Biomedical Imaging and Bioengineering
Telephone: 301-451-4772, Fax: 301-480-1614
Email: NIBIB-SBIR@mail.nih.gov
For administrative and business management questions, contact:
Mr. James Huff
National Institute of Biomedical Imaging and Bioengineering
Telephone: 301-451-4786, Fax: 301-451-5735
Email: huffj@mail.nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NCI 42
NATIONAL CANCER INSTITUTE (NCI)
Mission
The National Cancer Institute’s SBIR Development Center program is one of the nation’s largest
sources of financing for small businesses engaged in technology innovation. NCI’s funding,
mentoring, and networking assistance is offered to small businesses demonstrating promising
next-generation cancer cure technologies, with the ultimate goal being successful
commercialization and benefiting public health. NCI’s SBIR/STTR Programs offer funding for
therapeutic agents and devices; in vitro and in vivo diagnostics, including companion diagnostics
and imaging agents; agents and technologies for cancer prevention; tools for research in cancer
biology, cancer control, and epidemiology; digital health, including health information technology
and bioinformatics; and many more areas of interest to the NCI.
The goal of NCI’s SBIR/STTR program is to increase small business participation and private-
sector commercialization of novel technologies that can prevent, diagnose, and treat cancer.
The major NCI SBIR/STTR portfolio areas are listed below as a guide to general technology
areas funded through the program. However, NCI will accept any applications outside these
topic areas, and proposing innovative cancer-related technologies with strong commercial
potential is encouraged.
Budget Guidance
For budgetary, administrative, or programmatic reasons, NCI may decide not to fund an
application or may decrease the length of an award and/or the budget recommended by a review
committee. Total funding support (direct costs, indirect costs, fees) normally may not exceed the
amounts defined by the SBA, which can be found on the NIH SEED website. However, for certain
research topics, the U.S. Small Business Administration has approved a list of SBA-Approved
Waiver Topics for which the NCI generally will fund Phase I applications with higher budgets up to
$400,000 total costs combined over all years, and project periods up to 2 years; similarly, for
certain research topics, NCI will consider Phase II applications with higher budgets up to
$2,250,000 total costs combined over all years, and project periods up to 3 years.
Specific SBIR and STTR Program Information
NCI SBIR program is technology agnostic. We welcome all innovative solutions with
commercial potential that is relevant to the mission of the NCI and that reduces the burden on
cancer patients, their caregivers, and providers.
For up-to-date information on high-priority technology areas, and to learn about
programmatic initiatives and upcoming events, visit the NCI SBIR Development Center
homepage: http://sbir.cancer.gov.
In addition, please see the contact list at the end of the NCI section to identify the NCI SBIR/STTR
Program Director(s) that specializes in your technology area.
NCI will accept applications for support of clinical trials in the NCI priority area
mentioned above or any other areas that are relevant to the NCI’s mission.
Prior to Submission
Applicants are strongly encouraged to contact SBIR/STTR staff prior to submitting any

NIH, CDC, and FDA Program Descriptions and Research Topics
NCI 43
application. To schedule a meeting, please email ncisb[email protected]ov with a copy of your
specific aims page that includes answers to the following questions:
•
What is your product?
•
What would be the impact of your technology on cancer patients, providers, or
caregivers?
•
How is your product innovative and how is it different from the current standard?
•
What are your aims for the application? What will be your milestones or success
criteria?
•
Who is the end-user of your product? Who is the purchaser?
For NCI-related SBIR Information, visit http://sbir.cancer.gov.
Specific Funding Opportunities and Programs
1.
Commercialization Readiness Pilot (CRP) and Phase IIB Competing Renewal
Awards CRP
The National Cancer Institute (NCI) welcomes CRP applications from companies that have active
or completed NCI SBIR (Phase II and Phase IIB) or STTR (Phase II) awards (PAR-20-128).
Please note that NCI will provide funding support up to a maximum of $250,000 in total costs
(direct costs, indirect costs, and fee). Potential applicants are strongly encouraged to contact NCI
SBIR Program staff to discuss prior to submission the CRP activities for which support will be
requested. The NCI may decline funding of any application for reasons of program balance or
budget. NCI does not participate in the SBIR/STTR Commercialization Readiness Pilot Program
Technical Assistance and Late-Stage Development (SB1) NOFOs (PAR-20-129 or PAR-20-130).
For SBIR/STTR Phase II awardees in need of additional assistance for late-stage development
and/or clinical trials, the NCI recommends its SBIR Phase IIB program.
Phase IIB
The NCI does not accept applications for Phase IIB SBIR competing renewal awards through this
Omnibus solicitation. However, the NCI offers Phase IIB opportunities in the form of the NCI SBIR
Phase IIB Bridge Award, which is announced via a separate funding solicitation:
https://sbir.cancer.gov/bridge. Please note that NCI will provide funding support up to a maximum
of $4,500,000 in total costs (direct costs, indirect costs, and fee). The NCI Phase IIB Bridge Award
is designed to support the next stage of development for cancer-related technologies previously
funded under SBIR or STTR Phase II awards from any Federal agency. The purpose of this award
is to address the funding gap known as the "Valley of Death" between the end of the SBIR Phase
II award and the subsequent round of financing needed to advance a product or service toward
commercialization. To achieve this goal, the Bridge Award funding opportunity is specifically
designed to incentivize partnerships between federally funded SBIR Phase II awardees and third-
party investors and/or strategic partners. Competitive preference and funding priority will be given
to applicants that demonstrate the ability to secure substantial independent third-party investor
funds (i.e., third-party funds that equal or exceed the requested NCI funds).
To ensure that you will be notified upon the release of the NCI SBIR Phase IIB Bridge Award
solicitation, please sign up for the NCI SBIR mailing list: https://bit.ly/NciSbirEmailSignUp. If you
have any questions regarding the NCI SBIR Phase IIB Bridge Award, please contact your Phase
II program director.
NIH, CDC, and FDA Program Descriptions and Research Topics
NCI 44
2.
Technical and Business Assistance (TABA) Funding
NCI will consider well-justified Technical and Business Assistance (TABA) costs up to the
limits specified on the NIH SEED Website. These costs can be requested in addition to the Phase
I and II budget caps set by NCI. TABA costs must be requested under the original application. NCI
will not fund TABA costs as a supplement and does not participate in the administrative
supplement program for TABA (NOT-OD-21-062).
Clinical Trials
Applicants are strongly encouraged to contact SBIR/STTR staff prior to submitting any application
containing clinical trials.
Does NCI accept Clinical Trials through
the Omnibus/Parent Notices of
Funding Opportunities?
Yes
Does NCI accept Clinical Trials through
specific Notices of Funding
Opportunities?
Yes
https://seed.nih.gov/small-business-
funding/find-funding/sbir-sttr-funding-
opportunities
Does NCI support Clinical Trials through
NON-SBIR/STTR Notices of Funding
Opportunities?
Yes
R21, R01, P01, K08
https://www.cancer.gov/grants-training/grants-
funding/funding-opportunities
Research Topics
NCI will accept applications in any of the NCI priority areas mentioned below or any other areas
that are relevant to NCI’s mission. NCI is technology agnostic and will fund any technology as
long as it benefits cancer patients, providers, or caregivers. Applications proposing innovative
cancer-related technologies, with strong commercial potential, that fall outside these topic areas
are also welcomed through the Omnibus Solicitation. NCI accepts and encourages SBIR & STTR
applications to support clinical trials.
Major NCI SBIR/STTR Portfolio Areas:
•
Therapeutics (e.g., Small Molecules, Biologics, Radiomodulators, and Cell-based Therapies)
•
In Vitro and In Vivo Diagnostics (e.g., Companion Diagnostics and Prognostic Technologies)
•
Imaging Technologies (e.g., Agents, Devices, and Image-Guided Interventions)
•
Devices for Cancer Therapy (e.g., Interventional Devices, Surgical, and
Radiation and Ablative Therapies, Hospital Devices)
•
Agents and Technologies for Cancer Prevention
•
Technologies for Cancer Control (e.g., Behavioral Health Interventions,
Tools for Genetic, Epidemiologic, Behavioral, Social, and/or Surveillance
Cancer Research)
•
Tools for Cancer Biology Research
•
Digital Health Tools and Software Platforms for Cancer-Related Technologies
Diversity Statement from NCI SBIR
The statutory purpose of the SBIR program is to strengthen the role of innovative SBCs in Federally

NIH, CDC, and FDA Program Descriptions and Research Topics
NCI 45
funded research or research and development (R/R&D) (See the Policy Directive). Specific
program purposes are to: (1) stimulate technological innovation; (2) use small business to meet
Federal R/R&D needs; (3) foster and encourage participation by socially and economically
disadvantaged SBCs (SDBs), and by women-owned SBCs (WOSBs), in technological innovation;
and (4) increase private sector commercialization of innovations derived from Federal R/R&D,
thereby increasing competition, productivity, and economic growth. To understand more about
how the NCI SBIR/STTR program fosters and encourages participation by WOSBs and SDBs,
please check the following link: https://sbir.cancer.gov/diversity
Contact Information
For additional information about the NCI SBIR/STTR programs, please contact the NCI SBIR
Development Center:
National Cancer Institute
Small Business Innovation Research (SBIR) Development Center
9609 Medical Center Drive
Rockville, MD 20850
Website: http://sbir.cancer.gov
Email: NCIsbir@mail.nih.gov
Phone: 240-276-5300
For additional information on research topics, please contact a Program Officer with the relevant
area of expertise:
Michael Weingarten, MA
Director, NCI SBIR Development Center
Email: weingartenm@mail.nih.gov
Gregory Evans, PhD
Program Director and Team Leader
Email: evansgl@mail.nih.gov
Areas of Expertise: Therapeutics (Immunotherapy, Gene Therapy), Cancer Imaging, Cancer Control,
Tools for Cancer Biology Research, and Digital Health
Jonathan Franca-Koh PhD, MBA
Program Director and Team Leader
Email: jonathan.franca-koh@nih.gov
Areas of Expertise: Cancer Biology, Biologics, Small Molecules, and Cell-Based Therapies
Monique Pond, PhD
Program Director and Team Leader
Email: monique.pond@nih.gov
Areas of Expertise: Biologics, Small Molecules, Therapeutic Devices, Digital Health, Regulatory
Resources
Patricia Weber, DrPH
Program Director
Email: weberp[email protected]ov
Areas of Expertise: Digital Health and Therapeutics (Small Molecules, Biologics, Immunotherapy)
Xing-Jian Lou, PhD
Program Director
Email: loux@mail.nih.gov
Areas of Expertise: In Vitro Diagnostics and Therapeutics (Gene Therapy, Biologics, Small Molecules)

NIH, CDC, and FDA Program Descriptions and Research Topics
NCI 46
Ming Zhao, PhD
Program Director
Email: zhaoming3@mail.nih.gov
Areas of Expertise: In Vitro Diagnostics, Cancer Stem Cells, Molecular Imaging, Bioinformatics,
Therapeutics (Small Molecules, Biologics, Immunotherapy), and Cancer Control (Community -Based
Participatory Research)
William Bozza, PhD
Program Director
Email: william.bozza@nih.gov
Areas of Expertise: Biologics, Protein Therapeutics, Regulatory (CMC)
Sarra Djemil, PhD
Program Director
Email: sarra.d[email protected]ov
Areas of Expertise: Therapeutics, Digital Health, Mentoring
Saroj Regmi, PhD
Program Director
Email: saroj.reg[email protected]ov
Areas of Expertise: Therapeutics, Diagnostics, Imaging, Digital Health, Investor Initiatives, Small Business
Transition Grant, I-Corps
Swamy Tripurani, PhD
Program Director
Email: swamy.tripurani@nih.gov
Areas of Expertise: Therapeutics, Biologics, Small Molecules, Diagnostics, Devices, and Regulatory
(CMC and Nonclinical)
Melissa Li, PhD
Program Director
Areas of Expertise: Biologics, Small Molecules, Digital Health
Linda Zane, PhD
Program Director
Email: linda.zane@nih.gov
Areas of Expertise: Therapeutics, Diagnostics, Research Tools

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 47
EUNICE KENNEDY SHRIVER NATIONAL INSTITUTE OF CHILD HEALTH AND
HUMAN DEVELOPMENT (NICHD)
Mission
The mission of the NICHD is to lead research and training to understand human development, improve
reproductive health, enhance the lives of children and adolescents, and optimize abilities for all.
The NICHD has a broad and diverse research portfolio, including biological, behavioral, and clinical
research related to conception and pregnancy, normal and abnormal development in childhood,
reproductive health, rehabilitation, and population dynamics across the lifespan.
For up-to-date information on priority research areas of scientific interest to the NICHD, please visit
our home page at http://www.nichd.nih.gov.
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SEED website. It is strongly encouraged to
contact program staff prior to applying.
The NICHD received a budgetary guideline waiver from the Small Business Administration for
applications relating to a limited list of scientific topics in SBA-Approved Waiver Topics. For these the
NICHD will accept applications up to $400K total costs for Phase I and $2.25M for Phase II.
Applicants should propose a budget that is reasonable and appropriate for completion of the
research project and requests for these budge levels must be very well justified. Applicants are
encouraged to contact the listed NICHD Branch Contact Program Officer for scientific-related
questions about a project’s eligibility for a budgetary waiver.
For general budgetary questions, applicants should contact NICHD’s SBIR/STTR Grants
Management Coordinator. The NICHD may decrease the budget or length of an award or
decide not to fund an application for budgetary, administrative, or programmatic reasons.
Specific SBIR and STTR Program Information
The NICHD will generally consider clinical trial proposals consistent with the topics listed below
with the following exception:
Developmental Biology and Congenital Anomalies Branch
The DBCAB does not support clinical trials through the SBIR/STTR program.
Pre-submission Resources & Commercialization Assistance
There are several resources and programs available throughout the SBIR/STTR process. For more
information, please visit our Commercialization Resources webpage.
Specific Funding Opportunities and Programs
In addition to the Omnibus program announcement, for up-to-date NICHD releases on targeted
funding announcements and programmatic initiatives visit: https://seed.nih.gov/small-
business-funding/find- funding/sbir-sttr-funding-opportunities or
https://www.nichd.nih.gov/grants- funding/SBIR_STTR/Pages/default.aspx.
NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 48
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
The NICHD accepts Phase IIB SBIR Competing Renewal applications to support additional R&D
necessary for approval of a federal regulatory agency (e.g., FDA, FCC). Such products may include
medical implants, pediatric devices, drugs, vaccines, and new treatment or diagnostic tools
Applicants who received NICHD SBIR Phase II support and who are currently Phase II awardees are
eligible. If the project meets the criteria for a budgetary waiver (see SBA-Approved Waiver Topics),
the Phase IIB should not exceed $3M total costs for three years. The amount of award may vary year
to year depending on the research proposed. Funding priority will be given to those small
business concerns that show the ability to develop innovative products and demonstrate
growth towards independence from the SBIR/STTR programs.
Prospective applicants are strongly encouraged to contact NICHD program staff well in advance of
submitting a Phase IIB Competing Renewal application.
Clinical Trials
Does NICHD accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
Yes
Does NICHD accept Clinical Trials
through specific Notices of Funding
Opportunities?
Yes
RFAs
https://seed.nih.gov/small-business-
funding/find-funding/sbir-sttr-funding-
opportunities
Does NICHD support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
Check the NICHD website for active
NOFOs: https://www.nichd.nih.gov/grants-
funding/opportunities-mechanisms/active-
foa/Pages/default.aspx
Research Topics
The major NICHD research priority areas for each Branch are listed below. Investigator initiated
applications that have commercial potential that fall outside these topic areas but fall within the
research mission of the NICHD are also considered through this Omnibus solicitation.
A.
Child Development and Behavior Branch
The CDBB encourages innovative developmentally-sensitive, theoretically-grounded, and evidence-
based small business initiatives that develop technology and products addressing the psychological,
social and emotional, psychobiological, language, numerical, literacy, cognitive and intellectual
development and health of persons from infancy through the transition to adulthood, recognizing the
important role others have in contributing to the healthy development of an individual. Products that
target at-risk populations and/or exploit new technologies that can expand the effective reach or
inclusion of underserved populations in order to encourage healthy development and/or our
understanding of the influences of context and/or behavior on development are especially
encouraged. CDBB is also interested in research on innovative approaches to both imaging and
other non-invasive measurement approaches to capture real time brain activation activity in typical
and atypical infants and young children (birth to age three).
Foci of specific interest include, but are not limited to (please also see the CDBB description for
research priorities):
•
Enhancing Bilingual and Biliteracy Development: Adaptive learning technology to enhance
bilingual and/or biliteracy development in English-language learning children and youth.

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 49
•
Bi- or Multi-Lingual Measures of Neurodevelopment: Develop easy to administer
objective neurodevelopmental measures from evidence-based neurocognitive research
specific to typically developing infants through pre-K children from diverse language
homes that are shown to correlate with development of brain connectivity and activation.
Remote administration measures are a high priority.
•
Pediatric Primary Care Behavioral and Health Promotion Interventions: Facilitate
research on the impact of behavioral and health promotion interventions in pediatric
primary care and related clinical settings with a focus on child and adolescent health
outcomes.
•
Psychosocial Adjustment for Individuals in High-Risk Environments: Develop
measures to identify and tools to stimulate developmental factors and mechanisms which
promote short- and long-term psychosocial adjustment for children and adolescents
exposed to high-risk family and neighborhood environments.
•
School Readiness Skills in Economically and Socially Disadvantaged Children:
Develop mobile device apps and/or hand-held devices that assess and/or promote the
development of executive functioning (EF) and school readiness skills and abilities in
infancy and early childhood and in diverse populations of children as well as measures of
home, childcare and preschool environments and practices that are related to child
learning and development.
•
Reading, Writing, and Mathematics Struggling Learners: Develop assistive
technology to enhance learner outcomes for individuals that struggle to acquire literacy
and/or numeracy skills, grounded in current scientific understanding of these challenges.
•
Assessment and Enhancement of Reasoning Development: Develop validated and
specific assessment tools that are sensitive to contributing factors (e.g., biobehavioral,
environmental, cultural, academic, and cognitive factors) to facilitate research on and the
promotion of neurocognitive development of reasoning (e.g., quantitative, deductive,
inductive, causal) in typically developing populations.
•
Fostering inclusion of typically-developing or at-risk infants, toddlers and children in
neuroimaging activities: Develop products or new strategies to facilitate neuroimaging
of typically-developing or at-risk infants, toddlers and children.
B.
Contraception Research Branch
The CRB supports research on developing new and improved methods of fertility regulation as well
as research on the benefits and risks of contraceptive drugs, devices and surgical procedures.
Areas of interest include, but are not limited to:
•
Development of new and improved methods of fertility regulation, for men and women,
that are safe, effective, inexpensive, reversible and acceptable with priority given to non-
hormonal and on-demand methods
•
Synthesis and testing of novel chemical compounds that are potential contraceptives
•
Multipurpose prevention technologies designed to prevent sexually transmitted infections,
such as HIV, as well as pregnancy
C.
Developmental Biology and Congenital Anomalies Branch
The DBCAB supports biomedical research on the cellular, molecular, and genetic aspects of typical and
atypical embryonic development including early embryogenesis, organogenesis, as well as topics in
stem cell and regenerative biology. The overall goal is to promote research on developmental biology to
understand the causes of structural birth defects.
Areas of interest include but are not limited to:

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 50
•
Development of new model systems (animal or other) to study developmental
mechanisms and causes of structural birth defects
•
Innovative technologies for in vivo imaging of developmental processes (cell and tissue
dynamics) and gene expression
•
Development of antibodies, novel ligands, and other probes to facilitate our
understanding of typical and atypical embryonic development in model organisms
•
Technologies for quantitative measurement of physical properties of cells/tissues in vivo during
development
•
Innovative technologies for studying metabolomics in developing vertebrate embryos
•
Technologies to facilitate and advance systems biology approaches to the study of
embryonic development and structural birth defects
•
Technologies to facilitate and advance high throughput chemical screening (including
small molecules) for advancing structural birth defects research
•
Software development to facilitate the collection and analyses of data generated using
medium-high throughput screening platforms in model systems (model organisms, cell-
based models)
•
Software development to facilitate the collection, mining and analyses of genomic and
phenotypic data from children affected with structural birth defects, and cross-analysis
with model organism data
•
Development of user-friendly software for biomedical researchers with limited knowledge
of computational biology to analyze large-scale human and other datasets associated
with structural birth defects
•
Technologies/methodologies to generate, and software to mine, data related to wound
healing and regenerative responses across animal species
•
Novel reagents for activation and mobilization of endogenous/adult stem cells to
promote in vivo tissue regeneration
•
Methodologies to drive limb regeneration in higher vertebrates (including in mammals)
that might otherwise lack the capacity for regeneration.
•
Technologies for iPSC-based regenerative medicine in the context of structural birth defect
•
Screening technologies for small molecules in human Embryonic Stem (ES) Cells or
Induced Pluripotent Stem Cells (iPSCs) and disease specific iPSCs for targeted
modification of regulatory networks affected in structural birth defects
D.
Fertility and Infertility Branch
The FIB supports research on the reproductive processes of men and women and of animals with
similar reproductive systems related to developing safer and more effective means of regulating,
preserving or achieving fertility.
Areas of interest include but are not limited to:
•
Development of reagents and tools, such as high-resolution technologies to
facilitate study of reproductive and developmental processes, including gamete
and early embryo development, and reproductive track development
•
Development of techniques and identification of novel biomarkers to produce, identify,
and use healthy gametes as well as advancement on preservation of human gametes
•
Development of organoid cultures and physiomimetic systems ideal for study of
gametogenesis and normal or diseased reproductive tissues/organs
•
Development of improved methods of growing and differentiating stem cell lines in vitro,
including feeder cell-free approaches to facilitate reproductive research
•
Development of improved technologies for the reprogramming of cells, including
embryonic stem cells or adult cells, into eggs and sperm
•
Development of improved technologies for preimplantation genetic diagnosis
•
Development of omics technologies to diagnose impairments in sperm function, fertilization,
ovulation, implantation, decidualization and other aspects of reproductive processes

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 51
•
Use of genomics and proteomics to develop novel diagnostics and treatments for
reproductive diseases and disorders
•
Use of semen, vaginal or cervical fluid, or menstrual effluent to diagnose fertility
status or other health conditions
•
Development of novel assays, kits, and devices to monitor and treat infertility
•
Development of Artificial Intelligence techniques/methods for selection of best sperm cells,
oocytes, and embryos to generate better predictive models for in vitro fertilization
•
Development of innovative technologies for point-of-care testing for fertility/infertility and
reproductive diseases and disorders
•
Development of patient-specific treatment regimen for infertility diseases using Artificial
Intelligence methods/technologies
•
Development of tools, technologies or apps for diagnosis and treatment of infertility in
resource limited settings to increase community and individual resources to address
infertility
•
Development of tissue engineering technologies for uterine tissue regeneration and
reproductive track reconstruction for treatment of infertility
•
Identification and/or validation of putative male or female infertility targets
•
Development of novel drugs or devices to treat male or female infertility.
•
Development of high-throughput screening methodologies for small molecule drugs
addressing infertility
E.
Gynecologic Health and Disease Branch
The GHDB supports biomedical research related to gynecologic health throughout the reproductive
lifespan, beginning at puberty and extending through perimenopause.
Areas of interest include, but are not limited to:
•
Development of new diagnostic approaches and treatments for female pelvic floor
disorders, including drugs, and devices used for treatment of pelvic organ prolapse,
urinary incontinence, fecal incontinence, and other female pelvic floor disorders
•
Development of new diagnostic methods and novel surgical and non-surgical treatments
for uterine fibroids, endometriosis, adenomyosis, and benign ovarian cysts. Non-invasive
diagnostics and/or diagnostics that make use of menstrual effluent are of particular interest.
•
Production of marketable novel or improved methods, devices, and technologies for the
diagnosis, monitoring and therapy of gynecologic pain disorders including chronic pelvic
pain, vulvodynia/vestibulodynia, and dysmenorrhea
•
Generation of new approaches for the diagnosis, monitoring and treatment of abnormal
menstrual cyclicity
•
Surgical and non-surgical treatments for girls and women with reproductive tract
abnormalities, including congenital structural abnormalities and complications from
female genital cutting
•
Devices and/or technologies designed to address surgical challenges in gynecologic
surgeries, including hysterectomy
•
Technologies designed to apply -omics platforms (genomics, proteomics, metabolomics
etc.) to questions of gynecologic health and disease
F.
Intellectual and Developmental Disabilities Branch
The IDDB sponsors research aimed at preventing, diagnosing, and ameliorating intellectual and
developmental disabilities (IDD). Emphasis is on studies related to IDD, including common and rare
neurodevelopmental and neuromuscular disorders, such as autism spectrum disorders, Down,
Fragile X, and Rett syndromes, mitochondrial conditions, inborn errors of metabolism, and others.

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 52
Areas of interest include, but are not limited to:
•
Innovative tools, including molecular, imaging, statistical or behavioral tools, to
characterize the etiology and pathophysiology of abnormal nervous system development.
•
Methods and devices to delineate genetic, genomic, and epigenetic causes of IDD and
develop gene-based treatments.
•
Methods or devices designed to screen for, diagnose, treat, and manage IDD and other
conditions, particularly those identified or identifiable by newborn screening.
•
Assessment tools for use in the clinic or community settings to enable the accurate
measurement of change in response to interventions.
•
Development of early interventions leading toward the prevention, diagnosis,
treatment, and management of IDD.
•
Methods or devices to develop or adapt smart technologies (such as wearable devices,
mobile health applications (apps), and electronic medical records (EMR)-based tools) to
assist in remote health monitoring, to service as point-of-care diagnostic tools, and/or to
enhance screening, diagnosis, prevention, treatment, or management for individuals with
IDD to improve their quality of life.
•
Development of assessment measures or treatments for co-morbid symptoms in those
with IDD including disordered sleep, self-injurious behaviors, obesity, gastrointestinal
dysfunction, seizures/epilepsy, attention deficit/hyperactivity disorder, anxiety,
depression, psychosis, immune dysregulation, self-injurious behaviors, and ADHD and
other mental health disorders.
•
Innovative and new digital technologies and mHealth solutions for improving transition of
adolescents to adult healthcare providers by improving health literacy, enabling self -
management, and encouraging adherence to existing treatments among adolescents.
•
Methods and devices to facilitate inclusion of people with all levels of IDD in research and
clinical care – both research/care targeted toward IDD populations and research/care for
more general populations where people with IDD are typically categorically excluded.
G.
Maternal and Pediatric Infection Disease Branch
MPIDB supports domestic and international research on human immunodeficiency virus
(HIV)/acquired immune deficiency syndrome (AIDS) and other infectious diseases (such as CMV,
Syphilis, tuberculosis, hepatitis and malaria) in people of child bearing age, pregnant people,
mothers, fetuses, infants, children and adolescents. Specific areas of interest include but are not
limited to epidemiology, clinical manifestations, immune-pathology, pathogenesis, transmission,
treatment and prevention (including immune-therapeutics like monoclonal antibodies, vaccines and
other biomedical modalities) of HIV infection, severe acute respiratory syndrome coronavirus 2
(SARS-CoV-2) infection, and other pertinent infectious diseases in children, adolescents and
pregnant people, with a focus on prevention of vertical transmission of HIV and other congenital
infections, and HIV-related and other infectious-disease related complications in these populations.
Additional areas of interest include:
•
New technologies relevant to resource-limited countries for:
o
Screening, diagnosis, and management of infectious diseases in pregnant women,
infants, and children, including but not limited to HIV such as SARS-CoV-2, congenital
CMV, congenital Syphilis, tuberculosis, and Zika virus)
o
Rapid assays to monitor disease activity and response to therapy as well as immune
response to vaccinations against relevant infections in infants and children (e.g., malaria,
tuberculosis), which can be used at the individual level and/or as part of public health
campaigns (e.g., eradication of outbreaks and prevention of spread)
o
Diagnosis and treatment of HIV-related co-morbidities (e.g., diagnosis of tuberculosis,
STIs)
o
Diagnosis and treatment of SARS-CoV-2 infection-related outcomes in mothers and
infants

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 53
o
Simple and less technologically demanding point of care assays to monitor CD4 cell
percentage/count, HIV viral load, or other surrogate markers of HIV disease progression
in children
o
Simple and easy to use/at home use diagnostics and point of care assays to monitor
clinical symptomatology and prognosis of SARS-CoV-2 infection and recovery in children
Interventions designed to promote or optimize medication adherence
Child-friendly formulations (preferably not liquid preparations) of drugs used to treat or prevent
HIV infection, complications of HIV infection, and/or other high-priority infections such as
tuberculosis, hepatitis, Syphilis, CMV, and malaria relevant to children, particularly in resource-
limited countries; Fixed-dose drug formulations and innovative methodologies for development
of solid heat stable formulations capable of being administered to young children (e.g.,
sustained release beads, etc.) and/or improve pill or volume burden
•
Innovative long-lasting drug formulations for antiretroviral and other anti-infective drugs
that would allow less frequent drug administration (e.g., once daily, weekly, or monthly)
•
Simple, standardized, validated tools to evaluate neurodevelopmental outcomes in children
•
Innovative data collection and database development approaches to leverage and link
electronic medical records and/or other health information systems to better
understand treatment and prevention of infectious diseases among infants, children,
adolescents, and people of child-bearing age.
•
Biomedical modalities including vaccines and methods to assess efficacy of vaccines, to
prevent acquisition of HIV and other infectious diseases in children, adolescents, and
women.
•
Topical microbicide agents, wearable, implantable, or insertable devices releasing
medications alone or as part of multipurpose prevention technologies (MPTs), to
prevent sexual acquisition of HIV and other sexually transmitted infections in
adolescents, adult women, and pregnant or postpartum people.
•
New, non-invasive technologies to evaluate complications of antiretroviral drugs (e.g.,
mitochondrial toxicity, bone toxicity) in HIV-infected infants, children, adolescents,
pregnant people, and their fetuses.
•
New or improvements to existing technologies for measuring the HIV latent reservoir, or
other long-term effects of infectious diseases, including high-throughput, visualization
algorithms, and improvement in assay reliability and sensitivity in children.
H.
Obstetric and Pediatric Pharmacology and Therapeutics Branch
The OPPTB supports research and research training on the development and use of safe and
effective therapeutic drugs and therapeutic-related medical devices for children and pregnant and
lactating people, including during the postpartum period. The branch promotes basic, translational,
and clinical research to improve the safety and efficacy of therapeutics, primarily pharmaceutical
drugs and medical devices. It is responsible for developing and supporting a comprehensive national
effort to increase the knowledge base for understanding how to appropriately treat disease during
pregnancy, lactation, infancy, childhood, and adolescence using evidence-based therapeutic
approaches. This includes support for the development and validation devices to inform treatment
decisions and enhance precision drug delivery. The goal of these efforts is to assure that
medications are appropriately tested for dosing, safety, and effectiveness for individuals within their
target populations. Of note: NICHD considers applications for pediatric conditions that have
significant efforts at other NIH institutes (e.g., sickle cell disease, pediatric oncology, juvenile
diabetes) to be of lower programmatic priority.
Applications to advance the study of obstetric and pediatric therapeutics include but are not limited to:
•
Understanding Differences and Heterogeneity in Pediatric Disease Treatment. Research to
quantitatively understand differences in drug action and related pathophysiology between
childhood and adult disease and conditions unique to pediatrics. This includes developing tools
(e.g., biomarkers, outcome measures, and physiologically based
pharmacokinetic/pharmacodynamic models) to support pediatric drug discovery and

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 54
development and to facilitate the application of precision medicine approaches in children.
•
Pharmacology and Pathophysiology of Pregnancy. Developmental pharmacology research and
approaches that explore the intersections of physiological changes in pregnant people and
during fetal development with drug action (e.g., pharmacokinetic, pharmacodynamics, and
pharmacogenomics) and with molecular pathways that may serve as novel therapeutic targets
for disease-modifying therapies specific to these populations. Critical areas include pain
management in pregnant and lactating people and treatment of gestational diabetes,
preeclampsia, and prevention of preterm delivery.
•
Novel Alternatives to Traditional Pediatric and Obstetric Clinical Trials. Development of
innovative approaches and algorithms to determine drug dosing, safety, and
effectiveness in children and in women during pregnancy and lactation. This includes
artificial intelligence-driven modeling and simulation methods, novel approaches to
utilizing existing data and archived biosamples/biospecimens, and pragmatic trials.
•
Population- and Individual-Specific Diagnostic and Therapeutic Devices that can advance
precision medicine through individualized diagnosis, drug delivery, and non-drug therapy
appropriate for use in neonates, children, and obstetric and lactating people. This may
include 3D bioprinting, AI-enhanced pharmacometrics modeling, AI-driven diagnostic and
decision-making tools, novel drug delivery devices, and formulations.
•
New Uses for Drugs, Biologics, and Other Therapeutics. This includes the development
and use of preclinical experimental models (e.g., animal models and human biomimetics),
use of organotypic microphysiologic cell culture systems and strategies for assessing
pharmacologic and toxicologic effects of therapeutics, use of genetically diverse model
organisms to assess precision prescribing approaches for interindividual manifestation of
disease or response to therapeutic agents, and computation models or the accumulation
of real-world evidence in support of new therapeutic uses.
I.
Pediatric Growth and Nutrition Branch
The PGNB supports research designed to support short and long-term health so that children can
achieve their full potential through an expanded understanding of those factors that influence
metabolism, growth (body composition and linear growth) and neurodevelopment. An additional
focus is on those biological (e.g., genetic, nutritional, endocrinological) factors that contribute the
early life origins of non-communicable disease (e.g., obesity, diabetes, cardiovascular disease,
osteoporosis). The PGNB encourages research that focuses on detecting the biological antecedents
of these conditions during pregnancy, infancy, and childhood.
Areas of interest include, but are not limited to:
New research tools, improved measurement methods, and technologies that enhance our understanding
of:
•
Growth:
o
Physical growth, body composition, bone health, nutrition, and obesity
o
Determinants of normal bone mineral accretion and peak bone mass. Interactions of
muscle and bone during infancy and childhood
o
Neuroendocrinology of puberty, linear growth, body composition
o
Mechanisms of hormone action during linear growth, pubertal maturation, and other
aspects of physical development
•
Biological antecedents of childhood obesity and its short and long-term consequences:
o
Genetic and molecular mechanisms of obesity, psychosocial risks of obesity, and
therapeutic interventions for obesity in children and adolescents
o
Impact of early life exposures including infant feeding practices on short and long- term
health and development
•
Biology of nutrition as it pertains to health and development (physical and neurological function)
during pregnancy, infancy and childhood including discovery, development and deployment of
biomarkers for early detection of:
o
Mal-(over-/under) nutrition; including biomarkers of exposure, status, function and effect (i.e.,

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 55
impact on early life development including neurodevelopment)
o
Enhanced understanding of the role of human milk in child health and
development.
o
Maternal nutrition (pre-pregnancy, pregnancy, and lactation)
o
Novel approaches to enhanced infant feeding practices in term and pre-term
infants
•
Developmental origins of health and disease including:
o
Ascertain biomarkers early in life that predict the onset of chronic diseases such as
diabetes, osteoporosis, and the metabolic syndrome later in life. The PGNB emphasizes
the life course model to develop primary preventive approaches to chronic diseases.
o
Develop platforms for implementation of biomarkers of disease status, nutritional
status, and biological function from infancy through adolescence
J.
Pediatric Trauma and Critical Illness Branch
The PTCIB supports research and research training in pediatric trauma, injury prevention, and
critical illness across the continuum of care. These efforts include research focused on the
prevention, treatment, and management of physical and psychological trauma and the surgical,
medical, psychosocial, and systems interventions needed to improve outcomes for critically ill and
injured children and adolescents.
Additionally, the PTCIB supports basic, clinical, and translational research that explores short- and
long- term consequences of traumatic experiences such as exposure to disasters, all forms of
violence against children, exposure to critical illness environment, and experiences of
bereavement, grief, and loss.
Applications of interest include, but are not limited to the research and development of:
•
Technologies, devices, and equipment used by pediatric critical care, emergency care,
and trauma care personnel.
•
Novel technologies in caring for injured children prior to and during transport to treatment
settings.
•
Tools and technologies for screening and diagnosis of injuries related to forms of
child maltreatment.
•
Devices and innovative therapeutic technologies for management of medical conditions
and related problems stemming from critical illness and serious or life-threatening
injuries.
•
Preventive intervention tools, materials, and technologies designed to improve clinical
practice, parenting, and social system support for injured or traumatized children.
•
Tools, materials, and technologies designed to reduce pediatric trauma exposure and the
number and severity of pediatric injuries and deaths.
•
Tools and technologies to improve the environment of pediatric intensive care including
resources to promote patient safety and to enhance clinical education and training of
critical care personnel
•
Tools and technologies that support the diagnoses and treatment of critical illness in
children, including nosocomial infections and iatrogenic injury.
K.
Population Dynamics Branch
PDB supports research and research training in demography, reproductive health, and population
health. In demography, the Branch supports research on the scientific study of human populations,
including fertility, mortality and morbidity, migration, population distribution, nuptiality, family
demography, population growth and decline, and the causes and consequences of demographic
change. In reproductive health, the Branch supports behavioral and social science research on

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 56
sexually transmitted diseases, HIV/AIDS, family planning, and infertility. In population health, the
Branch supports data collection and research on human health, productivity, behavior, and
development at the population level, using such methods as inferential statistics, natural
experiments, policy experiments, statistical modeling, and gene/environment interaction studies.
Applications are encouraged, but are not limited to these areas:
•
Technological innovations or inventions to improve collection of biomarker and
anthropometric data in large population-representative surveys
•
Hardware or software to improve the collection of accurate cause of death information or
health diagnosis such as information related to infant and maternal morbidity and
mortality, in large population-representative surveys or in administrative data sets
•
Methods for integrating data science, including artificial intelligence and machine
learning, into demographic research
•
Methods for improving the collection, documentation, archiving, linking, and
dissemination of population representative data sets, especially data sets that are
complex, multilevel or multimodal
•
Methods for protecting and assuring confidentiality for human subjects when collecting,
archiving, linking, or disseminating population-representative data sets, especially data
sets that are longitudinal or that include both spatial and individual-level data
•
Methods for reducing the costs of collecting, linking, and disseminating large-population-
representative data sets
•
Development and dissemination of effective tools for prevention research and
intervention programs related to STIs/HIV; pregnancy; contraceptive use; adolescent,
young adult, and maternal mortality; child health; at-risk youth; and other health-related
topics relevant to PDB science
•
Innovative approaches and techniques for research design, measurement, and data
collection and analysis in the social and behavioral sciences, with particular attention to
methodology and measurement issues related to protecting research subjects, archiving
and disseminating complex datasets, and studying diverse populations and/or sensitive or
confidential behaviors
L.
Pregnancy and Perinatology Branch
The PPB supports research in the following areas: the physiology of pregnancy and labor; high- risk
pregnancies, including those with hypertensive disorders, diabetes or seizure disorders; fetal
pathophysiology; premature labor and birth; diagnostic, monitoring, and therapeutic devices and
instruments for newborn infants in the nursery and in Neonatal ICU setting; improving the existing
products or developing new products that would improve the routine and extended care of the
newborn infants; products and agents related to breastfeeding; hospital supplies specifically related
to use in the care of newborn infants; nanotechnology and its application for the care of newborn
infants; instruments and devices for assessing and monitoring the nursery environment (noise,
lighting, and odor); disorders of the newborn; sudden infant death syndrome; and biological and
behavioral antecedents of low birth weight.
The following topic areas are of high priority:
•
Non-invasive (or minimally invasive) methods to assess preeclampsia; gestational
diabetes; fetal well-being; spontaneous preterm birth; and stillbirth
•
Methods to characterize the bioactive components of human milk
•
Non-invasive methods to longitudinally identify predictors and indicators of placental
dysfunction including malperfusion, abnormal placental development, and impaired
placental function (nutrient transfer, metabolic function, exchange of respiratory gases, and
hormone production).
•
Devices, instruments, and tools to minimize bacterial colonization, reduce proclivity for

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 57
thrombus formation, and reduce healthcare associated infection risks
•
Lab-on-a-chip; specifically, non- or minimally-invasive approaches for assessing
metabolic profiles (e.g., glucose and lactate/pyruvate), ketone bodies, bilirubin
(unconjugated, free, indirect, and total), and other major analytes (Na+ Ca+ Cl+ K+ etc.)
•
Rapid methods for diagnosis of bacterial infections and the assessment of antibiotic
sensitivity
•
Improved syringes, needles, and injection set ups to help administer small doses of
medications over prolonged periods (e.g., insulin for treating hyperglycemia)
•
Methods to assess pain in the newborn, analgesia, and the evaluation of neonatal opioid
withdrawal syndrome
•
Non-invasive measures to assess brain energy utilization in the newborn, especially
glucose, oxygen, lactate, ketones, and other energy substrates
•
Improved devices and instruments for assisted ventilators for use in the neonatal ICU
M.
National Center for Medical Rehabilitation Research
This Center supports innovative research on the restoration, replacement, enhancement or adaptation of
function for people with chronic physical disabilities. This includes rehabilitative approaches across
etiologies and the lifespan, as well as the environmental and policy factors that promote full participation.
We encourage studies that integrate biomedical, engineering and/or psychosocial approaches to develop
practical and creative solutions to the daily functioning of people with disabilities and their families. The
mission of the NCMRR is to increase the effectiveness of medical rehabilitation practices through
research. Information about specific program areas within NCMRR can be found here.
Examples may include but are not limited to:
•
Adaptation and Plasticity: Develop non-invasive and surrogate measures of plasticity
that would be appropriate for use in a clinical setting to target rehabilitation therapies and
monitor treatment effectiveness (e.g., biomarkers, imaging)
•
Novel Technology: Orthotics, prosthetics, and robotics devices and interfaces; Assistive
technologies; Invasive and non-invasive biological sensors, prosthetic systems or implants
to improve function; New control methods and improved sensory feedback; Strategies for
controlling and adapting to the environment; Advanced wheelchair designs and
enhancements and other mobility devices; Biomaterials and tissue interfaces,
nanotechnology, bionics
•
Rehabilitation Interventions: Development and use of robotics; Gaming applications;
Virtual and Augmented Reality; Simulations; M-health and other approaches to promote
participation, understand and support healthy behaviors, reduce health disparities, and
enhance clinical compliance, especially in children with physical disabilities.
•
Chronic Symptom Management: Methods to increase screening for chronic conditions or
preventable secondary conditions in individuals with physical disability; Prevention and
treatment strategies for mitigating symptoms associated with multiple chronic conditions in
individuals with physical impairments, including persistent pain, symptoms of obesity,
diabetes, cardiovascular deconditioning, fatigue, symptoms of overuse injuries, pressure
ulcers, sleep disturbances, and depressive symptoms; Improving muscle c apacity in
chronic physical disability to include therapeutic or adaptive exercise and muscle
stimulation; muscle-disuse syndromes and contractures; Rehabilitation interventions for
improvement of physical disability and comorbid cognitive, sensory, or somatic
consequences of impairment, disease or injury; Autonomic function in the context of injury
or specific conditions.
•
Rehabilitation in the Community: Strategies to build or modify community and/or
environmental resources that provide effective rehabilitation and health promotion services
within the individual’s own community. Development of engineering, crowdsourcing, and social
science approaches to promote, monitor, and sustain outcomes in real world settings.

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 58
Investigators proposing budgets exceeding the guidelines are encouraged to contact program staff
six weeks prior to submitting the application.
Contact Information
Child Development and Behavior Branch
NICHD CDBB Small Business (SB) Program Director
Phone: 301-435-6855 OR 301-435-3602
Email: NICHDCDBB-SB-Program@mail.nih.gov
Contraception Research Branch
Steven C. Kaufman, MD, MS
Phone: 301-435-6989
Email: kaufmans@exchange.nih.gov
Developmental Biology and Congenital Anomalies Branch
Mahua Mukhopadhyay, PhD
Phone: 301-435-6886
Email: mukhopam@mail.nih.gov
Fertility and Infertility Branch
Clara Cheng, PhD
Phone: 301-435-6992
Email: clara.cheng@nih.gov
Gynecologic Health and Disease Branch
Candace M. Tingen, PhD
Phone: 301-435-6971
Email: candace.tingen@nih.gov
Intellectual and Developmental Disease Branch
Sujata Bardhan, PhD
Phone: 301-435-0471
Email: sujata.bardhan@nih.gov
Maternal and Pediatric Infection Disease Branch
Sai Majji, PhD
Phone: 301-402-8048
Email: sai.majji@nih.gov
Obstetric and Pediatric Pharmacology and Therapeutics Branch
Antonello Pileggi, MD, PhD,
MSCTI Phone:301-402-5019
Email: pileggia@nih.gov
Pediatric Growth and Nutrition Branch
Andrew A. Bremer, MD,
PhD Phone: 301-402-7886
Email: andrew.bremer@nih.gov
Pediatric Trauma and Critical Illness Branch
Tessie October, MD, MPH
Phone: 301-827-6757
Email: tessie.october@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NICHD 59
Population Dynamics Branch
Ronna Popkin, PhD
Phone: 301-827-5121
Email: ronna.popkin@nih.gov
Pregnancy and Perinatology Branch
Diane Gumina, PhD
Phone: 301-827-7304
Email: diane.gumina@nih.gov
National Center for Medical Rehabilitation Research
Toyin Ajisafe, PhD
Phone: 301-827-9242
Email: toyin.ajisafe@nih.gov
For additional SBIR/STTR program administrative information and research topics
contact: Toyin Ajisafe, PhD
Phone: 301-827-9242
Email: toyin.ajisafe@nih.gov
For additional financial/business management questions contact:
Mindy Bixby, Grants Management Specialist
Phone: 301-402-3204
Email: mindy.bixby@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 60
NATIONAL INSTITUTE ON DRUG ABUSE (NIDA)
Mission
NIDA is the lead federal agency supporting scientific research on drug use and its consequences. Our
mission is to advance science on the causes and consequences of drug use and addiction and to apply
that knowledge to improve individual and public health through 1) strategically supporting and conducting
basic and clinical research on drug use, its consequences, and the underlying neurobiological, behavioral,
and social mechanisms involved; and 2) ensuring the effective translation, implementation, and
dissemination of scientific research findings to improve the prevention and treatment of substance use
and addiction and enhance public awareness of addiction as a brain disorder.
Budget Guidance
Award budgets and project periods are listed in the Section II Award Information of the Omnibus/Parent
Notices of Funding Opportunities for SBIR and STTR. Total award budgets include direct costs, indirect
costs, and fees and are capped not to exceed the total award amounts listed.
NIDA also sets its budget limits for specific research topics that received a waiver from the U.S. Small
Business Administration to exceed the hard budget caps. The current list of approved NIDA topics can be
found in the SBA-Approved Waiver Topics. NIDA budget limit for Phase I on SBA-approved waiver topics
is $400,000 in total costs and a project period of up to 1 year. NIDA budget limit for Phase II on SBA -
approved waiver topics is $3,000,000 in total costs and a project period of up to 3 years.
In all cases, applicants should propose a budget and a project duration period that are reasonable and
appropriate for the completion of the research project. Applicants are strongly encouraged to contact NIDA
program officials early in the application process to receive assistance regarding the program.
SBIR and STTR Program Information
SBIR and STTR programs at NIH are primarily intended to encourage private-sector technology
commercialization and increase small business participation in federally funded research and
development (R&D). The goal of the NIDA SBIR/STTR program is to increase small business participation
and private-sector commercialization of novel products or services that can help manage, prevent,
monitor, diagnose, and treat acute substance use, chronic substance use, and substance use disorders.
The NIH/NIDA dual peer review system is mandated by statute. A Scientific Review Group (SRG)
composed primarily of non-federal scientists with expertise in relevant scientific disciplines and current
research areas carries out the first level of review. The National Advisory Council on Drug Abuse (NACDA)
performs the second level of review. Only applications that are recommended for approval by both the
SRG and the NACDA may be recommended for funding. Final funding decisions are made by the NIDA
Director.
NIDA funding decisions for small business programs are based on a combination of factors:
• Programmatic priorities and current portfolio balance (for funded projects, please search the NIH
RePORTER database at https://reporter.nih.gov/)
• Potential for commercialization and public health impact;
• For Phase II applications: results of Phase I (or equivalent) clearly indicating that both technical
feasibility and commercial feasibility were established, and the scientific merit and commercial
potential of the project proposed in Phase II;
• For applicants who received preceding SBIR and STTR grants: quality of prior performance and
evidence of Phase III activities;

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 61
• Peer review critiques and overall impact score;
• Security risk, as assessed by the HHS Due Dilligence Program;
• Availability of funds.
Disclosure Requirements Regarding Ties to Foreign Countries
(https://seed.nih.gov/small-business-funding/small-business-program-basics/foreign-disclosure-and-risk-
management)
Applicants to the SBIR and STTR programs are required to disclose all funded and unfunded relationships
with foreign countries, using the Required Disclosures of Foreign Affiliations or Relationships to Foreign
Countries Form (hereafter referred to as the SBIR STTR Foreign Disclosure Form), for all owners and
covered individuals. Applicants who do not submit the completed SBIR STTR Foreign Disclosure Form
during the NIDA JIT process will not be considered for funding. Details of the SBIR and STTR Foreign
Disclosure and Risk Management Pre-award and Post-Award Requirements can be found in NOT-OD-24-
029.
NIDA-specific SBIR and STTR Program Information
NIDA is the largest source of initial funding for research efforts to address the needs of acute substance
use, chronic substance use, and substance use disorder (SUD) markets, undertaken with the goal of
commercialization. Commercialization is a process of delivering new products or services into the market.
Historically, the engagement of large private biomedical enterprises in the SUD space was limited.
Therefore, NIDA is devoted to achieving meaningful impact through enabling small businesses and
startups to commercialize substance use and drug addiction products, services, or technologies. NIDA
acknowledges the importance of project management in successful new product development and
encourages the use of project management tools and terms by the grant applicants.
The SBIR and STTR programs are phased programs:
Phase I: Supporting Feasibility and Proof of Concept. The objective of this phase is to establish the
technical and commercial feasibility of the proposed research or R&D efforts and to determine the quality
of performance of the applicant (small business concern or SBC) before providing further federal support
in Phase II.
Phase II: Supporting Research and Development. The objective of this phase is to continue the research
or R&D efforts initiated in Phase I. Funding will be based on the results of Phase I and the scientific and
technical merit and commercial potential of the Phase II application.
Phase III. The objective of Phase III is for the small business concerns (SBC) to pursue commercialization
objectives resulting from the Phase I/II R&D activities. A Phase III is defined as an award that derives
from, extends, or completes prior SBIR effort and is funded with non-SBIR funds. NIDA encourages
grantees to seek commitment(s) of funds and/or resources from an investor or partner organization for the
commercialization of the product(s) or service(s) resulting from the SBIR/STTR Phase I/II grant. Phase III
funding may come from different sources: private investors, venture capital firms, strategic alliances,
research contracts, sales of prototypes, public offerings, state finance programs, non-SBIR-funded R&D,
or production commitments from industrial firms or a federal agency for use by the U.S. government. NIDA
monitors SBC efforts to pursue, with non-SBIR/STTR funds, the commercialization of Phase I and II
outputs.
While both the SBIR and STTR programs are organized to provide government funding only in Phase I
and Phase II, NIDA is committed to assuring that its grantees are also prepared to successfully execute
Phase III.
Feasibility and Milestones

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 62
This section provides additional requirements and instructions which relate to definitions of the terms
“feasibility” and “milestones” for NIDA-specific applications, as well as addressing funding for assistance
in developing and executing in these areas.
Feasibility
To improve the odds of reaching Phase III, NIDA suggests using a simple desirability-feasibility-viability
(DFV) framework. While envisioning the goal for the Phase I project, NIDA applicants are encouraged to
address both technical feasibility (e.g., “can this offering [product or service] be built?”) and commercial
feasibility (e.g., “should this offering be built?”), which may include the initial explorations into desirability
(e.g., “does anyone want or need this offering?”) and viability (e.g., “how can financial stability be
assured?”).
It is important to understand the resources that NIH allows to establish commercial feasibility. The NIH
Grant Policy (see section 18.5.5.1 Market Research), states that “NIH will not support market research,
including studies of the literature that lead to a new or expanded statement of work ”, under the SBIR or
STTR grant, with the exception of Technical and Business Assistance (TABA) funds (see “Technical and
Business Assistance (TABA)” section below). The policy defines market research as follows: “For
purposes of the SBIR/STTR programs “market research” is the systematic gathering, editing, recording,
computing, and analyzing of data about problems relating to the sale and distribution of the subject of the
proposed research. It includes various types of research, such as the size of potential markets and
potential sales volume, the identification of consumers most apt to purchase the products, and the
advertising media most likely to stimulate their purchases. However, “market research” does not include
activities under a research plan or protocol that include a survey of the public as part of the objectives of
the project to determine the impact of the subject of the research on the behavior of individuals”.
Milestones
In NIDA small business grant applications, a clear understanding of the term “milestone” needs to be
demonstrated. In project management, the milestone is used to define an important decision point at
which significant uncertainty for a given project is resolved. The quest to conduct research activities to
establish technical and commercial feasibility should culminate in reaching a significant milestone. The
NIH SF424 (R&R) Application Guide instructs the applicants to include milestones for every Phase I,
Phase II, Fast Track and SBIR direct to Phase II applications, as these will be used in the evaluation
process. Milestones are evaluated as part of Approach and Investigator(s) review criteria. Missing or
inadequate technical and commercial milestones can therefore negatively affect the application’s impact
score.
In NIDA small business applications, NIDA expects meaningful milestones and milestone deliverables,
associated with each milestone. Specific Aims and milestones are related but distinct elements in the
application. As stated by the SF424 (R&R) Application Guide, Specific Aims are goals, specific objectives,
and expected outcomes of the proposed research. These Specific Aims are the activities that enable a
company to reach a milestone (go / no-go decision point). Each Specific Aim in the application should
therefore have at least one milestone associated with it.
Moreover, milestones, e.g. decision points, are tied to deliverables which are always tangible. Accordingly,
NIDA expects its awardees to identify meaningful milestones and to develop tangible deliverables
associated with each milestone that are specific, measurable, achievable, relevant, and time-bound.
These can be written in the following format:
• Milestone A…
o Specific Aim A…
▪ Milestone Deliverable A (specific, measurable, achievable, relevant, time-bound)…
For example,
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 63
To establish Technical feasibility for a drug discovery and development project:
Milestone 1: In-vitro assay validation for SAR studies
Specific Aim 1: Validate and optimize in vitro assays (Potency and Selectivity)
Milestone deliverable 1 (specific, measurable, achievable, relevant, and time-bound): Within 2
months, achieve Z’ factor of ≥0.5, based on values from at least ½ plate of positive and negative
controls.
Milestone deliverable 2 (specific, measurable, achievable, relevant, and time-bound): Within 4
months, demonstrate that concentration response testing over at least 8 concentrations
generates reproducible IC50 values withing a 3-fold range for 3 standard compounds.
To establish Commercial feasibility for a medical device development project:
Milestone 1: Payor Feedback
Specific Aim 1: Obtain payor feedback regarding requirements for coverage and reimbursement.
Milestone deliverable (specific, measurable, achievable, relevant, and time-bound): Within 3
months of receiving the NIDA Notice of Grant Award, initiate the contact with The FDA CDRH
Early Payor Feedback Program (EPFP) and submit EPFP Overview Request; meet with at least
3 payors to obtain feedback about clinical study design and proposed endpoints necessary to
obtain payor coverage.
Fast-Track Application
The NIH Fast-Track process allows Phase I and Phase II grant applications to be submitted and reviewed
together. It expedites award decisions and funding of SBIR and STTR Phase II applications for
scientifically meritorious projects that have a high potential for commercialization. Fast-Track applications
receive a single rating. Before submitting applications for Fast-Track review, applicants are strongly
encouraged to consult with NIDA program staff to assure that Fast-Track is appropriate. NIDA encourages
Fast-Track mechanisms for those applications for which a high potential for commercialization is
established. For its Fast-Tracks, NIDA also encourages preliminary data that clearly supports technical
and commercial feasibility. If repurposing already-existing drugs or devices for SUD diagnosis or
treatment, preliminary data about existing drug/device and scientific rationale for the feasibility in the SUD
space is required. In addition, NIDA requires a commercialization plan that demonstrates a high probability
of commercialization and letters of Phase III support/interest, additional funding commitments, and/or
resources from the private sector or non-SBIR/STTR funding sources. Letters of interest from potential
commercial partners or investors and letters of commitment of funds or other resources that will enhance
the likelihood of commercialization should be placed following the letters of support for consultants and
collaborators.
Importantly, before Fast-Track Phase II could start, NIDA conducts an administrative review and evaluates
the achievement of the stated milestones. In addition to Approach and Investigator(s), Fast-Track
milestones are assessed in the peer review under Additional Review Criteria: Does the phase I
applications specify milestones that should be achieved prior to initiating Phase II? Applicant’s failure to
provide milestones and specific, measurable, achievable, relevant, and time-bound milestone deliverables
may be sufficient reason for the peer review to exclude the application form the Fast-Track review.
Fast-Track applicants must propose two separate sets of milestones and associate them with specific,
measurable, achievable, relevant, and time-bound milestone deliverables, one set for Phase I and another
set for Phase II. The timelines for milestone deliverables are to be completed sequentially. It is important
to clearly state the go / no-go milestones that will determine a transition to Phase II. Failure to adequately
address these criteria may negatively affect the application's impact score. The following format is
encouraged:

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 64
Phase I
o Specific Aim A…
▪ Milestone Deliverable A (specific, measurable, achievable, relevant, time-bound)…
Phase I Milestone A (go/no-go, pivot)
Phase II
o Specific Aim A…
▪ Milestone Deliverable A (specific, measurable, achievable, relevant, time-bound)…
Phase II Milestone A…
Based on peer review recommendations, NIDA Program Officers may negotiate the Phase I milestones
with applicants and potential Fast-Track awardees before they are included in the terms of the award.
Fast-Track applications will receive a secondary review by the NIDA advisory council. Phase II
applications will be selected for funding based on NIDA's assessment of the Phase I progress, and
determination that the Phase I milestones were achieved; an update and verification of the
Commercialization Plan and any commitment(s) for funds and/or resources from an investor or partner
organization, the project’s potential for meeting the mission of NIDA and for commercial success; and the
availability of funds. NIDA may find it appropriate for an outside reviewer(s) to be involved in the process
of administrative review. If NIDA staff determines that the progress has not been adequate during Phase I,
additional information may be requested. Because of the intricacy of the Fast-Track mechanism, NIDA
staff invites open communication with applicants regarding this complex mechanism. NIDA staff will
monitor program progress against proposed milestones and make non-competing award decisions based
on achieving milestones. If NIDA program staff determines that progress in Phase I has not been
adequate, milestones were not met, and the Fast-Track Phase II is not recommended for funding, that
decision is not appealable.
Technical and Business Assistance (TABA)
NIDA informs its applicants about additional capabilities that may assist in establishing technical and
commercial feasibility through the dedicated TABA funding. Through TABA, small business applicants may
request up to $6,500 per year for a Phase I project and up to $50,000 across all years for a Phase II
project to help address the development and commercialization of their new products and processes
resulting from such projects, including intellectual property protections. TABA funding could be requested
to assist with product sales, intellectual property protections, market research and/or validation,
development of regulatory or manufacturing plans, and access to technical and business literature
available through online databases. Importantly, TABA activities are conducted externally, and, if NIDA
applicants wish to utilize the outside TABA provider/vendor that is not associated with the small business
applicant, they are required to include the vendor as a consultant in the budget and to provide a det ailed
budget justification. All instructions in the SF424 (R&R) Application Guide must be observed. Fast-Track
applications are a combination of both Phase I and Phase II and small businesses can request TABA
funding in both phases within their Fast-Track application up to these amounts for each phase. NIDA does
not allow requesting TABA funding through an Administrative Supplement.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NIDA does not participate in the CRP program. NIDA will not accept Phase IIB applications through the
Omnibus or specific program announcements.
Specific Funding Opportunities and Programs Translational Research
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 65
NIDA offers additional funding opportunities and programs to accelerate the preclinical discovery and
development of new medical products for SUD patients, including pharmacotherapeutics and medical
diagnostic and therapeutic devices.
Blueprint MedTech:
(https://neuroscienceblueprint.nih.gov/neurotherapeutics/blueprint- medtech/blueprint-medtech)
The program aims to support innovators by accelerating the development of cutting-edge medical devices
to diagnose and/or treat disorders of the nervous system. The program provides: (a) non-dilutive funds to
support medical device development activities led by investigators, and (b) additional resources and
support services.
Contact: Leonardo Angelone, PhD, Blueprint MedTech Program Lead (leonardo.angelone@nih.gov)
Blueprint Neurotherapeutics Network (BPN) for small molecules:
(https://neuroscienceblueprint.nih.gov/neurotherapeutics/bpn-small-molecules)
The program provides both non-dilutive funding and additional resources for small molecule drug
discovery and development, from hit-to-lead chemistry through phase I clinical testing. The program offers
funding, access to NIH-funded contract research organizations (CROs), and access to consultants with
expertise in various aspects of drug discovery and development.
Contact: Elena Koustova, PhD, MBA, Director, NIDA Office of Translational Initiatives and Program
Innovations (koustovae@nida.nih.gov)
Both programs utilize the cooperative agreement (U44) mechanism, which is milestone-driven and
involves NIH program staff participation in developing the project plan, monitoring research progress, and
appropriate go/no-go decision-making. SBIR applicants considering projects involving translational
research are strongly encouraged to contact program staff well in advance of submission.
Clinical Trials
Does NIDA accept Clinical Trials through
the Omnibus/Parent Notices of Funding
Opportunities?
Yes
Does NIDA accept Clinical Trials
through specific Notices of Funding
Opportunities?
Yes
NIDA accepts and supports SBIR and STTR
clinical trial applications through specific
opportunities, which can be found on the
NIDA Funding Opportunities webpage:
https://nida.nih.gov/funding
Does NIDA support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
NIDA accepts and supports non-SBIR/STTR
clinical trial applications through specific
opportunities, which can be found on the
NIDA Funding Opportunities webpage:
https://nida.nih.gov/funding
Research Topics
The major NIDA SBIR/STTR portfolio areas of interest are listed below as a general guide. Applications
proposing innovative technologies in substance use and addiction with strong commercial potential that
fall outside these portfolio areas are also encouraged through this Omnibus solicitation.
1. Biomarker Development for SUDs
2. Personalized Medicine for SUDs
3. SUD Drug Discovery and Development
4. FDA-regulated Medical Therapeutic and Diagnostic Devices for Substance Use and Addiction
5. Harm Reduction Technologies
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 66
6. Technological Approaches to Decrease Stigma Associated with Substance Use and Addiction
7. Prevention Technology to Address Substance Use and Addiction in Various Underserved
Populations
Biomarker Development for SUDs
Currently, there are no biomarkers to assess or predict treatment efficacy or categorize SUDs into clinical
subtypes. Thus, it is impossible to design treatments for effective and long-term recovery by classifying
SUD patients into categories that have reproducible and predictive validity.
Long-term use of opioids and other substances alters the integrity of homeostasis, changing the
endogenous opioid, endogenous cannabinoid, and almost all receptor systems studied so far in the brain
and peripheral immune cells. Biomarkers and signatures in patients diagnosed with an SUD can be very
different from those observed in patients without SUDs. These biomarkers or potential predictive markers
could serve as objective prognostic indicators to develop SUD. In addition, they could act as response
predictors to SUD therapeutics in adults, or as diagnostic biomarkers for infants with neonatal abstinence
syndrome (NAS). Furthermore, artificial intelligence (AI)-related technologies are being investigated in
healthcare to analyze patients’ big data, such as electronic health records of historical and current patient
treatments, to create more effective and better patient outcomes and to identify new diagnostic tools and
novel analyses. Accordingly, AI-related tools are of interest to accelerate traditional and innovative areas
of SUD biomarker development.
The proposed biomarker research should emphasize the importance of biomarker signatures that can
intersect SUD and related conditions that are considered important to the mission of NIDA. Proposed
projects may include biomarkers that assess the probability of SUD or allow an assessment of the
treatment trajectory in patients under treatment for SUD. Specific projects may encompass, but not
restricted to:
• Biomarkers with high specificity and sensitivity for opioids, stimulants, cannabis and other
emerging substances;
• Biomarkers that can detect substance use in early stage;
• Development of biomarkers that provide objective measures of substance use.
• Biomarkers that can predict an individual’s response to different treatment modalities.
• Biomarkers suitable for longitudinal monitoring of substance use.
• Biomarkers capable of detecting concurrent use of multiple substances
Projects solely focused on biomarkers for pain and alcoholism are of limited interest.
Personalized Medicine for SUDs
Advancements in technology and our deepening understanding of underlying neurobiology have provided
us with the chance to target specific neurobiological processes and tailor interventions approaches to
individual patients based on their unique genetic, neurobiological, and environmental characteristics. This
personalized approach recognizes the significant variability among individuals in how they respond to
medications, therapies, and other interventions. Genetic variations can influence a person’s susceptibility
to developing SUD, and treatment response. Genetic testing would identify specific gene variants
associated with SUD risk, metabolism of drugs and treatment response. Further, neurobiological
differences in brain structure and function can impact how an individual’s experiences and respond to
different substances. Identifying aberrant brain connectivity patterns and assessing neurotransmitter
levels can guide the treatments to modulate the pathways. Finally, social, cultural, and environmental
factors play a significant role in the development and course of SUDs. Areas of interest include but are not
limited to: a) Identifying and leveraging existing social support networks and recognizing environmental
triggers that contribute to substance use and modifications to mitigate the impact and b) utilizing big-data
analytics and predictive modeling to identify patterns and predictors of treatment response and refine
personalized treatment approaches. Overall, personalized medicine holds promise for improving the
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 67
effectiveness and outcomes of SUD treatment by addressing the biological and social factors that
contribute to substance use disorders.
SUD Drug Discovery and Development
Pharmacotherapy offers an important means of treating SUDs. Currently, there are five
pharmacotherapies approved by the Food and Drug Administration (FDA) for the treatment of Opioid Use
Disorder (OUD) and mitigation of opioid withdrawal symptoms: methadone, buprenorphine, extended-
release naltrexone, naloxone, and lofexidine. In addition, varenicline is an approved drug for the treatment
of nicotine cessation. However, given the diverse nature of SUDs, many patients have limited responses
to available medications and, consequently, there is an urgent need for novel treatments. It remains of
program interest to identify and develop improved pharmacotherapeutics with clear advantages over our
current approved pharmacotherapeutics for OUD treatment and for nicotine cessation treatment.
Additionally, there are no FDA-approved medications for cocaine, methamphetamine, or cannabis use
disorders.
Broadly, novel pharmacotherapeutics are encouraged for the range of unmet medical needs in SUD, for
polysubstance use, and for emerging novel treatment modalities and mechanisms of action for SUD
treatments. Developing and evaluating new, more efficacious medications remains a high priority.
Candidate medications may include either novel or re-purposed compounds.
Specific areas of interest include medications that target one or more domains of the addiction cycle,
including reward, stress and negative affect, incentive salience, executive function, habituation, and
impulsivity/compulsivity. Proposed projects may include emerging technologies and platforms for SUD
medication development with a focus on products with the potential to minimize drug seeking, compulsive
behavior, overdose prevention, and reversal. Specific projects may include, but are not limited to:
• Early therapeutic discovery activities ranging from target identification and validation through lead
development;
• SUD phenotypic assay development (e.g., organoids, organ-on-a-chip technologies, and higher
content invertebrate models, ex vivo bioassays) with validation studies in animal models (e.g.,
rodent models).
• Preclinical and/or clinical drug development;
• Medications that would address specific symptoms of withdrawal, such as cravings, depression,
cognitive impairments, pain, and sleep problems;
• Medications (neurochemicals) involved in social bonding that also modulates key processes
associated with addiction, including reward and stress responses, and may enhance the efficacy
of psychosocial addiction treatments;
• Big-data analytics and machine-learning algorithms analysis yielding insight into behavioral and
biological markers of relapse risk;
• Artificial Intelligence (AI)-related tools in SUD drug discovery and development to increase
innovation and support a cost- and time-effective SUD drug development of pharmacotherapies.
• Combination of pharmacotherapeutics to improve SUD treatment adherence and decrease risk of
relapse. Molecules may include new and investigational compounds and repurposed approved
medications
Projects proposing to study compounds already extensively investigated or currently being studied in
patients with SUD, and projects solely focused on pain or on alcoholism not associated with SUD are of
limited interest.
FDA-regulated Medical Therapeutic and Diagnostic Devices for Substance Use and Addiction
Medical Devices, including Software as Medical Device (SaMD), offer promising means to monitor,
diagnose, and treat patients who use substances for medical purposes, in addition to patients with SUDs.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 68
Currently, there are only a few devices that are cleared by the FDA for the treatment of SUD. As such, the
investigation and development of new safe and effective medical devices intended to prevent, monitor,
diagnose, and treat substance use and addiction is a high priority. Applications in this area are expected to
address the needs of those who actively use substances, chronically use substances, or have a
diagnosed SUD, and their caregivers, to ensure access to high-quality, safe, and effective medical
devices. It is expected that proposed approaches will include activities that will lead to regulatory
submissions for pre-market clearance / approval, including interactions with the FDA via the following
pathways: pre-submission (Q-submission), Investigational Device Exemption, 510(k), DeNovo, or
Premarket Approval (PMA) application. Additional pre-clinical activities may include, but are not limited to,
a) bench testing or computational modeling studies; b) good laboratory practice animal studies; c) good
manufacturing practice studies; d) toxicology and biocompatibility studies; e) software verification and
validation; f) usability/user experience testing.
Specific areas of interest include, but are not limited to:
• Imaging devices intended to investigate brain function and enhance monitoring, diagnosis, and/or
treatment of SUD;
• Devices that directly diagnose and/or reduce craving and withdrawal symptoms;
• Devices that identify and/or treat NAS;
• SaMD focused on behavioral health interventions to alleviate the burden of SUD;
• Therapeutic devices (e.g., neuromodulation) intended to improve SUD treatment outcomes and
relapse prevention;
• Physiological monitoring devices, including remote detection (e.g., wearables, sensors, health
monitoring/emergency notification systems), specifically intended for use in patients affected by
substance use and addiction.
Harm Reduction Technologies
Harm reduction is an evidence-based public health approach that that directly engages people who use
drugs (PWUD) to prevent overdose, disease transmission, and other harms associated with drug use.
NIDA included harm reduction in its FY 2022-2026 Strategic Plan in Priority Scientific Area #2: Develop
and Test Novel Prevention, Treatment, Harm Reduction, and Recovery Support Strategies. Harm
reduction was also identified as a federal drug policy priority in the 2022 National Drug Control Strategy
from the White House Office of National Drug Control Policy (ONDCP) and is also one of the strategic
priorities of the U.S. Department of Health and Human Services (HHS) Overdose Prevention Strategy.
Decades of evidence has shown that strategies for harm reduction substantially reduce HIV and hepatitis
C infection among people who inject drugs, reduce overdose risk, enhance health and safety, and
increase the likelihood of PWUD to initiate substance use disorder (SUD) treatment (SAMHSA Harm
Reduction Framework 2023).
The ideology behind harm reduction is based on helping PWUD increase their quality of life even if they
are not yet ready to enter treatment. The ONDCP’s Guiding Principles on Harm Reduction are: 1)
supporting individuals and overcoming obstacles in accessing all types of care, from overdose prevention
strategies to medications and mental health services, 2) providing ongoing support to individuals once
harm reduction or treatment services are initiated, 3) creating connections for PWUD with caring staff or
volunteers as part of receiving health and social services, and 4) treating PWUD with respect and dignity
to help them achieve better outcomes. Harm reduction strategies can address safer practices, safer
settings, access to healthcare, transitions to care, sustainable infrastructure, and a sustainable workforce.
Applications addressing harm reduction principles include, but are not limited to, technologies for:
• Education about the value of harm reduction and reduction of stigma surrounding drug use;
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 69
• Prevention, treatment, recovery, and general health promotion for PWUD;
• Addressing overdose education, detection, and naloxone use;
• Promoting safer use (e.g., drug-checking, reducing infection risk);
• Prevention, testing, and treatment for sexually transmitted infections;
• Ensuring access to and assistance with nutrition, clothing, shelter, housing;
• Enabling peer support and the inclusion of people with lived experience in all aspects of care;
• Increasing access to low-barrier treatment services, including access to healthcare and oral
health services;
• Ensuring access to medication and treatment on-demand, including mobile buprenorphine and
methadone;
• Expanding telehealth and addressing low technology literacy;
• Ensuring coordination of care for individuals leaving carceral settings.
Technological Approaches to Decrease Stigma Associated with Substance Use and Addiction
Stigma is understood as a socially constructed phenomenon that occurs when members of a group
experience status loss or discrimination based on some shared characteristic that is deemed undesirable
by others. Its effects can occur through attitudes and beliefs internalized by impacted individuals (self-
stigma), through overt discrimination by others (experienced or enacted stigma), and through the fear of
such discrimination (felt stigma). The stigma around substance use and addiction represents a signific ant
public health problem, despite the growing understanding that substance use and addiction are complex
brain disorders with behavioral and physiological components. As for other disorders, medical care is
often necessary to facilitate recovery and prevent adverse outcomes, including overdose. Patients can
recover from substance use and addiction and lead healthy lives; however, stigma limits successful
access to care. Stigma often may be related to multiple conditions, such as SUD, mental illness, or
infectious disease; behaviors such as specific drug use practices (e.g., opioid injection); or identity
statuses related to gender, sexual orientation, sexual identity, race/ethnicity, or socioeconomic factors,
such as personal income. It is expected that leveraging state-of-the-art technologies and the latest
science will allow to develop and commercialize the products and services aimed at reducing the stigma
around substance use and addiction.
Applications in this topic may propose projects demonstrating how latest technology and evidence-based
science could meaningfully reduce the stigma associated with substance use and addiction. Applications
may address individual (internalized, anticipated, or enacted), interpersonal, organizational, and/or
structural levels of stigma.
Applications and focus can be on any entry point along the continuum of care. Areas of specific research
interest and substance use and addiction service contexts include, but are not limited to:
• Providing anti-stigma training for medical professionals;
• Targeting stigma reduction of non-medical providers (social workers, criminal justice, family
members, and educators);
• Enhancing both employee well-being and effectiveness of a drug-free and stigma-free workplace
program;
• Anti-stigma training specific to adolescent substance use and prevention;
• Digital certification program for nonprofessional care givers who provide support services for
patients with SUD;
• Virtual employee assistance programs with focus on SUD and mental health.
Additionally, examples of technological approaches include, but are not limited to:
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 70
• Natural language processing, computer vision, and other machine learning tools to detect and
analyze provider behaviors and medical records reflecting stigma around substance use and
addiction alone and intersectional stigma;
• Digital compassion (anti-stigma) coaching for medical professionals delivering treatment to SUD
patients exploring immersive technologies such as extended reality;
• Ecological momentary sampling and other digital phenotyping patient-centered tools to detect
points of vulnerability and counteract internal stigma supporting the whole-person model of
recovery;
• Neural activity-based tools and services to help develop and disseminate the most effective anti-
stigma campaign.
Prevention Technology to Address Substance Use and Addiction in Various Underserved
Populations
Differences in race, socioeconomic status, sex, and geography have created inequities in care for
substance use and addiction. Alternatives in healthcare that emerged during the pandemic, such as virtual
doctor visits, along with new tools to facilitate telehealth, may help address some of the barriers to SUD
care for currently underserved populations.
There are three categories for which prevention technology can address substance use and addiction in
various underserved populations despite the aforementioned differences. Primary prevention provides
tools to intervene before health effects occur. Secondary prevention provides screening tools to identify
diseases in the earliest stages before the onset of signs and symptoms. Tertiary prevention provides tools
to manage disease post-diagnosis to slow or stop disease progression.
Applications may address micro- (individual, internalized, anticipated, or enacted), or macro-
(interpersonal, organizational, and/or structural) levels of health inequities related to various underserved
populations. The focus of applications can be on any entry point along the continuum of substance use
and addiction care. Examples include:
• Primary prevention through measures such as altering risky behaviors (cannabis and/or tobacco
use), and banning substances known to be associated with a disease or health condition.
• Secondary prevention through measures such as SBIRT (Screening, Brief Intervention, Referral
to Treatment).
• Tertiary prevention through measures such as rehabilitation, and medication assisted therapy.
Areas of specific research interest with respect to substance use and addiction service contexts include,
but are not limited to:
• Providing prevention education and behavior change training for medical professionals; targeting
knowledge awareness and behavior change for non-medical providers (social workers, criminal
justice, family members, and educators);
• Digital or non-digital behavior change interventions enhancing both employee well-being and
effectiveness of a drug-free workplace program;
• Digital or non-digital prevention education training specific to adolescent substance use and
prevention;
Additionally, examples of technological approaches include, but are not limited to:
• Machine learning tools (e.g., natural language processing) for provider/medical record substance
use and addiction bias;
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDA 71
• Digital mindfulness coaching for medical professionals delivering substance use and addiction
services with virtual reality;
• Ecological momentary sampling and other digital phenotyping patient-centered tools to detect
points of vulnerability and address them with a whole-person model of recovery.
Contact Information
Prior to Submission: Applicants are strongly encouraged to request a technical assistance meeting with
NIDA SBIR/STTR staff prior to submitting any application. To schedule a meeting, please email:
Ernestine Lenteu
Program Specialist
NIDA Office of Translational Initiatives and Program Innovations
NIDASBIR@mail.nih.gov.
For additional information on research topics, please contact a Program Officer with the relevant
area of expertise:
Leonardo Angelone, PhD
Program Officer
Email: leonardo.angelone@nih.gov
Areas of expertise: FDA-regulated therapeutic and diagnostic devices for substance use and addiction.
Morris Flood, DHSc
Program Officer
Email: morris.flood@nih.gov
Areas of expertise: Consumer products and approaches that address the health-related social needs that
impact substance use and addiction; and digital health technology translation to commercial substance
use and addiction products that address the overall social determinants of health.
Stacie Gutowski, PhD
Program Officer Email: stacie.gutowski@nih.gov
Areas of expertise: FDA-regulated therapeutic and diagnostic devices for substance use and drug
addiction, including neuromodulation.
Saravanan Karuppagounder, PhD
Program Officer
Email: saravanan.karuppagounder@nih.gov
Areas of expertise: Therapeutics for Stimulant Use Disorders and Marijuana or Cannabis Use Disorder,
Biomarker Development and Validation.
Yordan Kostov, PhD
Program Officer
Email: yordan.kostov@nih.gov
Areas of expertise: FDA-regulated monitoring and diagnostic devices for substance use and addiction,
including target-agnostic detection.
Tam Nguyen, PhD
Program Officer
Email: tam.nguyen@nih.gov
Areas of expertise: Pharmacotherapeutics for opioid use disorder, overdose reversal, and smoking
cessation.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCD 73
NATIONAL INSTITUTE ON DEAFNESS AND OTHER COMMUNICATION
DISORDERS (NIDCD)
Mission
The NIDCD supports research on the normal mechanisms of, as well as on diseases and
disorders of hearing, balance, smell, taste, voice, speech and language. The Institute also
supports research related to disease prevention and health promotion. The NIDCD addresses
special biomedical and behavioral problems associated with people who have communication
impairments or disorders. The NIDCD also supports efforts to create and refine devices, as well
as develop cellular-based applications that may replace or substitute for lost and impaired
sensory and communication functions. For more information about areas of interest, please visit
our home page at http://www.nidcd.nih.gov/ and the NIDCD Strategic Plan website
(https://www.nidcd.nih.gov/about/strategic-plans). Potential applicants are encouraged to
contact the program staff noted below early in the process of preparing the application.
Budget Guidance
Total funding (direct costs, indirect costs, fees) normally may not exceed the amounts defined by
the SBA, which can be found on the NIH SEED website. Applicants considering a requested
budget greater than these limits are strongly encouraged to contact program staff before
submitting the application. The Small Business Administration has allowed NIDCD to make
awards that exceed these amounts for the areas noted in the SBA-Approved Waiver Topics. For
topics listed in the SBA-Approved Waiver Topics, the NIDCD generally will not fund Phase I
applications greater than $385,000 total costs or Phase II applications greater than $3,000,000
total costs. All applications must contain sufficient detail to justify the requested budget, and
NIDCD may decrease the length of an award and/or the budget as recommended by a review
committee or administrative review.
Specific SBIR and STTR Program Information
NIDCD will accept applications for support of clinical trials in any of the areas noted above.
NIDCD does not accept applications for Phase IIB competing renewal awards.
NIDCD participates in the SBIR/STTR Commercialization Readiness Pilot (CRP) Program.
Clinical Trials
Does NIDCD accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
Yes
Does NIDCD accept Clinical Trials
through specific Notices of Funding
Opportunities?
No
Does NIDCD support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
NIDCD accepts and supports non-SBIR/STTR
clinical trial applications through specific
opportunities, which can be found on the
NIDCD Funding Opportunities webpage:
https://www.nidcd.nih.gov/research/clinical-
studies/researchers-professionals/know-what- is-
available

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCD 74
Research Topics
The NIDCD accepts a broad range of small business applications that are significant,
innovative, and relevant to its mission. Some examples of research topics within the NIDCD
mission areas include topics shown below; further example can be found on the NIDCD
Strategic Plan website (https://www.nidcd.nih.gov/about/strategic-plans).
Priority is given to meritorious applications that are likely to develop innovative technologies, provide
clear evidence of effectiveness, and bring novel products to the commercial marketplace.
Hearing and Balance Program
Development of treatment modalities to prevent or lessen the effects of hearing disorders;
development of new hearing aids, over the counter hearing aids, cochlear implants, and other
assistive devices; development of improved screening technologies to assess hearing loss, in
adults as well as in neonates and infants. Development of technologies that provide self-fitting,
self-adjusting, or other features that increase performance, accessibility, or affordability of
hearing aids; development of new outcome measures for assessing the efficacy of treatments for
hearing disorders. Development of technologies for the diagnosis and treatment of tinnitus.
Development of technologies for the diagnosis and treatment of otitis media including non-
invasive diagnostics to identify middle ear pathogens, novel antibacterial strategies, and
prophylactic anti-microbial strategies. Development of technologies for the study, diagnosis and
treatment of noise-induced and age-related hearing loss.
Development of technologies for the study, diagnosis and treatment of balance disorders,
particularly for the elderly; development of clinical tests and instruments to assess
balance/vestibular function; development of instruments and tests measuring head stability and
vestibular function during natural stimulation of the vestibular system; development of perceptual
reporting techniques and psychological indices for clinical assessment of the balance-disordered
patient; development of tests and new outcome measures for assessing the efficacy of physical
rehabilitative regimens for balance disorders; and development of assistive devices for balance
disorders, including neural prostheses for the vestibular system.
Development of new research tools to aid in the study of the auditory and/or balance systems
that can provide an improved understanding of fluctuating patterns of neural circuit structure and
function over time and across large assemblies of neurons; new animal models of impaired
function; improved diagnostic tools for inner ear function, including DNA-based assays and
biochemical markers of disease; innovative tests and instruments to screen for and diagnose
inner ear function. Development of technologies to enable gene transfer to the inner ear,
including viral vectors and cell type specific markers and probes to examine cell lineage in inner
ear regeneration. Development of innovative in vivo imaging capabilities to significantly advance
visualization, diagnosis, and treatment of disorders in the clinic.
Voice, Speech, and Language Programs
NIDCD is interested in the development of technologies for the study of communication
disorders: nature, causes, diagnosis, treatment, and prevention. These communication
disorders include but are not limited to: aphasia, apraxia, developmental language disorders,
dysarthria, dysphonia, and stuttering. In addition, research is needed for communication
challenges that may accompany individuals with autism, deafness, hearing loss, or the inability
to rely on spoken language as a primary means of communication. NIDCD is particularly
interested in projects that employ a user-centered design or similar approach that engages the
target end user throughout the development and research process. In addition, these
technologies should be accessible to culturally and linguistically diverse populations.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCD 75
The emphasis of responsive projects may include the development of technologies such as:
augmentative and alternative communication (AAC) devices; assistive device enhancements
that better simulate natural speech (e.g. age, gender, emotion); brain computer interface (BCI)
communication prosthesis; mobile health applications; gender affirming voice care; flexible and
adaptable treatment delivery systems or intervention protocols that can be easily tailored to the
needs of an individual; improved artificial larynges and tracheoesophageal shunts; artificial
intelligence computer models that simulate normal and disordered communication;
virtual/augmented reality approaches to treatment; and technologies that assist in the access to
or delivery of healthcare during a public health crisis.
Taste and Smell Program
Development of easily administered diagnostic tools for testing human chemosensory function
throughout the lifespan; development of intervention strategies and targeted drugs for the
treatment of taste and smell disorders; preventive measures to limit the harmful effects of
infections, airborne toxins, radiation, chemotherapy and other drugs on chemosensory function;
novel therapies to stimulate regeneration of mature sensory neurons in damaged and/or aged
tissue; development of biomarkers for neurodegenerative disease; development of tools to
facilitate chemosensory research including improved neuroimaging techniques and visualization
at structural and cellular levels.
Contact Information
For administrative and business management questions,
contact:
Ms. Samantha Tempchin
Grants Management Officer
National Institute on Deafness and Other Communication
Disorders 301-435-0713, Fax: 301-451-5370
Email: tempchins@nidcd.nih.gov
For additional information on the following research topics, contact:
Hearing and Balance, BCI Communication Prostheses, Taste and Smell Programs
Roger L. Miller, Ph.D.
National Institute on Deafness and Other Communication
Disorders 301-402-3458
Email: roger.miller@nih.gov
Language Program
Holly L. Storkel, Ph.D.
National Institute on Deafness and Other Communication
Disorders 301-451-6842, Fax: 301-402-0390
Email: holly.storkel@nih.gov
Voice & Speech Program
Lisa M. Kopf, Ph.D.
National Institute on Deafness and Other Communication
Disorders 240-678-2544, Fax: 301-402-0390
Email: lisa.kopf@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCR 76
NATIONAL INSTITUTE OF DENTAL AND CRANIOFACIAL RESEARCH
(NIDCR)
Mission
The NIDCR advances fundamental knowledge about dental, oral, and craniofacial (DOC) health
and disease, and translates these findings into prevention, early detection, and treatment
strategies that improve overall health for all individuals and communities across the lifespan. For
more specific information about areas of interest to the NIDCR, please visit our home page at
http://www.nidcr.nih.gov.
NIDCR’s small business programs are highly focused on maximizing translational science
opportunities – moving rapidly and translating basic dental and orofacial discoveries into useful
products.
Budget Guidance
Total funding (direct costs, indirect costs, fees) normally may not exceed the amounts defined by
the SBA, which can be found on the NIH SEED website. Applicants considering a requested budget
greater than these limits are strongly encouraged to contact program staff before submitting the
application. The Small Business Administration has allowed NIDCD to make awards that exceed
these amounts for the areas noted in the SBA-Approved Waiver Topics. All applications must
contain sufficient detail to justify the requested budget, and NIDCR may decrease the length of an
award and/or the budget as recommended by a review committee or administrative review.
Specific SBIR and STTR Program Information
Special Statement regarding clinical trials:
NIDCR does not support clinical trials through SBIR/STTR programs. Small business concerns
proposing a clinical trial must use the UG3/UH3 program. Projects seeking to propose technology
validation studies within SBIR/STTR applications that involve human subjects research must
provide a detailed justification describing that the funds available through these awards can
adequately support the proposed human subject study especially if the study is testing a drug,
device or biologic in support of an investigational new drug (IND) or investigational device (IDE)
application.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NIDCR does not accept Phase IIB renewal applications. NIDCR participates in the CRP program but
does not support clinical trials through the CRP program.
Clinical Trials
Does NIDCR accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
No
Does NIDCR accept Clinical Trials
through specific Notices of Funding
Opportunities?
No

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCR 77
Does NIDCR support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
NIDCR Clinical Trial Planning and
Implementation Cooperative Agreement
(UG3/UH3 Clinical Trial Required) and
subsequent reissues
NIDCR Behavioral and Social Intervention
Clinical Trial Planning and Implementation
Cooperative Agreement (UG3/UH3 Clinical
Trial Required) and subsequent reissues
Research Topics
Translational Developmental Biology and Mammalian Genetics and Genomics
Priorities are to 1) understand the development of craniofacial complex and 2) elucidate the
mechanisms underlying dental, oral, and craniofacial (DOC) conditions and disorders. The ultimate
goal is to enable early prevention, diagnoses, and treatments of DOC conditions and disorders on
individual basis. Interests in this area include but are not limited to:
A.
Develop advanced assays and reagents that allow robust and scalable throughput to
genetically engineer and functionally characterize organisms in craniofacial development
and genetics studies.
B.
Develop novel or improved methods and devices that are minimally or non-invasive, cost
effective, and sensitive, for early detection of DOC conditions and disorders using
imaging, multi-omic, and other state-of-the art technologies and approaches. Methods
and devices suitable for Point of Care, at home, and telemedicine uses are encouraged.
C.
Develop methods that are applicable to early treatments (as early as in utero or
perinatal, or at a later developmental stage) for human DOC conditions and disorders.
Translational Dental, Oral, and Craniofacial Data Science
Priorities are to 1) maximize the utility of Big Data to accelerate DOC research and 2) better
enable evidence-based and data science-driven clinical practices. Interests in this area include but
are not limited to:
A.
Develop advanced analytics to retrieve diverse, multi-dimensional data from data
repositories, knowledgebases, literature, electronic health/dental/medical/records, and
other sources, and infer relations between data elements to inform basic and clinical
DOC research. Machine Learning/Deep Learning/Artificial Intelligence (ML/DL/AI) and
natural language processing tools are considered highly relevant.
B.
Develop phenotyping, data curation, and data analysis web interfaces for clinicians to
support clinical decision making.
C.
Develop devices, including those for Point of Care, at home, and telemedicine uses, for the
diagnoses of DOC conditions and disorders. Examples include, but are not limited to,
imaging and AI based facile devices.
Infectious Diseases and Immunity
Research relating to the etiology, pathogenesis, prevention, diagnosis and treatment of infectious
diseases of the oral cavity is supported by the NIDCR. This includes research on practical ways to
effectively use the host immune system to prevent or treat oral infectious diseases and microbial-
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCR 78
induced inflammation. Infectious diseases of the oral cavity include caries, periodontitis, candidiasis,
peri- implantitis, pulpitis, and various viral, bacterial, and fungal infections of the oral mucosa and
research on the diagnosis and prevention of oral manifestations and malignancies of HIV infection
and AIDS. Specific examples of technology development needs include but are not limited to:
A.
Develop ways to overcome or eliminate the risk of oral infections in persons who smoke
or chew tobacco, drink alcohol, or are immunosuppressed, have diabetes, are
malnourished, or are psychologically stressed.
B.
Explore novel methods or agents to eradicate oral biofilms (dental plaque) on teeth, oral soft
tissues, and dental implants without adversely affecting the normal oral flora.
C.
Isolate, synthesize or prepare new antibiotics and antimicrobial agents that can overcome
bacterial and fungal resistance to current compounds. Formulate combinatorial drug
regimens to attack microbes growing in oral biofilms (dental plaque).
D.
Develop controlled release systems for local delivery of synthetic peptides, recombinant
proteins, or other chemical or immunotherapeutic agents to prevent, control, and/or treat
oral infectious diseases, or the oral manifestations of HIV infection.
E.
Develop biological response modifiers or other immunological approaches to reduce or
eliminate microbial-induced chronic inflammation or the tissue destruction associated
with chronic inflammation in the oral cavity.
F.
Develop ways to interfere with microbial colonization and growth through the use of
antimicrobial agents and chemotherapy.
G.
Identify and exploit the structural features of oral biofilms for increased therapeutics delivery.
H.
Develop computer programs and apply systems biology approaches to model
biologically active peptide regions of oral components that have anti-fungal, anti-
bacterial and anti-viral activities.
I.
Develop substitutes of naturally occurring chemicals (phytochemicals) known to have
a role in controlling opportunistic infections induced by HIV.
J.
Develop synthetic peptides and recombinant proteins of oral components with anti-
fungal, anti- bacterial and anti-viral activities including those against HIV and oral
opportunistic pathogens.
K.
Develop oral topical formulations with combined microbicide, analgesic, and anti-
inflammatory activities to enhance oral mucosal defenses and prevent and/or control oral
infections and lesions in HIV-infected and/or immunosuppressed subjects.
L.
Discover, test, standardize, and validate novel biomarkers present in oral
biospecimens for screening and clinical diagnosis of HIV, and oral opportunistic
pathogens infections and AIDS malignancies. Apply similar strategies as listed below
for oral, oropharyngeal and salivary gland cancers to AIDS malignancies.
M.
Develop the next generation of rapid tests and point of care devices to detect, quantify,
screen, and diagnose HIV and oral opportunistic pathogens. Develop novel assays to
quantify oral mucosal reservoirs for oral viruses, oral immune responses to viral
prophylactic and therapeutic vaccines, and viral changes due to anti-viral treatments.
N.
Develop safe and effective targeted diagnostic and therapeutic technologies in
response to endemic and pandemic infections.
Oral, Oropharyngeal and Salivary Gland Cancers
Emphasis is on molecular mechanisms of oral epithelial cell deregulation that lead to oral
cancers. Research related to early detection, diagnosis, and prevention, and treatment of oral
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCR 79
cancers is of particular interest. Examples include but are not limited to the following areas:
A.
Develop imaging techniques for the early detection, diagnosis and prognosis of pre-
malignant lesions.
B.
Develop effective pharmacological, immunological and radiological modalities for
treatment of pre- malignant and malignant lesions in preclinical models.
C.
Develop novel technologies for the genetic and molecular-targeted therapy (e.g. siRNAs,
peptide- based therapies) in preclinical models.
D.
Develop genetic animal models of oral cancer premalignancy and oral cancer progression
that mimic human oral cancers, including HPV-associated oropharyngeal cancers.
E.
Develop animal models to facilitate the testing of therapeutic and chemopreventive
agents for oral cancers.
Temporomandibular Disorders and Orofacial Pain
Emphasis is on research for chronic disabling painful diseases of the oral-craniofacial-dental areas
including chronic pain, neuropathies, and diseases of the temporomandibular joint. NIDCR
encourages applications that include but are not limited to:
A.
Develop improved methods and technologies for measuring nociceptive,
chemosensory, tactile, kinesthetic, or proprioceptive function involving craniofacial
structures. Such measures may be useful in screening for deficits, improving
diagnosis, or for evaluating responses to orofacial treatments or interventions.
B.
Develop improved biomarkers for neuropathic pain conditions affecting oral-craniofacial
tissues or structures.
C.
Develop assays facilitating reliable evaluations of relationships between biological and
other risk factors as they relate to onset, and exacerbation of pain and for examining
transition from acute pain to chronic pain conditions.
D.
Identify and develop novel pharmacologic or biological agents, and non-pharmacologic
methods/approaches, including but not limited to small molecules, peptides,
recombinant proteins, nucleic acids, electrical stimulation, and others which could
be grouped broadly to electromagnetic induction to modulate mood/nerves, to
prevent, control, and/or treat orofacial pain.
E.
Develop animal models to facilitate testing of therapeutic agents for orofacial pain.
Saliva, Salivary Diagnostics, and Salivary Gland Diseases
Emphasis is on salivary gland physiology and pathophysiology and in the repair and restoration of the
damaged gland. Examples include but are not limited to:
A.
Develop viral, non-viral and gene therapy-based approaches to address compromised
salivary gland function. Develop cell and tissue-based strategies and technologies for
restoration of damaged or destroyed salivary gland function.
B.
Develop novel compounds or materials that protect and preserve salivary glands from
head and neck cancer irradiation therapy.
C.
Develop non-invasive methods for the determination of efficacy and safety of
artificial saliva, sialogogues, and their delivery vehicles used in addressing the
diminution or lack of saliva (xerostomia) due to Sjögren’s Syndrome or head and
neck cancer irradiation therapy.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCR 80
D.
Develop biomarker-based technologies for the identification of Sjögren’s Syndrome using
blood or saliva as body fluids.
E.
Identify biomarkers derived from oral fluids that are predictive of the onset,
progression and recurrence of oral diseases and conditions, such as periodontal
diseases, caries, and oral, oropharyngeal and salivary gland cancers.
F.
Develop immunological strategies and immunotherapy-based approaches for addressing
xerostomia from Sjögren’s Syndrome.
G.
Improve existing or develop new tools for early detection of salivary gland cancers.
Biotechnology, Biomaterials, and Applications for Regeneration and Restoration of Oral, Dental
and Craniofacial Tissues
Emphasis is placed on the development of a broad range of technologies targeted at regeneration
and restoration of diseased and injured hard and soft tissues of the oral and craniofacial complex
and on translating these applications to the clinic. Tissues of interest include craniofacial and
alveolar bone, the periodontal ligament, TMJ bone and cartilage, oral mucosa, facial skeletal
muscle, vasculature and nerves. Also of interest are multi-tissue composites and organs, such as
vascularized and innervated bone and muscle, salivary gland, tooth, periodontium, bone-
periodontal ligament-cementum interface and osteochondral complexes. Specific topics could
include but are not limited to:
A.
Develop technologies for design, fabrication, and manufacturing of biomimetic and
biocompatible biomaterials and a range of structurally complex scaffolds, including
nanomaterials and self-assembling nano-scaffolds, for tissue engineering and
regenerative medicine applications. Projects need to include assessments
demonstrating the ability of biomaterials and scaffolds to support generation and
regeneration of mineralized tissues that replicate the mechanical, physical and biological
properties of dentin, enamel, cementum, or bone.
B.
Develop cell-based technologies, including stem cell-based technologies. These include,
designing strategies for isolation, purification, differentiation, scaled up production,
manufacturing, standardization and quality control of stem and progenitor cells and their
differentiated progenies, derivation of efficient and predictable methodologies for cellular
reprogramming, and advancing technologies for reconstruction of stem cell niches for
augmenting tissue regeneration.
C.
Develop bioreactor systems to facilitate design, fabrication, and manufacturing of soft and
hard tissues of dental, oral and craniofacial complex. These bioreactors may be able to
mimic biophysical forces, such as mechanical and electrical forces that normally guide
tissue morphogenesis in vivo. Among other desirable features of the bioreactors are
maintenance of tissue construct oxygenation and real-time tissue assessment to
encompass metabolites, gene expression, or proteomics evaluation, in addition to
morphology and spatial imaging by labeling capabilities.
D.
Develop improved dental composite materials and bonding agents, including biomimetic
and self-healing materials and adhesive sealants. These include but are not limited to
materials to replace Bis-GMA resin-based systems that are suitable for restoring crowns of
posterior teeth and exposed roots of the teeth. Any novel dental composite restorative
components or systems must include assessments in a physiologically relevant test
system that mimics microbial and physicochemical conditions found in the oral cavity.
E.
Develop methods, materials, and devices for orthodontic, prosthetic, periodontic,
endodontic and craniofacial applications including those that can be used for craniofacial
bone distraction, reconstruction, hard and soft craniofacial tissue healing and regeneration,
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCR 81
and scarless craniofacial tissue repair.
F.
Develop miniaturized artificial tissue and organ mimics/tissue chips and organoids that can
be adapted to high-throughput formats for a broad range of applications, such as analysis of
biomaterial and tissue function, drug efficacy and toxicology assays, biocompatibility
assays, genetic screening and elucidating mechanisms of dental, oral and craniofacial
development and disease.
G.
Develop mathematical, computational, and bioinformatics approaches for modeling oral
and craniofacial tissues and organ function and physiology to address needs of systems
biology, synthetic biology, and single cell analysis. Develop new approaches for utilizing
novel biomolecules, including growth factors, cytokines, small molecules, siRNAs, and
others for counteracting diseases and injuries of oral and craniofacial tissues and
promoting their healing and regeneration.
H.
Develop new approaches to study molecular or cellular interactions between hard and soft
tissues such as between the nervous system and mineralized tissues. Approaches can
include development of new technologies or application of existing technologies that are
newly applied to the dental and craniofacial field.
I.
Develop advanced viral and non-viral based biomolecule delivery approaches, including
nanotechnology-based technologies that can precisely deliver and release therapeutic
proteins, nucleic acids, small molecules, or combinations thereof with predictable temporal
kinetics to target specific tissue sites.
J.
Develop imagining diagnostics to accelerate clinical implementation of reliable,
reproducible, highly specific and sensitive diagnostic instruments for various applications,
including but not limited to dental caries, cracked teeth, pulp vitality, bone quality, and
periodontal disease.
K.
Develop imaging diagnostics to accelerate clinical implementation of reliable, reproducible,
highly specific and sensitive diagnostic instruments for various applications, including but
not limited to dental caries, cracked teeth, pulp vitality, bone quality, and periodontal
disease.
L.
Develop safe and effective biosensors for noninvasive, dynamic real-time monitoring of
physiological processes in the human body using the oral cavity as the sensing site. These
biosensors will be able to assess health and disease states and receive feedback from body
fluids and clinical compounds that are found in or pass through the oral cavity and in certain
cases, will be able to communicate these outputs wirelessly and remotely.
M.
Develop safe and effective biosensors, monitoring devices and systems, data driven and
computer science tools for automated detection, diagnosis and treatment of dental, oral and
craniofacial disease.
N.
Develop effective multimodal breakthrough technologies for real-time detection of proven
disease biomarkers, viruses and/or pathogens with high sensitivity and specificity that
integrate detection technologies, such as optical spectroscopy, electrical impedance, radio
frequency, acoustic, and immunosensing, with multiplexing capability.
Preclinical Research
A.
Preclinical research and development activities for dental and craniofacial technologies
including the translation of innovations devices, drugs, biologic and combination products
(reconstructive materials, regenerative products, pharmaceuticals, therapeutics, vaccines,
digital health technologies) that require review and approval by the FDA as a regulated
product before commercial distribution.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCR 82
Biomedical Clinical Research
Emphasis is on development of methods, drugs and materials to diagnose or treat oral and craniofacial
diseases and conditions. Areas of interest include but are not limited to projects that:
A.
Develop improved methods to detect and predict progression of dental caries, periodontal
disease, reversible and irreversible pulpitis.
B.
Develop improved methods or materials to prevent dental, oral, and craniofacial diseases or
conditions.
C.
Develop new or improved methods or materials to enhance oral and craniofacial surgery.
This would include both intraoral and extra-oral surgery.
D.
Develop improved methods or materials to mechanically and/or biologically repair or treat
tooth structure damaged by dental caries or periodontal disease.
E.
Develop, customize, and validate data-driven technologies coupled with automated high
throughput tools that accelerate development and regulatory evaluation of novel
biomaterials.
F.
Develop improved appliances to aid suckling and improve speech production by newborn infants
with cleft palate and cleft lip.
G.
Develop safe and efficacious methods to diagnose caries, pulp vitality and / or periodontal
diseases utilizing non-ionizing radiation.
H.
Develop technologies for local delivery of drugs to treat oral and craniofacial diseases or
disorders.
I.
Develop novel non-opioid pharmacological medications for management of acute dental pain.
J.
Develop safe and efficacious methods or medications to manage complications of head and
neck cancer treatment.
K.
Develop tools for implementation of precision medicine in the oral cavity.
L.
Develop methods and tools to detect soft tissue pathologies in the oral cavity.
M.
Develop oral devices and materials for monitoring local and systemic conditions.
Behavioral Clinical Research
Provides support for the development of evidence-based products related to behavioral and
social aspects of oral health, oral health prevention or treatment interventions, and other patient-
oriented aspects of oral health. This includes support for clinical trials and patient-oriented
research to establish safety and initial efficacy of products. NIDCR is especially interested in
applications that significantly improve oral health by 1) being broadly applicable to many
populations, 2) contributing to meaningful oral health improvements for a specific population, 3)
expediting translation of research findings into oral health improvements, and/or 4) equipping oral
health care providers, educators or researchers with tools to improve public oral health.
Examples of studies of interest include, but are not limited to, the following:
A.
Develop and test the effectiveness of innovative teaching or educational tools or curricula to
inform oral health professionals and dental students regarding the role of genetics and
genomics, including the oral microbiome, in oral diseases, conditions and oral health care;
and/or oral cancer prevention and early detection.
B.
Develop and test digital health, connected technologies, and approaches to improve time-
sampled monitoring of behavioral adherence with preventive, condition management, or
therapeutic regimens specifically relevant to oral diseases/conditions. Such devices or

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDCR 83
methods could be utilized in a variety of settings, including naturalistic settings, within
clinical trials, within oral health care delivery systems, etc.
C.
Develop and test novel compliance and survey measures or tools to identify the underlying
causes of insufficient preventive dentistry for specific underserved populations.
D.
Develop and test for safety, efficacy, and/or effectiveness of measures or materials for
diagnosing, preventing, or treating oral, dental, and craniofacial conditions and disorders.
E.
Develop or adapt for use in new populations or settings: Novel measures or methods for
identifying individual, family, group, or other processes that explain oral health behavior;
oral health interventions utilizing technology to improve efficiency of delivery (e.g.,
management of chronic pain related to temporomandibular joint disorders, etc.);
interventions addressing health behaviors highly associated with oral health (e.g., tobacco,
alcohol, and other drug use; management of diabetes, HIV infection, or other chronic
illnesses; etc.).
F.
Develop and test innovative methods for facilitating collaborations, referrals, and/or ongoing
follow- ups between oral health professionals and other health care professionals across
primary and specialty practices.
G.
Develop technologies or modules beyond existing web-based platforms to improve
preventive oral health hygiene for children and adolescents (e.g., social marketing via app-
and web- based interaction, virtual reality “worlds”, “massively multiplayer online games”,
etc.).
H.
Develop and test web-based training or other innovative approaches for oral health care
professionals to accelerate accurate translation of new knowledge regarding oral diseases
and their effective prevention or treatment into clinical or public health practice.
Contact Information
For additional information on research topics,
contact: Orlando Lopez, Ph.D.
Dental and Craniofacial Small Business Coordinator
Division of Extramural Research
National Institute of Dental and Craniofacial Research
NIH
6701 Democracy Blvd., Rm. 612
Bethesda, MD 20892-4878
(301) 402-4243
Email: orlando.lopez@nih.gov
For administrative and business management questions,
contact: Gabriel Hidalgo, MBA
Acting Chief Grants Management Officer
National Institute of Dental and Craniofacial Research
6701 Democracy Blvd., Rm. 656
Bethesda, MD 20892-4878
(301) 827-4630
Email: gabriel.hidalgo@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 84
NATIONAL INSTITUTE OF DIABETES AND DIGESTIVE AND KIDNEY
DISEASES (NIDDK)
Mission
The mission of the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) is to
conduct and support medical research and research training and to disseminate science-based
information on diabetes and other endocrine and metabolic diseases; digestive diseases, nutritional
disorders, and obesity; and kidney, urologic, and hematologic diseases, to improve people’s health
and quality of life. For additional information about areas of interest to the NIDDK, please visit our
home page at http://www.niddk.nih.gov. See our SBIR/STTR page at
https://www.niddk.nih.gov/research-funding/research-programs/small-business.
Please also find the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK)
Strategic Plan for Research, published in December 2021, at https://www.niddk.nih.gov/about-
niddk/strategic-plans-reports/niddk-strategic-plan-for-research.
Budget Guidance
For budgetary, administrative, or programmatic reasons, the NIDDK may not fund an application or
may decrease the length of an award and/or the budget recommended by a review committee.
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts defined
by the SBA, which can be found in the “Award Budget” section of the current Omnibus Solicitations.
With appropriate justification from the applicant, the NIDDK may consider budgets that exceed these
amounts to support research that aligns with an approved waiver topic (see SBA-Approved Waiver
Topics).
The NIDDK generally considers:
•
Phase I budgets up to $350,000 total costs or project periods up to 2 years.
•
Phase II applications up to $2,200,000 total costs or project periods up to 3 years
(Phase II budgets generally should not exceed $1,100,000 total costs in any
year).
•
Phase IIB (see below) applications up to $3,000,000 total costs or project periods up to
3 years (Phase IIB budgets generally should not exceed $1,100,000 total costs in any
year).
Applicants considering a requested budget greater than these limits are strongly encouraged to contact
program staff with a draft of their Specific Aims page before submitting an application.
The NIDDK also participates in the SBIR/STTR Commercialization Readiness Pilot (CRP) Program.
Applicants should review the Award Budget section of relevant Notices of Funding Opportunities. For
Phase II awardees, especially those developing products that require clinical evaluation or approval
by a Federal regulatory agency, the NIDDK strongly encourages potential applicants to apply to
NIDDK’s Phase IIB program.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 85
Specific SBIR and STTR Program Information
NIDDK will accept clinical trials in most of the areas within the mission of the NIDDK. NIDDK does
not support clinical trials in hematologic diseases.
Final Progress Reports
As detailed in NOT-OD-17-085, the NIH has implemented the Final Research Performance Progress
Reports (Final RPPR) for SBIR/STTR Final Progress Reports.
The NIDDK is interested in tracking the progress of the small business concerns it funds and the
products they develop. Funding priority will be given to those small business concerns that show not
only their ability to develop products but also their growth as a small business concern towards
independence from the SBIR/STTR program.
Additional Programs and Services for NIDDK SBIR/STTR Awardees
The NIDDK encourages awardees to apply to participate in programs NIH offers to support the
development of their products (https://seed.nih.gov/support-for-small-businesses). The NIDDK may
offer additional programs throughout the year, and awardees are encouraged to keep their contact
information current so that they receive announcements regarding these programs.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NIDDK will accept Phase IIB SBIR/STTR Competing Renewal grant applications (only) from NIDDK-
supported Phase II awardees that propose to continue the process of developing products that
ultimately require 1) clinical evaluation, 2) approval by a Federal regulatory agency, and 3)
continuing refinements to durable medical equipment (DME) designs such as cost reduction, testing
for safety, durability, and reliability, and meeting or establishing standards. This renewal grant should
allow small businesses to get to a stage where interest and investment by third parties is more likely.
Such products include, but are not limited to biological products, devices, drugs, medical implants,
etc. related to the mission of the NIDDK. These awards are intended to support completion of
research needed for an Investigational New Drug (IND) application or Investigational Device
Exemption (IDE). Applicants must provide evidence that they have consulted formally with the U.S.
the Food and Drug Administration (FDA) concerning the research needed for the development of a
drug, biologic or medical device and that the proposed research will address these regulatory
requirements. Such evidence should include FDA correspondence from a pre-IND meeting for an
IND application or a pre-IDE meeting for an IDE application, and the status of the project in a
timeline related to Federal regulatory approval processes. Applicants should describe these
consultations and the outcomes in the Progress Report section of the Research Strategy and
provide FDA correspondence or meeting minutes as part of Just-in-Time procedures.
Examples of research that would be considered responsive to this announcement are listed below for
illustrative purposes and are not exclusive of other appropriate activities.
•
Completion of studies as required by the FDA for an IND or Radioactive Drug Research
Committee (RDRC) application.
•
Assessment of devices with regard to performance standards related to the FDA approval process.
•
Clinical studies in support of an application for clearance or approval by the FDA. See the
table below and review NIDDK’s Policies for Clinical Researchers
(https://www.niddk.nih.gov/research- funding/human-subjects-research/policies-clinical-
researchers) when considering an application involving human subjects.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 86
The NIDDK also participates in the SBIR/STTR Commercialization Readiness Pilot (CRP) Program.
Please review CRP NOFOs for NIDDK’s participation. For Phase II awardees, especially those
developing products that require clinical evaluation or approval by a Federal regulatory agency, the
NIDDK strongly encourages potential applicants to apply to NIDDK’s Phase IIB program.
Clinical Trials
Does NIDDK accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
Yes
The NIDDK accepts SBIR, but not STTR
applications with NIH-defined clinical trials.
Does NIDDK accept Clinical Trials
through specific Notices of Funding
Opportunities?
No
Does NIDDK support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
Small businesses are eligible to apply for several
non-SBIR/STTR funding opportunities.
Comprehensive information on Human Subjects
Research at NIDDK can be found here:
https://www.niddk.nih.gov/research-
funding/human-subjects-research
The NIDDK has a R01 funding opportunity for
pilot & feasibility clinical trials and accepts low-
risk clinical trials within the mission of the
NIDDK through the parent NIH NOFOs for clinical
trials.
For high-risk clinical studies (see NOT-DK-20-
051: https://grants.nih.gov/grants/guide/notice-
files/NOT-DK-20-051.html) that involve more
than one research centers, NIDDK uses a two-
part process that usually requires use of a U34
planning grant followed by a separate application
for the clinical trial or study (U01). Potential
applicants are strongly encouraged to
contact NIDDK staff to discuss their proposed
study and determine the most appropriate
mechanism for submitting their application.
Clinical Trials Allowed and Optional funding
opportunities to which NIDDK is subscribed can
be found on NIDDK’s Current Funding
Opportunities page:
https://www.niddk.nih.gov/research-
funding/current-opportunities.
Research Topics
Diabetes, Endocrinology and Metabolic Diseases
The Division of Diabetes, Endocrinology and Metabolic Diseases supports SBIR/STTR projects in
the areas of type 1 and type 2 diabetes, endocrine disorders, and neuroendocrinology. High priority
topic areas are listed below:
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 87
II.
Sensors, Hormone Replacement, Delivery Devices, and Other Technologies for Diabetes
Treatment:
A.
Novel accurate, reliable, and user-friendly continuous monitoring sensor technologies
relevant to diabetes treatment and monitoring. Preferably, these sensors should have
long functional life, and for glucose sensing be accurate at all glycemic ranges,
particularly at concentrations below 54 mg/dl.
B.
Use of Artificial Intelligence, Machine Learning (AI/ML) tools to enable fully automated
closed loop pancreatic hormone delivery systems in response to multi-analyte
physiological input.
C.
Novel insulin and glucagon formulations showing improved kinetics and stability.
D.
Telemedicine/remote monitoring approaches that can be incorporated as
components/and or adjuvants of closed loop systems for better diabetes self -
management.
E.
Technologies that may promote and facilitate adherence/compliance by users of
diabetes monitoring and control devices.
F.
More reliable and efficient biocompatible infusion sets for automated hormone delivery
and improved kinetics.
G.
New implantable and easy to replace technologies that may mimic the beneficial effect
of gastric bypass/bariatric surgery for the treatment of diabetes without the need of a
major invasive surgical procedure.
III.
Diabetic Wound Healing and Diabetic Neuropathy:
A.
Drugs, biologic therapies, and novel delivery systems that accelerate healing of
diabetic foot ulcers and prevent recurrences.
B.
Off-loading devices that improve patient acceptability and adherence.
C.
Diagnostic and predictive biomarkers, including improved outcome measures, for
diabetic foot ulcers that can be used to diagnose biofilms, predict healing, select
treatment strategies, or determine risk of primary or secondary occurrence of foot
ulcers. The biomarkers may use biosamples, images or sensors.
D.
Educational approaches and new technologies that increase adherence to
preventative measures for diabetic foot ulcers in high-risk patients or increase
adherence to off-loading and other recommended treatment regimens for diabetic
foot ulcers.
E.
Disease-modifying therapies for the prevention and treatment of diabetic neuropathy.
F.
Sensors, algorithms, and patient interfaces that can provide feedback to diabetic
individuals with insensate feet to prevent diabetic foot ulcers.
G.
Biomarkers to monitor disease progression and response to therapy for diabetic
neuropathy, including peripheral sensory, autonomic, and painful diabetic
neuropathy.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 88
IV.
Immune Modulation and Cell Replacement Therapies:
A.
Development of immunomodulation/tolerance strategies, including cell-based, to
prevent, revert or slow progression of type 1 diabetes.
B.
Development and optimization of engineered islet cell replacement sources with
improved transplant graft attributes, including but not exclusive to: graft function
durability under transplantation and metabolic stress; graft survival with lowered or no
systemic immunosuppression and non-invasive quantitative monitoring of graft mass.
C.
Novel biomimetic and immuno-engineering strategies for the development of immune
evasive cells/islets and biomaterials/devices for successful long-term engraftment
with no need of systemic immunosuppression.
D.
Development of reproducible methods that improve yield/viability/function of
islets/insulin producing cells and allow their ex-vivo expansion for transplantation.
V.
Prediction, Screening, Diagnostics, and Monitoring:
A.
Development of methodologies, products, or biomarkers useful for predicting,
preventing or delaying progression of pre-diabetes or diabetes, including tests for
identifying patients at risk, and methods of monitoring disease progression.
B.
Validated tests for autoantibody detection, auto-reactive T-cells and other
immune/metabolic parameters for type 1 diabetes early diagnosis and monitoring.
Improvements could include higher throughput - point of care technologies (reliable,
accurate, cost-effective, highly sensitive, and standardized with rapid turnaround
time).
C.
Multiplexed assays for peptides and proteins that are used as biomarkers in diabetes
and metabolic diseases (e.g., insulin, pro-insulin, glucagon, c-peptide, HbA1c..etc).
D.
Development of non-invasive technologies such as imaging for the in vivo
measurement/evaluation of pancreatic islet’s cell mass, function and inflammation.
E.
Artificial Intelligence, Machine Learning, and Deep Learning driven methods and
technologies that may optimize prediction, diagnosis, monitoring and treatment of
diabetes, endocrine and metabolic disorders.
VI.
Pre-Clinical Research and Disease Modeling:
A.
Development and optimization of microphysiological/organ on chip platforms in the
application of pre-clinical testing and/or modeling of physiological and
pathophysiological aspects of diabetes, endocrine and metabolic disorders.
B.
Development of methods utilizing replenishable cell sources, that generate functional
islet like cells/tissues that can be successfully tested in microphysiological systems
and/or in vivo models of the disease.
C.
Development and testing of in silico/simulation models with predictive capability to
complement and/or replace in vitro and in vivo pre-clinical testing.
VII.
Tools for Measuring Peripheral Neurotransmitters and Neuromodulation:
A.
Devices that modulate or control the hepatic or pancreatic branches of the vagus
nerve with the aim of relieving diabetes or other metabolic disorders. Projects
concerned with the liver should be focused on the regulation of glucose or lipid
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 89
metabolism. Technologies would include closed- or open-loop neural stimulators of
sensory or motor nerves originating from or terminating in the endocrine pancreas or
liver.
B.
Tools that provide high spatio-temporal resolution of neurotransmitter release in the
endocrine pancreas or liver.
C.
Tools that measure autonomic activity in the liver, endocrine pancreas, or adipose
tissue in animal models or humans.
Digestive Diseases and Nutrition
The Division of Digestive Diseases and Nutrition supports research in diseases and disorders of
the digestive tract; esophagus, stomach, intestine, colon, anorectum, pancreas, liver, gallbladder,
and biliary tract; as well as research in nutrition and obesity. Innovative investigator-initiated
projects that are not mentioned below are also encouraged. Examples of areas that may be of
interest to small businesses include, but are not limited to:
I.
Gastrointestinal
A.
Development of new diagnostic techniques and tests, including non-invasive tests and
imaging for detecting Barrett’s esophagus, GERD, and other intestinal disorders.
B.
Development of agents and techniques to measure, diagnose, stimulate regeneration
of enteric neurons, and treat motility disorders.
C.
Development of novel therapies to modulate/enhance GI lymphatic function for the
treatment of GI pathologies.
D.
Development of gut-derived biomarkers of neurodegenerative brain disease.
E.
Development of approaches to simultaneously interrogate or modulate the central
nervous system (CNS) and the gastrointestinal system.
F.
Development and validation of neurotechnologies that improve the association of
symptoms, pathophysiology, and function for gastrointestinal disorders.
G.
Development of novel proteomic or metabolomic technologies designed to study
digestive diseases and their complications.
H.
Development of assays and screening methods for detection of biomarkers for
diagnosis, grading and staging digestive diseases.
I.
Development of Live Biotherapeutic Products (LBPs), such as probiotic organisms for
the prevention or treatment of gastrointestinal conditions, or to enhance the
nutritional properties of dietary components. These LBPs would not include vaccines,
oncolytic bacteria, or gene therapy agents.
II.
Liver
A.
Development of novel antifibrotic therapies for chronic progressive liver diseases.
B.
Development of quantitative tests of hepatic “reserve” for assessment of therapeutic
intervention, transplantation, or surgical risk in patients with liver disease.
C.
Development of point-of-care, serologic, and rapid tests for rapid diagnosis, treatment
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 90
requirements and genotyping of hepatitis.
D.
Development of rapid, reliable, and inexpensive tests for genetic screening and risk
markers important in liver disease.
E.
Development of sensitive and reliable non-invasive techniques to detect and monitor
liver fibrosis and other chronic liver diseases and the associated complications.
F.
Creation of bio-artificial organs for temporary hepatic support in patients with acute liver
failure.
III.
Pancreas
A.
Development of and validation of therapeutic interventions for treatment of pancreatitis
and its complications.
B.
Development of more accurate, non-invasive approaches to the diagnosis of chronic
pancreatitis by functional, radiologic, endoscopic, or pathologic/cytologic means.
IV.
Nutrition/Obesity
A.
Development of novel methods and tools to accurately evaluate nutritional status,
physical activity, and energy expenditure.
B.
Development of non- or minimally invasive technologies that allow access and/or
delivery to discrete regions of the digestive tract.
C.
Development of novel breath, urine, or blood tests to accurately measure dietary
intake.
D.
Development of non-invasive neurotechnologies to stimulate and/or modulate
hormone/peptide release from the gastrointestinal system for the treatment of
metabolic disorders such as obesity.
Kidney, Urologic and Hematologic Diseases
The Division of Kidney, Urologic, and Hematologic Diseases provides research funding and
support for basic, translational, and clinical research studies of the kidney, urinary tract, and
disorders of the blood and blood-forming organs. Projects may include development of tools to
improve understanding of the physiology, pathophysiology, and diseases of the kidney, urinary
tract, and blood and blood forming systems, or to develop rational diagnostics, treatments, and
prevention strategies for these diseases. Projects may be to develop tools/technologies to
support clinical care, population health and/or pragmatic research to improve health outcomes in
populations with kidney diseases and/or urologic conditions. Projects to develop tools or
technologies to address health disparities or promote health equity are encouraged. NIDDK
encourages research that takes a holistic perspective of human health by considering biological,
behavioral, and social contributors to the scientific exploration, prevention, and management of
these diseases/conditions. Development of -omics, bioinformatics, and multi-scale technologies
for the study of these systems, especially where these systems interact, is also encouraged.
Research opportunities that may be of interest to small businesses include, but are not limited to:
I.
KIDNEY DISEASES
Areas of research include chronic kidney disease, end-stage renal disease, diabetic kidney
disease, polycystic kidney disease, hypertensive kidney injury, acute kidney injury, kidney
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 91
donation (delayed graft function and chronic rejection), congenital kidney disorders,
glomerular and tubulointerstitial diseases, IgA nephropathy, hemolytic uremic syndrome, fluid
and electrolyte disorders, kidney repair and regeneration, and normal and abnormal kidney
development and physiology.
Dialysis, Devices and Medical Technologies
A.
Development of innovative forms of renal dialysis which improve efficiency, have
lower associated morbidity (e.g., tissue engineered artificial kidneys, implantable or
wearable dialyzers), reduce side effects and constraints of dialysis treatment, and/or
improve access, experience, and quality of life.
B.
Development of functional nephrons for transplantation.
C.
Development of pharmacological agents, devices, techniques, or diagnostics that
enhance maturation and longevity of a vascular access.
D.
Development of dialysis membrane technologies with enhanced biocompatibility and
anti- fouling properties.
E.
Development of a means to provide continuous anticoagulation to permit renal
replacement therapy.
F.
Development of reliable, non-invasive, wearable or online monitoring systems for real-
time assessment and adjustment of treatment parameters such as blood volume,
access flow, and urea clearance.
G.
Development of hemodialysis or peritoneal dialysis catheters using improved
biomaterials, which reduce catheter-related infections, the foreign body response,
biofouling, and biofilm formation.
H.
Development of novel methods to generate dialysate for hemodialysis or peritoneal
dialysis.
I.
Development of devices or techniques to enhance the long-term success of kidney
transplantation (e.g., techniques for repairing kidneys or for kidney storage and
preservation).
J.
Development of technologies to improve kidney biopsies (i.e., to improve safety or
tissue acquisition).
Health Information Technologies
K.
Development of health information technologies or mobile technologies that enhance
delivery of care, population health management, health equity, and/or research for
patients with kidney diseases.
L.
Development of applications or application programming interfaces that use health
data standards (e.g., Fast Healthcare Interoperability Resources [FHIR], clinical
terminologies) to improve accessibility, accuracy, and/or completeness of real-world
health, behavioral, and societal/contextual data for research and care of individuals
with kidney diseases.
M.
Development of technologies to engage patients with kidney diseases in their care or
to support interaction with caregivers.
N.
Development of innovative technologies or platforms to facilitate kidney research
training and education, which could include software or simulation tools.
Diagnostics and Imaging
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 92
O.
Development of clinical assays that enable biopsychosocial precision medicine
approaches to treating kidney diseases.
P.
Development of technologies that use artificial intelligence/machine learning (AI/ML)
or other advanced statistical approaches to integrate disparate data types to inform
diagnosis of kidney diseases. AI/ML approaches should leverage data from diverse
populations and apply equity considerations to ensure resulting models do not further
embed structural racism or discrimination.
Q.
Development of platforms for pre-analytical preparation, imaging, and automated
analysis of kidney tissue.
R.
Development of non- or minimally invasive methods for evaluating kidney functions,
including in individuals with congenital genitourinary conditions.
1.
Reliable, non-invasive, non-radioactive methods of measuring glomerular filtration
rate (GFR) or tubular functions.
2.
Translation of biomarkers of acute kidney injury or chronic kidney disease with
clinical utility into commercial assays.
3.
Translation of biomarkers for early detection of kidney diseases or prediction of
kidney disease progression, recovery, or drug response.
S.
Development of improved renal imaging techniques, differential renal function
assessment, diagnostic assessment of non-malignant kidney diseases, or
measurement of perinatal nephron endowment.
T.
Development of technology to improve collection of real-time data (e.g., biomarkers,
diet, physical activity, patient reported outcomes, vital signs, patient experience of
kidney or urologic disease or its treatment, social or environmental factors which
affect the development or progression of kidney disease), patient outcomes, and
adherence for clinical studies.
U.
Development of imaging or molecular analysis technologies to enhance information
extraction from renal biopsies and development of antibodies or other probes for
unique cell types of the kidney.
Therapeutics Discovery and Development
V.
Lead optimization and preclinical development of pharmacological agents that might
be used to intervene in acute or chronic renal disorders and in disorders of renal
hemodynamics, blood pressure, electrolyte metabolism, and extracellular volume
regulation.
W.
Development of drugs or biologics designed to specifically target kidney cell types.
X.
Development of drugs or biologics to stimulate productive kidney repair or
regeneration.
Y.
Development of technologies to enhance the validation of kidney disease targets or to
screen compounds for efficacy or toxicity (e.g., kidney organoids or tissue chips, more
relevant animal models of acute kidney injury).
Z.
Development of data and cell banks (e.g., of diabetic kidney disease families and
polycystic kidney disease families) for use by the research community.
AA.
Development of preventative measures for acute kidney injury (e.g., during coronary
artery bypass grafting, sepsis, or treatment with nephrotoxic agents).
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 93
II.
UROLOGIC DISEASES
Areas of research include benign prostatic hyperplasia, lower urinary tract symptoms (LUTS)
including urinary incontinence, urinary tract infections, urinary stone disease, erectile
dysfunction, urologic chronic pelvic pain syndromes (including interstitial cystitis and chronic
prostatitis), congenital urologic disorders, repair and regeneration of lower urinary tract
organs, normal and abnormal lower urinary tract development, and physiology of the urinary
system and male genital organs (excluding applications targeting male fertility).
Diagnostics and Imaging
A.
Translation of blood or urine biomarkers in the lower urinary tract or other urologic
disorders into commercial assays with clinical utility.
B.
Development of non-invasive or minimally invasive methods to diagnose bladder
inflammation or changes in the urothelium that are not of a cancerous origin.
C.
Development of new technologies for rapid clinical diagnosis and characterization of
urinary tract infection (UTI).
D.
Development of new technologies or methods with reduced radiation dose for
evaluating vesico-ureteral reflux in children and infants.
E.
Development of diagnostic modes to clinically and non-invasively or minimal-
invasively measure bladder outlet obstruction before and after surgical or
pharmaceutical intervention.
F.
Development of objective diagnostic devices or methods for the assessment of
urinary storage and voiding disorders, including stress, urge, and mixed incontinence,
in both adults and children.
G.
Development of wireless and non-invasive or minimally invasive measurement
technologies for real-time assessment of lower urinary tract function, which can
include neuro- pharmacological/neuro-physiological urodynamics.
H.
Development of radiation-free and accurate imaging technologies for urinary stone
disease.
I.
Development of technologies that use artificial intelligence/machine learning (AI/ML)
to integrate disparate data types to inform diagnosis of urologic diseases. AI/ML
approaches should leverage data from diverse populations and apply equity
considerations to ensure resulting models do not further embed structural racism or
discrimination.
J.
Development of platforms for pre-analytical preparation, imaging, and automated
analysis of genitourinary tissues.
Drug and Device (Therapeutic) Interventions
K.
Lead optimization and preclinical development of pharmacological agents for
treatment or prevention of urinary stone disease, urological chronic pelvic pain
syndromes, urinary tract infections, or other urologic diseases or conditions within
NIDDK’s mission.
L.
Development of novel neuromodulation devices, which restore function or mitigate
pain conditions of the lower urinary tract.
M.
Development of urinary catheters which reduce the incidence of infection in the
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 94
urinary tract and decrease urethral and bladder inflammation.
N.
Development of technologies for treatment of bladder outlet obstruction.
O.
Development of bioengineered materials or structures, including cell-laden structures,
for the repair or regeneration of genitourinary organs.
Health Information Technologies
P.
Development of health information technologies or mobile technologies that enhance
delivery of care, population health management, health equity, and/or research for
patients with urologic diseases or conditions.
Q.
Development of applications or application programming interfaces that use health
data standards (e.g., Fast Healthcare Interoperability Resources [FHIR], clinical
terminologies) to improve accessibility, accuracy, and/or completeness of real-world
health, behavioral, or social data for research and care of individuals with urologic
diseases or conditions.
R.
Development of technologies to engage patients with urologic diseases or conditions
in their care or to support interaction with caregivers.
S.
Development of innovative technologies or platforms to facilitate urology research
training and education, which could include software or simulation tools.
Research Tools
T.
Development of tools for elucidating the role of urinary or gut microbiome in urinary
stone disease or other urologic diseases or conditions within NIDDK’s mission.
U.
Development of novel models of benign prostatic hyperplasia.
V.
Development of technology to improve collection of real-time data (e.g., biomarkers,
diet, physical activity, vital signs, psychological parameters, and social or
environmental factors), patient- reported outcomes, and adherence for clinical studies
(e.g., studies of gene-environment interactions in the manifestation of urologic
diseases).
III.
HEMATOLOGIC DISEASES
The NIDDK hematology research program focuses on understanding basic cellular and
molecular mechanisms that underlie the production and function of blood cells in health and
disease. The program emphasizes translational applications of new insights and knowledge
gained from basic research in these areas toward the development of novel or improved
approaches for the diagnosis, stratification, and treatment of hematologic diseases. This
includes the development of disease biomarkers, gene targeted therapies, or hematopoietic
stem cell transplantation for acquired and heritable blood diseases (e.g., hemoglobinopathies,
such as sickle cell disease or thalassemia; hemochromatosis, iron overload, porphyrias,
amyloidosis, iron deficiency anemia, and cytopenias resulting from bone marrow failure
disorders, congenital dyserythropoietic anemias, Schwachman-Diamond syndrome,
myelodysplastic syndrome, neutropenias, myelofibrosis, essential thrombocythemia, or
polycythemia vera), and the measurement and chelation of tissue iron in iron overload
disorders. The NIDDK hematology research program provides resources for basic and
preclinical development efforts leading up to IND or IDE submissions but does not fund
clinical trials. The program has a particular focus on myeloid lineage and hematopoietic stem
NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 95
cells, including the effects of aging on hematopoiesis.
Drug Discovery and Development
A.
Establishment of robust in vitro or animal models of benign hematologic
diseases for drug discovery or development.
B.
Development of therapeutics that target elements of hematopoietic stem cell
niches (e.g., stromal cells, osteoblasts, endothelium, macrophages, pericytes,
nerve cells).
C.
Development of novel bone marrow conditioning regimens that promote
hematopoietic stem cell homing, engraftment, and hematopoiesis.
D.
Development of therapeutics that modulate blood cell production from
hematopoietic stem cells and progenitors based upon understanding of physical
and chemical regulatory pathways.
E.
Development of therapeutics that modulate metabolism, storage, and transport
of iron or heme.
Cell Therapies
F.
Development of equipment, chemically defined reagents, and methods for high
volume ex vivo expansion, isolation, and/or differentiation of highly purified
human hematopoietic stem and progenitor cells.
G.
Development of equipment, chemically defined reagents, and methods for
selective removal or destruction of diseased human hematopoietic stem and
progenitor cells (e.g., in myelodysplastic syndrome, MDS). Treatment of malignant
clones and blood cancers are not within the scope of the NIDDK Hematology
mission.
H.
Development of therapeutics that induce fetal hemoglobin synthesis by
chemical means, genome editing, or other means.
I.
Development of therapeutics that target blood cell membrane structure.
Diagnostics and Imaging, Medical Technologies, and Research Tools
J.
Development and validation of sensitive, specific, reproducible, quantitative,
and clinically applicable assays for measuring levels or expression of iron
regulatory molecules or for measuring misfolded or aggregate amyloid
proteins such as amyloid A transthyretin or immunoglobulin light chain in
blood.
K.
Development of technologies to track, purify, monitor or assay single-cells in vivo or in vitro.
L.
Development of non-invasive systems for monitoring circulating blood cells,
blood chemistry or blood cell production.
M.
Development of imaging technology for the non-invasive measurement of
bone marrow cellularity, fibrosis, and function.
N.
Development of imaging technology for the non-invasive measurement of
tissue iron loading and distribution.
O.
Development of technologies to understand the roles of mitochondria in non-
malignant hematologic diseases.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 96
P.
Development of technologies that use artificial intelligence/machine learning
(AI/ML) to integrate disparate data types (e.g., histomorphology, karyotyping,
next generation sequencing, immunophenotyping, and flow cytometry) to
inform diagnosis of non- malignant hematologic diseases. AI/ML approaches
should leverage data from diverse populations and apply equity
considerations to ensure resulting models do not further embed structural
racism or discrimination.
Q.
Development of platforms for pre-analytical preparation, imaging, and
automated analysis of the bone marrow.
R.
Development of innovative technologies or platforms to facilitate hematology
research training and education, which could include software or simulation
tools.
Contact Information
For additional information on research topics, contact:
DIABETIC TECHNOLOGY, TYPE 1 DIABETES AND ENDOCRINE DISEASES
Dr. Guillermo Arreaza-Rubín
National Institute of Diabetes and Digestive and Kidney
Diseases Phone: 301-594-4724
Email: arreaza-rubing@niddk.nih.gov
DIABETIC WOUND HEALING AND TYPE 2 DIABETES
Dr. Yan Li
National Institute of Diabetes and Digestive and Kidney
Diseases Phone: 301-435-3721
Email: yan.li5@nih.gov
NEUROMODULATION, DIABETIC NEUROPATHY, AND TYPE 2 DIABETES DRUG DISCOVERY
Dr. Bradley M. Cooke
National Institute of Diabetes and Digestive and Kidney
Diseases Phone: 301-480-9896
Email: brad.cooke@nih.gov
GASTROINTESTINAL, OBESITY, AND NUTRITION DISORDERS AND DISEASES
Dr. Robert Lunsford
National Institute of Diabetes and Digestive and Kidney
Diseases Phone: 301-402-1397
Email: robert.lunsford@nih.gov
LIVER, PANCREATIC, AND NEUROBIOLOGICAL DISORDERS AND DISEASES
Ms. Christine Densmore
National Institute of Diabetes and Digestive and Kidney
Diseases Phone: 301-402-8714
Email: christine.densmorec@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIDDK 97
KIDNEY, UROLOGIC AND HEMATOLOGIC DISEASES
Dr. Daniel R. Gossett
National Institute of Diabetes and Digestive and Kidney
Diseases Phone: 301-594-7723
Email: daniel.gossett@nih.gov
For administrative and business management questions,
Ms. Christina Coriz
National Institute of Diabetes and Digestive and Kidney
Diseases Phone: 301-594-8848
Email: christina.coriz@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 98
NATIONAL INSTITUTE OF ENVIRONMENTAL HEALTH SCIENCES (NIEHS)
Mission
The mission of the National Institute of Environmental Health Sciences www.niehs.nih.gov is to discover
how the environment affects people in order to promote healthier lives, with a vision of providing global
leadership for innovative research that improves public health by preventing disease and disability.
NIEHS achieves its mission and vision through a multidisciplinary biomedical research program,
prevention and intervention efforts, and a communication strategy that encompasses training,
education, technology transfer, and community outreach.
Budget Guidance
For all NIEHS research interest topic areas other than Hazardous Substances Remediation and Site
Characterization SBIR Program included in this PHS 2024 Omnibus SBIR/STTR Solicitation, NIEHS will
accept SBIR/STTR application total funding support (direct costs, indirect costs, and fee) requests up to
$306,872 for Phase I and $2,045,816 for Phase II. NIEHS will not fund applications at budget levels
exceeding these hard cap budget guidelines, except through specific RFAs. For budgetary,
administrative, or programmatic reasons, NIEHS may decide not to fund an application or may
decrease the length of an award and/or the budget. In all cases, applicants should propose a budget
that is reasonable and appropriate for completion of the research project, and the budget request must
be well justified. The Hazardous Substances Remediation and Site Characterization SBIR Program has
different limits on budget requests for both Phase I and Phase II, check under that topic below for
details.
NIEHS will consider well justified Technical and Business Assistance (TABA) costs up to the limits
specified on the NIH SEED website. These costs can be requested in addition to the Phase I and II
limits indicated above.
Specific SBIR and STTR Program Information
For additional information about NIEHS's Small Business Programs, please visit Small Business
Innovation Research & Small Business Technology Transfer Grants (SBIR/STTR, R41, R42, R43, R44)
(nih.gov). NIEHS DOES NOT Fund technologies for the detection and remediation of pathogens in
the environment.
Final Progress Reports
As detailed in NOT-OD-17-085, the NIH has implemented the Final Research Performance Progress
Reports (Final RPPR) for SBIR/STTR Final Progress Reports.
The NIEHS is interested in tracking the progress of the small business concerns it funds and the
products they develop. It is expected that small businesses who have received previous SBIR/STTR
grants have had success in commercializing their previously supported technologies. Small businesses
that are primarily interested in research and development (and not commercialization) should consider
other grant mechanisms at NIH, rather than the SBIR/STTR program. Funding priority will be given to
those small business concerns that demonstrate their ability to develop and commercialize products.
Specific Funding Opportunities and Programs
In addition to this omnibus program announcement, the NIEHS releases targeted SBIR/STTR
Notices of Funding Opportunities (NOFOs); signup for the listserv ( Subscribe or Unsubscribe to
the SBIR-NIEHS List (nih.gov) ) to be notified of NOFOs.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 99
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NIEHS does not intend to support any new Phase II B grants in this funding period. NIEHS
currently participates in PAR-23-219: SBIR/STTR Commercialization Readiness Pilot (CRP)
Program Technical Assistance and Late Stage Development (SB1, Clinical Trial Not Allowed).
Clinical Trials
Does NIEHS accept Clinical Trials
through the
Omnibus/Parent Notices of
Funding Opportunities?
Yes
Does NIEHS accept Clinical
Trials through specific Notices of
Funding Opportunities?
No
Does NIEHS support Clinical
Trials through NON-SBIR/STTR
Notices of Funding Opportunities?
Yes
For non-SBIR/STTR clinical trials funding mechanisms
for which small businesses are eligible, please visit the
https://www.niehs.nih.gov/funding/grants/announceme
nts/request/index.cfm.
Research Topics
NIEHS Non-Clinical Trials Topics:
Exposure Assessment Tools
The NIEHS Exposure Biology and the Exposome Program supports technologies to better understand
how a person’s environment contributes to their health. Sensor technologies, computational
approaches, improved biomarkers, and biomonitoring capabilities, are needed to measure, analyze, and
predict a wide range of internal and external exposures and health outcomes across diverse geographic
populations. These tools should be designed fit-for-purpose in collaboration with the stakeholders (e.g.,
community engagement programs, citizen scientists, disaster response personnel, epidemiologists, or
clinical researchers).
Examples include:
Sensors and Other Exposure Assessment Tools
•
Technologies and methodologies to assess personal exposure to specific or
combined air pollutants in population studies, including wearable monitors and sensor
networks.
•
Devices for collecting exposure measurements across multiple stressors and scales,
with an emphasis on high sensitivity and specificity and low-cost devices, when
feasible. High-priority analytes include contaminants of emerging concern (e.g.,
perfluorinated compounds, and toxins produced in harmful algal blooms) as well as
ultrafine particulates, microplastics, pesticide exposures, and industrial chemicals.
•
Novel sampling technologies to enable subsequent targeted and untargeted laboratory
analysis
•
Sensor technologies that can be integrated into existing smart devices for sensing
personal environment as well as provide chemical speciation data.
•
Tools and approaches for identifying and characterizing contaminants in drinking water
that may pose a risk to human health, with a particular emphasis on new contaminants

NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 100
or compounds that are of emerging concern.
Note that identification of environmental pathogens in drinking water is not within
the NIEHS mission.
Computational and Informatics-based Tools and Methods for Exposure Assessment
•
Informatics tools and platforms to organize, store, retrieve, extract, and integrate data
on exposures and health effects.
•
Application of machine learning methods and natural language processing for
extracting and integrating diverse data types and for generating causal networks from
experimental data and public knowledgebases
•
Computational and statistical approaches to integrate exposure data from different
sources, including publicly available databases and information from monitoring
approaches (e.g., sensors, remote sensing, and biomonitoring), to provide quantitative
exposure estimates, identification, and characterization of adverse effects on human
health.
•
Adapting or developing new methods and tools for automating environmental health-related
literature and systematic reviews, including article selection and prioritization, data
extraction, study quality evaluation, and summarization of for environmental health impacts
Additional information can be found on the NIEHS Exposure Biology and the Exposome Program
website Exposure Biology and the Exposome (nih.gov)
Nano Environmental Health and Safety
The NIEHS Nano Environmental Health and Safety (Nano EHS) program is interested in the detection of
engineered nanomaterials (ENMs) in the environment, in consumer products, and in biological samples,
and is interested in technologies or methods that can predict toxicity potential of ENMs.
High priority engineered nanomaterials of interest are those with a potential for human exposure.
Examples include:
•
Sensors, tools or technologies that can detect metal and carbon-based engineered
nanomaterials or micro/nanoplastics in air, water, and consumer products, and provide a
contextual assessment of the toxicological potential.
•
Mid- to high-throughput and high-content assays using in vitro or tissue chip technologies to
screen and rank toxicity of emerging engineered nanomaterials for cytotoxicity, genotoxicity,
and metabolic or other human relevant toxicity.
•
Methods and tools to assess leaching of engineered nanomaterials from nanotechnology-
based water filtration systems.
•
Technologies to assess the life cycle of nanomaterials from nano-enabled products in the
market
•
Development of tools and technology platforms for the isolation, quantification,
physical and chemical characterization of various forms of micro/nanoplastics from
diverse media including biological samples, aqueous sources, air and food samples
and assessment of their toxicity potential and human health effects

NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 101
Information on the Nano EHS program can be found at Nano Environmental Health and Safety (nano
EHS)
Toxicity Screening, Testing, and Modeling
NIEHS supports research to identify the hazards, as well as the mechanistic understanding, of the
effects of environmental stressors on biological systems that can lead to adverse human health
outcomes. To increase the ability to characterize or predict the toxicity and hazard of environmental
stressors, the National Toxicology Program (NTP) Home - National Toxicology Program (nih.gov) at
NIEHS is interested in technologies to support the goals and initiatives of the Tox21 Program Tox21
(nih.gov).
Technologies that support Tox21 and other NTP goals may include the development and/or application
of in vitro physiologically relevant cell-based systems that effectively model responses in humans or
animals and may be used to reduce or replace in vivo animal use. High priority areas are the
development of metabolically competent in vitro screening models and assay systems for various tissue
types (e.g., cardiac, neurological, liver, GI tract, kidney, mammary gland, lung, and immune function) for
assessing the effects of the environmental stressors.
Toxicity Screening Approaches
•
Improved or new approach methodologies (NAMs) including human organotypic culture
models (OCM), and microphysiological systems (MPS) that more accurately predict in vivo
function for characterizing toxicity and/or related disease processes. Priority areas are
improved capability for generating more mature cells from embryonic stem (ES) or induced
pluripotent (iPS) cells for organotypic models, integrating multiple MPS together under
physiologically relevant conditions, and the ability to conduct in vitro pathology studies
using OCM, MPS or 3D culture models.
•
Organotypic models using cells from rat or mouse models or other experimental animal
models, with a focus on comparisons between in vivo and in vitro toxicity endpoints.
•
Approaches to characterize and integrate key molecular and cellular changes related to
effects of toxicant exposures in carcinogenicity, developmental neurotoxicity,
cardiotoxicity or immune functions.
•
In vitro model systems that incorporate barrier functionality and transport functions
into tissue models (e.g., kidney, placenta, or blood-brain barrier)
•
Enhanced lower organism models (e.g., zebrafish or C. elegans) for toxicity screening.
•
Stem cell models and assays for evaluating the effects of toxicants on cell
differentiation with multiple functional endpoints.
•
Screening systems that incorporate genetic diversity into toxicology testing (e.g., panels
of tissue- specific human iPS cells or rodent stem cells)
•
In vitro systems that focus on responses to mixtures of xenobiotics, chronic exposure
studies, or provide insights into the molecular characteristics of multiple chemical-
biological interactions and toxicodynamics.
•
Short-term tests, assays, or systems designed specifically to reduce or replace existing
regulatory animal studies for acute toxicity (oral or inhalation), reproductive or
developmental toxicity, carcinogenicity, or ocular toxicity.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 102
•
Cage-based technologies to monitor physiological and behavioral changes in experimental
animals in chemical toxicology studies.
Computational Approaches for Predictive Toxicology
•
New computational systems and tools for integrating toxicity data, including in vivo
and in vitro data, to analyze and visualize data across different screening systems
and predict chemical hazard/risk.
•
Computational tools to integrate and visualize transcriptomic and metabolomic data in
affected signaling and biochemical pathways.
•
Improved computational tools for in vitro to in vivo extrapolation of xenobiotic
exposures and modeling metabolic transformation of xenobiotics.
•
Advanced computational approaches (e.g., artificial intelligence/machine learning) to
integrate and develop multi-omics classifiers for exposures and pathology image analysis
tools for environmentally induced diseases.
Other Technologies Focused on Enhancing Toxicology Testing
•
Alternative or improved methods for fixing and preserving tissues that maintain cellular
structure for histopathology while minimizing degradation of nucleic acids (RNA, miRNA,
DNA, methylated DNA), proteins or metabolites, so that archival tissue blocks can be
better used for molecular analysis.
•
Liquid biopsy methods for isolation and novel assays of circulating nucleic acids that
reflect environmental chemical exposures or toxicity. These could include exosome-
packaged or cell-free nucleic acids altered by environmental exposures.
•
Alternative or improved methods for extracting high quality RNA, miRNA, DNA,
methylated DNA, proteins, or metabolites from existing archived tissues.
•
Tools and technologies for isolation and characterization of exosome and/or
extracellular microvesicles from biological fluids
Biomarkers of Exposure and Response
To better understand the risks to human health from environmental agents, NIEHS supports the
development and validation of biomarkers of exposure, including improved measures of internal dose,
DNA adduct identification, and untargeted analysis for metabolite identification, and biomarkers of
response, including assays that can distinguish reversible from irreversible changes in target organs or
surrogate tissues. Examples include:
Biomonitoring Technology
•
Personal or point-of-care monitoring technologies for rapid detection of multiple
exposures in biospecimens using non- or minimally invasive approaches.
•
Improved methods to detect DNA or protein adducts resulting from exogenous exposures.
•
Exposure assessment methods in novel matrices or small volumes
NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 103
Biological Response Markers
•
Markers of oxidative stress, inflammation, DNA damage response, immune function,
mitochondrial dysfunction, or altered epigenetic regulation.
High priority human biomarkers include, but are not limited to inflammation biomarkers, plasma-
or
serum-based markers that reflect altered RNA, protein expression, or metabolite profiles, markers
developed in exhaled breath, buccal cells, or other easily accessible, non-invasive biological samples,
miRNA or other exosome biomarkers, and epigenetic markers in surrogate tissue reflecting
modifications in target tissues.
Intervention Technologies
NIEHS supports efforts to prevent or reduce exposures to environmental chemical stressors that affect
human health. Technologies to reduce exposure may include:
•
Technologies for detecting and/or removing contaminants from drinking water, primarily
for home use.
•
Approaches for use in the home, workplace, and school settings for reducing volatile
compounds and other inhaled toxicants. Examples may include improved air filtration
systems as well as technologies to monitor the efficacy of filtration systems.
•
Technologies and applications that can provide real-time alerts about relevant
environmental exposure levels for sensitive populations (such as asthmatic
populations)
Education and Participatory Science
As part of its Partnerships for Environmental Public Health (PEPH) Program, NIEHS is interested in
developing tools that build capacity, improve environmental health literacy, and support participatory
science endeavors. These approaches or resources should be fit for purpose to meet the needs of the
following audiences: community members, health care and public health professionals, educators, and
students of all ages. Approaches may include:
•
Mobile applications that provide environmental health information about exposures of
concern in food, air, drinking water, or consumer products. These may include
•
Interactive apps that provide the context and risks of exposures such as single or
multiple, interacting exposures, level of exposure, frequency and proximity to
source and health risks
•
Apps that can be adapted for various age groups, races, ethnicities and/or
languages.
•
Devices for collecting and reporting information on exposures in environmental
samples for educational purposes in schools or communities.
•
Systems that can utilize public and voluntary population data from sensors, activity
trackers, GIS enabled devices, social communications, and surveillance cameras; for
example, to assist disaster response and communication.
•
Educational resources and tools related to environmental health in school settings or
community education programs.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 104
•
Training materials for wider dissemination of risk information (e.g., resources for
high school students or community leaders to build capacity of other community
residents)
Information on the PEPH program can be found at Partnerships for Environmental Public Health
(PEPH) (nih.gov).
Other Areas of Interest
Exposure and Response to Vaping and Electronic Nicotine Delivery Systems (ENDS)
NIEHS is interested in technologies to assess exposure to aerosols from e-cigarettes and other vaping
devices, including analyses of the chemical constituents in these aerosols. In addition, approaches to
test the toxicity and biological responses to ENDS aerosol constituents are of interest.
Disaster Response
NIEHS is interested in sensors and informatics tools that can be rapidly deployed after disasters,
including extreme weather events or climate change-related events. These tools can be used by
researchers to follow emergency response workers and individuals in the community to help understand
dermal, water and/or airborne exposure levels, locations, and times.
•
Environmental sensors that can be rapidly deployed during or after a disaster to track
exposures.
•
Informatic tools to rapidly build environmental health disaster research protocols similar
to the NIEHS RAPIDD Protocol Disaster Research Response (DR2) Program (nih.gov)
from existing information, tools, and platforms (e.g., PhenX, PROMIS, and Disaster
Research Response DR2 Repository) to support rapid research response efforts
•
Data management tools for disaster response that enable rapid collation and integration
of data from stationary sources and personal exposure monitors and survey information
collected from individuals.
•
Mobile devices and applications for collecting information on environmental exposures
from study participants involved in disaster research responses.
Hazardous Substances Remediation and Site Characterization SBIR Program
The NIEHS Superfund Research Program (SRP) "Hazardous Substances Remediation and Detection
Program" supports Small Business Innovation Research Grants (SBIR R43, R44) to foster the
commercialization of novel, cost-competitive technologies, products, and devices for remediation and
detection of hazardous substances in the environment. The SRP is specifically interested in proposals
applying new engineering, materials science, and biotechnology approaches. In addition, applicants are
encouraged to develop sustainable strategies such as offering low carbon footprint, reduced energy
consumption, utilization of renewable energy sources, resilient to weather extremes, and with reuse /
regeneration capabilities.
Remediation
•
Novel technologies for in situ remediation of contaminated sediments, soils, and
groundwater with testing/modeling to optimize product for long-term stability
•
Innovative bioremediation technologies including development and culturing/propagation
of novel plants, bacterial strains, or fungal species for implementing bioremediation
NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 105
•
Technologies to remediate chemical mixtures in environmental media
•
New strategies for delivery of reagents/amendments for groundwater
remediation and/or recovery/extraction of contaminants in groundwater
•
New amendments to stabilize contaminants and/or to use in caps for soil and sediment
remediation
•
New technologies and strategies to cleanup large complex sites with multiple sources
•
Resilient novel remediation approaches capable of withstanding climate change-related
impacts such as: fire, flooding groundwater level fluctuation, land use changes, and
other catastrophic events
•
Sustainable, energy efficient approaches with a net lifecycle benefit such as net zero
emission technologies; technologies that reduce waste generation; processes that
recycle/reuse/regenerate active components; long-term remediation approaches
equipped with solar or wind energy
Detection Technologies
•
Machine learning, computational, geographical information system-based, or modeling
products for predicting fate and transport of contaminants, rates of remediation,
bioavailability, or for identifying contamination sources
•
Real-time, field deployable, on-site analysis: soil, surface water, groundwater,
subsurface, sediments, air (such as volatile releases from sites), including
•
rapid, portable monitoring and screening of contaminants
•
multi-analyte sampling
•
remote monitoring/data capture/data processing capabilities such as time-
integrated and/or repeated measures
•
Accurate and reliable new passive sampler devices
•
Products that allow for rapid sample clean-up/preparation for analysis of environmental
samples and/or technologies for rapid extraction or processing of soil for incremental
sampling methodologies (ISM)
•
Non-targeted or multi-analyte field sampling devices or kits, including sample collection
products that can sequester a suite of analytes for later analysis
•
Novel techniques, sensors, and field analytical methods and real-time mapping/data
visualization for development of subsurface conceptual site models
•
Innovative tracer technologies for tracking contaminant migration/pathways
Examples of remediation and detection technology needs:
•
Vapor Intrusion: Improved technologies for predicting/anticipating time-periods for
occurrence of reasonable maximum indoor exposure(s) in impacted buildings, during
which sampling is recommended.
•
PFAS: Soil, sediment, and groundwater remediation technologies for mixtures and
degradation byproducts of per- and polyfluoroalkyl substances (PFAS); including
technologies for complete PFAS destruction; sustainable solutions with low energy input

NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 106
and/or minimal secondary waste generation; and/or PFAS removal technologies for
heterogenous water chemistries; rapid sensors to aid in site monitoring and/or prioritizing
site sampling protocols.
•
Novel, sustainable, nontoxic chemistries or processes to aid regeneration,
reuse, and/or reactivation of spent treatment residues (e.g., from granular
activated carbon).
•
Development of adsorption and concentration materials to reduce the volume of
material to be treated and/or to further concentrate the waste stream generated
from standard treatment technologies (e.g., granular activated carbon, reverse
osmosis) as part of a “treatment train”.
•
Development of polishing treatments tailored for specific PFAS (e.g.,
shorter chain, emerging PFAS replacements).
•
Development of novel catalysts or other additives to lower needed
temperature for complete thermal destruction.
•
Development of novel air pollution control technologies as a polishing step to
reduce emissions from PFAS management or treatment facilities (e.g., thermal
destruction, air sparging, Supercritical water oxidation (SCWO), hazardous
waste landfill facilities, etc.).
•
Development of novel materials or processes for solid waste and/or biosolids
treatment and/or stabilization.
• Mining: Active or passive remediation technologies for mining influenced water;
technologies to mitigate effects from acid drainage; portable neutralization treatment
systems; strategies to target remediation of sources such as mining waste piles; and
separation technologies that remove elements or compounds of concern from water and/or
reclaim potentially valuable critical elements dissolved in contaminated fluids
•
Complex Site/Geology:
•
Site characterization techniques and strategies for complex geology (fractured
bedrock, karst, and heterogeneous layered deposits) including understanding the fate
of contaminants within rock matrices and properties that affect back diffusion
•
Improved technologies for treating low permeability and heterogeneous lithology,
including amendment delivery methods
•
Devices to detect and measure non-aqueous phase liquids (NAPLs) in the subsurface
•
In-well real-time and/or continuous monitoring tools to assess the efficacy of
remediation; presence/absence of key factors required for remediation (e.g.,
biological, geological, chemical); and/or to identify rebound events
•
Robotic sampling for highly contaminated / remote sites
•
Disaster Response: Technologies for measuring/treating environmental contamination as
part of a disaster response effort
Applicants must demonstrate that the proposed technologies are relevant to Superfund and/or other
sites impacted by hazardous substances. Per program mandates described in the Superfund
Amendment Reauthorization Act (SARA), SRP does not accept applications targeting oil or gas site
characterization/remediation. Applicants are strongly encouraged to stay within total funding support
(direct costs, indirect costs, and fees) does not exceed $183,460 for Phase I awards and $1,223,064 for
Phase II awards. Applicants are encouraged to contact NIH program officials prior to submitting any
award budget for the "Hazardous Substances Remediation and Site Characterization Small Business
Innovation Research Program” in excess of these amounts. For budgetary, administrative, or
programmatic reasons, NIEHS may decide not to fund an application or may decrease the length of an
award and/or the budget. NIEHS will consider well justified Technical and Business Assistance (TABA)
costs up to the limits specified on the NIH SEED website. These costs can be requested in addition to

NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 107
the Phase I and II limits indicated above. These costs can be requested in addition to the Phase I and II
limits indicated above. Please note: the NIEHS Superfund Research Program (SRP) "Hazardous
Substances Remediation and Site Characterization Small Business Innovation Research
Program" no longer accepts Small Business Technology Transfer Grant (STTR: R41, R42)
applications. Funding decisions will be made based on programmatic need with an emphasis on novel
technologies distinct from current or recently-funded SBIR grants that are applicable to Superfund
and/or other sites impacted by hazardous substances.
Please visit the “Small Business Innovative Research Grants (R43/R44) Hazardous Substances
Remediation and Site Characterization SBIR Program” section on the following webpage:
https://www.niehs.nih.gov/research/supported/centers/srp/funding.
Worker Training Program
The major objective of the NIEHS Worker Training Program (WTP) is to prevent work related harm by
training workers in how best to protect themselves and their communities from exposure to hazardous
materials. The NIEHS WTP is interested in the development of e-Learning Technology- Enhanced
Training Products from a variety of delivery methods to assist both students and instructors in the
training and education process. These Technology-Enhanced Training Products are for the health and
safety training of hazardous materials (HAZMAT) workers; waste treatment personnel; skilled support
personnel associated with an emergency/disaster; emergency responders in biological hazard
response, infectious disease response and medical waste cleanup; emergency responders in
disasters; and worker resiliency training. Technology-Enhanced Training Products as defined by the
WTP includes, but are not limited to, online training, mobile device training, augmented reality (AR),
virtual reality (VR), and serious gaming. These advanced technologies complement all aspects of
training that can enhance, supplement, improve, and provide health and safety training for hazardous
materials workers. WTP accepts solicitations via requests for applications (RFA). Please contact
Kathy Ahlmark (ahlmark@niehs.nih.gov) for information on the next solicitation date, which differs from
the standard receipt dates of this NIH omnibus.
Information on the WTP program can be found at About the Worker Training Program (WTP) - Training for
Workers in Hazardous Environments (nih.gov)
NIEHS Clinical Trials Topics:
NIEHS will accept SBIR/STTR applications that propose clinical trials related to:
•
Development and testing of sensor technology, biomarkers, or biomonitoring technologies,
including field testing of new technologies for exposure assessment and biological
responses to environmental exposures.
•
Evaluation of tools or approaches for education and dissemination of information on
environmental hazards, including evaluation of changes in behavior.
Contact Information
For additional information on research topics, contact:
Dr. Daniel Shaughnessy
National Institute of Environmental Health Sciences
Division of Extramural Research and Training
POB 12233 (K3-12)
Research Triangle Park, NC 27709
(984) 287-3321
Email: shaughn1@niehs.nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIEHS 108
Dr. Lingamanaidu V. Ravichandran
National Institute of Environmental Health Sciences
Division of Extramural Research and Training
POB 12233 (K3-05)
Research Triangle Park, NC 27709
(984)-287-3309
Email: lingamanaidu.ravichandran@nih.gov
For information on the NIEHS Superfund Research Program -
Hazardous Substances Remediation and Site Characterization
SBIR Program, contact:
Dr. Heather Henry
National Institute of Environmental Health Sciences
Division of Extramural Research and Training
POB 12233 (K3-12)
Research Triangle Park, NC 27709
(984) 287-3268
Email: henryh@niehs.nih.gov
For administrative and business management questions contact:
Mr. Clark A. Phillips
National Institute of Environmental Health Sciences
Division of Extramural Research and Training
Grants Management Branch
(984) 287-4037
Email: clark.phillips@nih.gov
NIH, CDC, and FDA Program Descriptions and Research Topics
NEI 109
NATIONAL EYE INSTITUTE (NEI)
Mission
The mission of the NEI is to conduct and support research, training, health information
dissemination, and other programs with respect to blinding eye diseases, visual disorders,
mechanisms of visual function, preservation of sight, the special health problems and
requirements of the blind, and providing eye health care to underserved populations.
For up-to-date information on priority research areas of scientific interest to the NEI, please visit
our home page at http://www.nei.nih.gov.
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SEED website. For budgetary,
administrative, or programmatic reasons, NEI may decrease the length of an award and/or the
budget recommended by a review committee, or not fund an application. For topics listed in
SBA-Approved Waiver Topics, NEI does not generally fund Phase I applications greater than
$300,000 total costs or project periods greater than 2 years; or Phase II applications greater than
$2,000,000 total costs or project periods greater than 3 years. Applicants are strongly
encouraged to contact program officials prior to submitting any application in excess of the hard
caps listed above and early in the application planning process.
Specific SBIR and STTR Program Information
The NEI's programs are described in more extensive detail in documents which are available
from the Institute. For additional information about the research programs of the NEI, please
visit our home page at http://www.nei.nih.gov.
Phase IIB Competing Renewal Awards and Commercial Readiness Pilot (CRP)
The NEI will only accept SBIR Phase IIB Competing Renewal grant applications from Phase II
SBIR awardees to continue the process of developing technologies that ultimately require
federal regulatory approval or require extraordinary time and effort in the Research and
Development phase. Such technologies include, but are not limited to, pharmacologic agents,
biological products, and devices. These technologies should be clearly related to the mission of
the NEI. This renewal grant should allow small businesses to reach a stage in the project where
interest and investment by third parties is more likely. The NEI expects that the Phase IIB grant
will accelerate the transition of SBIR Phase II projects to the commercialization stage. The NEI
encourages applicants to establish business relationships with third-party investors and/or
strategic partners who can provide substantial financing to help accelerate the commercialization
of promising new products and technologies that were initiated with SBIR funding.
The Competing Renewal application must be a logical extension of a previously completed Phase
II (R44) SBIR grant. NEI grantees seeking SBIR Phase IIB Competing Renewal funding must
submit an application within a period no later than the first six receipt dates following expiration
of the previous Phase II budget period. Cumulative budgets should not exceed $1,800,000 total
costs, or time periods beyond three (3) years.
Although matching funds are not required, the NEI strongly encourages that applicants obtain
significant private investment. Competitive preference and funding priority will be given to
NIH, CDC, and FDA Program Descriptions and Research Topics
NEI 110
applicants that demonstrate the ability to secure substantial independent third -party investor
funds.
Applicants are strongly encouraged to contact the NEI Program Officer, Dr. Paek Lee (contact
information provided below) prior to submitting any application in excess of the hard caps listed
above and early in the application planning process.
Clinical Trials
Does NEI accept Clinical Trials through
the Omnibus/Parent Notices of Funding
Opportunities?
Yes
Minimal risk clinical trials only.
See details below.
Does NEI accept Clinical Trials through
specific Notices of Funding
Opportunities?
Yes
U44 PAR-24-066
Does NEI support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
R01 PA-20-183
UG1 PAR-21-042, PAR-21-043, PAR-21-041
U01 PAR-22-149
NEI accepts clinical trial applications submitted under SBIR and STTR Omnibus/Parent Clinical Trial
Required Notices of Funding Opportunities that include human subjects prospectively assigned to
one or more interventions that are minimal risk as defined by 45 CFR 46. Minimal risk means that the
probability and magnitude of harm or discomfort anticipated in the research are not greater in and of
themselves than those ordinarily encountered in daily life or during the performance of routine
physical or psychological examinations or tests. As part of the review process, the application must
include a clear, detailed plan for monitoring safety that is commensurate with the risks to study
participants. In addition to the minimal risk designation from the Institutional Review Board, other
reporting to the NEI may be required and will be outlined in the Notice of Award Terms and
Conditions.
Applicants who wish to submit greater-than-minimal risk, complex, large-scale, high-resource or
safety-risk clinical trials that propose to test efficacy, effectiveness or safety should not submit to
these NOFOs. Instead, early-stage clinical trial applications with greater than minimal risk to subjects
must be submitted to PAR-24-006 NEI Small Business Innovation Research (SBIR) Cooperative
Agreement for Early-Stage Clinical Trials with Greater than Minimal Risk (U44-Clinical Trial
Required). Complex, large-scale, high-resource clinical trials must be submitted to one of the Clinical
Research cooperative agreement NOFOs listed here: https://www.nei.nih.gov/grants-and-
training/funding-opportunities/current-funding-opportunities.
Research Topics
A.
General Research and Development Topics: NEI is interested in providing support for the
development of new technologies, strategies, research tools, reagents and methods that can be
applied to basic and translational research which will benefit vision health. This encompasses
research and development of innovative enabling technologies in areas of genomics, proteomics
and nanotechnology. More specific topics include drug and high throughput assays; drug delivery
systems; gene therapy, cell-based therapy and regenerative medicine; development of in vitro
and in vivo disease models; surgical devices and materials; telemedicine, mobile health, and
health education; and design/fabrication of new or improved ophthalmic instruments for diagnosis
and treatment of eye disorders.
B.
Retinal Diseases: New therapeutic approaches for inflammatory and degenerative diseases and
for inhibition of abnormal angiogenesis in the retina and choroid; Better methods of diagnosing
and treating diabetic retinopathy and other vascular diseases; Non-invasive techniques for early

NIH, CDC, and FDA Program Descriptions and Research Topics
NEI 111
diagnosis of macular degeneration and other retinal degenerative diseases; Instruments and
procedures for improved surgical management of retinal detachments; Retinal prostheses to help
restore visual function; Gene therapy/optogenetic methods for light sensitivity restoration in the
retina; Better methods for cell or tissue transplantation; New animal models/systems that better
mimic human retinal disease.
C.
Corneal Diseases: New diagnostic tools, therapeutic agents and drug delivery methods for the
treatment of corneal injury, infection, dry eye, ocular pain, and other ocular surface disorders;
New biomaterials for corneal prostheses and corneal transplants; Instruments and procedures for
correcting the refractive power of the cornea and/or measuring the cornea's optical properties or
other physiological properties.
D.
Lens and Cataract: New approaches in the post-operative management of cataract surgery; New
surgical instruments for cataract extraction and new biomaterials for replacement of the natural
lens; Design/fabrication of aspheric, toric, multifocal and accommodating intraocular lenses.
E.
Glaucoma and Optic Neuropathies: New therapeutic agents, instruments, and procedures for
the diagnosis and treatment of glaucoma; Non-invasive methods to measure changes in the optic
nerve head and retinal fiber layer.
F.
Strabismus, Amblyopia, and Refractive Error: New approaches to detect and treat strabismus,
amblyopia, and myopia; New tools and techniques for vision screening; New or improved methods
and materials for correcting the refractive power of the eye and/or measuring the eye's optical
properties or other physiological properties; New materials and manufacturing processes for
eyeglasses and contact lenses; prosthetic devices (both cortical and subcortical) for vision
restoration.
G.
Visual Impairment and Blindness: Instruments and methods to better specify, measure, and
categorize residual visual function; New or improved devices, systems, or programs that meet the
rehabilitative, adaptive, and everyday living needs of visually impaired/blind people.
Contact Information
For more information on research
topics, contact:
Paek Lee, Ph.D.
Program Director, Small Business
SBIR/STTR Division of Extramural Science Programs
National Eye Institute
6700B Rockledge Drive
Bethesda, MD 20817
301-435-8164
Email: paek.lee@nih.gov
Tony D. Gover, Ph.D.
Program Director, Small Business SBIR/STTR (Anterior Segment)
Division of Extramural Science
Programs National Eye
Institute 6700B
Rockledge Drive
Bethesda, MD 20817
301-529-7370
Email: tony.gover@nih.gov
For administrative and business management questions, contact:

NIH, CDC, and FDA Program Descriptions and Research Topics
NEI 112
Ms. Karen Robinson Smith
Chief Grants Management Officer
Grants Management Branch
Division of Extramural Activities
National Eye Institute, NIH, DHHS
6700B Rockledge Drive, Suite 3400
Bethesda, MD 20892
301-451-2020, Fax: 301-496-9997
Email: kyr@nei.nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIGMS 113
NATIONAL INSTITUTE OF GENERAL MEDICAL SCIENCES (NIGMS)
Mission
NIGMS supports basic research that increases our understanding of biological processes and
lays the foundation for advances in disease diagnosis, treatment, and prevention. NIGMS-
funded scientists investigate how living systems work at a range of levels—from molecules and
cells to tissues and organs—in research organisms, humans, and populations. Additionally, to
ensure the vitality and continued productivity of the research enterprise, NIGMS provides
leadership in training the next generation of scientists, enhancing the diversity of the scientific
workforce, and developing research capacity throughout the country.
All NIH institutes and centers support basic research that’s relevant to the diseases, organ
systems, stages of life, or populations within their mission areas. In contrast, NIGMS supports
fundamental research that doesn’t focus on those specific areas. Its research mission is aimed
at understanding the principles, mechanisms, and processes that underlie living organisms,
often using research models.
NIGMS also supports the development of fundamental methods and new technologies to
achieve its mission. Supported research may utilize specific cells or organ systems if they serve
as models for understanding general principles.
NIGMS also supports research in specific clinical areas that affect multiple organ systems:
anesthesiology and peri-operative pain; sepsis; clinical pharmacology that is common to multiple
drugs and treatments; and trauma, burn injury, and wound healing.
The Institute is organized into divisions that support research, research training, and capacity building
in a range of scientific fields. To find out more information on the participating divisions and the
research supported, please visit the following links:
Division of Biophysics, Biomedical Technology, and Computational Biosciences
(BBCB) Division of Pharmacology, Physiology, and Biological Chemistry (PPCB)
Division for Research Capacity Building (DRCB)
Division of Training Workforce Development, and Diversity (TWD)
Budget Guidance
According to statutory guidelines, total funding support (direct costs, indirect costs, fee) normally
may not exceed the amounts defined by the SBA, which can be found on the NIH SEED website.
NIGMS will not accept applications with budget requests exceeding this hard cap with the
exception of projects that fit within the SBA-Approved Waiver Topics list for awards over the
statuary budget limitations. NIGMS sets its own budget limits for the specific research topics that
are granted a waiver to exceed the U.S. Small Business Administration hard budget caps. The
NIGMS budget limit for a Phase I project on an approved topic is $350,000 in total costs with a
project period up to 1 year. The budget limit for a Phase II project on an approved topic is
$2,500,000 in total costs for a project period up to 3 years. If considering a project with a budget
exceeding the hard cap, applicants are strongly encouraged to contact NIGMS program officials
prior to submission, and preferably in the early stages of application preparation. In all cases,
applicants should propose a budget that is reasonable and appropriate for completion of the
research project.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIGMS 114
Specific SBIR and STTR Program Information
https://www.nigms.nih.gov/grants-and-funding/research-funding/small-business-research
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NIGMS will accept Phase IIB SBIR-only Competing Renewal grant applications to continue the
process of developing products that ultimately require 1) clinical evaluation, 2) approval by a
Federal regulatory agency, or 3) continuing refinements that include but are not limited to cost
reduction, testing for performance, safety, reliability and/or durability, and meeting or establishing
standards, particularly for basic or clinical research instrumentation or durable medical
equipment (DME) designs. This renewal grant should enhance the likelihood that small business
will attract interest and investment by third parties. Such products include, but are not limited to
research equipment, biological products, devices, drugs, medical implants, etc. within the
mission of the NIGMS. Budgets for this Phase IIB Competing Renewal opportunity must follow
the guidelines for Phase II applications (described above). For awards that are intended to
support completion of research needed to obtain an Investigational New Drug application (IND)
or Investigational Device Exemption (IDE), applicants must provide evidence that they have
consulted formally with the FDA concerning the research needed for the development of a drug,
biologic or medical device and that the proposed research will address these regulatory
requirements.
Such evidence should include FDA correspondence from a pre-IND meeting for an IND
application or a pre-IDE meeting for an IDE application, and the status of the project in a
timeline related to Federal regulatory approval processes. Applicants should describe these
consultations and the outcomes in the Progress Report section of the Research Strategy and
provide FDA correspondence or meeting minutes as part of Just-in-Time procedures.
Prospective applicants considering a Phase IIB Competing Renewal application are strongly
encouraged to contact either the Program person of record for the Phase II award or NIGMS
contacts listed at the end of this NIGMS topics announcement.
To assist NIGMS in planning for Phase IIB applications, it is helpful for prospective applicants to
submit to the NIGMS Small Business Strategy Coordinator (listed below) a letter of intent that
includes the following information:
•
Descriptive title of the proposed research
•
Phase II grant number
•
Name, address, and telephone number of the Principal Investigator
•
Names of other key personnel
•
Participating institutions
•
Notice of Funding Opportunity Number
The letter is non-binding and does not enter the review process. It is anticipated that only a small
number of NIGMS SBIR Phase II awards will be eligible for a Phase IIB Competing Renewal
award.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIGMS 115
Clinical Trials
Does NIGMS accept Clinical Trials
through the Omnibus /Parent Notices of
Funding Opportunities?
No
Does NIGMS accept Clinical Trials
through specific Notices of Funding
Opportunities
No
Does NIGMS support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
PA-20-206 https://grants.nih.gov/grants/guide/pa-
files/PA20-206.html
PA-20-183 https://grants.nih.gov/grants/guide/pa-
files/PA20-183.html
Contact Information
For general information on the NIGMS Small Business program, contact:
Eddie Billingslea, Ph.D., NIGMS Small Business Strategy Coordinator
Email: NIGMS_SmallBusiness@nigms.nih.gov
For scientific questions about NIGMS-funded SBIR/STTR research, contact:
DIVISION OF PHARMACOLOGY, PHYSIOLOGY, AND BIOLOGICAL
CHEMISTRY
Pharmacological and Physiological
Sciences Sailaja Koduri, Ph.D.
Email: Sailaja.Kodur[email protected]ov
Biochemistry and Bio-related
Chemistry Kadir Aslan, Ph.D.
Email: kadir.aslan@nih.gov
DIVISION OF BIOPHYSICS, BIOMEDICAL TECHNOLOGY, AND COMPUTATIONAL BIOSCIENCES
Ashley Barnes, Ph.D.
Email: ashley.barnes@nih.gov
DIVISION OF TRAINING, WORKFORCE DEVELOPMENT, AND DIVERSITY
Sailaja Koduri, Ph.D.
Email: Sailaja.Koduri@nih.gov
NIH, CDC, and FDA Program Descriptions and Research Topics
NIGMS 116
DIVISION FOR RESEARCH CAPACITY BUILDING
Krishan Arora, Ph.D.
301-435-0763
Email: arorak@nigms.nih.gov
Research and Development in Science Education
Tony Beck, Ph.D.
301-480-4623
Email: beckL@mail.nih.gov
For administrative and business management questions, contact:
ADMINISTRATIVE AND BUSINESS MANAGEMENT
Mr. Brian Iglesias
301-451-5903
Email: iglesiab@mail.nih.gov
Ms. Ilene Glassman
301-594-4648
Email:
ilene.glassman@nih.go
v
Ms. Alania Foster
301-451-8255
Email: Alania.foster@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NHLBI 117
NATIONAL HEART, LUNG, AND BLOOD INSTITUTE (NHLBI)
Mission
The NHLBI plans, conducts, and supports research, clinical trials, demonstration, and educational
projects related to the causes, prevention, diagnosis, and treatment of heart, lung, blood, and sleep
disorders. It also supports research on the clinical use of blood products and all aspects of the
management and safety of blood resources. The NHLBI SBIR/STTR programs foster basic, applied,
and clinical research on all product and service development related to the mission of the NHLBI. The
NHLBI has four extramural program divisions, described below. For more information on the NHLBI
Strategic Vision, visit: https://www.nhlbi.nih.gov/sites/default/files/2017-11/NHLBI-Strategic-Vision-
2016_FF.pdf.
For the most up-to-date information on the NHLBI Small Business Programs, please visit the
NHLBI SBIR/STTR website: ( https://www.nhlbi.nih.gov/grants-and-training/funding-opportunities-
and-contacts/small-business-program) and subscribe to our listserv (http://bit.ly/NHLBI-SBIR-
Updates). You can also follow us on X at @NHLBI_SBIR. NHLBI encourages potential applicants
to contact us at: http://bit.ly/ContactNHLBIsbir.
Budget Guidance
Total funding support (direct costs, indirect costs, and fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SEED website. For budgetary, administrative, or
programmatic reasons, the NHLBI reserves the right to disapprove funding or decrease the length of
an award and/or the budget recommended by a review committee. NIH has received a waiver from
SBA, as authorized by statute, to exceed the statutory budget limitations set by the SBA for specific
topics relevant to the NHLBI that can be found in the SBA-Approved Waiver Topics. It is strongly
recommended for applicants who have a project that falls under one of the SBA-Approved Waiver
Topics to reference the corresponding topic in their Budget Justification document. For most projects
falling under one of the SBA-approved waiver topics, the NHLBI does not fund Phase I applications
greater than the current SBA-mandated budget guidelines (up to $306,872 total costs for Phase I
and up to $2,045,816 total costs for Phase II applications). However, the NHLBI may occasionally
fund projects above these guidelines for proposals falling under one of the approved waiver topics if
one or more of the following criteria have been fulfilled:
1.
The application involves the clinical testing of therapeutics, devices, imaging agents,
biologics, diagnostics, clinical and rehabilitation tools, and/or technologies.
2.
The application involves the use of a large animal model. For the purpose of this guidance,
large animal model species include: swine (porcine), sheep (ovine), cattle (bovine), cat
(feline), dog (canine), ferret, and nonhuman primates.
Applicants with budget requests exceeding these caps should be prepared to negotiate their
budget down if their proposal does not fall into one of the above categories.
Furthermore, all applicants with budget questions, or considering requesting a budget greater than
the set budget limitation amounts, are strongly encouraged to contact the NHLBI SBIR office at
http://bit.ly/ContactNHLBIsbir before submitting an application. For more information on the NHLBI
Budget Policy for SBIR/STTR grants, please refer to NOT-HL-24-008.

NIH, CDC, and FDA Program Descriptions and Research Topics
NHLBI 118
Specific SBIR and STTR Program Information
The NHLBI encourages applications proposing innovative technologies related to any area within the
NHLBI mission.
The NHLBI maintains a list of Notices of Special Interest (NOSIs) and funding opportunities that are
specific to the Institute. Instructions for submitting applications in response to these topics are posted on
the web page. The list is revised throughout the year, so please check regularly for updates.
For more information, contact the NHLBI Small Business team at http://bit.ly/ContactNHLBIsbir or
the Division contact associated with your technology area listed at the end of the NHLBI section.
Technical and Business Assistance
Applicants can request funding for technical and business assistance (TABA) in their application.
Small businesses should include this budget request as part of the application and provide a detailed
description of the vendor and services in the Budget Justification. TABA costs may be requested in
addition to the Phase I and II budget caps set by the NHLBI. The NHLBI may also fund TABA costs
through the administrative supplement program (PA-20-272). For more information about requesting
TABA funding through administrative supplements, please contact Dr. Jain Krotz at
jain.krotz@nih.gov.
Interim and Final Progress Reports
As detailed in NOT-OD-17-085, the NIH has implemented the Final Research Performance
Progress Reports (Final RPPR) for SBIR/STTR Final Progress Reports.
The NHLBI is interested in tracking the progress of the small business concerns it funds and the
products they develop. Funding priority will be given to those small business concerns that show
both their ability to develop products and their growth as a small business concern towards
independence from the SBIR/STTR program.
Additionally, the NIH requires all SBIR/STTR grantees to submit their Life Cycle Certification
document with their Interim Research Performance Progress Reports (Interim RPPR). For more
information on this requirement, please reference NOT-OD-19-025.
Specific Funding Opportunities and Programs
The NHLBI encourages applications focused on innovative technologies that align with its
Mission (https://www.nhlbi.nih.gov/about/mission-statement) through the Omnibus Notices of
Funding Opportunity (NOFOs). In addition to this Omnibus program announcements, the NHLBI
releases targeted NOFOs and Notices of Special Interest (NOSIs) throughout the year. Sign up
for the listserv (http://bit.ly/NHLBI- SBIR-Updates) to be notified of new NOFOs and NOSIs.
Please note that NOFOs and NOSIs can be released or expire at any time throughout the year;
please refer to the NHLBI SBIR/STTR website for active announcements supported by NHLBI.
Programs and Services for NHLBI Small Business Awardees
The NHLBI offers free assistance to applicants and awardees regarding intellectual property,
commercialization, and business plan development. Please visit https://nhlbi.my.salesforce-
sites.com/ to request services.
The NHLBI hosts “Small Biz Hangouts” - a free educational series covering the basics of biomedical
technology development. Previous Hangouts are archived on the NHLBI YouTube channel Small
Business Resources playlist. Sign up for the NHLBI listserv (http://bit.ly/NHLBI-SBIR-Updates) to learn
about upcoming live events, program announcements. Learn more about available resources at

NIH, CDC, and FDA Program Descriptions and Research Topics
NHLBI 119
http://www.nhlbi.nih.gov/about/org/dera/otac/resources.
Regulatory and Commercialization Guidance for Phase II and Fast-Track Applicants
The NHLBI strongly encourages applicants to include a robust regulatory strategy with
corresponding milestones in Phase II and Fast-Track applications. Applicants are also
encouraged to include letters of support or other evidence documenting their regulatory
strategy. Furthermore, the NHLBI also strongly encourages applicants to describe the
following elements in their commercialization plan: management team, market size/opportunity,
competitive advantage, intellectual property, potential impact on healthcare costs and
outcomes, pricing and reimbursement strategy, the clinical trial plan (if applicable), and the
proposed go-to-market strategy for the technology. The NHLBI will consider the strength of
these elements when making funding decisions for Phase II and Fast-Track applications.
For more information on additional factors that are involved with making funding decisions for
Phase II SBIR/STTR applications and other applications funded through the Zone of
Consideration, please visit https://www.nhlbi.nih.gov/current-operating-guidelines.
For assistance regarding the Phase II commercialization plan, watch the “Small Biz Hangout” for
advice on Writing Your Phase II Commercialization Plan (http://bit.ly/Ph2CommPlanHangout) and
contact Stephanie Davis (nhlbi_sbir@mail.nih.gov) with specific questions.
Phase IIB Competing Renewal Awards
NHLBI only accepts Phase IIB SBIR/STTR Competing Renewal applications through specific
opportunities. These opportunities can be found on the NHLBI Funding Opportunities webpage:
https://www.nhlbi.nih.gov/grants-and-training/funding-opportunities-and-contacts/small-business-
program. Applicants are strongly encouraged to contact Dr. Jain Krotz at jain.krotz@nih.gov for
additional information on Phase IIB funding opportunities.
Commercialization Readiness Pilot (CRP)
The NHLBI welcomes the submission of Commercialization Readiness Pilot (CRP) program
applications from current or past Phase II/IIB awardees. The CRP aims to facilitate the transition of
previously or currently funded SBIR and STTR Phase II and Phase IIB projects to the
commercialization stage by providing additional support for technical assistance and later-stage
research and development (R&D) not typically supported through Phase II or Phase IIB grants or
contracts. NHLBI will accept budgets up to $500,000 total costs (direct costs, indirect costs, fee)
across up to two years. NHLBI participates in the following CRP NOFOs below:
•
SBIR/STTR Commercialization Readiness Pilot (CRP) Program Technical Assistance
and Late Stage Development - Clinical Trial Not Allowed (PAR-23-219)
•
SBIR/STTR Commercialization Readiness Pilot (CRP) Program Technical Assistance
and Late Stage Development - Clinical Trial Required (PAR-23-220)
Clinical Trials
Does NHLBI accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
Yes
Please reach out to program staff at least 30 days
before the application deadline to discuss your
project if submitting a Clinical Trial application.
Does NHLBI accept Clinical Trials
through specific Notices of Funding
Opportunities?
Yes
Please reach out to program staff at least 30 days
before the application deadline to discuss your
project if submitting a Clinical Trial application.
NIH, CDC, and FDA Program Descriptions and Research Topics
NHLBI 120
Does NHLBI support Clinical Trials
through NON-SBIR/STTR Notices
of Funding Opportunities?
Yes
For information on non-SBIR/STTR clinical trials
funding mechanisms for which small businesses
are eligible, please visit the NHLBI clinical trials
website
Research Topics
Cardiovascular Sciences
The Division of Cardiovascular Sciences (DCVS) supports basic, clinical, population, and health
services research on the causes, prevention, and treatment of cardiovascular diseases. The
research programs of the Division encompass investigator-initiated research, Institute-initiated
research in targeted areas of research need and scientific opportunity, specialized centers of
research focused on selected research topics, and clinical trials. Research supported by the Division
is concerned with the etiology, pathogenesis, prevention, diagnosis, and treatment of coronary artery
disease and atherothrombosis; pediatric and structural heart disease; heart failure and arrhythmias;
and hypertension and vascular diseases. DCVS also supports investigations into the development
and use of medical devices, imaging devices, software programs, AI/ML technologies, molecular
technologies and other tools to improve cardiovascular health. A broad array of epidemiological
studies is supported by the DCVS to describe disease and risk factor patterns in populations and to
identify risk factors for disease. Also supported are clinical trials of interventions to prevent and treat
disease; studies of genetic, behavioral, sociocultural, and environmental influences on disease risk
and outcomes; and studies of the application of prevention and treatment strategies to determine
how to improve clinical care and public health. If you would like to learn more about the scientific
areas covered by the DCVS and connect with a program officer, please visit the division website:
Division of Cardiovascular Sciences | NHLBI, NIH.
Lung Diseases
The Division of Lung Diseases (DLD) supports research on the causes, diagnosis, management,
prevention, and treatment of lung diseases and sleep disorders. Research is funded through
investigator- initiated and Institute-initiated grant and contract programs in areas including
asthma, bronchopulmonary dysplasia, chronic obstructive pulmonary disease, cystic fibrosis,
respiratory neurobiology, critical care and acute lung injury, developmental biology, pediatric and
neonatal pulmonary diseases and care, immunologic and fibrotic pulmonary disease, rare lung
disorders, pulmonary vascular disease, and pulmonary complications of AIDS and tuberculosis.
Also supported are mechanistic and non-mechanistic clinical trials to predict, prevent and treat
pulmonary disease; digital health including mobile / tele-health, wearable devices, respiratory
surgical devices, aerosol drug or gas delivery, supplemental oxygen, bioinformatics, mechanical
ventilation, imaging devices, personalized medicine and AI/ML to help inform clinical decision
making in pulmonary medicine. If you would like to learn more about the scientific areas covered
by the DLD and connect with a program officer, please visit the division website: Division of Lung
Diseases | NHLBI, NIH.
Sleep and Circadian Biology
The National Center for Sleep Disorders Research (NCSDR) supports research on the causes,
prevention, and treatment of sleep disorders and the promotion of sleep health. Research is
funded through investigator- initiated and Institute-initiated, grant, and contract programs in

NIH, CDC, and FDA Program Descriptions and Research Topics
NHLBI 121
sleep and circadian biology research projects. The NCSDR is interested in funding projects
related to the regulation of sleep and sleep disorders including circadian disorders, insomnia and
obstructive sleep apnea. The NCSDR is also interested in research focused on the development
of tools, devices and data science approaches for the early prediction, detection, and treatment
of sleep deficiency and sleep and circadian disorders. If you would like to learn more about the
scientific areas covered by the NCSDR and connect with a program officer, please visit the
website: National Center on Sleep Disorders Research | NHLBI, NIH.
Blood Diseases and Resources
The Division of Blood Diseases and Resources (DBDR) supports research on the causes,
prevention, and treatment of nonmalignant blood diseases, including anemias, sickle cell disease,
hemophilia and thalassemia; premalignant processes such as myelodysplasia and myeloproliferative
disorders; and other abnormalities of hemostasis and thrombosis; and immune dysfunction.
Research supported by the Division encompasses a broad spectrum of topics ranging from basic
biology and mechanism of action, to medical management of blood diseases. The Division has a
major responsibility for research to improve the adequacy and safety of the nation's blood supply. It
also plays a leading role in transfusion medicine and blood banking, including research to evaluate
blood donation screening, manufacturing, processing technologies and storage. The Division also
has a major responsibility supporting research in hematopoiesis and stem cell biology and disease. It
also supports hematopoietic stem cell transplantation research and the application of stem cell
biology findings to the development of new cell-based therapies to repair and regenerate human
tissues and organs. If you would like to learn more about the scientific areas covered by the DBDR
and connect with a program officer, please visit the division website: Division of Blood Diseases and
Resources | NHLBI, NIH.
Center for Translation Research and Implementation Science
The Center for Translation Research and Implementation Science (CTRIS) plans, fosters, and
supports an integrated and coordinated program of research to understand the multi-level
processes and factors that are associated with successful integration of evidence-based
interventions within specific clinical and public health settings such as worksites, communities,
and schools; identifies and makes readily available to implementation and dissemination
practitioners emergent knowledge about the late phases of translation research, especially the
"T4" phase, for rapid and sustained adoption of effective interventions in real world settings;
leads the NHLBI effort in the rigorous, systematic evidentiary reviews and subsequent NHLBI
participation in the collaborative model for clinical practice guidelines development; supports
training and career development of personnel in "T4" translation research and health inequities
relating to heart, lung, and blood diseases; provides a focal point for advice and guidance on
matters pertaining to minority health, health inequities and minority participation in research;
represents the NHLBI to other governments, other Federal Departments and agencies,
international organizations, and the private sector on global health issues; and provides data
analytics and portfolio analysis to evaluate and inform future directions of implementation
research programs. If you would like to learn more about the scientific areas covered by the
CTRIS and connect with a program officer, please visit the division website: Center for
Translation Research and Implementation Science | NHLBI, NIH.
Contact Information
SBIR OFFICE
For general questions about the NHLBI SBIR/STTR grant program,
please contact:

NIH, CDC, and FDA Program Descriptions and Research Topics
NHLBI 122
Stephanie Davis, Ph.D.
Division of Extramural Research Activities
Phone: 1-301-496-8412
Email:mailto: nhlbi_sbir@mail.nih.gov
For administrative and business management questions, please contact:
Andre Walker
Division of Extramural Research
Activities Phone: 1-301-827-8061
Email: walkera@nhlbi.nih.gov
CARDIOVASCULAR SCIENCES
Rahul Thakar, Ph.D.
Division of Cardiovascular Sciences
Advanced Technologies and Surgery Branch
Phone: 301-827-8151
Email: rahul.thakar@nih.gov
LUNG DISEASES AND SLEEP AND CIRCADIAN BIOLOGY
Lei Xiao, M.D., Ph.D.
Division of Lung Diseases
Restrictive and Vascular Lung Diseases Branch
Phone: 301-827-7852
Email: lei.xiao@nih.gov
Sidd Shenoy, Ph.D.
Division of Lung Diseases
Acute and Infectious Lung Diseases Branch
Phone: 301-827-4751
Email: sidd.shenoy@nih.gov
For sleep-related proposals, please contact:
Shilpy Dixit, Ph.D. Division of Lung Diseases
National Center for Sleep Disorders Research
Phone: 301-402-9064
Email: shilpy.dixit@nih.gov
BLOOD DISEASES AND RESOURCES
Ron Warren, Ph.D.
Division of Blood Diseases and Resources
Translational Blood Science and Resources Branch
Phone: 301-827-8288
Email: ronald.warren@nih.gov
Ilana Goldberg, Ph.D.
Division of Blood Diseases and Resources
Molecular, Cellular & Systems Blood Science
Branch Phone: 301-496-8587
Email: ilana.goldberg@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NHGRI 124
NATIONAL HUMAN GENOME RESEARCH INSTITUTE (NHGRI)
Mission
The National Human Genome Research Institute (NHGRI) has been guided, since the inception
of the Human Genome Project in 1990, by a sequential series of plans, each of which has been
developed with considerable input from the scientific community. These plans have always laid
out ambitious goals and measurable objectives to gauge progress. The Institute recently
examined the current state of genomics and gathered input on its future directions, which
resulted in a 2020 strategic plan to guide NHGRI and research at the forefront of genomics
(Green et al. Strategic vision for improving human health at The Forefront of Genomics. Nature
586, 683–692 (2020).
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SEED website, unless the application fits an
SBA-approved NHGRI waiver topic. For topics listed in the SBA-Approved Waiver Topics, the
NHGRI generally will not fund Phase I applications to the Omnibus greater than $400,000 total
costs or Phase II applications greater than $2,150,000 total costs (not including the costs of
technical and business assistance). For budgetary, administrative, or programmatic reasons, the
NHGRI may not fund an application or may decrease the length of an award and/or the budget
recommended by a review committee. Applicants with budget questions or considering
requesting a budget greater than these amounts are strongly encouraged to contact
program staff before submitting an application.
Specific SBIR and STTR Program Information
Information about the NHGRI Small Business Program and specific funding announcements is
available online. Applicants are strongly encouraged to discuss their research plans with NHGRI
Program Staff prior to submitting their applications.
Clinical Research Support
The National Human Genome Research Institute (NHGRI) will accept applications designated
as clinical trials for all program areas supported by the Institute as outlined below for non-clinical
trials small business grants. The broadened definition of clinical trials as defined in NOT-OD-15-
015 and on the NIH website is not intended to expand the scope of applications accepted by
NHGRI beyond studies that have a major genomic or Ethical, Legal and Social Implications
(ELSI) component and relate clearly to NHGRI’s mission. Information on areas of research
interest is available on the NHGRI Research Funding Divisions homepage and the ELSI
Research Domains website.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NHGRI does not accept applications for Phase IIB competing renewal awards through this
Omnibus solicitation. NHGRI participates in the CRP program.
NIH, CDC, and FDA Program Descriptions and Research Topics
NHGRI 125
Clinical Trials
Does NHGRI accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
Yes
Does NHGRI accept Clinical Trials
through specific Notices of Funding
Opportunities?
No
Does NHGRI support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
Research Topics
A.
Technology and Methods Development
Technology development in DNA sequencing, genotyping, and single-cell analysis are
examples of activities that have changed the nature of what scientific research questions are
practical to address, have enabled new approaches, and have facilitated the development of
new community resource data sets. Many areas of critical importance to the realization of
the genomics-based vision for biomedical research require continued technological and
methodological developments before pilots and then large-scale approaches can be
attempted. Accordingly, the NHGRI will continue to support the development of new,
fundamental technologies in all areas of genomics. Important areas in which technology
development applications would be responsive include (but are not limited to) experimental
technologies and computational methods to analyze gene expression and other molecular
phenotypes; discovery and characterization of genetic variation; identification of the genetic
contributions to health, disease, and drug response; statistical analytic methods for
understanding human genomic variation and its relationship to health and disease; and
chemical genomics. There is also continued need to support technology development for the
comprehensive discovery of functional elements in the human and model organism
genomes and new nucleic acid sequencing technology. Many of these assays would benefit
from the ability to work with very small amounts of starting material down to the level of
single cells and subcellular compartments, along with minimally invasive human specimens
that are easy to collect, handle, and store. As these technologies mature, emphasis should
be on high throughput, cost-effective methods that consistently produce very high- quality
data.
The Institute also places high priority on contributing selectively to the development of new
and needed technology in related areas, such as proteomics and systems biology research,
when NHGRI funding can be used to further a truly unique development that will have a
significant impact on the field.
Further information on opportunities related to technology and methods development is
available on the NHGRI Genome Technology Program website.
B.
Bioinformatics, Computational Genomics, and Data Science
The ongoing development of new sequencing technologies has dramatically increased the
amount of data produced for genomics in basic science and translation to medicine. NHGRI
encourages new computational approaches for the analysis, visualization, and integration of
genomic information in basic and clinical research and in applications to improve its utility in
healthcare. These approaches may include the development of methods for processing,

NIH, CDC, and FDA Program Descriptions and Research Topics
NHGRI 126
annotating, interpreting, analyzing, and sharing of sequencing data with associated
phenotypes and other large-scale genomic data sets such as haplotype maps, genetic
variants, transcriptome measurements, functional elements, and, in some cases, protein
interactions. New tools for population-based analysis using the pangenome reference are of
interest. NHGRI also encourages the development of better computational solutions for
storage, access, compression, secure sharing, privacy, and transfer of large genomic
datasets by biomedical researchers.
NHGRI will support projects to improve informatics tools to make them more easily adopted
by any biomedical research laboratory that wishes to use genomic technologies to address
biomedical questions. This may include making them more efficient, reliable, robust, well-
documented, and well- supported, or deploying them in containers or at scale in a cloud-
based platform.
Where possible, existing or emerging community data standards, models, and methods for
data representation and exchange should be used in the development of these new
methods and tools as well as other approaches to enhance reproducibility. Standards-based
approaches such as GA4GH are also encouraged to integrate and share genomics and
phenotype data for data mining with other sources including for clinical application. Projects
that will make genomic digital objects Findable, Accessible, Interoperable, Reusable (FAIR)
in the broader community are highly recommended.
Further information on programs related to NHGRI supported research in these areas is
available on the Computational Genomics and Data Science Program website.
C.
Population Genomics and Genomic Medicine
Advances in the understanding of genomic variation across human populations and the functional
consequences of variants independently, in combination, and in different environmental contexts
have significantly impacted how genomic information can be used in both public health and
clinical practice settings, alternatively known as genomic medicine. An existing challenge is how
to capture, interpret, and return genomic information at high volumes and in a cost-effective
manner. Innovative technologies and methods are needed to allow information on genomic
variation to be used broadly in clinical settings while meeting regulatory requirements, to inform
public health efforts, and to accurately convey genomic risk profiles to a lay audience.
Biotechnology and informatics have enhanced our ability to survey the entire genome within and
among populations. This progress has allowed for improved inferences about evolution of the
genome and better characterization of populations, key elements of populations genomics. An
existing challenge is how to assemble and analyze multiple genomes using computational
methods to identify patterns of genomic divergence. Technology is needed to enable nuanced
incorporation of population-based discovery with detailed investigation of disease-based cohorts
and prospective variant evaluation. Population genomic information can be used to understand
disease process, improve risk prediction, and apply the results in patient care.
The research scope of Population Genomics and Genomic Medicine at NHGRI includes:
characterizing the spectrum and distribution of genetic variation in humans and other biomedically
relevant organisms; developing statistical and computational methods for comparing genomes
and genome function within and across species as well as for relating genetic variation to health-
and disease-related traits; developing resources and statistical methods for observational studies
and clinical trials incorporating advanced genomic technologies; conducting proof -of-principle
studies that apply genomic technologies to epidemiologic and clinical research; developing
research methods and infrastructure needed for future epidemiologic and clinical studies of
genetic and environmental contribution to disease; investigations of whether and how clinical

NIH, CDC, and FDA Program Descriptions and Research Topics
NHGRI 127
genome variation impacts disease prevention diagnosis, and treatment; studies of approaches to
improve the identification and interpretation of genomic variation for dissemination in clinical
settings; assessing phenotypic manifestations of genetic variation through electronic medical
records (EMRs); integrating genomic results and clinical decision support into EMRs; studies that
address current barriers to the implementation of clinical genome sequencing; and assessing the
impact of genetic information on clinical utility, health outcomes, and delivery of care.
For additional information about Genomic Medicine at NHGRI, please visit the Division of Genomic
Medicine website.
D.
Ethical, Legal and Social Implications
NHGRI, through the ELSI Research Program, supports research studies that examine
and address the ethical, legal, and social implications of genomics. These studies may
focus on issues associated with genomic research, genomic healthcare, the interplay
between the field of genomics and organizations, institutions, or other organized
stakeholders, and broader values and societal effects that shape and are shaped by
genomics.
More detailed information on specific ELSI research priorities within each of these broad
areas is available on the ELSI Research priorities website.
E.
Genomic training and education
NHGRI supports educational activities and curriculum development that increase genomics
knowledge of students, trainees, and genomics professionals. The goal of these activities is to
provide an avenue for entry and pursuit of genomics careers. The widespread impact of
genomics creates a need to train diverse groups of people to develop innovative and impactful
genomic research approaches and resources. Training opportunities may be proposed at the
undergraduate, postbaccalaureate, graduate, postdoctoral, or professional level.
For more information on genomic training and education at NHGRI, please visit the Training
Program website.
Contact Information
For more information on research topics,
contact:
Ian Nova, Ph.D.
Small Business Program Co-Lead (SBIR)
Phone: 240-987-2885
Email: ian.nova@nih.gov
Renee Rider, M.S., J.D., LCGC
Small Business Program Co-Lead (STTR)
Phone: 301-443-4336
Email: renee.rider@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIMH 128
NATIONAL INSTITUTE OF MENTAL HEALTH (NIMH)
Mission
The mission of the National Institute of Mental Health (NIMH) is to transform the understanding and
treatment of mental illnesses through basic and clinical research, paving the way for prevention,
recovery, and cure. Mental disorders constitute an immense burden on the U.S. population, with
suicide as one of the leading causes of death in the US, major depression the leading cause of
disability in the U.S., and schizophrenia, bipolar disorder, and obsessive-compulsive disorder ranked
among the ten leading causes of disability. NIMH also takes a leading role in understanding the
impact of behavior on HIV transmission and pathogenesis, and in developing effective behavioral
preventive interventions. The NIMH conducts a wide range of research, research training, research
capacity development, as well as public information outreach and dissemination to fulfill its mission.
The NIMH Strategic Plan (http://www.nimh.nih.gov/about/strategic-planning-reports/index.shtml) and
the National Advisory Mental Health Council’s workgroup report “From Discovery to Cure”
http://www.nimh.nih.gov/about/advisory- boards-and-
groups/namhc/reports/fromdiscoverytocure_103739.pdf present key scientific priorities across these
domains, and describe the need for technologies to realize these priorities. Research priorities for
the NIMH further include aspects of HIV/AIDS prevention, treatment, and care, in accordance with the
Trans-NIH Plan for HIV-Related Research (https://www.oar.nih.gov/hiv-policy-and-
research/strategic-plan).
For the Institute to continue fulfilling this vital public health mission, it must foster innovative thinking
and ensure that a full array of novel scientific perspectives is used to further discovery in the evolving
science of brain, behavior, and experience. In this way, breakthroughs in science can become
breakthroughs for all people with mental illnesses.
The NIMH SBIR/STTR programs support small businesses to develop technologies that can advance
the mission of the Institute, including in basic neuroscience research relevant to mental disorders,
translational and clinical research of mental disorders, clinical diagnosis or treatment of mental
disorders, and dissemination of evidence-based mental health care.
For additional information about areas of interest to the NIMH, please visit our home page at
http://www.nimh.nih.gov and the NIMH SBIR/STTR program priorities page at
https://www.nimh.nih.gov/funding/sbir/nimh-sbir-sttr-program-priorities.shtml. NIMH has released a notice
of considerations regarding the use of animal neurobehavioral approaches, please see NOT-MH-19-053:
https://grants.nih.gov/grants/guide/notice-files/NOT-MH-19-053.html. NIMH has specific requirements for
clinical trials, please visit: https://www.nimh.nih.gov/funding/opportunities-announcements/clinical-trials-
foas/index.shtml. A 2020 NAMHC Workgroup on Drug Development provides further guidance on
pharmacologic trial designs.
NIMH has additional guidance on areas of interest in stress biology research, please see NOT-MH-
18-058: https://grants.nih.gov/grants/guide/notice-files/NOT-MH-18-058.html. NIMH has released a
notice of considerations for preclinical research involving psychedelics and related compounds:
https://grants.nih.gov/grants/guide/notice-files/NOT-MH-23-125.html. The NIMH SBIR/STTR
programs will only consider preclinical psychedelics and/or related compound projects.
The NIMH SBIR/STTR website provides guidance and resources for applicants:
https://www.nimh.nih.gov/funding/sbir/.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIMH 129
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SEED website. NIH has received a waiver from
the SBA, as authorized by statute, to exceed the statutory budget limitations set by the SBA for
specific topics relevant to the NIMH that can be found in the SBA-Approved Waiver Topics. Please
note, for budgetary, administrative, or programmatic reasons, the NIMH may not fund an application
or may decrease the length of an award and/or the budget recommended by a review committee.
Applicants with budget questions or considering requesting a budget greater than the SBA
defined amounts are strongly encouraged to contact program staff before submitting an
application.
Specific SBIR and STTR Program Information
1.
Potential SBIR/STTR applicants should contact NIMH prior to submitting an application to
ensure the application is of priority/interest to NIMH. Please see the Contacts section.
2.
An additional criterion that the federal government considers in supporting a small
business with SBIR/STTR funds, is past commercialization performance. It is expected that
small businesses who have received previous SBIR/STTR grants, have had success in
commercializing their previously supported technologies. Small businesses that are mostly
interested in research and development (and not commercialization) should consider other
grant mechanisms at NIH, rather than the SBIR/STTR program. Program staff at NIMH
can help identify the most appropriate grant mechanism to use.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
The NIMH will accept Phase IIB SBIR/STTR Competing Renewal grant applications in two
categories: 1) to continue research and development of technologies that ultimately require federal
regulatory approval, and 2) to continue research and development of complex instrumentation,
clinical research tools, or behavioral/digital health interventions and treatments.
Technologies in the former category (those that ultimately require federal regulatory approval) include
but are not limited to pharmacologic agents and drugs, biological products, medical devices,
vaccines, etc. related to the mission of the NIMH. Phase IIB SBIR/STTR Competing Renewal grants
for such technologies should allow small businesses to move research and development to a stage
where interest and investment by third parties is more likely.
Companies that are developing technologies that do not focus on drug development, but that require
federal regulatory approval prior to commercialization, may be eligible to submit a Phase IIB
Competing Renewal application.
For both technology areas, Phase IIB applications may be submitted through the Omnibus
SBIR/STTR Notice of Funding Opportunity. Generally, for this opportunity, budget limits of $3 million
total costs and time periods up to 3 years may be requested. These budget allowances have been
approved by the SBA through a waiver. For budgets higher than $3 million, contact NIMH program staff
prior to submitting the application.
The following examples would make appropriate topics for proposed NIMH SBIR/STTR Phase IIB
Competing Renewal projects. These are meant for illustrative purposes only and are not exclusive of
other appropriate activities:
•
Preclinical studies, including pharmacology and toxicology, beyond those conducted under the
Phase I (R43/R41) and initial Phase II (R44/R42) grants. Some in vivo or in vitro studies would
be expected to have been carried out in Phase I or the initial Phase II grant.
NIH, CDC, and FDA Program Descriptions and Research Topics
NIMH 130
•
Completion of studies as required by the Food and Drug Administration (FDA) for
Investigational New Drug (IND) or Radioactive Drug Research Committee (RDRC)
application.
•
Studies in normal healthy volunteers to determine a drug’s safety profile, metabolism, etc.
•
Assessment of devices with regard to performance standards related to the FDA approval process.
•
Safety and effectiveness studies of novel medical devices.
•
Evaluation of novel imaging approaches for diagnostic purposes.
•
Clinical studies in support of Pre-Market Approval for biomarkers/medical devices by the FDA.
Although technologies in the latter category listed above (complex instrumentation, clinical research tools,
or behavioral interventions/treatments) may not require federal regulatory approval, extraordinary time
and effort is needed for their research and development. Therefore, NIMH supports Phase IIB Competing
Renewal awards of existing Phase II grants for such technologies. The Phase IIB Competing Renewal
award for these would provide up to an additional three years of support at total cost funding levels of up
to $3 million (generally) for the project. These budget allowances have been approved by the SBA
through a waiver.
Please contact the Program Director in the appropriate Division or Dr. Margaret Grabb (listed
below) before beginning the process of putting an application together. In addition, prospective
applicants are encouraged to submit to the program contact a letter of intent that includes the
following information:
•
Descriptive title of the proposed research
•
Name, address, and telephone number of the Principal Investigator
•
Names of other key personnel
•
Participating institutions
•
Notice of Funding Opportunity (e.g., PA-19-273).
Although a letter of intent is not required, is not binding, and does not enter into the review of a
subsequent application, the information that it contains allows NIH staff to estimate the potential
review workload and plan the review. It is expected that only a portion of NIMH SBIR Phase II
awards will be eligible for a Phase IIB Competing Renewal grant.
Clinical Trials
Does NIMH accept Clinical Trials
through the Omnibus/Parent Notices
of Funding Opportunities?
Yes
NIMH will prioritize funding for SBIR/STTR
applications with a clinical trial focus that are
consistent with the stated research goals and priorities
relevant to clinical trials as outlined in the clinical trials
NOFOs. For more information
see: https://www.nimh.nih.gov/funding/sbir/sbir-
amp-sttr-funding-opportunity-announcements
Does NIMH accept Clinical Trials
through specific Notices of Funding
Opportunities?
Yes
NIMH will prioritize funding for SBIR/STTR
applications with a clinical trial focus that are
consistent with the stated research goals and priorities
relevant to clinical trials as outlined in the clinical trials
NOFOs. For more information
see: https://www.nimh.nih.gov/funding/sbir/sbir-
amp-sttr-funding-opportunity-announcements

NIH, CDC, and FDA Program Descriptions and Research Topics
NIMH 131
Does NIMH support Clinical Trials
through NON-SBIR/STTR Notices
of Funding Opportunities?
Yes
NIMH will prioritize funding for SBIR/STTR
applications with a clinical trial focus that are
consistent with the stated research goals and priorities
relevant to clinical trials as outlined in the clinical trials
NOFOs.
For more information
see: https://www.nimh.nih.gov/funding/sbir/sbir-
amp-sttr-funding-opportunity-announcements
And:
•
First in Human and Early Stage Clinical Trials of
Novel Investigational Drugs or Devices for
Psychiatric Disorders (U01 Clinical Trial
Required) PAR-21-133
•
Early Stage Testing of Pharmacologic or
Device-based Interventions for the Treatment of
Mental Disorders (R61/R33- Clinical Trial
Required) PAR-21-137
•
Early Stage Testing of Pharmacologic or
Device-based Interventions for the Treatment of
Mental Disorders (R33- Clinical Trial Required)
PAR-21-136
•
Development of Psychosocial Therapeutic and
Preventive Interventions for Mental Disorders
(R61/R33- Clinical Trial Required) PAR-21-135
•
Development of Psychosocial Therapeutic and
Preventive Interventions for Mental Disorders
(R33 Clinical Trial Required) PAR-21-134
•
Confirmatory Efficacy Clinical Trials of Non-
Pharmacological Interventions for Mental
Disorders (R01 Clinical Trial Required) PAR-21-
132
•
Pilot Effectiveness Trials for Treatment,
Preventive and Services Interventions (R34-
Clinical Trial Required) PAR-21-131
•
Clinical Trials to Test the Effectiveness of
Treatment, Preventive, and Services
Interventions (R01 Clinical Trial Required) PAR-
21-130
•
Clinical Trials to Test the Effectiveness of
Treatment, Preventive, and Services
Interventions (Collaborative R01 - Clinical Trial
Required) PAR-21-129
Research Topics
Division of Neuroscience and Basic Behavioral Science (DNBBS)
The Division of Neuroscience and Basic Behavioral Science provides support for research programs
in the areas of basic neuroscience, genetics, basic behavioral science, research training, resource
development, technology development, drug discovery, and research dissemination. The Division has
the responsibility, in cooperation with other components of the Institute and the research community,
for ensuring that relevant basic science knowledge is generated and then harvested to create
improved diagnosis, treatment, and prevention of mental and behavioral disorders.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIMH 129
In this Division, the SBIR and STTR programs support research and the development of tools
related to basic brain and behavioral science, genetics, and drug discovery and development
relevant to the mission of the NIMH. Such tools include software (such as informatics tools and
resources and tools for analyzing data); hardware (such as the development of instrumentation or
devices); wetware (such as the use of iRNAs or other bioactive agents as research tools or
molecular imaging agents or genetic approaches to label neural circuits or modify circuit functions);
and drug discovery related technologies such as high throughput screening (HTS) or computational
pharmacology approaches. Assay development projects should follow the best practices laid out in
the Assay Guidance Manual: https://www.ncbi.nlm.nih.gov/books/NBK53196/.
AREAS OF EMPHASIS
•
Novel imaging probes to study brain structure and function at all levels, from the
molecular level to the whole organ, using any imaging modality (PET, fMRI, optical, etc.)
in animal or human studies.
•
Drug discovery/drug development of novel compounds which act on molecular pathways
(receptors, enzymes, second messengers, etc.) that are not typically targeted with currently
available psychiatric drugs, and that have a strong biological justification as a novel
mechanism for treatment of psychiatric disorders.
•
First in human drug trials.
•
Novel screening assays for high throughput acquisition and analysis of data about behavior
and the brain, from the level of genes to behavior.
•
Novel technologies that would enable researchers to study how populations of neural
cells work together within and between brain regions, in order to understand how changes
in neural activity contributes to mental disorders, using animals or when applied to
humans.
•
Develop informatics tools to facilitate the analysis and sharing of data between laboratories
about behavior and the brain. This could include common data element efforts but is not
limited to that area.
•
Technologies consistent with the goals of the BRAIN Initiative:
http://www.braininitiative.nih.gov/, including human/ clinical-based technologies.
Prospective applicants are strongly encouraged to contact Dr. Margaret Grabb and Ms. Paige
Anderson (listed below) with questions about the relevance of their interests to the mission of this
division.
Division of Translational Research (DTR)
The DTR directs, plans, and supports programs of research and research training that translate
knowledge from basic science to discover the etiology, pathophysiology, and trajectory of mental
disorders and develops effective interventions for children and adults. DTR supports integrative,
multidisciplinary research on the following areas: the phenotypic characterization and risk factors for
psychiatric disorders; neurobehavioral mechanisms of psychopathology; trajectories of risk and
resilience based on the interactive influences of genetics, brain development, environment, and
experience; and design and testing of innovative psychosocial, psychopharmacologic, and somatic
treatment interventions.
In this Division, the SBIR and STTR Programs support research aimed at facilitating the validation
and commercialization of new methods of assessing psychopathology and measuring treatment

NIH, CDC, and FDA Program Descriptions and Research Topics
NIMH 130
response to therapeutic agents. In addition, the SBIR and STTR Programs support the clinical
development of interventions, including novel pharmacologic agents or brain stimulation devices
as well as technology development used to deliver novel psychosocial approaches to the treatment
of mental illness in adults, pediatrics and geriatrics. For more information on NIMH supported
clinical trials and requirements, see: https://www.nimh.nih.gov/funding/opportunities-
announcements/clinical-trials-foas/index.shtml
AREAS OF EMPHASIS
•
Develop valid measures of the various constructs in the Research Domain Criteria (RDoC)
matrix (see https://www.nimh.nih.gov/research/research-funded-by-nimh/rdoc/index.shtml),
e.g., behavioral tasks, psychometrically sophisticated self-report measures, and measures of
physiological and neural activity, into a commercial product.
•
Conduct early stage, proof of concept clinical trials to advance the development of novel
therapeutics. The clinical trials are expected to include biological/behavioral data to assess
target engagement and to help determine potential success or failure of the compound
before moving on to larger clinical trials (see NOT-MH-11-015
http://grants.nih.gov/grants/guide/notice-files/NOT-MH-11-015.html).
•
Develop, test and perform initial validation of reliable and stable biomarkers that can identify
at-risk individuals prior to disease onset, improve diagnosis and classification, predict
treatment response, or to measure disease progression. Biomarkers are also needed in
clinical trials to identify dose ranges, to identify a specific subpopulation of subjects to enroll
in a treatment trial, or to measure efficacy or toxicity/side effects. Biomarkers in psychiatry
will initially be appropriate as clinical research tools, and only after significant technical and
clinical validation, could move toward diagnostic utility or other context of uses.
•
Development of novel diagnostic tools and innovative measures of treatment response and
disease progression, preclinical or clinical efficacy testing, or toxicity measures for drug
development.
•
Development of hardware and software tools to enable refined physiological and
behavioral assessment of normal and atypical neurodevelopment focused on pediatrics,
adult and geriatric age ranges.
•
Web-based tools and biosensors to enhance prevention, early identification and treatment of
pediatric mental disorders by various educational and health professionals.
•
Development of hardware and software tools to support operations of multi-site clinical trials.
•
Development of novel methods to enhance efficiency of early phase clinical trials.
•
Novel technologies and data analytic tools to enable quantification of behavioral data that is
relevant to research or clinical trials in mental disorders and/or autism.
•
Development of imaging technologies that can reveal specific pathologies in major mental
disorders.
Prospective applicants are strongly encouraged to contact Dr. Margaret Grabb (listed below) with
questions about the relevance of their interests to the mission of this division.
Division of AIDS Research (DAR)
The NIMH DAR supports scientific research to understand and alleviate the consequences of HIV
NIH, CDC, and FDA Program Descriptions and Research Topics
NIMH 131
disease on the central nervous system, and research to strengthen the provision and outcomes of
HIV/AIDS prevention and treatment. Examples of high-priority research areas for SBIR/STTR
applications are described below.
•
Develop and test novel, non-invasive diagnostic approaches (instrumentation, imaging,
biomarkers, central nervous system [CNS] cell-based in vitro models) to comprehend HIV-
1 associated CNS dysfunction and innovative technologies to study the mechanisms
involved in HIV-1 associated neuropathogenesis and persistence of HIV-1 in the CNS.
•
Design and test novel therapeutic interventions aimed at amelioration of HIV-1
associated CNS dysfunction, and/or eradication of HIV-1 from CNS reservoirs, and/or
strategies to prevent viral resurgence in the CNS upon cessation of anti-retroviral
therapy.
•
Tools to assess neurotoxicity profiles of antiretroviral medications and pharmacological
strategies to reduce adverse effects of anti-retroviral drugs (neuropsychiatric side effects
and drug-drug interactions).
•
Develop new tools/ techniques to aid in deciphering the complex neuro-immune
interactions at a molecular and cellular level in the context of HIV.
•
Develop or adapt neurological/neuropsychological/neurobehavioral assessments to
evaluate HIV-1 associated abnormalities in adults or children in resource limited
environments that are adaptable to different cultures and languages.
•
Build and optimize informatics tools to aid in analyzing and characterizing the phenotype
of CNS disease modalities associated with HIV by using machine learning, big data and
systems biology- based approaches.
•
Develop technologies and tools to increase regular HIV testing and support uptake,
adherence, and persistence to biomedical HIV prevention regimens among persons
behaviorally vulnerable to HIV or to biomedical HIV treatment regiments among people
newly diagnosed with HIV.
•
Develop innovative tools and approaches that use existing patient-level data, such as
electronic medical records and prescription refill or claim data, to improve engagement in
HIV care or HIV treatment adherence to strengthen sustained viral suppression, including
development and testing of predictive algorithms to identify those at risk for future non-
adherence.
•
Develop approaches that seamlessly integrate tools for mental health screening and
treatment into HIV healthcare or increase the capacity of HIV clinics to address mental
health concerns.
•
Develop decision support tools that help individuals, couples, and clinicians make informed
choices about the increasing number of proven and available HIV prevention and
treatment regimens, including long-acting regimens and multipurpose prevention
technologies (MPTs).
•
Develop innovative wireless technologies, remote sensing devices, biomarkers, assays, or
other novel methods to improve scientific measurement of HIV exposure due to sexual
behavior, or scientific measurement of social determinants that influence HIV treatment and
prevention. Assessment approaches could occur retrospectively (not a clinical trial – using
existing data, such as electronic medical records).
•
Develop and improve digital communication technologies to raise HIV awareness and

NIH, CDC, and FDA Program Descriptions and Research Topics
NIMH 132
promote accurate and timely health information to users, groups, and geographic regions
most impacted by HIV.
•
Develop and test tools, curricula, and strategies that seek to reduce documented HIV-
related disparities and health inequities (e.g., age, sex, gender identity, sexual
orientation, race, ethnicity, socioeconomic circumstance, etc.) in HIV incidence and HIV
prevention, treatment, and cure outcomes.
•
Develop innovative long-acting systemic and non-systemic multipurpose prevention
technologies that prevent HIV infection and pregnancy (hormonal and non-hormonal
methods) in adolescents and young women.
Prospective applicants are strongly encouraged to contact Dr. Vasudev R Rao (listed below) with
questions about the relevance of their interests to the mission of this division.
Division of Services and Intervention Research (DSIR)
The Division of Services and Intervention Research (DSIR) SBIR/STTR supports two critical areas of
research for people with or at risk for mental illness:
•
Intervention research to evaluate the efficacy and effectiveness of pharmacologic,
psychosocial, somatic, rehabilitative, sequential and combination interventions on mental
and behavior disorders- including acute and longer-term therapeutic effects on functioning
across domains for children, adolescents, and adults.
•
Mental health services research to improve the access, continuity, equity, value, quality and
outcomes of mental health care, as well as to improve the dissemination of information about
and the implementation of effective interventions, to strengthen the public health impact of
NIMH research.
The intervention research program aligns with NIMH Strategic Objectives 3.2 and 3.3 and addresses
the efficacy/effectiveness of treatment and preventive interventions in usual practice and community
settings with the purpose of informing clinicians, patients, families, and health policy makers on
evidence-based practices. In funding decisions, special emphasis is placed on the potential clinical
and/or public health impact of the research activities and on the implications of the research findings
for improving community practice and health outcomes. Types of interventions include the full range
of behavioral, psychotherapeutic, pharmacologic, and non-pharmacologic somatic or
complementary/alternative interventions, as well as rehabilitation or other adjunctive services, e.g.,
integrated approaches to chronic mental illness. Examples of areas of interest are:
•
Analyses of naturalistic databases to evaluate the effectiveness of preventive and
treatment interventions.
•
Randomized clinical trials evaluating the effectiveness of preventive and treatment
interventions that have been augmented or refined with the intent to enhance their clinical
potency or efficiency.
•
Identifying moderators and mediators of intervention effects as a step to design and test
personalized interventions.
•
Moderator/mediator identification could occur retrospectively (not a clinical trial – using EHR).
•
Moderator/mediator identification could occur prospectively (within the context of a clinical trial).
•
Evaluating the effectiveness of predictive algorithms to improve identification and
intervention of individuals at elevated risk of mental illness and suicide.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIMH 133
•
Evaluating the combined or sequential use of interventions.
•
Evaluation of combined/sequential interventions could occur retrospectively (not a
clinical trial – using EHR).
•
Evaluation of combined/sequential interventions could occur prospectively (within the
context of a clinical trial).
•
Determining the optimal duration, frequency and intensity of an intervention to optimize
improvements in symptoms and functioning, establishing the utility of preventive intervention
or continuation or maintenance treatment (that is, for prevention of relapse or recurrence).
•
Evaluation of the optimal length of an intervention could occur retrospectively (not a
clinical trial – using EHR).
•
Evaluation of the optimal length of an intervention could occur prospectively (within the
context of a clinical trial).
•
Evaluating the long-term impact of preventive and therapeutic interventions on
symptoms, functioning, and quality of life.
•
Evaluation of the optimal length of an intervention could occur retrospectively (not a
clinical trial) – using EHR or survey data).
•
Evaluation of the optimal length of an intervention could occur prospectively (within the
context of a clinical trial).
Services research covers all mental health services across the lifespan for all mental health
disorders, includes clinical trial and non-clinical trial designs, and aligns with NIMH Strategic
Objective 4 , which includes but is not limited to:
•
Service settings at the patient, provider, health system, and cross system levels to include
primary care, specialty mental health, emergency departments, integrated care, general
medical, and other delivery settings (such as employment, educational, veteran, military,
criminal justice, child welfare, juvenile justice and other community settings).
•
Enhanced capacity for conducting services research by developing and utilizing
innovative and established methodologies, including health economics, to inform decisions
about the organization, delivery and financing of care.
•
The clinical epidemiology of mental disorders to include development and use of data
sets from health surveillance activities, decision support tools, administrative claims,
mobile apps and similar technologies, electronic health record (EHR), disease registries,
and other databases where epidemiological data (to include big data) reside.
•
Interventions and other research to improve access, continuity, engagement, quality,
uptake, equity, efficiency, and cost of care.
•
Research that reduces disparities and advances equity in mental health interventions,
services, and outcomes for racial and ethnic minority groups, individuals limited by
language or cultural barriers, sexual and gender minorities, individuals living in rural areas,
socioeconomically disadvantaged persons and other underserved groups.
•
The dissemination of information about and implementation of evidence-based
interventions, programs, support tools, or other practices or technologies into
service settings.

NIH, CDC, and FDA Program Descriptions and Research Topics
NIMH 134
For both interventions and services research, DSIR supports the development and testing of digital
health tools. These tools include technology-assisted approaches to assessment (e.g., technology-
assisted screening and diagnosis) and intervention (e.g., m-health and other technology platforms to
support the delivery of preventive, therapeutic, and services interventions). DSIR encourages efforts
to employ technology-assisted approaches to expand the reach, efficiency, continuity, quality, and/or
boost the therapeutic benefit of research-informed strategies, rather than mere translation of
research-supported strategies onto new or emerging technology platforms. Collaboration with NIMH-
supported researchers for the development of software for new analytic techniques and/or decision-
making algorithms is encouraged. Also supported is research and the development or adaptation of
tools and technologies to be used to enhance the training and development of new generations of
researchers and practitioners and to keep established researchers and practitioners up-to-date on
the findings, implementation, and methods of interventions and services research.
Prospective applicants are strongly encouraged to contact Dr. Adam Haim (listed below) with
questions about the relevance of their interests to the mission of this division.
Contact Information
Margaret Grabb, Ph.D. (general questions about the NIMH SBIR program, Phase IIB program,
DNBBS, DTR divisional interests)
National Institute of Mental Health
6001 Executive Boulevard, Room 7133
Rockville, MD 20852 Telephone: 301-443-3563
Email: mgrabb@mail.nih.gov
Adam Haim, Ph.D. (DSIR divisional interests)
Division of Services and Intervention Research
6001 Executive Boulevard
Room 7273
Rockville, MD 20852 Telephone: 301-445-3593
Email: haima@mail.nih.gov
Maggie Sweeney, Ph.D. (DSIR divisional interests; send initial inquiries to Dr. Adam Haim above)
Division of Services and Intervention Research
6001 Executive Boulevard
Telephone: 301-480-1582
Email: maggie.swee[email protected]v
Dr. Vasudev R Rao M.B.B.S, M.S. (DAR divisional interests)
Division of AIDS Research
5601 Fishers Ln
Rockville MD 20852
Telephone: 240-669-5609
Email: vasudev.rao@nih.gov
Paige Anderson, M.S. (preclinical assay development)
6001 Executive Boulevard, Room 7120.3
Bethesda, MD 20892-9645
Rockville, MD 20852 (for express/courier
service) Telephone: 301-827-6550
Email: paige.anderson@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NIMHD 132
NATIONAL INSTITUTE ON MINORITY HEALTH AND HEALTH DISPARITIES
(NIMHD)
Mission
The mission of the National Institute on Minority Health and Health Disparities (NIMHD) is to
promote minority health and to lead, coordinate, support, and assess the National Institutes of
Health (NIH) efforts to improve minority health and reduce and ultimately eliminate health disparities.
In this effort, the NIMHD conducts and supports basic, clinical, social and behavioral research;
facilitates the development of research infrastructure and training; fosters emerging programs; and
reaches out to racial/ethnic minority populations and other U.S. populations with health disparities,
defined in section 464z-3(d)(1) of the Public Health Service Act, 42 U.S.C. 285t(d)(1) as “health
disparity populations” based on higher overall rates of disease incidence, prevalence, morbidity,
mortality, or survival rates as compared to the health status of the general population. NIH-
designated U.S. health disparity populations currently include Blacks/African Americans,
Hispanics/Latinos, American Indians/Alaska Natives, Asian Americans, Native Hawaiians and other
Pacific Islanders, socioeconomically disadvantaged populations, underserved rural populations, and
sexual and gender minorities.
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SEED website. For budgetary, administrative,
or programmatic reasons, the NIMHD may not fund an application or may decrease the length of
an award and/or the budget recommended by a review committee. NIH has received a waiver from
SBA, as authorized by statute, to exceed the statutory budget limitations set by the SBA for specific
topics relevant to the NIMHD that can be found in the SBA-Approved Waiver Topics. Applicants
with budget questions or considering requesting a budget greater than these amounts are
strongly encouraged to contact program staff before submitting an application.
Specific SBIR and STTR Program Information
The Small Business Innovation Research (SBIR) Program and the Small Business Technology
Transfer (STTR) Program enable the Nation’s small businesses to apply their unique research and
development capabilities toward accomplishing NIMHD’s mission. NIMHD has developed a
research framework and small businesses are encouraged to consider the factors operating within
and across the frameworks’ multiple ecosocial levels and domains before initiating the design of
products for potential research and development by NIMHD (see the NIMHD Research Framework
for more information). The framework, initially developed for researchers, can also inform small
businesses’ research and development of new technologies, products, and services for improving,
sustaining or enhancing minority health and extending longevity and for reducing or eliminating
health disparities. The factors identified in the framework are known to contribute to the creation
and perpetuation of poor minority health and health disparities over time and place. Entrepreneurs
are encouraged to consider these and other factors when conceptualizing, designing, and
prototyping novel products seeking NIMHD SBIR and STTR funding. Minority health and health
disparity academic researchers are encouraged to consider partnering with small businesses to
assist in translating NIMHD- or NIH-funded research findings into potentially commercializable
products for improving minority health or eliminating health disparities within one or more levels or
domains of influence.
Through small business Phase I, Phase II, and Fast-track awards, NIMHD supports multi- and
trans- disciplinary research and development leading to novel and or improved products capable of
contributing to NIMHD’s mission. Research and development informed by the NIMHD Research
NIH, CDC, and FDA Program Descriptions and Research Topics
NIMHD 133
Framework or other framework may proceed or be initiated at the molecular, cellular, individual,
community or population level. Funding support for focus groups, phase I/II clinical trials, and other
studies involving human participants needed to develop and test the proposed product may be
requested. Additionally, NIMHD seeks innovative strategies for improving minority health,
eliminating health disparities, and enhancing health and well-being where small businesses
engage, collaborate or partner with health disparity communities from conception, application
submission, and through completion of NIMHD funding periods and beyond. The NIMHD Research
Framework acknowledges the value of small businesses partnering with community-based or -
located organizations or small businesses and with health care providers and health care-
organizations. Applications partnering with community health centers or other patient providers are
encouraged and of interest. Applications developing innovative technologies or services for
enhancing minority health and well-being through partnerships with community-based small
businesses, such as beauty salons, barbershops, pharmacies, etc., that engage with racial and
ethnic minority or health disparity populations on a regular basis, are also of interest.
Technology that leverages indigenous community advisors and supporters in health promotion or
prevention efforts may contribute to overall community health improvement and well-being through
the processes of community empowerment and increased community cohesion.
An overarching objective of NIMHD’s investments in SBIR/STTR programs is to ensure that racial
and ethnic minorities and health disparity populations benefit equally from innovations in health
promotion, educational and medical curricula, prevention interventions, biotechnology, imaging
technologies, technologies for computational biology and informatics, including, for example,
systems and structural biology; and technologies designed to advance personalized medicine and
health, electronic health records, and systems, etc. New or improved instruments, devices, and
related methodologies to facilitate biomedical or behavioral research and efforts that seek to
simplify via redesign or the design of new instruments, devices, and methods likely to increase
access, reduce costs, and improve quality of care and outcomes are of special interest.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NIMHD welcomes Phase IIB Competing Renewal Applications for Phase II grants and contracts
via the Omnibus Solicitation for SBIR or STTR Grant Applications, and as indicated by other
NIMHD Notices of Funding Opportunities (NOFOs). Standard NIMHD Phase II funding policy
applies unless otherwise stated in the NOFO. Non-NIMHD Phase II awardees must contact
NIMHD prior to submission to confirm programmatic interest.
Clinical Trials
Does NIMHD accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
Yes
Does NIMHD accept Clinical Trials
through specific Notices of Funding
Opportunities?
Yes
https://seed.nih.gov/small-business-
funding/find-funding/sbir-sttr-funding-
opportunities
Does NIMHD support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
Research Topics
Applicants are encouraged to engage in research and development that results in a product,

NIH, CDC, and FDA Program Descriptions and Research Topics
NIMHD 134
process or service that will improve minority health and eliminate health disparities and that by
design targets or involves any of the topics listed in the NIMHD waiver list or otherwise will
contribute to the NIMHD mission. For additional information about research areas of interest to
the NIMHD, please visit our website at https://www.nimhd.nih.gov/programs/extramural/research-
areas/.
Contact Information
For additional information on research topics, contact:
Division of Community Health and Population Science
LCDR Michael Banyas, USPHS,
M.P.A, M.A, Program Manager
National Institute on Minority Health and Health
Disparities, NIH Email: michael.banyas@nih.gov
Phone: 301-827-7478

NIH, CDC, and FDA Program Descriptions and Research Topics
NINDS 135
NATIONAL INSTITUTE OF NEUROLOGICAL DISORDERS AND STROKE
(NINDS)
Mission
The mission of NINDS is to reduce the burden of neurological disease—a burden borne by every
age group, by every segment of society, by people all over the world
(https://www.ninds.nih.gov/About- NINDS/Who-We-Are/Mission). To this end, the Institute
supports and conducts research on the healthy and diseased brain, spinal cord, and peripheral
nerves. The NINDS SBIR/STTR (https://www.ninds.nih.gov/Funding/Small-Business-Grants)
program funds small business concerns to conduct innovative neuroscience research and/or
development (R/R&D) that has both the potential for commercialization and public benefit.
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SEED website, unless the application fits an
SBA-approved waiver topic. For topics listed in the SBA-Approved Waiver Topics, NINDS generally
will not fund Phase I applications to the Omnibus greater than $700,000 total costs, with no more
than $500,000 total cost in an year or project periods greater than 2 years; or Phase II applications
greater than $3,000,000 total costs, with no more than $1,500,000 or project periods greater than 3
years. For budgetary, administrative, or programmatic reasons, the NINDS may not fund an
application or may decrease the length of an award and/or the budget recommended by a review
committee. Information about the NINDS budget guidelines can be found on the NINDS SBIR
webpage https://www.ninds.nih.gov/funding/ninds-small-business-program/submitting-small-
business-application.
For all other funding opportunities, applications should follow the guidelines in the Award Budget
section of those announcements carefully.
All applicants are strongly encouraged to contact program staff before submitting an application.
Specific SBIR and STTR Program Information
NINDS Priorities
NINDS priorities are given to meritorious research proposals with the greatest potential to advance
the NINDS mission (see https://www.ninds.nih.gov/About-NINDS/Who-We-Are/Mission). NINDS is
especially interested in:
1.
Novel and innovative technologies that are new to the SBIR or STTR programs.
2.
Technologies coming to the SBIR or STTR programs for their first indication
or market opportunity.
3.
Companies and applicants that are new to the SBIR and STTR programs.
4.
NINDS Cooperative Agreement (U44) Translational Programs. NINDS has specific
translational programs that utilize the SBIR cooperative agreement mechanism (U44)
as noted below. If eligible, companies are encouraged to apply through these
programs.
NINDS SBIR and STTR funding decisions are based on a combination of factors:
NIH, CDC, and FDA Program Descriptions and Research Topics
NINDS 136
1.
potential for high impact on advancing the NINDS mission and the other programmatic
priorities described in NOT-NS-18-002 (https://grants.nih.gov/grants/guide/notice-
files/NOT-NS-18- 002.html);
2.
potential for commercialization;
3.
portfolio balance (to determine whether similar projects have already been funded,
search NIH Reporter http://projectreporter.nih.gov/reporter.cfm);
4.
the quality of the previous performance of the applicant and/or company in the SBIR
and/or STTR program, including evidence of Phase III activities;
5.
for Phase II applicants, the results of the Phase I;
6.
the peer review scores and critiques; and
7.
availability of funds.
Research Topics General Areas of Interest
The NINDS accepts a broad range of small business applications that are significant, innovative,
and relevant to its mission. Examples of research topics within the mission of NINDS are shown
below. This list is not all inclusive and some research areas fall into multiple categories.
1.
Therapeutics and Diagnostics Development for Neurological Disorders, including
biomarker and diagnostic assays, therapeutics (drugs, biologics, and/or devices) for
treatment of neurological disorders, and technologies/methodologies to deliver
therapeutics to the nervous system.
2.
Clinical and Rehabilitation Tools, including intraoperative technologies for
neurosurgeons, rehabilitation devices and programs for neurological disorders, and
brain monitoring systems
3.
Technology and Tools, including technologies to image the nervous system, neural
interfaces technologies, and tools for neuroscience research and drug development.
More information on NINDS areas of interest for the SBIR and STTR programs can be found
here: https://www.ninds.nih.gov/funding/ninds-small-business-program/ninds-small-business-
program-areas-interest.
Clinical Trials
Does NINDS accept Clinical Trials
through the Omnibus/Parent Notices
of Funding Opportunities?
No
Does NINDS accept Clinical Trials
through specific Notices of Funding
Opportunities?
Yes
NINDS may accept and support SBIR and
STTR clinical trial applications through specific
opportunities, which can be found on the
NINDS Funding Opportunities webpage:
https://www.ninds.nih.gov/Funding/Find-
Funding-
Opportunities.
Does NINDS support Clinical
Trials through NON-SBIR/STTR
Notices of Funding Opportunities?
Yes
NINDS accepts and supports SBIR and STTR
clinical trial applications through specific
opportunities, which can be found on the
NINDS Funding Opportunities webpage:
https://www.ninds.nih.gov/Funding/Find-
Funding- Opportunities.
NINDS Clinical Trials Topics:

NIH, CDC, and FDA Program Descriptions and Research Topics
NINDS 137
NINDS is committed to identifying effective treatments for neurological disorders by supporting
well- executed clinical trials. NINDS may accept and support SBIR and STTR clinical trial
applications within the NINDS mission through specific opportunities. Other human subjects
research can be submitted through the SBIR and STTR Parent (Clinical Trials Not Allowed)
solicitation. However, NINDS may decline funding of any application that includes human subjects
for programmatic or administrative reasons. SBIR applicants considering projects involving human
subjects research are strongly encouraged to contact program staff in advance of submission.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NINDS only accepts Phase IIB SBIR/STTR Competing Renewal applications through specific
opportunities that focus on the commercialization of SBIR and STTR developed technologies.
These opportunities can be found on the NINDS Funding Opportunities webpage:
https://www.ninds.nih.gov/Funding/Find-Funding-Opportunities. Contact
ninds_sbir@ninds.nih.gov for additional information.
Specific Funding Opportunities and Programs
NINDS Translational Research Programs
The NINDS offers a variety of specific funding opportunities and programs to accelerate the
preclinical discovery and development of new therapeutic interventions for neurological disorders.
These programs have specific funding opportunities and allow for budgets over the hard cap.
Several programs utilize the cooperative agreement (U44) mechanism, which is milestone-driven
and involves NIH program staff participation in developing the project plan, monitoring research
progress, and appropriate go/no-go decision-making. SBIR applicants considering projects
involving translational research are strongly encouraged to contact program staff well in advance of
submission.
•
Blueprint Neurotherapeutics Network for Biologics (BPN-Biologics) is dedicated to
biotechnology product- and biologics-based therapies, which broadly include modalities
such as peptides, proteins, oligonucleotides, gene therapies, and cell therapies. The
program supports lead optimization, IND-enabling studies for the candidate, and early-
phase clinical trials. Contact: Chris Boshoff (chris.boshoff@nih.gov)
•
Blueprint MedTech Program provides support for innovators developing
groundbreaking medical device technologies. The mission of the program is to
catalyze the translation of novel technologies from early-stage development to
readiness for first-in-human clinical studies. Contact: NINDS-Devic[email protected]v;
Blueprint-MedTech@nih.gov
•
Blueprint Neurotherapeutics Network (BPN) provides both funding and non-dilutive
support for small molecule drug discovery and development, from hit-to-lead chemistry
through phase I clinical testing. The program offers funding, access to NIH-funded
contract research organizations (CROs), and access to consultants with expertise in
various aspects of drug discovery and development. Contact: Charles Cywin
(charles.cywin@nih.gov)
•
NIH Countermeasures Against Chemical Threats (CounterACT) supports
research to understand the fundamental mechanisms of toxicity caused by chemical
threat agents and the development of new and improved therapeutics for reducing
mortality and morbidity caused by these agents. Chemical threats are toxic
compounds that could be used in a terrorist attack against civilians, or released at

NIH, CDC, and FDA Program Descriptions and Research Topics
NINDS 138
toxic levels by accident, such as from industrial production, storage, or shipping, or
natural disaster. NINDS supports partnerships between small business and not-for-
profit laboratories engaged in research related to the NIH CounterACT program that
falls within the NINDS mission, including devices that could be used during a
chemical emergency involving mass casualties, as well as some research on
therapeutics. The CounterACT program only supports research on chemical threats
that have been identified by the United States Government (USG) as Chemicals of
Concern (CoC). There are over 200 CoCs, grouped by primary mechanism of action
into toxidromes. The toxidromes that fall under the NINDS mission (with chemical
threat examples) are: Anticoagulants (e.g., brodifacoum, bromadiolone, diphacinone),
Blood/Metabolic (Knockdown) agents (e.g., hydrogen cyanide, hydrogen sulfide,
arsenic trioxide), Cholinergic (Organophosphate Nerve Agents and Insecticides)
(e.g., sarin, soman, parathion, phorate, aldicarb), and Convulsants (e.g., picrotoxin,
TETS, strychnine). All potential applicants should contact program staff to ensure
their chemical threat is of interest to the program. Contact: Shardell Spriggs
(shardell.spr[email protected]v)
•
Office of the Neural Exposome and Toxicology (ONETOX) supports research on
exposures that have an impact on neurological disease and disorders, and overall
nervous system health. The exposures described in the exposome not only include
environmental chemical and biological toxins, but also psychosocial factors and
internal factors such as diet and the microbiome, and these exposures may occur
anytime from in utero to late in life. NINDS only support studies within the NIND
mission space. Contact: David Jett (david.jett@nih.gov)
•
Translational Neuroscience Biomarker Program focuses on improving the quality
and efficiency of neurotherapeutic clinical research by supporting rigorous biomarker
development and validation. In order to achieve this goal, the program: 1) promotes
rigorous biomarker identification and validation through milestone-driven funding
opportunities, and 2) facilitates the validation of biomarkers and biomarker signatures as
fit for purpose tools for use in clinical trials to accelerate therapeutic development and
aid in decision making for patient care. Contact: Carol Taylor-Burds (carol.taylor-
burds@nih.gov).
•
Stroke Preclinical Assessment Network (SPAN) seeks to conduct late-stage
preclinical studies of putative neuroprotectants combined with reperfusion. SPAN
utilizes a novel, adaptive, secured system for parallel testing of promising interventions
designed to extend the treatment time window and/or improve outcome compared to
reperfusion when combined with thrombolysis, thrombectomy or both. Contact:
Francesca Bosetti (frances@mail.nih.gov)
Information about these and other programs can be found at
https://www.ninds.nih.gov/Current- Research/Research-Funded-NINDS/Translational-
Research.
Trans-NIH Initiatives
Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative
The Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative is a
Presidential project aimed at revolutionizing our understanding of the human brain. NIH is
one of several federal agencies involved in the BRAIN Initiative. Planning for the NIH
component of the BRAIN Initiative is guided by the long-term scientific plan, “BRAIN 2025: A

NIH, CDC, and FDA Program Descriptions and Research Topics
NINDS 139
Scientific Vision,” which details seven high-priority research areas. This report can be found
at https://braininitiative.nih.gov/.
NIH has a number of specific Notices of Funding Opportunities through the BRAIN Initiative that
are targeted to small business concerns. These funding opportunities can be found at
https://www.braininitiative.nih.gov/funding/. Applicants are encouraged to consider if these
funding opportunities may be appropriate to their research. Contact ninds_sbir@ninds.nih.gov
for additional information.
Helping to End Addiction Long-term (HEAL) Initiative
The Helping to End Addiction Long-term (HEAL) Initiative is an aggressive, trans-agency
effort to speed scientific solutions to stem the national opioid public health crisis. Further
information on the HEAL Initiative can be found at https://heal.nih.gov/. NIH has several
specific Notices of Funding Opportunities through the HEAL Initiative that are targeted to
small business concerns. These funding opportunities can be found at
https://heal.nih.gov/funding. Applicants are encouraged to consider if these funding
opportunities may be appropriate to their research. Contact ninds_sbir@ninds.nih.gov for
additional information.
Alzheimer's Disease and Related Dementias (AD/ADRD)
NINDS collaborates with NIH’s National Institute on Aging (NIA), the lead NIH Institute for
Alzheimer’s disease (AD) research and for NIH's response to the National Plan to Address
Alzheimer’s Disease, to establish research priorities and fund biomedical research to
decrease the burden of dementia on individuals, families, and communities. Learn more
about AD/ADRD funding opportunities here: https://www.ninds.nih.gov/current-
research/focus-disorders/focus-alzheimers-disease-and-related-dementias. Applicants are
encouraged to consider if these funding opportunities may be appropriate to their research.
Contact ninds_sbir@ninds.nih.gov for additional information.
For additional information about NINDS funding opportunities, please visit the NINDS Funding
Opportunities webpage at: https://www.ninds.nih.gov/Funding/Find-Funding-Opportunities.
Contact Information
Scientific/Research Contact
Emily Caporello, Ph.D.
Director, NINDS Small Business Program
Phone: 301-496-1779
Email: emily.caporello@nih.gov
Program-wide Email: ninds_sbir@ninds.nih.gov
Financial/Grants Management Contact:
Chief Grants Management Officer
National Institute of Neurological Disorders and Stroke (NINDS)
Email: ChiefGrantsManagementOfficer@ninds.nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NINR 140
NATIONAL INSTITUTE OF NURSING RESEARCH (NINR)
Mission
The National Institute of Nursing Research (NINR) leads nursing research to solve pressing
health challenges and inform practice and policy – optimizing health and advancing health equity
into the future.
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SBIR website. For budgetary, administrative,
or programmatic reasons, the NINR may not fund an application or may decrease the length of an
award and/or the budget recommended by a review committee. NIH has received a waiver from
the SBA, as authorized by statute, to exceed the statutory budget limitations set by the SBA for
specific topics relevant to the NINR that can be found in the SBA-Approved Waiver Topics. The
NINR will generally not fund:
•
Phase I applications to the Omnibus greater than $385,000 total costs or project
periods greater than 2 years, or
•
Phase II applications greater than $2,500,000 total costs or project periods greater than 3
years.
Applicants that are considering requesting a budget greater than these amounts are strongly
encouraged to contact program staff before submitting an application.
Specific SBIR and STTR Program Information
The NINR SBIR program is technology agnostic and welcomes all innovative solutions with
commercial potential that are relevant to the NINR mission and research topics listed below.
Technical and Business Assistance (TABA). NINR will consider well justified TABA costs up to
the limits specified on the NIH SEED Website. Applicants can request TABA funding within your
award or apply for an NIH TABA program. Applicants cannot receive both.
For additional information about NINR’s Small Business Programs, please visit NINR’s Small
Business Funding.
Specific Funding Opportunities and Programs
NINR will also consider SBIR/STTR application related to other NINR funding initiatives that is
relevant to the Institute’s mission. These funding opportunities are revised throughout the year, so
please subscribe to receive regular updates
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NINR does not accept Phase IIB applications.
NINR does not participate in the Commercialization Readiness Pilot (CRP) Program.
NIH, CDC, and FDA Program Descriptions and Research Topics
NINR 141
Clinical Trials
Does NINR accept
Clinical Trials through
the Omnibus/Parent
Notices of Funding
Opportunities?
Yes
Does NINR accept
Clinical Trials through
specific Notices of
Funding
Opportunities?
Yes
https://www.ninr.nih.gov/researchandfunding/fundingopportunities
Does NINR support
Clinical Trials through
NON-SBIR/STTR
Notices of Funding
Opportunities?
Yes
https://www.ninr.nih.gov/researchandfunding/fundingopportunities
Research Topics
NINR has developed a research framework that takes advantage of what makes the Institute
unique by focusing on a holistic, contextualized approach to optimizing health for all people,
rather than on specific diseases, life stages, or research topics. The framework builds on the
strengths of nursing research, spans the intersection of health care and public health, and
encompasses the clinical and community settings where nurses engage in prevention, treatment,
and care—including hospitals and clinics, schools and workplaces, homes and long-term care
facilities, justice settings, and throughout the community. The framework encourages research
that informs practice and policy and improves health and quality of life for all people, their families
and communities, and the society in which they live. NINR’s research framework includes guiding
principles and research lenses that promote innovative and rigorous multilevel study designs that
look upstream, midstream, and downstream to discover solutions to the nation’s most pressing
and persistent health challenges. NINR’s guiding principles describe the qualities that
investigators should emphasize in all NINR-supported research. In considering awards for
funding, the extent to which studies reflect these principles will factor into our decisions.
NINR will prioritize research that:
•
Tackles today’s pressing health challenges and stimulates discoveries to prepare for,
prevent, or address tomorrow’s challenges.
•
Discovers solutions across clinical, community, and policy settings to optimize
health for individuals, families, communities, and populations.
•
Advances equity by removing structural barriers from research, cultivating diversity in
perspectives and ideas, and fostering inclusion and accessibility in designing,
conducting, and participating in research; and
•
Is innovative, develops or applies the most rigorous methods, and has the
potential for the greatest impact on health.
NINR identified five complementary and synergistic research lenses that best leverage the
strengths of nursing research and promote multilevel approaches, cross-disciplinary and -
sectoral collaboration, and community engagement in research. It is important to note that the
lenses are not research topics, but rather perspectives through which to consider the full
spectrum of nursing research topics that encompass health and illness within the context of
people’s lived experiences. These lenses allow nursing research to examine new topics while
also allowing scientists to take a different look at long- standing areas of interest. The research
lenses are:

NIH, CDC, and FDA Program Descriptions and Research Topics
NINR 142
•
Health Equity: Reduce and ultimately eliminate the systemic and structural inequities
that place some at an unfair, unjust, and avoidable disadvantage in attaining their full
health potential.
•
Social Determinants of Heath: Identify effective approaches to improve health and
quality of life by addressing the conditions in which people are born, live, learn, work,
play, and age
•
Population and Community Health: Address critical health challenges at a macro
level that persistently affect groups of people with shared characteristics
•
Prevention and Health Promotion: Prevent disease and promote health through the
continuum of prevention – from primordial to tertiary
•
Systems and Models of Care: Address clinical, organizational, and policy challenges
through new systems and models of care
NINR encourages researchers to view the health equity and social determinants of health
lenses as primary foci through which to consider the population and community health,
prevention and health promotion, and systems and models of care lenses.
See NINR’s Strategic Plan for more information on each of the research lenses.
NINR does not generally support the development of technologies in the following topic areas:
•
Technologies designed for staffing purposes.
•
Technologies designed to provide nurse educational and professional training
(e.g., VR simulations).
•
Technologies that assess or limit exposure to occupational health stressors.
•
Technologies for sanitization or decontamination in a clinical setting.
NINR is particularly interested in applications from socially/economically disadvantaged small
businesses (SDB), women-owned small business (WOSB), and small businesses located in
under- represented states. Responsive entrepreneurs should aim to develop technologies that align
with the research topics above.
Applicants do not need to have a nursing background to apply.
NINR Clinical Trials Topics:
NINR accepts clinical trials in any of the topic areas above.
Contact Information
For additional information please contact:
Dr. Karen Kehl
Director, Small Business Innovation Research Program
National Institute of Nursing Research (NINR)
Division of Extramural Science Program (DESP)
6701 Democracy Blvd, Room 727
Bethesda, MD 20892-4870
Office: (301) 594-8010
Email: karen.kehl@nih.gov
NIH, CDC, and FDA Program Descriptions and Research Topics
NCATS 143
NATIONAL CENTER FOR ADVANCING TRANSLATIONAL SCIENCES
(NCATS)
Mission
NCATS is transforming translational science to improve human health; it relies on the power of data,
new technologies and teamwork to develop, demonstrate and disseminate innovations that reduce,
remove or bypass costly and time-consuming bottlenecks in translational research. For additional
information please visit the NCATS Strategic Plan webpage -
https://ncats.nih.gov/strategicplan/introduction
NCATS small business funding is designed specifically to transform the translational science process
so that new treatments and cures for diseases can be delivered to patients more quickly. The Center
supports the development of technologies, assays, drugs, devices, instruments, and methodologies
that may have broad application to any stage of the translational process from preclinical
development to clinical research and to implementation science in patient care and public health. For
additional information, please visit http://www.ncats.nih.gov.
NCATS is committed to supporting small business Phase I, Phase II, Fast-track and Phase IIB Competing
Renewal awards through the Small Business Innovation Research (SBIR) and Small Business
Technology Transfer Programs (STTR). For additional information, please visit
http://ncats.nih.gov/smallbusiness.
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts defined
by the SBA, which can be found on the NIH SEED website. For budgetary, administrative, or
programmatic reasons, NCATS may decide not to fund an application or may decrease the length of
an award and/or the budget recommended by a review committee.
For certain topical areas, there is an SBA-Approved Waiver Topics list for which the NCATS generally
will not fund:
•
Phase I applications greater than $350,000 total costs or project periods greater than 2 years
•
Phase II applications greater than $2,150,000 total costs or project periods greater than 3 years
Applicants considering a requested budget greater than these limits are strongly encouraged to contact
program staff before submitting an application.
Specific SBIR and STTR Program Information
NCATS Clinical Trials Topics
NCATS will not accept SBIR and STTR applications that propose clinical trials under the Omnibus
solicitation.
Specific Funding Opportunities and Programs
There are more than 6,500 identified rare and neglected diseases, yet only about 250 treatments
are available for these conditions. The limited numbers of patients can make gathering information
and designing drug studies difficult. As a result, scientists often know little about the symptoms and
biology of these conditions. Also, some private companies may find it difficult to justify the cost of
developing drugs for such small rare disease markets.

NIH, CDC, and FDA Program Descriptions and Research Topics
NCATS 144
The Therapeutics for Rare and Neglected Diseases (TRND) program is designed to combat these
challenges. Its mission is to encourage and speed the development of new treatments for diseases
with high unmet medical needs. TRND stimulates therapeutic development research collaborations
among NIH and academic scientists, nonprofit organizations, and pharmaceutical and biotechnology
companies working on rare and neglected illnesses. The program provides expertise and resources,
working with research partners to move therapeutics through preclinical testing, including plans for
clinical trials and submission of an IND application to the Food and Drug Administration. These
efforts effectively “de-risk” therapeutic candidates and make them more attractive for adoption by
outside business partners. To learn more about the TRND program goals, please visit
https://ncats.nih.gov/trnd/about/goals
Bridging Interventional Development Gaps (BrIDGs)
The Bridging Interventional Development Gaps (BrIDGs) program enables research collaborations to
advance candidate therapeutics for both common and rare diseases into clinical testing. Investigators
do not receive grant funds through this program. Instead, selected researchers partner with NCATS
experts to generate preclinical data and clinical-grade material through government contracts for use
in Investigational New Drug (IND) applications to a regulatory authority such as the Food and Drug
Administration (FDA). In general, BrIDGs provides synthesis, formulation, pharmacokinetic and
toxicology expertise and resources to its collaborators.
NIH contractors conduct preclinical studies under the direction of NCATS staff. NCATS, along with any
co-funding NIH Institutes and Centers, supports contract costs. The decision to collaborate on a
proposed project is based on an internal assessment of scientific merit, programmatic fit and the
availability of NIH funds. To find out how to submit a proposal to BrIDGs, please visit
https://ncats.nih.gov/bridgs/work
As of fall 2015, BrIDGs has generated data to support 18 investigator-initiated INDs that have been
cleared by the FDA and one clinical trial application cleared by Health Canada. A total of 14 projects
have been evaluated in clinical trials. Five BrIDGs-supported agents have been evaluated in Phase II
human clinical trials, in which researchers give an experimental therapy to a group of patients to
evaluate the effectiveness and safety of a treatment. Third-party organizations have licensed or
invested in 10 agents during or after their development by BrIDGs. To learn more about active and
completed BrIDGs projects, please visit https://ncats.nih.gov/bridgs/projects
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
Occasionally, NCATS may accept Phase IIB SBIR Competing Renewal grant applications of
NCATS supported Phase II awards to continue research and development of products that have a
potential to address bottlenecks in the translational process, and where additional time and effort is
needed to reach a stage where interest and investment by third parties would be likely. Such
products are expected to have broad applicability and be consistent with the mission of NCATS.
Applicants are strongly encouraged to speak to NCATS Program staff prior to submitting their
Phase IIB application. Budgets for Phase IIB grant applications must be approved by NCATS
Program staff prior to submission.
NIH, CDC, and FDA Program Descriptions and Research Topics
NCATS 145
Clinical Trials
Does NCATS accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
No
Does NCATS accept Clinical Trials
through specific Notices of Funding
Opportunities?
No
Does NCATS support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
U01 – however the SBC can only participate if
repurposing an existing drug or biologic
(therapeutics) that have already completed at
least a Phase l trial for a different indication by
the time an award is made. These pharma drugs
and biologics are listed in PAR-18-332.
https://grants.nih.gov/grants/guide/pa-files/PAR-
18-332.html
Research Topics
Preclinical Drug Discovery and Development
•
Innovative platforms for identification and prioritization of targets for therapeutic
intervention with clear clinical impact; such as those that are: implicated for disease,
have genetic variations that have been identified in functional regions of receptor targets,
and/or have high potential for biased signaling that would promote the beneficial effects
of receptor signaling and reduce the unwanted effects
•
Tools and technologies to enable high throughput screening of compound activity on
currently “non- druggable” targets
•
Assays for high-throughput screening of rare-diseases-related targets
•
Co-crystallization high-throughput screening techniques
•
Fluorescence probes to replace antibodies for determination of cellular protein translocation
•
Phenotypic assay development, including stem cell technology platforms for human
“disease-in-a- dish” applications and the evaluation of toxicity
•
Interventions that target molecular pathways or mechanisms common to multiple diseases
•
Platforms for non-antibody biologics, cell-based therapies and gene therapy discovery
•
Small molecule and biologics analytical characterization
•
Accelerated bioengineering approaches to the development and clinical application of
biomedical materials, devices, therapeutics and/or diagnostics
•
Development of novel technologies for enzyme replacement therapies (e.g.,
new cell culture/expression system) to solve major bottlenecks in rare
diseases research
•
Innovative methods to determine alternative uses for existing therapeutic interventions
for high priority areas, such as rare diseases and pain.
•
Tools and technologies that increase the predictivity or efficiency of medicinal chemistry,
biologic or other intervention optimization
•
Technologies to deliver nucleic acid therapeutics to tissues other than the liver
•
Methodologies and technologies to increase efficiencies of manufacturing therapeutics
•
Development of novel high-throughput technologies that focus on making translational
research more efficient
•
GMP production of exosome/extracellular vesicles
•
Generation of producer lines for large scale production of exosomes/extracellular vesicles
•
Extracellular RNA-based biomarkers and therapeutics of human diseases
•
Approaches to targeting the human microbiome for therapeutic or diagnostic purposes
NIH, CDC, and FDA Program Descriptions and Research Topics
NCATS 146
•
Scale up, manufacturing and characterization of IPS cells
•
3D printing technologies
•
Technologies to substantially improve the efficiency and reduce the cost of clinical
grade gene therapy vector manufacturing
•
Development of in vitro human tissue models (organs, 3D printing)
•
Technologies to allow therapeutic proteins other than lysosomal enzymes to be secreted
and taken up by other cells via cross-correction
•
Novel strategies to prevent deleterious immune responses to gene therapy, to improve
efficiency genome editing and/or enzyme replacement therapy
•
Establishing more robust phenotypic screens that may help prioritize candidate
compounds for further testing
•
Innovative technology for non-small molecule delivery
•
High-throughput epigenetics screening/characterization tools and technologies
•
Microphysiological systems (MPS)/Tissue Chips, including MPS/Tissue Chips that
incorporate known functional variants, e.g., ACMG 59 or CPIC A alleles, for study
comparison using the same derived genetic background across a set of tissue chips with
the functional variant
•
Volatile organic compounds (odors, scents) as biomarkers for disease
•
Bacteriophage-based therapeutics for disease and as modulators of the microbiome and
microbiome natural products
•
High throughput Surface Plasmon Resonance devices for detecting protein small molecule
interactions
•
New class of quantum-enabled sensing technologies for advancing translational sciences (e.g.,
point of care ultrasensitive high-throughput technologies for diagnostics, and miniaturized
benchtop technologies for chemical/bioanalyte characterization).
•
Development of diagnostics or useful drug targets for rare diseases by using Pangenome
data.
Biomedical, Clinical, & Health Research Informatics
•
Searchable access to information about research resources, facilities, methods, cells,
genetic tests, molecules, biologic reagents, animals, assays, and/or technologies with
evidence about their use in research studies
•
Cloud-based tools and methods for meaningful sharing, re-use and integration of research data
•
Novel platforms, technologies and tools for: (1) enabling clinical and translational
research, particularly those with mechanisms for inclusion of patient-reported data
and (2) integration of patient data collected from multiple devices and
multiple/diverse clinical studies
•
Development of personalized phenotypic profiling (as well as personalized intervention)
based on patient-centered integration of data from multiple data sources, including
social media
•
Development of predictive models for translational science
•
Digital applications and tools (including telemedicine platforms) that facilitate/enhance
translational research and medicine in rural populations
•
Generic Disease Registry template platforms that can be reused for multiple diseases.
•
Mobile device validation tools to ensure data from different brands or versions have
compatible results.
•
Tools to assess with algorithms developed with artificial intelligence, machine learning.
•
Tools that allow for persistent identifier and attribution for data contributors that give
credit to the data producers while ensuring that shared data has not been altered
•
Patient Mobile Tool Platforms that facilitate tool developers to build “apps” that integrate

NIH, CDC, and FDA Program Descriptions and Research Topics
NCATS 147
into their medical records.
•
Tools and environments that enable an easy interrogation of publicly available data
•
Innovative approaches like nanoneedles and nano particles to help improve early disease
detection and targeted therapeutic delivery
•
Tools and technologies that leverage AI and real-world data for enhanced product
evaluation and surveillance that could be used to provide a resource for rapid response to
public health needs
Clinical, Dissemination and Implementation Research
•
Tools and technologies that:
o
increase the efficiency of human subjects research, that facilitate the rapid
diagnosis and/or clinical trial recruitment and subject tracking, institutional review
board evaluation and/or regulatory processes
o
evaluate and improve the process of informed consent
o
address medication adherence in clinical settings
o
address and improve community engagement
o
address the rapid diagnosis and/or clinical management of rare diseases
o
help characterize human disease states and assist in assessing the impact of interventions
o
support unique approaches to advance diversity, equity and inclusion.
o
mine published data and generate usable knowledge and analytics to advance research
•
Increased efficiency of clinical research conduct, including but not limited to regulatory
decision support, patient eligibility analysis and recruitment and retention tracking
•
Educational tools for clinical and translational science
•
Computational or web-based health research methods, including:
o
Platforms for generally applicable and scalable multi-disease registries and
natural history studies
o
Clinical trial designs and analyses (e.g., for pragmatic clinical trials)
•
Approaches, tools, platforms and environments that:
o
Integrate data in novel ways for development of new biomarkers that can be
tested in translational research paradigms for which there are barriers or
bottlenecks
o
Engage prospective research participants who are from under-represented
communities and impacted by disparities and the digital divide
•
Strategies to enhance the quality of and accelerate the conduct of dissemination and
implementation research
•
Sustainable solutions for effective tools and environments in translational research
•
Development and validation of patient reported outcomes, clinician-reported outcomes and
biomarkers for rare diseases that are not already supported by a disease-specific NIH
Institute or Center
•
Patient empowerment tools/apps that allow users to compare their treatment and
outcomes to normative populations existing treatment guidelines
•
Telemedicine or digital health applications that focus on research in rural populations
•
Tools and technologies that enhance the quality, safety, efficiency, effectiveness of new
innovations in community settings
Contact Information
For additional information on research topics, please contact:
Krishna ‘Balki’ Balakrishnan, PhD,
MBA NCATS SBIR/STTR Acting
Program Director
National Center for Advancing Translational
Sciences, NIH Phone: 301-827-7149
Email: NCATS-SBIRSTTR@mail.nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NCATS 148
Meena U. Rajagopal, PhD
NCATS SBIR/STTR Program Officer
National Center for Advancing Translational
Sciences, NIH Phone: 301-827-1921
Email: NCATS-SBIRSTTR@mail.nih.gov
Mayra A. Alvarez Lopez, MS
NCATS SBIR/STTR Program Analyst
National Center for Advancing Translational
Sciences, NIH Phone: 301-827-7146
Email: NCATS-SBIRSTTR@mail.nih.gov
For Administrative, business management and grants policy questions, please contact:
Ms. Imoni Williams
Grants Management Specialist, SBIR/STTR Project Liaison
Phone: 301-435-2939
E-mail: imoni.williams@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
NCCIH 149
NATIONAL CENTER FOR COMPLEMENTARY AND INTEGRATIVE HEALTH
(NCCIH)
Mission
The mission of NCCIH, as described in the Center’s Strategic Plan, is to define, through rigorous
scientific investigation, the usefulness and safety of complementary and integrative health
interventions and their roles in improving health and health care.
The following narrative indicates the scope of projects suitable for the Small Business Innovation
Research (SBIR)/Small Business Technology Transfer (STTR) programs that fit within the mission of
NCCIH. For additional information about areas of interest to NCCIH and a listing of NCCIH’s currently
funded applications, please visit https://www.nccih.nih.gov/research. Business concerns interested in
exploring SBIR/STTR grant opportunities with NCCIH are encouraged to visit the NCCIH SBIR
website and contact NCCIH program directors prior to submitting an application.
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts defined
by the Small Business Association (SBA), which can be found on the NIH SEED website. NCCIH
policy on grant duration is described in NOT-AT-20-017.
There is an SBA-Approved Waiver Topics list, for which NCCIH generally will not fund:
•
Phase I applications greater than $325,000 total costs per year or project periods
greater than 2 years.
•
Phase II applications greater than $2,000,000 total costs for the duration of the project
or project periods greater than 3 years.
Applicants considering a requested budget greater than these limits are strongly encouraged to contact
program staff before submitting an application.
Specific SBIR and STTR Program Information
NCCIH supports the development and validation of innovative technology to advance fundamental
understanding, enhance and monitor usage, or determine the usefulness and safety of a variety of
complementary and integrative health approaches, including natural products and/or mind and body
approaches.
Specific Funding Opportunities and Programs
NCCIH does not accept applications for Phase IIB.
NOSI-AT-21-001 Development and/or Validation of Devices or Electronic Systems to Monitor or
Enhance Mind and Body Interventions (SBIR/STTR)
NOSI-AT-20-015 Methods Development in Natural Products Research (SBIR/STTR)

NIH, CDC, and FDA Program Descriptions and Research Topics
NCCIH 150
Clinical Trials
Does NCCIH accept clinical trials
through the Omnibus/Parent Notices of
Funding Opportunities?
Yes
For applications involving clinical studies that fall
within the NIH definition of a clinical trial, NCCIH
will not support clinical trials aiming to test
efficacy/effectiveness (meaning the study is
powered on a primary outcome that is a clinical
assessment used in clinical diagnosis of disease
or monitoring of disease severity) of an
intervention as a part of an SBIR/STTR Phase I
application. Applicants seeking to conduct
efficacy or effectiveness clinical trials should
pursue funding via other Notices of Funding
Opportunities (NOFOs) such as the Omnibus
SBIR/STTR Phase II and Fast-Track.
NCCIH recognizes a difference between “clinical
trials” that are designed to answer specific
questions about the clinical effect of
interventions and mechanistic studies that have
the primary goal of understanding how an
intervention works.
A clinical outcome study has the objective of
determining the clinical safety, tolerability,
feasibility, efficacy, and/or effectiveness of
pharmacologic, nonpharmacologic, behavioral,
biologic, surgical, or device (invasive or
noninvasive) interventions.
A mechanistic study has the objective to
understand the mechanism(s) of action of an
intervention, a biological or behavioral process,
or the pathophysiology of a disease/condition.
NCCIH continues to accept clinical trials of
all types on Omnibus SBIR/STTR Phase II
and Fast-Track applications.
See NOT-AT-19-012 for “NCCIH Policy for SBIR
and STTR Phase I Applications Proposing
Clinical Trials to the Omnibus Solicitations”
Does NCCIH accept clinical trials
through specific Notices of Funding
Opportunities?
Yes

NIH, CDC, and FDA Program Descriptions and Research Topics
NCCIH 151
Does NCCIH support clinical trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes
Notice of Special Interest (NOSI): Fundamental
Science Research on Complementary and
Integrative Health Approaches, Including Natural
Products or Mind and Body Interventions
Mechanisms Underlying the Contribution of
Sleep Disturbances to Pain (R01 Clinical Trial
Optional)
Mechanisms Underlying the Contribution of
Sleep Disturbances to Pain (R21 Clinical Trial
Optional)
Center of Excellence for Research on
Complementary and Integrative Health (P01
Clinical Trial Optional)
Investigator Initiated Clinical Trials of
Complementary and Integrative Interventions
Delivered Remotely or via mHealth (R01
Clinical Trial Required)
Notice of Special Interest: Exploring the
Mechanisms Underlying Analgesic Properties of
Minor Cannabinoids and Terpenes
Research Topics
Natural Products (including botanicals, herbs, probiotics, prebiotics, dietary supplements, special
medicinal diets, and microbiome- /microbial-based therapeutics):
•
Development and validation of technologies for standardization and characterization of
biologically active ingredients in natural products.
•
Development and validation of technologies for taxonomic identification of botanical raw
materials or detection of adulterants.
•
Development and validation of technologies for the identification and characterization of
bioactive metabolites derived from oral consumption of natural products.
•
Development and validation of methods for the sustainable production of low-yield natural
products of commercial interest.
•
Development of novel analytical tools and technologies to study the microbiome,
including its composition, genetics, and bioactivity, that can help clarify associations
between the human microbiome and brain function and health.
•
Development of gut microbiome monitoring assays for validating safety and functional
analysis of genomic and microbiota interactions.
•
Development of complementary and integrative therapeutic approaches to modify and
balance the gut microbiota in healthy populations and individuals with disrupted microbiota
and related diseases.
•
Clinical testing of natural products for the management of hard-to-treat symptoms such as
pain, sleep disorders, or mild-to-moderate anxiety and depression to allow development of an
evidence base that would accelerate U.S. Food and Drug Administration (FDA) approval of
a drug indication for the natural product.
Mind and Body Approaches (including meditation, mindfulness, hypnosis, yoga, tai chi, acupuncture,
manual therapies, and music/art therapies):
NIH, CDC, and FDA Program Descriptions and Research Topics
NCCIH 152
•
Development, testing, and validation of appropriate objective and/or quantitative
measures and instruments to assess or monitor mind and body approaches in different
contexts (e.g., classrooms, families, child welfare, juvenile justice systems).
•
Development, testing, and validation of measures and tools to assess training or
fidelity of implementation of mind and body approaches in different settings (e.g., health
care, community, families, schools, child welfare, juvenile justice systems).
•
Development and testing of technologies for the implementation of mind and body
approaches in group or individual settings and/or self-care strategies. Examples may
include but are not limited to the use of mobile health technologies such as smartphone
apps, sensors, online delivery, or phone- based delivery.
•
Development and validation of methods for standardization and characterization of
the active components of mind and body approaches.
•
Development and validation of methods for standardization of multimodal interventions to
study whole person health.
•
Development and validation of imaging tools or instruments for studying manual therapies,
including but not limited to massage, acupuncture, or spinal manipulation.
•
Development and testing of innovative technologies for multisensory delivery of mind
and body approaches.
•
Development, testing, and validation of innovative technologies to enhance
sensory-based (temperature, light, olfaction, etc.) therapies.
•
Development, testing, and validation of innovative technologies to facilitate delivery of
music/art- based interventions and to identify novel outcome measures and biomarkers for
these interventions.
General Tool/Technology Development:
•
Development and validation of biomarkers that correlate with efficacy of complementary and
integrative health approaches.
•
Development and validation of standardized, reliable, and cost-effective tools that correlate
with brain imaging in response to mind and body interventions.
•
Development and validation of tools, technologies, and instruments, including gaming and
virtual reality technologies, for the accurate assessment of adherence and/or fidelity to the
use of mind and body practices and interventions.
•
Development and validation of tools to improve patient-reported outcome measures of
importance in clinical studies of complementary and integrative health approaches.
•
Development, pilot testing, and validation of wireless technologies for real-time data
collection and monitoring of brain activity or other physiological signals for mind and body
approaches.
•
Development or adaptation of biochemical or epigenetic monitoring devices for
complementary and integrative health approaches.
•
Development and validation of tools to improve biological and physiological outcome
measures for use in clinical studies of complementary or integrative health approaches.
•
Development or adaptation of technologies for objective assessment of pain with relevance
to complementary and integrative health approaches.
•
Development of sleep monitoring technologies or biomarker panels to assess sleep
deprivation, sleep deficiency, circadian rhythm dysregulation, and connection of sleep
disturbances with health risks.
•
Development and testing of in vivo labeling technology of tissues or cells responsible for
generating signals in response to different internal senses (e.g., mechanical force,
temperature, osmolarity, oxygen levels).
•
Development and testing of technology or methods for quantifying biomechanical forces
applied to internal tissues or cells.
•
Development and testing of mobile health technology or nonmobile technology and methods
to monitor or quantify physical and/or emotional well-being, breathing, or sleep.

NIH, CDC, and FDA Program Descriptions and Research Topics
NCCIH 153
Contact Information
For additional information on research topics, please contact:
Emrin Horgusluoglu, PhD
Program Director, Division of Extramural Research
National Center for Complementary and Integrative Health (NCCIH)
6707 Democracy Boulevard, Suite 401
Telephone: 240-383-5302
Email: emrin.horgusluoglu-moloch@nih.gov
NIH, CDC, and FDA Program Descriptions and Research Topics
NLM 154
NATIONAL LIBRARY OF MEDICINE (NLM)
Mission
The National Library of Medicine (NLM) offers support for research and development projects in
biomedical informatics and data science. Biomedical informatics and data science research applies
computer and information sciences to improve the access, storage, retrieval, management,
dissemination and use of biomedical information. Grants are made to U.S. small businesses that seek
to undertake informatics research and development leading to commercialization. Critical research
areas include: representation of medical knowledge in computers; organization and retrieval issues for
image databases; enhancement of human intellectual capacities through virtual reality, dynamic
modeling, artificial intelligence, and machine learning; medical decision-making; linguistic analyses of
medical languages and nomenclatures; investigations of topics relevant to health information or library
science; biotechnology informatics issues; and informatics for disaster management. For additional
information about areas of interest to NLM and a listing of NLM funded applications, please visit
http://www.nlm.nih.gov/ep. Business concerns interested in exploring SBIR/STTR grant opportunities
with NLM are encouraged to contact the NLM representatives prior to submitting an application.
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SEED website. For budgetary,
administrative, or programmatic reasons, the NLM may not fund an application or may decrease
the length of an award and/or the budget recommended by a review committee. NIH has
received a waiver from SBA, as authorized by statute, to exceed the statutory budget limitations
set by the SBA for specific topics relevant to the NLM that can be found in the SBA-Approved
Waiver Topics. Applicants with budget questions or considering requesting a budget
greater than these amounts are strongly encouraged to contact program staff before
submitting an application.
Specific SBIR and STTR Program Information
NLM Clinical Trials Topics
NLM will not accept SBIR applications that propose clinical trials, and all of the topics listed must
be for projects that do not propose clinical trials.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
NLM does not accept Phase IIB applications and does not participate in the CRP program.
Clinical Trials
Does NLM accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
Yes
Does NLM accept Clinical Trials through
specific Notices of Funding
Opportunities?
Yes
Does NLM support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
Yes

NIH, CDC, and FDA Program Descriptions and Research Topics
NLM 155
NLM Research Topics
NLM’s SBIR/STTR grant programs are focused on areas of particular interest from small
business. The following narrative indicates the scope of projects suitable for the
SBIR/STTR program that fit within the mission of NLM. They are not listed in priority order.
1.
Development of new, innovative tools and methods for annotating, curating, and
managing biomedical data resources
2.
Advanced information retrieval and knowledge discovery from very large and/or
heterogeneous data sets to support biomedical research
3.
Development of novel approaches enabling analysis and discovery at scale
across biomedical domains, research institutions, or health care sectors, including
those leveraging high-performance cloud computing and federated learning
4.
Novel platforms, technologies, tools, and techniques enabling multimodal data
analysis to support health research or health care applications
5.
Multi-level, reusable, data analytic models, simulations, information visualization,
and presentation approaches to enhance decisions, learning or understanding of
biological and clinical processes
6.
Computational approaches integrating structured and unstructured data, natural
language processing, automated metadata assignment to support biomedical
research
7.
Data science methods, approaches, or tools that enhance the quality,
confidentiality, security, and/or utility of biomedical data, including personal health
information
8.
Artificial Intelligence techniques for characterizing and minimizing the impact of
errors, incompleteness, missingness, within health-related data sets
9.
Novel data-driven methods to facilitate real-time decision-making in clinical
practice and public health
10.
Analytical tools, technologies, and other data-driven strategies to track disease
outbreaks, epidemics, and pandemics
11.
Tools and technologies for understanding and predicting climate and
environmental effects on human health
Contact Information
For additional information on research topics, contact:
Dr. Rebecca Moen
Program Officer
Division of Extramural Programs
National Library of Medicine
301-594-7593
Email: rebecca.moen@nih.gov
For administrative and business management questions, contact:
Ms. Samantha Tempchin
Grants Management Officer
Extramural Programs Division
National Library of Medicine
301-496-4222,
Fax: 301-402-0421
Email: samantha.tempchin@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
ORIP 156
DIVISION OF PROGRAM COORDINATION, PLANNING, AND STRATEGIC
INITIATIVES (DPCPSI), OFFICE OF RESEARCH INFRASTRUCTURE
PROGRAMS (ORIP)
Mission
ORIP supports high-quality, disease-free animal models and specialized animal research facilities to
help meet the needs of biomedical researchers to understand, detect, treat, and prevent a wide
range of human diseases. This support enables discoveries at molecular, cellular, and organ levels
that lead to animal-based studies which then are translated to patient-oriented clinical research,
aiming to find treatments to ameliorate or cure common and rare diseases. Through the small
business Phase I, Phase II, Fast-track, Direct Phase II and Phase IIB awards, ORIP is especially
interested in funding research to develop preclinical methods and technologies to reduce or replace
animal use with novel alternative methods, improve animal models of human diseases, and the
care, use, and management of laboratory animals. ORIP also encourages the development and
implementation of technologies to directly benefit the welfare of research animals and to directly
improve animal facilities that support biomedical and behavioral research.
A list of some potential ORIP program topics follows the description of our Phase IIB Competing Renewal
Awards. For additional information about ORIP, please visit our home page at https://orip.nih.gov/.
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed the amounts
defined by the SBA, which can be found on the NIH SEED website. For budgetary, administrative, or
programmatic reasons, ORIP may not fund an application, may decrease the length of an award,
and/or the budget recommended by a review committee. NIH has received a waiver from SBA, as
authorized by statute, to exceed the statutory budget limitations set by the SBA for specific topics
relevant to ORIP that can be found in the SBA-Approved Waiver Topics. Applicants who have
budget questions or are considering requesting a budget greater than these amounts are
strongly encouraged to contact program staff before submitting an application.
Specific SBIR and STTR Program Information
ORIP Clinical Trials Topics
ORIP will not accept SBIR applications that propose clinical trials. All the topics listed below are for
projects that do not propose clinical trials.
Specific Funding Opportunities and Programs
In addition to the Omnibus program announcement, ORIP has targeted Notices of Funding
Opportunities (NOFOs). Please visit our ORIP Funding Opportunities webpage to view the latest
targeted NOFOs.
Phase IIB Competing Renewal Awards and Commercialization Readiness Pilot (CRP)
ORIP will only accept Phase IIB SBIR Competing Renewal grant applications of ORIP-supported Phase II
awards to continue research and development of methods, technologies, tools, and devices for basic or
translational research where extraordinary time and effort are needed for completion of these projects.
The Phase IIB Competing Renewal award is intended to allow small businesses the opportunity to

NIH, CDC, and FDA Program Descriptions and Research Topics
ORIP 157
reach a stage where interest and investment by third parties would be more likely. Such products are
expected to have broad applicability, consistent with the mission of ORIP. Budgets that do not exceed
$1 M per year in total costs (for up to 2 years), may be requested for this Phase IIB Competing
Renewal opportunity; however, it is expected that, in most cases, the requested budget would not
exceed the final year budget of the applicant’s previous Phase II award. This opportunity is available
for the SBIR program only.
Please contact your Program Officer before beginning the process of preparing a Phase IIB
Competing Renewal application. In addition, prospective applicants are strongly encouraged to
submit to the Program Contact (listed after each section) a letter of intent by email that includes the
following information:
•
Descriptive title of the proposed research
•
Name, address, and telephone number of the Principal Investigator
•
Names of other Key Personnel
•
Participating organizations
•
Notice of Funding Opportunity Number (e.g., PA-22-XXX)
A letter of intent is not required, is not binding, and does not enter into the review of a subsequent
application. It is expected that only a few of ORIP SBIR Phase II awards will be eligible for a Phase
IIB Competing Renewal grant.
ORIP does not participate in the Commercialization Readiness Pilot (CRP) program at this time.
Clinical Trials
Does ORIP accept Clinical Trials
through the Omnibus/Parent Notices of
Funding Opportunities?
No
Does ORIP accept Clinical Trials
through specific Notices of Funding
Opportunities?
No
Does ORIP support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
No
Research Topics
ORIP Non-Clinical Trials Topics:
Division of Comparative Medicine
A.
Development of in vitro animal cell culture techniques, microphysiological systems
(organs- on-a-chips, organoids) or computational (in silico) methods to reduce the
number of animals used in studies and to replace certain tests conducted in animal
models with novel alternative methods (NAMS).
B.
Development of improved reagents and cost-effective methods and technologies to
accurately screen and diagnose selected diseases of laboratory animal, and to perform
overall assessments of animal quality and health status. An urgent need currently exists
for the development of improved methods for detection of active tuberculosis in
nonhuman primates (NHPs).
C.
Development of improved reagents, techniques, and devices/tools for genomic
and transcriptomic analysis and data mining from tissue or single cells of laboratory
NIH, CDC, and FDA Program Descriptions and Research Topics
ORIP 158
animals and animal models of human diseases.
D.
Development of new technologies for rapid or high throughput animal
characterization and deep phenotyping (at tissue, organ, cellular or molecular
levels) .
E.
Development of nonintrusive, wearable technologies to identify or assess biomarkers in well
validated animal models.
F.
Development of prophylactics or new therapeutic agents to prevent and/or control
infectious diseases of laboratory animals. One high priority need is to develop
methods to control and prevent monkey B virus (Macacine alphaherpesvirus-1)
infections.
G.
Identification, development, and characterization of spontaneous (naturally
occurring) and engineered vertebrate animal models for studies of various human
diseases (excluding most random mutagenesis projects performed on rodents).
H.
Development and refinement of high-throughput technologies, cryoprotectants and
devices for the cryopreservation, vitrification, long-term maintenance, and revival of
cells, tissues and organs, as well as laboratory animal embryos and gametes,
especially for Drosophila, aquatic models of biomedical value, swine, and NHPs.
I.
Development of technologies, tools, and devices for the effective monitoring of
frozen, cryopreserved cells and biological materials/tissues as well as laboratory
animal embryos and gametes (including monitoring conditions during their
distribution).
J.
Development of technologies for improved embryo transfer within a single animal
species or of intraspecific embryo transfer to allow preservation of rare or unique animal
species that may have unique value in biomedical research as animal models for human
disease.
K.
Development of improved reagents, artificial intelligence/machine learning
technologies, tools, devices, and high-throughput technologies to perform, analyze,
capture, and process data gathered in “omics” studies (genomics, transcriptomics,
epigenomics, proteomics, lipidomics, glycomics, metabolomics, and phenomics,
among others) in normal, diseased, and intervention conditions in animal/biological
models to support or validate pre-clinical analyses.
L.
Development of biological tools and reagents for reconstruction, remodeling, repair,
and regeneration of tissues damaged by injury or disease. Development of the
technologies and procedures to test efficacy and safety of these experiments in
animal models. Approaches to detect and track survival of implanted cells and
tissues in vivo.
M.
Development of reagents (including antibodies), assays, and technologies that will
facilitate research using aquatic biomedical models, such as zebrafish or Xenopus,
for understanding basic aspects of development, physiology, or genetics.
N.
Development of reagents (including antibodies), assays, and technologies that will
facilitate research using NHPs for understanding basic aspects of development,
physiology, or genetics. High priority needs include reagents for NHP species other
than the rhesus macaque.
O.
Development of rapid and sensitive technology for the accurate detection and
diagnosis of polymicrobial infections in biomedical laboratory animal models, including
those agents involved in vertical transmission of diseases into embryos and larvae.
P.
Technologies for improved sex determination of embryonic, neonatal, and juvenile
stages of animals, with one high priority need being nonmammalian species.
NIH, CDC, and FDA Program Descriptions and Research Topics
ORIP 159
Q.
Development of rapid and sensitive technology for the detection and study of emerging
human pathogens in animal models.
R.
Development of non-invasive, micro-sensor technologies (e.g., embedded sensors,
devices, microchips) for NHPs and other live animal models to collect data related to
neuroimaging, behavioral and cognitive assessments, metabolism, microbiomes, and
other biomedical research areas. Of special interest are wearable, swallowable
sensitive and selective probes/sensors for detecting physiological fluctuations in
living animals, with the capability of monitoring at deep tissues level.
S.
Development of technologies for cell-based therapies that could be used as
implantable biocomputers in animal models of human disease, to perform
complex logic computations that integrated signals from multiple
metabolites/pathways. These include remote-controlled switches and natural,
nontoxic, highly soluble, and potentially beneficial to health trigger molecules.
T.
Development of technologies and tools (biosensors, imaging approaches, and
reagents such as antibodies (especially nanobodies)), to facilitate validation
and use of multiple model organisms in research studies to enhance the rigor,
and reproducibility of animal research and their clinical and translational
research potential.
Division of Construction and Instruments
The Division of Construction and Instruments supports the development and implementation of
technologies that enhance and improve the welfare and research facilities of animal models in
biomedical and behavioral research. In particular, the areas being supported include novel tools
and equipment that improves and facilitates the care and monitoring of animals. One area of
interest is the integration of technological features for detecting, measuring, monitoring,
recording, analyzing, and reporting physiological, biobehavioral, and environmental conditions to
allow for documenting these physiological parameters and extrinsic factors and enhancing
scientific rigor and reproducibility in animal studies. Another area of interest encompasses the
improvement of laboratory equipment to maintain the environmental conditions and upkeep of the
infrastructure within animal facilities. Of special importance is the employment of green
technologies. Examples of topics of special interest include (but are not limited to) the
development of better, more reliable, and more efficient:
A.
Equipment such as vacuum cleaners, air filters, hoods, snorkels, and autoclaves
for animal research facilities, for barrier facilities, and other facilities with special
needs and requirements.
B.
Equipment/devices to distribute water and food and to monitor and record their
intake by research animals.
C.
Equipment to increase the quality of life and prevent injuries of research
animals, staff, and investigators.
D.
Technology to monitor and protect the well-being of animals, including IT-supported tools.
E.
Equipment and its use for maintenance of disease-free colonies and healthy animals.
F.
Equipment to disinfect devices, furnishings, and other apparatus in animal
facilities such as aquaria, cages, tunnels, and racks.
G.
Cost-effective husbandry and colony management techniques, equipment,
and/or new approaches to improve laboratory animal welfare and assure
efficient and appropriate research use.
H.
Specialized equipment and caging for laboratory animals to permit optimal

NIH, CDC, and FDA Program Descriptions and Research Topics
ORIP 160
environmental control, and operational efficiency, including improvements in caging,
identification/tagging of animals and remote monitoring in animal facilities.
I.
Specialized equipment to permit integrated environmental factor measuring,
monitoring, reporting, recording and documentation (such as for air quality,
temperature, humidity, lighting, sound, and vibration level).
Contact Information
For additional information on DCM research topics, contact:
Miguel Contreras, Ph.D.
Division of Comparative Medicine,
Office of Research Infrastructure Programs,
Division of Program Coordination, Planning, and Strategic Initiatives,
Office of the Director
Phone: 301-451-7845,
Email: miguel.contreras@nih.gov mailto:contre1@mail.nih.gov
For additional information on DCI research topics, contact:
Xiang-Ning Li, Ph.D.
Director
Division of Construction and Instruments,
Office of Research Infrastructure Programs,
Division of Program Coordination, Planning, and Strategic Initiatives
Office of the Director Phone: 301-435-0766
Email: xiang-ning.li@nih.gov
For administrative and business management questions, contact:
Ms. Donna James
Grants Management Specialist,
National Heart, Lung, and Blood Institute (NHLBI),
Office of Research Infrastructure Programs (ORIP),
6705 Rockledge Drive, 202-G
Bethesda, MD 20817
Phone: 301-827-8063
Email: donna.james@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
ORWH 161
OFFICE OF RESEARCH ON WOMEN’S HEALTH
Mission
ORWH is the first Public Health Service office dedicated specifically to promoting women's
health research within and beyond the NIH scientific community. ORWH was established in
September 1990. Congress assigned a far-reaching leadership role for ORWH by mandating
that the ORWH Director:
• Advise the NIH Director and staff on matters relating to research on women's health
• Strengthen and enhance research related to diseases, disorders, and conditions that
affect women
• Ensure that research conducted and supported by NIH adequately addresses issues
regarding women's health
• Ensure that women are appropriately represented in biomedical and bio-behavioral
research studies supported by the NIH
• Develop opportunities and support for recruitment, retention, reentry, and advancement
of women in biomedical careers
• Support and advance rigorous research that is relevant to the health of women
• Ensure NIH-funded research accounts for sex as a biological variable (SABV)
ORWH crafts and implements the NIH-Wide Strategic Plan for Women's Health Research in
partnership with NIH Institutes and Centers (ICs) and co-funds research on the role of sex and
gender on health. ORWH also collaborates with NIH ICs, the NIH Office of Extramural
Research, and the NIH Office of Intramural Research to monitor adherence to NIH's inclusion
policies, which ensure that women and minorities are represented in NIH-supported clinical
research.
Budget Guidance
ORWH does not award grants but co-funds women’s health-related applications that have
received an award from one of the participating NIH Institutes and Centers (ICs) listed in the
announcement. Budgets should be discussed with Institute or Center program staff, not ORWH.
Specific SBIR and STTR Program Information
The NIH Office of Research on Women’s Health is technology agnostic and welcomes
applications focused on diseases and health conditions that predominantly affect women (e.g.,
autoimmune diseases; depressive disorders, Alzheimer’s disease (AD) and Alzheimer’s
disease-related dementias (ADRD), gender-based-violence), present and progress differently in
women (e.g., cardiovascular disease; HIV; reproductive aging and its implications), or are
female-specific (e.g., uterine fibroids; endometriosis; menopause).
Specific Funding Opportunities and Programs
In addition to the Omnibus program announcement, for a current list of funding announcements
and programmatic initiatives visit: https://orwh.od.nih.gov/research/funded-research-and-
programs/funding-opportunities-and-notices
Research Topics

NIH, CDC, and FDA Program Descriptions and Research Topics
ORWH 162
ORWH will consider co-funding for applications that have a central focus on the health of
women, as demonstrated through specific aims that EITHER explicitly address a particular
condition in women OR focus on one of the high priority topics below. In either
approach, intersectional and/or multidimensional approaches that consider the health of women
in context (e.g., projects accounting for social and structural variables—
including race, ethnicity, socioeconomic status, and State and Federal policies— that affect
women’s health) are strongly encouraged. While projects are not required to exclusively focus
on women for ORWH co-funding, studies that include more than one sex or gender should be
designed and powered to make generalizable conclusions about women and enable sex or
gender difference comparison.
ORWH intends to prioritize interdisciplinary research that:
• Addresses the influence of sex-linked biology, gender-related factors, or their
intersections on health.
• Addresses how physical, mental, and psychological health outcomes interact with
structural factors to either mitigate or exacerbate health disparities and aims to create
behavioral interventions to address these issues.
• Advances the translation of research advancements and evidence in women’s health
into practical benefits for patients and providers.
• Informs and develops multi-sector partnerships to advance innovation in women's health
research.
• Increases public awareness of the need for greater investment in and attention to
women’s health research, as well as women’s health outcomes across the lifespan.
• Advances research to reduce health disparities and inequities affecting women’s health,
including those related to race, ethnicity, age, socioeconomic status, disability, and
exposure to environmental factors and contaminants that can directly affect health.
• Increases uptake of evidence-based interventions that advance women’s health.
• Addresses topics identified in the Women’s Health Innovation Opportunity Map.
• Reduces violence, stigma, and trauma related to HIV.
• Develops woman-centered self-tests for HIV viral load monitoring, including in breast
milk.
• Develops topical microbicide agents, wearable, implantable, or insertable devices
releasing medications alone or as part of multipurpose prevention technologies (MPTs),
to prevent sexual acquisition of HIV, other sexually transmitted infections and/or
unplanned pregnancy across the lifespan including in pregnant or postpartum people.
• Promotes the use of advanced statistical modeling, data visualization, artificial
intelligence (AI), and machine learning (ML) methods for research on the health of
women.
• Develops methods, tools, or technologies to increase data sharing and improve data
management practices to align with the Findability, Accessibility, Interoperability, and
Reusability (FAIR) data principles and enhances the utility of new and existing data on
the health of women.
• Methods for integrating data science, including artificial intelligence and machine
learning, into research on the health of women.
• Develops cutting-edge computational tools and technologies to facilitate screening for
prevention, diagnosis, and treatment of diseases that affect women.
• Improves estimates of the impact of chronic conditions in women, including projects to
diagnose and reduce misclassification of female-specific and gynecologic conditions,
conditions that predominantly impact women or affect women differently, and projects to
characterize differences in chronic condition presentation by gender, race and ethnicity,

NIH, CDC, and FDA Program Descriptions and Research Topics
ORWH 163
and the various structural and social determinants that these women experience or are
affected by.
• Explores the role of hormonal fluctuations on the development of chronic conditions in
women, including the influence of age of menarche; the role of menstrual cycle
ir/regularity, length, and phase; the length of the reproductive window; the link between
adverse pregnancy outcomes and development of chronic conditions later in life; and the
effect of exogenous hormones on the development of chronic conditions
• Develops new and better approaches for addressing the symptoms that affect women
during perimenopause, menopause, and postmenopause
• Improves early and accurate detection and diagnosis of chronic conditions in women,
including the diagnosis of multiple chronic conditions
• Elucidates gender differences in access and use of health care services, including
methods for assessing discrimination (e.g., sexism, racism, ageism, and homophobia)
encountered by women when accessing health care services for chronic conditions.
Contact Information
For additional information on ORWH priorities, please contact:
Elizabeth Barr, Ph.D.
ORWH Associate Director for Interdisciplinary Research
Office of Research on Women’s Health, NIH
Phone: 301-402-7895
Email: Elizabeth.Barr@nih.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 164
CENTERS FOR DISEASE CONTROL AND PREVENTION (CDC)
CDC is the nation’s leading science-based, data-driven, service organization that protects the
public’s health. For more than 70 years, we’ve put science into action to help children stay healthy
so they can grow and learn; to help families, businesses, and communities fight disease and stay
strong; and to protect the public’s health.
Vision
Equitably protecting health, safety, and security.
Mission
CDC works 24/7 to protect America from health, safety, and security threats, both foreign and in the
U.S. Whether diseases start at home or abroad, are chronic or acute, curable, or preventable, human
error or deliberate attack, CDC fights disease and supports communities and citizens to do the same.
CDC increases the health security of our nation. As the nation’s health protection agency, CDC
saves lives and protects people from health threats. To accomplish our mission, CDC conducts
critical science and provides health information that protects our nation against expensive and
dangerous health threats and responds when these arise.
The 2022-2027 CDC Strategic Plan advances science and health equity and affirms the agency’s
commitment to one unified vision — equitably protecting health, safety, and security. The plan
leverages five core capabilities of the agency to include: a diverse public health workforce, world-
class data and analytics, state-of-the-art laboratories, rapid response to outbreaks at their source,
and strong global capacity and domestic preparedness. Our work is underscored by the agency’s
Pledge to the American People and dedication to use timely data and science to drive and
communicate customer- centered, high-impact public health action.
Budget Guidance
Total funding support (direct costs, indirect costs, fees) normally may not exceed $306,872 for
Phase I awards and $2,045,816 for Phase II awards. These are the amounts currently defined by
the U.S. Small Business Administration (SBA), which can also be found on the CDC SBIR
website. Applicants considering a requested budget greater than these limits are strongly
encouraged to contact CDC program staff before applying. For budgetary, administrative, or
programmatic reasons, the CDC may not fund an application or may decrease the length of an
award and/or the budget recommended by a review committee. CDC has received a waiver from
SBA, as authorized by statute, to exceed the statutory budget limitations set by SBA for specific
topics relevant to CDC that can be found in the SBA-Approved Waiver Topics Over Statutory
Budget Limitations document.
Before considering and/or preparing an application to the CDC SBIR program, applicants are
encouraged to review the websites of CDC’s participating components and to contact the SBIR
program staff listed below.
Specific SBIR and STTR Program Information
CDC does not accept STTR applications.
CDC does not accept Direct to Phase IIs, Fast-Track, or Phase IIB.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 165
For additional information about CDC, please visit: http://www.cdc.gov.
Specific Funding Opportunities and Programs
Clinical Trials
Does CDC accept Clinical Trials through
the Omnibus/Parent Notices of Funding
Opportunities?
Yes
Does CDC accept Clinical Trials
through specific Notices of Funding
Opportunities?
Yes
Research Topics/Specific Areas of Interest
Additional information on the awarding components (CIOs) and their research interests is available
electronically on the home pages shown throughout the “Research Topics/Specific Areas of
Interest” sections below.
CDC encourages investigator-initiated applications that focus on support for the prevention,
detection,and response to emerging public health threats. CDC also encourages investigator-
initiated applications that align with the agency’s overall mission and the research areas of interest
of the participating CDC components below.
Contact Information
General questions about the CDC SBIR program should be directed to:
Small Business Innovation Research Program (SBIR)
Office of Science (OS)
Centers for Disease Control and Prevention (CDC)
Email: SBIR@cdc.gov
GLOBAL HEALTH CENTER (GHC)
Mission and Research Areas of Interest
CDC’s global health mission is to improve and protect the health, safety, and security of Americans
while reducing morbidity and mortality worldwide. CDC’s Global Health Center works to protect
Americans from dangerous and costly public health threats, including COVID-19, vaccine-
preventable diseases, HIV, TB, and malaria—responding when and where health threats arise.
Goals of the center include:
-
Protecting Americans and populations across the globe by strengthening global public health
prevention, detection, and response (Health Security)
-
Saving lives, improving health outcomes, and fostering healthy populations globally (Health
Impact)
-
Leading and influencing the advancement of global public health science and practice and
serving as a leading source of credible scientific information (Public Health Science Leadership)
GHC seeks to accomplish its goals by:
-
Leveraging the latest technologies and advanced analytics to accelerate public health impact
-
Eliminating health disparities and achieve optimal health for all
-
Establishing and strengthening sustainable country, regional, and international public health
systems

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 166
-
Fostering health diplomacy in coordination with other U.S. Government agencies through its
longstanding bilateral and multilateral partnerships, engagement with the private sector, and
ongoing collaborations with academic institutions and foundations
-
Availing its workforce to address the most urgent global public health threats
Additionally, GHC encourages submission of research applications with innovative research
technologies designed to reduce health disparities and promote health equity.
Please visit the GHC website at: https://www.cdc.gov/GlobalHealth/.
Description of Research Areas of Interest
Areas of focus include:
-
Achieving an AIDS-Free generation
-
Innovating TB and Malaria prevention and treatment
-
Optimizing the impact of pneumococcal conjugate vaccine
-
Eradicating polio and combating vaccine-preventable diseases
-
Eliminating epidemic meningitis in Africa
-
Improving country capacity to detect and respond to outbreaks and prevent future ones
-
Training a multidisciplinary, global workforce
-
Improving in-country emergency response capacity
-
Maintaining a rapid response capacity at CDC to respond to international public
health emergencies
-
Analyzing data to best inform programming in limited resource settings
-
Rapidly translating data to improve public health outcomes
Examples of specific research areas of interest include, but are not limited to:
-
Advanced tools and techniques for detecting new microbes, biomarkers, and cases
-
Non-traditional surveillance tools and methods for real-time detection of emerging
public health threats, including COVID-19, vaccine-preventable diseases, HIV, TB,
and malaria
-
Virtual reality equipment and software to support the development of virtual
laboratory environments to assess practical laboratory skill competency
-
Technologies to detect diseases faster
Impact and Commercialization Potential:
This research could lead to the development of tools for the detection of diseases and improve
the ability to rapidly respond to emerging public health threats. Successful and novel innovation
that will lead to the development of these tools and technologies will have huge commercial
potential across many markets.
For GHC programmatic information, contact:
Lata Kumar, MS, MPH, MBA
Office of the Director
Center for Global Health Center
Centers for Disease Control and Prevention (CDC) Mail Stop H21-9
1600 Clifton Road NE Atlanta, GA 30329
Phone: (404) 639-7618
Email: LKumar@cdc.gov
For grants specific, administrative information, contact:

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 167
Angie Willard, Team Lead
Office of Grant Services
Office of Financial Resources
Office of the Chief Operating Officer
Centers for Disease Control and Prevention (CDC)
Mail Stop TV-2
2939 Flowers Road NE
Atlanta, GA 30341
Phone: 770-498-2596
Email: AEN4@cdc.gov
NATIONAL CENTER ON BIRTH DEFECTS AND DEVELOPMENTAL
DISABILITIES (NCBDDD)
Mission and Research Areas of Interest
The mission of CDC’s National Center on Birth Defects and Developmental Disabilities (NCBDDD) is
to promote the health of babies, children, and adults and to enhance the potential for full, productive
living. To achieve its mission, the Center works to identif y the causes of birth defects and
developmental disabilities, helps children to develop and reach their full potential, and promotes
health and well-being among people of all ages with disabilities, including blood disorders. NCBDDD
seeks to accomplish these goals through research, partnerships, and prevention and education
programs. Our efforts in surveillance, research, and health promotion programs provide information
used to identify causes of birth defects and infant disorders, find opportunities to prevent them, and
improve the health of those with these conditions. Additionally, NCBDDD encourages submission of
research applications with innovative research technologies designed to reduce health disparities
and promote health equity.
Please visit the NCBDDD website at: http://www.cdc.gov/ncbddd/index.html.
Description of Research Areas of Interest
Goals of the center include:
-
Improving the health and developmental outcome of children and adults with or at
risk of disabilities
-
Preventing complications and improving the management of blood disorders
-
Addressing preventable causes of birth defects, infant disorders, and related conditions
-
Promoting and improving the health of people living with birth defects, infant disorders,
and related conditions
Examples of specific research areas of interest include, but are not limited to:
-
Improved methods for screening for newborn heart disorders
-
Technology-based solutions to improve access to evidence-based interventions to
manage attention-deficit/hyperactivity disorder and tic disorders
-
Tools and technologies to improve the life of people living with birth-defects
-
Technologies that improve access of individuals with intellectual disabilities to services
and evidence-based management interventions
Impact and Commercialization Potential:
This research could lead to the development of tools for the management of birth defects and
developmental disabilities, improve access to effective interventions among people with disabilities,

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 168
and lead to the development of devices and methods for the detection and management of birth
defects. Effective tools and technologies have the potential for uptake by the intended populations
and their caregivers.
For NCBDDD programmatic information, contact:
Natalie Brown, MPH
Office of the Director
National Center for Chronic Disease Prevention and Health Promotion
Centers for Disease Control and Prevention (CDC)
Mail Stop S107-8
4770 Buford Highway
Atlanta, GA 30341
Phone: 404-639-4601
Email: NBrown3@cdc.gov
For grants specific, administrative information, contact:
Brownie Anderson-Rana
Office of Grant Services
Office of Financial Resources
Office of the Chief Operating Officer
Centers for Disease Control and Prevention (CDC)
Mail Stop K-75
2920 Brandywine Rd.
Atlanta, GA 30341
Phone: 770-488-2771
Email: BAndersonRana@cdc.gov
NATIONAL CENTER FOR CHRONIC DISEASE PREVENTION AND HEALTH
PROMOTION (NCCDPHP)
Mission and Research Areas of Interest
CDC’s National Center for Chronic Disease Prevention and Health Promotion (NCCDPHP) carries
out a variety of activities that improve the nation’s health by preventing a range of chronic diseases
such as arthritis, cancer, diabetes, heart disease, obesity, and stroke, while promoting health and
wellness in the areas of reproductive health, oral health, nutrition, and physical activity. The Center’s
activities include supporting states’ implementation of public health programs; public health
surveillance; translation research; and developing tools and resources for organizations at the
national, tribal, state, and community levels. NCCDPHP has identified the following Social
Determinants of Health (SDoH) as priorities: built environment, clinical-community linkages, social
connectedness, tobacco control policy, and food and nutrition security. Additionally, the Center
encourages submission of research applications with innovative technologies designed to reduce
health disparities and promote health equity.
Please visit the NCCDPHP web site at: http://www.cdc.gov/chronicdisease/index.htm.
Description of Research Areas of Interest
The center’s areas of focus include:
-
Development, implementation, and promotion of effective strategies for preventing and
controlling cancer

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 169
-
Prevention of diabetes and diabetes complications
-
Improving cardiovascular health for all, reducing the burden, and eliminating
disparities associated with heart disease and stroke
-
Supporting healthy eating, active living, and obesity prevention
-
Promoting optimal and equitable health in women and infants
-
Supporting efforts to protect the public’s health from the harmful effects of tobacco use
-
Improving symptoms and quality of life for people with conditions such as Alzheimer’s
disease, arthritis, epilepsy, and lupus
-
Improving oral health by promoting proven interventions
-
Working through schools to implement effective school-based strategies that help youth become
healthy, successful adults
Examples of specific research areas of interest include, but are not limited to:
-
Tools to increase access to health care, decision support tools to improve
prevention, measurement, and management of chronic diseases
-
Applications to improve nutrition among children and adults
-
Implementation tools to improve use of evidence-based interventions such as chronic
disease self-management programs
-
Design aids to increase physical activity through the built environment
-
Tools to promote physical activity and physical activity education among children
-
Methods to improve access to screening
-
Applications to reduce smoking and support quitting
-
Interventions to promote prevention and early detection of cancers
Impact and Commercialization Potential:
This research could lead to the development of solutions to increase uptake of prevention
interventions and increase access to screening services, as well as lead to the development of
tools to monitor and manage chronic diseases, address health disparities among specific sub-
populations, increase linkage to clinical and other support services, and result in prevention and
better management of chronic diseases. Practical, easy to use, and effective solutions will have
potential for uptake by large segments of the target populations.
For NCCDPHP programmatic information, contact:
Natalie Brown, MPH
Office of the Director
National Center for Chronic Disease Prevention and Health
Promotion
Centers for Disease Control and Prevention
(CDC)
Mail Stop S107-8
4770 Buford Highway
Atlanta, GA 30341
Phone: 404-639-4601
Email: NBrown3@cdc.gov
For grants specific, administrative information, contact:
Angie Willard, Team Lead
Office of Grant Services Office of Financial Resources
Office of the Chief Operating Officer
Centers for Disease Control and Prevention (CDC)
Mail Stop, T2939 Flowers Road NE Atlanta, GA 30341

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 170
Phone: 770-498-2596
Email: AEN4@cdc.gov
NATIONAL CENTER FOR EMERGING AND ZOONOTIC INFECTIOUS
DISEASES (NCEZID)
Mission and Research Areas of Interest
The National Center for Emerging and Zoonotic Infectious Diseases (NCEZID) aims to prevent
disease, disability, and death caused by a wide range of infectious diseases. NCEZID focuses on
diseases that have been around for many years, emerging diseases (those that are new or just
recently identified), and zoonotic diseases (those spread from animals to people). Work is guided in
part by a holistic "One Health" strategy, which recognizes the vital interconnectedness of microbes
and the environment. Through a comprehensive approach involving many scientific disciplines,
better health for humans and animals and an improved environment can be attained. Research to
address reducing health disparities and increasing health equity is strongly encouraged.
Please visit the NCEZID website at: http://www.cdc.gov/ncezid.
Description of Research Areas of Interest
(1)
Develop a Rapid, Portable, Point-of-Care Antifungal Therapeutic Drug Monitoring Tool
Background: Every year, an estimated 150 million severe fungal infections occur worldwide,
resulting in approximately 1.7 million deaths. Antifungals drugs for treating severe fungal infections
are limited to just 3 classes, and antifungal resistant infections are a rising global public health threat,
underscoring an urgent need to optimize antifungal use to prevent mortality and minimize resistance
selection pressure. Antifungal therapeutic drug monitoring is a critical component of individualized,
precision treatment for fungal infections and for antifungal stewardship. It can help optimize individual
patients’ antifungal treatment regimen by measuring antifungal drug concentrations in a patient's
biologic fluids and applying well-described pharmacokinetic principles to adjust drug dosing.
Infectious Diseases Society of America's Guidelines for treatment of fungal diseases like candidiasis,
aspergillosis, histoplasmosis, coccidioidomycosis, and blastomycosis recommend therapeutic drug
monitoring for certain antifungals, particularly for triazole antifungals (e.g., itraconazole, voriconazole,
Posaconazole). Antifungal therapeutic drug monitoring can help improve outcomes through
minimizing toxicity associated with supratherapeutic drug levels and prevent treatment failure from
subtherapeutic levels. Also, in vitro studies have shown that suboptimal drug levels can lead to
antifungal resistance, supporting therapeutic drug monitoring's potential role in preventing the
emergence of antifungal-resistant organisms. Measurement of antifungal concentrations is usually
performed by analytical assays, such as high-performance liquid chromatography or liquid
chromatography-mass spectrometry which are usually only available in large academic centers;
bioassays are available for certain azole antifungals (e.g., itraconazole), but are difficult to interpret
and can be inaccurate.
Recent studies suggest that antifungal therapeutic drug monitoring is underutilized, leading to missed
opportunities to address sub- and supratherapeutic drug levels and improve patient outcomes. An
analysis of a hospital discharge data set showed that only 16% of therapeutic drug monitoring-eligible
patients receiving itraconazole, Posaconazole, or voriconazole had drug level monitoring performed.
Among 55 predominately academic medical centers, only 41% of patients receiving isavuconazole,
NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 171
Posaconazole, or voriconazole for treatment or prophylaxis of invasive fungal infections had
therapeutic drug monitoring performed.
A recent survey found that there are many challenges encountered in performing antifungal
therapeutic drug monitoring; the most common being long turnaround times for send-out tests. Other
barriers noted were difficulty coordinating testing logistics, difficulty interpreting results, and cost.
Specific Research Area(s) of Interest: The major research priorities areas for NCEZID are listed below.
Investigator initiated applications that have commercial potential that fall outside these topic areas but fall
within the research mission of NCEZID will also be considered through this Omnibus solicitation.
Research to address reducing health disparities and increasing health equity is strongly encouraged.
The goals for the proposed research are to address fungal infections being treated with antifungals
requiring therapeutic drug monitoring. Prioritized antifungals would be itraconazole, voriconazole, and
Posaconazole.
Examples of specific research areas of interest include, but are not limited to:
• Development of rapid and novel strategizes for measuring in vivo antifungal level in patient for
therapeutic efficacy.
Impact: This research will lead to the development of practical, timely and innovative solutions to
address barriers to performing antifungal therapeutic drug monitoring (e.g., long turnaround times,
difficulty in coordinating logistics) to improve patient outcomes when being treated with prioritized
antifungals.
Commercialization Potential: Current antifungal therapeutic drug monitoring is limited to large academic
centers. Successful and novel innovation will improve patient outcomes and prevent toxicity and
potentially resistance while having a large market with an increasingly population susceptible to fungal
infections. Rural and urban, community and academic hospitals will be able to procure the innovative
tools.
(2)
Finding the cause of Guillain-Barré syndrome
Background: Guillain-Barré syndrome (GBS) is a rare disorder where the body’s immune system
attacks part of the peripheral nervous system, resulting in symptoms ranging from brief weakness to
devasting paralysis. The global incident rate of GBS is about 1-2 cases per 100,000 people per year,
and the CDC estimates that about 3,000 to 6,000 cases of GBS occur every year in the United
States. The cause of GBS is not fully understood, but is most frequently associated with
Campylobacter jejuni infections, with about 1 in 1,000 people with Campylobacter infection
developing GBS in the United States. Other risk factors include respiratory illnesses, viral infections,
and vaccination. Finding an accurate cause for GBS is challenging, since symptom onset can occur
weeks after the initial infection. In outbreaks of GBS that have occurred globally, determining the
cause of GBS for accurate diagnosis and surveillance has proved to be difficult, especially in regions
with limited resources.
Specific Research Area(s) of Interest: The major research priorities areas for NCEZID are listed below.
Investigator initiated applications that have commercial potential that fall outside these topic areas but fall
within the research mission of NCEZID will also be considered through this Omnibus solicitation. Research
to address reducing health disparities and increasing health equity is strongly encouraged.
The goal of the proposed research is to develop innovative methods to determine the cause of GBS.
Examples of specific research areas of interest include, but are not limited to:
• Developing cost-effective diagnostic tests for Campylobacter species from GBS patient samples.
• Enhancing surveillance by leveraging novel technologies to sequence type associated with specific
serotypes of Campylobacter jejuni.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 172
• Determining the cause of illness during a GBS outbreak.
Impact: This research will lead to finding innovative ideas to diagnose and perform surveillance on
illnesses with ambiguous causes that can be difficult to determine using traditional methods.
Commercialization Potential: The innovative solutions developed through this research will have
significant commercial potential in improving public’s health both domestically and globally.
(3)
Developing Novel Diagnostic Testing for Typhoid Disease
Background: Typhoid fever is a significant global health concern, particularly in regions with limited
access to healthcare facilities and resources. Traditional diagnostic methods for typhoid, such as blood
culture and serological tests, have limitations in terms of accuracy, cost, and turnaround time. Therefore,
there is an urgent need to develop novel diagnostic testing approaches that are rapid, affordable, and
highly sensitive and deployable in resource limited settings.
Specific Research Area(s) of Interest: The major research priorities areas for NCEZID are listed below.
Investigator initiated applications that have commercial potential that fall outside these topic areas but fall
within the research mission of NCEZID will also be considered through this Omnibus solicitation.
Research to address reducing health disparities and increasing health equity is strongly encouraged.
The goal of this proposal is to develop innovative methods to identify the cause of illness for suspected
Typhoid fever. The proposed test method should improve the accuracy of existing diagnostic methods
and be deployable in resource limited settings.
Examples of specific research areas of interest include, but are not limited to:
• Identify appropriate markers specific and sensitive for Salmonella enterica serotype Typhi
detection from suspected cases of typhoid fever. This may involve techniques to analyze specific
bacterial genomes or bacterial antigens.
• Design and optimize an assay based on the identified biomarkers using cutting-edge
technologies that are user-friendly, require minimal equipment, field deployable, and provide rapid
results.
Impact: This research will lead to innovative diagnostic approaches to address a challenging infectious
disease that causes illness both domestically and internationally.
Commercialization Potential: Successful approaches will lead to faster and more accurate diagnoses
and reduced disease, disability, and death and will have huge commercial potential across many
markets.
(4)
Develop new or enhanced diagnostics for Leptospira spp.
Background: Leptospirosis, caused by over 35 different species of pathogenic and intermediate
Leptospira species, is one the most common zoonotic/environmental diseases in the world. Leptospira
spp. are shed in animal urine and are capable of surviving in soil and water. Globally it is estimated that
there are up to 500,000 severe cases of leptospirosis annually, although this is likely a severe
underestimate due to non-specific clinical presentations, poor availability of reliable tests, and lack of
surveillance systems. Cases of leptospirosis increase after flooding events, tends to affect individuals in
economically poor areas, and is responsible for disproportionately high global burdens both economically
and in disability adjusted life years for affected individuals. Due to climate change and the associated
extreme weather events, it is expected that cases of leptospirosis will continue to increase at a high rate.
Specific Research Area(s) of Interest: The major research priorities areas for NCEZID are listed below.
Investigator initiated applications that have commercial potential that fall outside these topic areas but fall
within the research mission of NCEZID will also be considered through this Omnibus solicitation.
Research to address reducing health disparities and increasing health equity is strongly encouraged.
Development of better diagnostic and environmental detection tools is critical to increase positive patient

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 173
outcomes and identify at risk populations.
Examples of specific research areas of interest include, but are not limited to:
• Development of new or enhanced diagnostics for Leptospira spp. including serological assays
with better specificity/sensitivity and are easier to transfer to partners than current assays, rapid
point of care diagnostics, and molecular assays capable of detecting intermediate pathogenic
species in addition to pathogenic species.
• Develop diagnostic tests that can easily differentiate between species and/or serovars of
Leptospira spp.
• Develop methods for better detection and genetic characterization of Leptospira spp. from
environmental matrices.
Impact: Leptospirosis is vastly underreported in the US, and cases are likely to increase due to extreme
weather events related to climate change. The proposed research should lead to practical solutions
related to the detection of these pathogens in the environment and the effective diagnosis of disease in
humans.
Commercialization Potential: Products related to the diagnosis and detection of Leptospira spp. will
have significant commercial potential both in the domestic and international markets and will improve both
surveillance and patient outcomes.
(5)
Vector Borne Diseases: Detection, Prevention, Diagnosis and Response
Background: Bacterial and viral vector borne diseases are transmitted to humans primarily through
vectors such as an infected mosquito, tick, or flea. Some of these diseases have long been present in
the United States while others have recently emerged.
Vector-borne diseases are a major public health concern. Lyme disease causes over 300,000
estimated human illnesses annually in the U.S. tick-borne rickettsial diseases, such as Rocky
Mountain spotted fever, ehrlichiosis, and anaplasmosis, are responsible for over 4,000 U.S. cases
each year, including some that result in death. Dengue fever causes millions of cases worldwide,
including thousands of cases in Puerto Rico each year. Outbreaks of arboviral diseases such as
West Nile encephalitis and Chikungunya fever have been reported in recent years. Less common,
but often deadly threats such as Yersinia pestis causes the ancient disease plague. Local plague
outbreaks occur in the southwestern U.S., and it is a significant health threat in Africa and Asia.
Specific Research Areas of Interest: The goal of this project is to encourage research that will enhance
prevention, detection, diagnosis, and response capabilities to vector borne diseases through funding
innovative solutions that address the following:
1)
Mitigate the spread and impact of vector borne diseases
2)
Improve our ability to prevent, detect, and respond to outbreaks of vector borne diseases
3)
Develop diagnostic tests to differentiate among vector borne diseases
4)
Develop vaccines effective against vector borne diseases
Impact: Given the large number of individuals affected by these diseases, and the challenges to
public health for their containment, improved detection through better diagnostic tests and improved
prevention through vaccination would have a great impact on the health of the nation. The proposed
research should lead to the development of practical solutions for the detection, prevention, and
diagnosis of vector-borne diseases.
Commercialization Potential: The products and innovations developed through this process will
have significant commercial potential and will improve public health and the healthcare system’s
response to vector-borne diseases.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 174
(6) Antibiotic Resistant Healthcare-Associated Infections
Background: Healthcare-associated infections (HAI) are a threat to patient safety. CDC provides national
leadership in surveillance, outbreak investigations, laboratory research, and prevention of healthcare-
associated infections. CDC uses knowledge gained through these activities to detect infections and
develop new strategies to prevent healthcare-associated infections. Healthcare-associated infections
(HAIs) can be found to effect 1 in 25 hospitalized patients on any given day in the United States, leading
to an annual burden of 722,000 infections and 75,000 deaths. Meanwhile, among 18 antibiotic resistant
(AR) organisms identified by CDC in 2013 as urgent, serious, and concerning threats, nearly half are
primarily healthcare-associated. Whereas 1 in 7 HAIs in hospitals overall are caused by AR-threat
bacteria, in some types of hospitals, AR-threat bacteria cause 1 in 4 infections. In all cases, HAIs caused
by AR-threats are more difficult to treat and some are now untreatable. There are three broad, current
strategies that clinicians and nurses need to employ to prevent these AR HAIs: following
recommendations for preventing invasive device and surgical procedure-related infections, preventing
cross-transmission of AR HAI pathogens, and practicing optimal antibiotic stewardship.
Specific Research Area(s) of Interest:
The major research priorities areas for NCEZID are listed below. Investigator initiated applications that
have commercial potential that fall outside these topic areas but fall within the research mission of
NCEZID will also be considered through this Omnibus solicitation. Research to address reducing health
disparities and increasing health equity is strongly encouraged. The goals for the proposed research are
to address antibiotic resistant healthcare-associated infections.
Examples of specific research areas of interest include, but are not limited to:
• Development of novel diagnostics that either: 1) offer a more rapid and definitive diagnosis of
whether a patient does or does not require an antibiotic (alternatively whether it is safe to stop an
antibiotic), or 2) better detect (i.e., earlier, more rapidly, and more accurately) whether a patient is
infected or colonized (and thereby may transmit) with an AR HAI pathogen.
• Novel therapeutics and preventatives based upon preservation or restoration of the human
microbiome.
• Predictive surveillance analytics that harness genomic-scale (public) data for assessing potential
trajectories and forecasting de novo risk analysis of bacterial strains/clonal types for epidemic
spread, hypervirulence, and antibiotic resistance.
Impact: This research will lead to the development of practical and innovative solutions to address the
matrix of complex problems caused by antibiotic resistant healthcare- associated infections.
Commercialization Potential: Successful and novel innovation that will reduce disease, disability, and
death will have huge commercial potential across many markets.
For NCEZID programmatic information, contact:
Amy Yang, PhD
National Center for HIV, Viral Hepatitis, STD, and TB Prevention
Centers for Disease Control and Prevention (CDC)
Phone: 404-718-8836
Email: VDZ9@cdc.gov
For grants specific, administrative information, contact:
Angie Willard, Team Lead
Office of Grant Services Office of Financial Resources
Office of the Chief Operating Officer
Centers for Disease Control and Prevention (CDC)
Mail Stop TV-2

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 175
2939 Flowers Road NE Atlanta, GA 30341
Phone: 770-498-2596
Email: AEN4@cdc.gov
NATIONAL CENTER FOR ENVIRONMENTAL HEALTH (NCEH)
Mission and Research Areas of Interest
National Center for Environmental Health (NCEH) plans, directs, and coordinates a program to
protect the American people from environmental hazards. We promote a healthy environment
and prevent premature death, avoidable illness, and disability caused by non-infectious, non-
occupational environmental and related factors. We are especially committed to safeguarding the
health of vulnerable populations ― such as children, the elderly, and people with disabilities ―
from certain environmental hazards.
NCEH encourages submission of research applications with innovative research
technologies designed to reduce health disparities and promote health equity.
Please visit the NCEH web site at: https://www.cdc.gov/nceh/.
Description of Research Areas of Interest
The major research priorities areas for NCEH are listed below. Investigator-initiated applications
that have commercial potential that fall outside these topic areas but fall within NCEH’s research
mission will also be considered through this Omnibus solicitation. Research to address emerging
health threats, health disparities, and health equity are strongly encouraged.
(1) Tools or technologies to assess or estimate indoor air quality and health risks
Background: Poor indoor air quality resulting from high levels of dust mite allergen, mold, and
particulate matter less than 2.5 microns (PM2.5) is a well-established risk factor for development
of respiratory diseases, including asthma. Because these agents are microscopic, occupants
often do not know how to decrease exposure. Improved exposure assessment methods are an
important tool in helping individuals decrease their exposure and prevent negative health
outcomes resulting from poor indoor air quality related to these agents.
For this topic, NCEH has a specific interest in tools or technologies to assess indoor air quality
related to particulate matter (PM2.5), allergens (such as those from dust mites), and mold (which
contains allergenic and irritant components) implicated in the development or exacerbation of
asthma; and in tools or technologies to assess building-related health risks that increase
exposure to these agents.
PM2.5: Particulate matter less than 2.5 microns (PM2.5) is also microscopic and associated with
asthma attacks. PM2.5 is generated from smoke associated with cooking, hair dryers, and other
smoke generating devices, including those in an occupational setting, and can accumulate to
levels high enough to trigger asthma attacks without activating a smoke detector. An early
warning system is needed before smoke detector activation. Such an early warning system could
be connected to building systems (e.g., fans/filtration devices) which can turn on automatically to
decrease PM2.5 exposures prior to smoke detector activation to prevent the onset of asthma
attacks. For additional information about indoor air triggers such as dust mites or mold, please

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 176
visit our web site at https://www.cdc.gov/asthma/pdfs/home_assess_checklist_P.pdf .
Dust mites: Exposure to dust mites can result in significant clinical illnesses including rhinitis,
allergic asthma, atopic dermatitis, and conjunctivitis. The disparities in childhood asthma in the
United States are well-known; approximately 16% of black children and 7% of white children have
asthma (CDC Vital Signs). In the U.S., a population-based study found that 15-20% of
participants were allergic to dust mites (Salo et al. 2011). Additional research showed that 25% of
children with asthma have allergies to dust mites (Stevenson et. al., 2001).
Furthermore, dust mite allergic sensitization varies by region, being more prevalent in the
Southeastern U.S. (Salo et al. 2014). Although present in many residences and other buildings,
dust mites are unnoticed because of their microscopic size and translucent bodies, making them
difficult to detect and remove from homes, schools, and businesses.
Mold: In 2009, the World Health Organization concluded that early mold exposure was
associated with the development of asthma in some children, particularly among children who
may be genetically susceptible to asthma development, and that selected interventions that
improve housing conditions can reduce morbidity from asthma and respiratory allergies. Mold
allergy and exposure to mold has been found to be strongly associated with childhood
emergency department visits in inner-city environments (Brosnan, 2013). Mold is also
microscopic. During investigations of possible mold-related disease (e.g., allergy, asthma, and
infections), costly and labor-intensive sampling by trained technicians and analysis in
laboratories can delay action for mold remediation due to the misidentification of “mold -like”
substances which are not mold (e.g. soot, rust, and algae).
Building related risk factors can contribute to poor health outcomes from asthma triggers:
Several well-established housing characteristics are associated with poor indoor air quality. For
example, age of housing, presence of basements, type of housing structure, number of
stories/floors, and presence of crawl spaces have been associated with greater risk of mold
exposure. Although the American Housing Survey is a large population-based survey of the
U.S., it only conducts a sample. Only tax assessor data contains this type of information for each
building across the country. With tax assessor data (not centrally managed but managed by
each county or state), geographic information system (GIS) mapping can be used to overlay
these data with health data to better elucidate the relationships and building- related risk factors
between indoor air quality and adverse health outcomes. NCEH is interested in better
understanding how the gentrification of neighborhoods can modify such relationships between
housing-related factors and adverse health.
Project Goal: The goal for the proposed research is to develop tools or technologies to assess
indoor air quality related to PM2.5, dust mite, or mold exposure, and to develop data-linkage
platforms to identify building-related features that are associated with increased risk for poor indoor
air quality from buildings within the U.S.
Examples of specific research areas of interest include, but are not limited to, the development of an:
-
Early warning system integrated with building systems (e.g., fans/filtration
devices) to automatically detect smoke related PM2.5 levels prior to smoke
detector activation.
-
Application to alert individuals that they are in a high-risk dust mite area.
-
Application that can view mold-like substances on a surface, and determine if the
substance is mold (e.g., using computerized image analysis).
-
Machine learning capable data-linkage platform that can extract building information
from tax assessor datasets and enable granular assessments of building-related risk
factors from data spanning several years.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 177
Impact: Asthma resulting from indoor air exposures to allergens and particulate matter is a
significant public health concern, resulting in acute and chronic clinical disease for millions of
Americans.
Innovative technologies to rapidly detect and prevent these exposures would have a beneficial
impact on public health, especially for underserved populations experiencing residential or
occupational housing inequities and at increased risk for exposure to indoor air pollution. The
technologies sought for development have the potential for commercial application across many
markets, including residential and occupational settings, schools, and healthcare facilities.
A building air quality data-linkage platform can enable more granular assessments of building-
related risk factors that decrease indoor air quality and subsequently affect health. More accurate
assessments of building-related risk factors will enable future researchers to better understand
place- based determinants of health impacting asthma and many other poor health outcomes
(e.g., cardiorespiratory disease) resulting from exposures in residential and/or occupational
settings.
Commercialization Potential: Commercial application of this technology could allow state and
local jurisdictions, businesses, and public health practitioners address housing related
environmental justice issues in a focused manner instead of only using broad levels of building
characteristics (e.g., multi-family homes vs. single-family homes).
For NCEH programmatic information, contact:
Tonia Linley
Program Specialist
National Center for Injury Prevention and Control
Centers for Disease Control and Prevention CDC
Phone: 770-488-3593
Email: IMX9@cdc.gov or NCIPC_ERPO@cdc.gov
For grants specific, administrative information, contact:
Brownie Anderson-Rana
Office of Grant Services Office of Financial Resources
Office of the Chief Operating Officer
Centers for Disease Control and Prevention (CDC)
Mail Stop K-75
2920 Brandywine Rd.
Atlanta, GA 30341
Phone: 770-488-2771
Email: BAndersonRana@cdc.gov
NATIONAL CENTER FOR HIV, VIRAL HEPATITIS, STD, AND TB PREVENTION
(NCHHSTP)
Mission and Research Areas of Interest
NCHHSTP is committed to our vision of a future free of HIV/AIDS, viral hepatitis, STDs, and TB.
We are responsible for public health surveillance, prevention research, and programs to prevent
and control HIV and AIDS, other STDs, viral hepatitis, and TB. NCHHSTP’s Strategic Plan

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 178
articulates a vision, guiding principle, and overarching goals and strategies to influence and
enhance our programs. The three overarching goals highlighted in this plan are to decrease:
•
incidence of infection,
•
morbidity and mortality, and
•
health disparities.
Every year, millions of Americans are infected with HIV, viral hepatitis, STDs, or TB, and tens of
thousands die from their infection. Most of these infections share commonalities, from modes of
transmission to demographic, social, and economic conditions that increase risk. As a prevention
leader, NCHHSTP focuses on high impact prevention and control efforts to reduce incidence,
morbidity, mortality, and health disparities due to these infections.
Please visit the NCHHSTP website at: http://www.cdc.gov/nchhstp/.
Description of Research Areas of Interest
(1) Improved Diagnostic Tests for HIV, STDs, Hepatitis, and TB
Background: For many individuals infected with HIV, Hepatitis C, STDs and TB, they do not
receive timely diagnosis due to stigma associated with these infections. Improving diagnostic
technology and offering alternatives that can be done at the point of care or self -testing has the
potential to decrease morbidity and transmission. For individuals that are at risk and uninfected, it
allows them to focus on prevention. For those that are infected, treatment can be started earlier.
Furthermore, since many of the infections share common routes of transmission or have high rates
of infections in specific populations, there is the potential for overlapping infections also known as
syndemics.
Whereas there is tremendous value in testing and diagnosis for each of the described diseases,
some at risk individuals are never tested or do not receive their tests results and often times are
only tested for one of the diseases when testing for a combination of the diseases would be more
beneficial both for the individual and for public health. Prognostic tests (e.g., viral load, drug
resistance monitoring) also play an important role in improving health outcomes for infected
individuals.
Specific Research Areas of Interest: The major research priorities areas for NCHHSTP are
listed below. Investigator initiated applications that have commercial potential that fall outside
these topic areas but fall within the research mission of NCHHSTP will also be considered through
this Omnibus solicitation. Research to address reducing health disparities and increasing health
equity is strongly encouraged.
The major goal of the project is development of diagnostic reagents, tests or testing platforms, that will
further improve diagnosis or monitoring of HIV hepatitis C, syphilis, gonorrhea or TB. The specific area of
interest is innovative approaches or novel technology that would allow for easier screening for or
diagnosis of HIV hepatitis C, syphilis, gonorrhea or TB alone or in any combination using a single test
device or platform.
Examples of specific research areas of interest include, but are not limited to: The preferred test
format or technology would facilitate testing that allows for rapid results (preferably less than one hour), is
affordable (comparable to currently available tests) and can be performed at the point of care or by an
individual (self-test). Tests that include an option for self-collection for submission to a laboratory are also
desired. Likewise, the ability to combine detection and markers for drug resistance gonorrhea, and TB
specifically) is highly desired as it would allow for better treatment outcomes and reduce the spread of
drug resistant pathogens.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 179
Impact: It is known that early diagnosis and treatment of HIV infection can improve health
outcomes for individuals infected with HIV. Furthermore, such testing has the potential for
decreasing transmission of HIV. Likewise, improved diagnosis of HCV can lead to c urative
treatment. For syphilis, improved diagnostic tools have the potential to decrease congenital syphilis
and promote timely treatment to prevent spread to sexual partners. Improved TB and gonorrhea
testing that includes easy detection of drug resistance is important to prevent the spread of drug
resistant strains and improve treatment. Improving diagnosis of all of these pathogens has a
potential for a significant decrease in morbidity. Faster turnaround times, lower cost, and more
efficient detection would be highly impactful for these individuals, their partners and the
community.
Commercialization Potential: Diagnostic reagents, tests or testing platforms, that will further
improve diagnosis or monitoring of HIV, hepatitis C, syphilis, gonorrhea or TB, would be in great
demand by the healthcare and public health systems as well as other sectors engaged in using
diagnostics to treat or prevent transmission in impacted populations.
For NCHHSTP programmatic information, contact:
Amy Yang, PhD
Extramural Research Program Office
Office of the Associate Director for Science
National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention
Centers for Disease Control and Prevention (CDC)
Phone: 404-718-8836
Email: VDZ9@cdc.gov
For grants specific, administrative information, contact:
Angie Willard, Team Lead
Office of Grant Services Office of Financial Resources
Office of the Chief Operating Officer
Centers for Disease Control and Prevention (CDC)
Mail Stop TV-2
2939 Flowers Road NE Atlanta, GA 30341
Phone: 770-498-2596
Email: AEN4@cdc.gov
NATIONAL CENTER FOR IMMUNIZATION AND RESPIRATORY DISEASES
(NCIRD)
Mission and Research Areas of Interest
The mission of the National Center for Immunization and Respiratory Diseases (NCIRD) is the prevention
of disease, disability, and death through immunization and by control of respiratory and related diseases.
NCIRD balances its efforts in the domestic and global arenas as well as accommodates the specific
needs of all populations at risk of vaccine preventable diseases from children to older adults. Research to
address reducing health disparities and increasing health equity is strongly encouraged.
Please visit the NCIRD website at: http://www.cdc.gov/ncird/.
Description of Research Areas of Interest

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 180
(1)
Prevention and Diagnosis of Acute Respiratory Infections in the US and Globally
Background: Acute respiratory infections kill an estimated 3.9 million people annually
and in developing countries are the leading cause of mortality in children under 5 years
of age. Specific respiratory virus infections such as influenza, SARS-CoV-2, and
respiratory syncytial virus, are major contributors to this burden of disease, as are
other respiratory bacterial and viral pathogens. Respiratory virus infections are
frequent events in all age groups and impose a substantial burden on social and
healthcare delivery systems.
Specific Research Areas of Interest: The goal of this research includes, but is not limited to,
activities that support the development and evaluation of tools for: 1) the prevention of acute
respiratory infections such as pneumonia, influenza, COVID-19, and Legionnaire’s disease, 2) rapid
recognition and containment of outbreaks, and 3) advanced diagnostic technologies including point-
of-care testing, advanced molecular detection, and whole genome sequencing.
Impact: This research will lead to the development of practical solutions for the prevention and
diagnosis of vaccine preventable diseases that have a substantial impact on the economy, health,
and wellbeing of society. The goal of the research supported through this mechanism is expected to
begin shifting viral and bacterial infections from common occurrences to rare events and to reduce
the disproportionate burden of COVID-19 on populations at increased risk for infection, severe
illness, and death.
Commercialization Potential: The innovative technologies and solutions developed through this
program will make it possible to improve the public health and healthcare system’s response in a
variety of settings, thus making the commercialization potential unlimited.
For NCIRD programmatic information, contact:
Amy Yang, PhD
Extramural Research Program Office
Office of the Associate Director for Science
National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention
Centers for Disease Control and Prevention (CDC)
Phone: 404-718-8836
Email: VDZ9@cdc.gov
For grants specific, administrative information,
contact:
Angie Willard, Team Lead
Office of Grant Services Office of Financial
Resources Office of the Chief Operating Officer
Centers for Disease Control and Prevention (CDC)
Mail Stop TV-2
2939 Flowers
Road NE Atlanta,
GA 30341
Phone: 770-498-2596
Email: AEN4@cdc.gov

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 181
NATIONAL CENTER FOR INJURY PREVENTION AND CONTROL (NCIPC)
Mission and Research Areas of Interest
For 30 years, CDC’s National Center for Injury Prevention and Control (NCIPC) has helped protect
Americans from injuries and violence. We are the nation’s leading authority on injury and violence.
We study violence and injuries and research the best ways to prevent them, applying science and
creating real-world solutions to keep people safe, healthy, and productive. NCIPC will prioritize
funding meritorious applications that address the NCIPC program topics listed in this program
announcement. NCIPC may also consider meritorious applications that address current NCIPC
research priorities.
To learn more about NCIPC, please visit the NCIPC website at: http://www.cdc.gov/injury/index.html.
Description of Research Areas of Interest
NCIPC encourages submission of research applications with innovative research technologies
designed to reduce health disparities and promote health equity.
The major research priorities areas for the NCIPC are listed below. Investigator initiated applications
that have commercial potential that are outside these topic areas but within the research mission of
NCIPC will also be considered through this solicitation. Research to address emerging health
threats, health disparities, and health equity are strongly encouraged.
To learn more about NCIPC research priorities, please visit our web site at:
https://www.cdc.gov/injury/researchpriorities/index.html.
(1)
Prevention and Management, and Recovery of Traumatic Brain Injury
Background: There were nearly 70,000 traumatic brain injury (TBI)-related deaths in 2021 and more
than 200,000 TBI-related hospitalizations. TBIs affect how the brain works and can range from mild
concussions to serious and debilitating injuries with long-term effects. They are caused by a bump, blow,
or jolt to the head or a penetrating injury (e.g., a gunshot) and are often the result of falls, firearms, motor
vehicle crashes, or assaults. While TBIs affect individuals of all ages, research suggests that some
individuals may be at greater risk for experiencing a TBI and more severe consequences of a TBI.
Individuals at higher risk often include racial and ethnic minorities, service members, people living in rural
communities, and survivors of intimate partner violence, among others.
The trajectory of recovery from a TBI is influenced by multiple factors, including the overall severity of the
injury, number of prior TBIs, and receiving an appropriate diagnosis, referrals, and tailored care.
Individuals are generally eager to return to prior activities following a TBI. However, there can be
considerable complexity in decision-making around when to return to school, work, physical activity,
driving, or other daily tasks. Careful monitoring and tracking of symptoms can help inform this dec ision-
making, as well as having access to easy-to-use evidence-based resources and supports to guide one’s
recovery path. Additionally, due to symptoms experienced and uncertainty around recovery timeline, TBI
recovery can be particularly challenging from not only a physical health perspective but also a social,
emotional, and mental health perspective (e.g., anxiety, sadness). Therefore, ensuring that strong, active
support networks exist for individuals with TBIs is a critical component of the recovery process.
Project Goal: CDC is interested in research to develop technology that can assist persons with a TBI in
better assessing when they can return to normal activities, including school, work, physical activity,
driving, and/or other daily activities. The technology can help improve understanding and confidence
among individuals with a TBI in tracking their path to recovery, as well as preventing future TBI.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 182
The technology could aid those who have experienced a TBI by helping them better understand what to
expect, track their symptoms throughout their recovery to help inform decision-making around returning to
activities, access recommendations each step of the way to guide them towards recovery and
reintegration in activities, and access support networks or linkage to support networks to help manage
social, emotional, and mental health during recovery. Knowledge of what to track and look for at each
step of the recovery process, support tips and information to help guide individuals (including
consideration of when to transition from rest to light, moderate, or full activity, how to handle recovery
delays, and when to seek additional healthcare), and connections to active supports and communities to
ensure care of not only physical but also social and emotional health during the recovery period are
critical needs. The technology can also help prevent future, repeated TBI by providing individuals with
information and tools centered around TBI risks and effective prevention approaches.
Given disparities in TBI, the technology should consider tailoring and framing to populations at highest
risk. These populations could include, but are not limited to, military service members and veterans, racial
and ethnic minorities, people living in rural communities, survivors of intimate partner violence, and
people of lower incomes and without access to healthcare.
For Phase I, the awardee is expected to develop, and beta test new technology designed to support
individuals on their road to recovery from a TBI. The technology should be intuitive and easy to use.
Impact and Commercialization Potential: The availability of technology that can ease one’s road to
recovery from a TBI can help improve recovery timelines and improve quality of life and social, emotional,
and mental health. Such technology can also help individuals better understand, track, manage, and
recover from their symptoms and reintegrate into normal activities, including school, work, physical
activity, driving, and other daily activities. Additionally, the availability of technology could assist in
reducing health disparities among vulnerable populations who tend to have worse health outcomes (a
greater likelihood of dying from a TBI or living with long-term problems that resulted from the injury).
Development of this technology has commercial viability. Commercial applications of this technology may
be of interest to adolescents, adults, caregivers, healthcare providers, health insurance companies, and
youth sports personnel, among others.
(2)
Technological Innovations to Reduce Deaths and Injuries from Motor Vehicle Crashes
Background: Motor vehicle crashes are a leading cause of death among those aged 1-54 years in
the United States, killing over 40,000 people every year and injuring 3 million more. Motor vehicle
crashes can result from a single or combination of environmental, human behavioral, and vehicle-
related risk factors including hazardous road conditions, driving too fast for the environment, driver
perception deficits, non-compliance with vehicle safety devices, impaired driving, lack of seat belt
use, lack of helmet use, distracted or drowsy driving, and sub-optimal vehicle performance.
Reducing any of these risk factors can lower the likelihood of a crash and increase the chance of
survival in the event of a crash.
Adaptive technologies can generate feedback loops about the road and environment, driver fitness,
and vehicle performance. Applications of these adaptive technologies in both private and commercial
vehicles can reduce risks associated with motor vehicle crashes. Currently, there are a limited
number of adaptive technologies to warn drivers of potential dangers associated with driving; most of
these technologies focus on vehicle related performance (e.g., collision warning, electronic stability
control, and lane departure warning). Innovative adaptive technologies are sought that can assist in
alerting drivers to risks associated with the road or environment and vehicle performance, and that
can facilitate drivers to modify personal risk behavior, including impaired or drowsy driving. These
adaptive technologies can result in the development of tools or systems that will reduce the
likelihood and severity of motor vehicle crashes and assist drivers in making potentially life-saving
decisions more quickly and more intuitively.
Project Goal: CDC is interested in research to address the development of improved

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 183
technologies that have the potential to further reduce motor vehicle crashes and resulting
injuries to all road users.
These technologies can address driver risks such as excessive speed, distracted driving,
impaired driving, drowsy driving, non-compliance with use of vehicle safety equipment (e.g., seat
belts), environmental conditions (including road quality), vehicle performance and other factors
that may impact driving. Technologies could also address risks to individuals walking, biking,
riding, or rolling, such as crowdsourcing information on unsafe infrastructure (e.g., sidewalk gaps,
unsafe walking surfaces and compliance with the Americans with Disabilities Act). For Phase I,
the awardee is expected to develop and pilot test new technology designed to reduce motor
vehicle crashes and resulting injuries. Testing could also consider and explore acceptability of the
technology to road users.
Impact and Commercialization Potential: The availability of technologies to reduce motor vehicle
crashes and resulting injuries can save lives. Technologies that reduce motor vehicle crashes and
resulting injuries can help improve health equity and reduce injury disparities among vulnerable road
users. Development of these technologies has commercial viability. Commercial applications of this
technology might be of interest to motor vehicle manufacturers, insurance companies, clinicians, health
systems, departments of transportation, parents of young drivers, and all road users.
(3)
Electronic Tools to Assist Older Adults at Risk for Falls
Background: Unintentional falls are the leading cause of fatal and nonfatal injuries in older
adults aged 65 years and older. Falls result in 3 million emergency department visits and more
than 900,000 hospitalizations each year. About 38,000 older adult deaths occur each year
because of a fall. This risk of falls and their consequences is not equally distributed across the
population. Research indicates that fall risk is highest among adults of older ages (e.g., 80+),
American Indian/Alaska Native and White non-Hispanic populations, individuals with numerous
chronic health conditions (e.g., depression, diabetes, cardiovascular disease, stroke), and those
with neurological disorders (e.g., dementia, Parkinson’s disease). Falls often lead to reduced
mobility and loss of independence; therefore, reducing fall risk is conducive to maintaining
independence.
An example of a resource for information about falls prevention is CDC’s Stopping Elderly
Accidents, Deaths, and Injuries (STEADI) initiative. CDC developed the STEADI initiative based
on the American and British Geriatric Societies Guideline for the Prevention of Falls in Older
Persons to provide training, resources, and tools for healthcare providers to use to incorporate
older adult fall prevention into routine clinical care. STEADI has three core components –
SCREEN older adults at least annually to determine who is at risk for a fall, ASSESS those at
risk to determine their modifiable risk factors, and INTERVENE by recommending evidence-
based strategies for each identified risk factor. Modifiable risk factors (and associated strategies
to address them) include strength and balance limitations (physical therapy); medications with
side effects that increase risk (medication management); vision problems (vision exam), foot
problems (podiatrist exam), orthostatic blood pressure (medication management or referral to a
specialist); home hazards (home assessment with an occupational therapist), and comorbidities
(medication management or referral to a specialist).
A fourth potential core component is patient FOLLOW-UP. Older adults are more likely to follow
these recommendations with continuous follow up from their healthcare provider and when
receiving reminders and continuous ongoing support (e.g., from their network of support persons
and/or caregivers). Older adults are also more likely to follow recommended fall prevention
activities when the activities are affordable and easily accessible. Finally, recommendations
utilizing a positive framing regarding preserving mobility and thriving as we age can be
particularly effective (e.g., CDC’s “Still Going Strong” campaign). Technologies targeted at older
adult healthcare providers, as well as older adults and their families, caregivers, and support
persons may help facilitate long-term uptake of interventions and strategies to prevent falls and

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 184
maintain mobility within a positive framing.
Project Goal: CDC is interested in research to develop technology that will help healthcare providers
incorporate STEADI-based fall prevention into their clinical practice and/or support older adults and
their families, caregivers, or support persons in understanding and supporting active implementation
of recommended strategies to reduce older adults’ fall risk, maintain mobility, and support healthy
aging.
One way this can be accomplished is through technology that incorporates elements of an evidence-
based program, such as those described in CDC’s STEADI tools and resources and inspired by a
positive, optimal mobility approach such as those used in CDC’s Still Going Strong campaign or
CDC’s MyMobility Plan tools. For example, the technology could support healthcare providers’ use of
the STEADI algorithm that guides them through the three core components.
Alternatively, the technology could integrate STEADI, Still Going Strong, MyMobility Plan, or other
patient education materials and resources to educate older adults and their families, caregivers, and
support persons about falls and how to prevent them, maintaining strength and health as one ages,
and adhering to fall prevention recommendations.
Given disparities in falls risk, the technology should consider tailoring and framing to populations at
highest risk, as noted above. For Phase I, the awardee is expected develop and beta test the
technology. The technology should incorporate applicable healthcare information privacy regulations,
be intuitive and easy to use by the target audience, and include an interface designed with the target
audience in mind.
Impact and Commercialization Potential: The availability of a technological tool to assist clinicians or
older adults, their families, and support persons in preventing falls and maintaining mobility can help
ensure that healthcare provider recommendations and evidence-based strategies are followed. By
facilitating fidelity to recommendations and improving communication about fall risk, healthy aging, and
prevention between older adults, families, and providers the expected public health benefit is a reduction
in older adult falls and increased support for healthy aging.
Development of a technological tool that reduces risk for older adult falls has commercial viability.
Commercial applications of this technology may be of interest to older adults and their families,
healthcare providers, community support staff, health insurance companies, and other stakeholders
invested in preventing older adult falls and supporting healthy aging.
(4)
Innovative Technologies to Help Prevent Drug Overdose
Background: The national drug overdose crisis continues to evolve in the United States, causing
increasing numbers of deaths and warranting innovative interventions. In 2022, over 107,000
people died from drug overdoses and 76% of those deaths involved opioids. Over 26% of
overdose deaths in 2022 involved cocaine, and almost one-third (32%) involved psychostimulants
with abuse potential (e.g., methamphetamine). Further, recent data suggest that overdoses
involving multiple substances are increasing. Given the evolving nature of the overdose crisis,
there is a need for innovative technological approaches to support the prevention of drug
overdose. These technologies can focus on: 1) using data to inform action (i.e., linking various
data platforms, including those that monitor and track nonfatal or fatal drug overdoses or
emerging drug threats to inform overdose prevention and response efforts; 2) developing or
implementing digital interventions to deliver drug overdose prevention); 3) helping people in need
of substance use treatment or harm reduction services identify and access services; and 4)
developing and implementing applications or platforms to monitor the provision of care and harm
reduction services or materials or track usage and/or outcomes.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 185
Innovative solutions are needed to support new opportunities to effectively leverage technology
and data systems to improve drug overdose prevention and response efforts, link and retain
people who use drugs to care and harm reduction services and monitor the provision of harm
reduction services or materials and track usage and outcomes of these services. To support
these objectives, we are soliciting proposals to create innovative and user-friendly, electronic
tools, applications, or technologies that address at least one of the following prevention activities:
1.
Using data to inform action: Aggregate and synthesize information from
various data sources that track nonfatal or fatal drug overdoses or document
emerging drug threats (e.g., toxicology or drug seizure data) to inform and
implement overdose prevention and response efforts (e.g., using data to
identify geographical “hotspots” that can benefit from naloxone distribution,
fentanyl test strips, or overdose prevention campaigns).
2.
Develop or implement digital drug overdose prevention: Digitally deliver
overdose prevention materials (e.g., information on how to recognize signs and
symptoms of drug overdose, how to respond to a suspected drug overdose,
where to obtain naloxone) using innovative technology.
3.
Helping people in need of substance use treatment or harm reduction services
identify and access services: Develop or implement a mechanism for people
who use drugs (PWUD) and their family or friends to identify and access local
substance use disorder (SUD) treatment or harm reduction support services.
This could include developing an application or tool to help identify providers
based on geography or services provided or help identify and remedy barriers
to accessing services (e.g., transportation, childcare services, etc.).
4.
Developing and implementing applications or platforms to monitor the provision
of care and harm reduction services or materials or to track usage and/or
outcomes: To better monitor the provision of care and harm reduction services
or materials to clients, develop an innovative solution to monitor metrics on
service delivery and uptake or to track usage and/or outcomes (e.g., linkage
and retention or use of services).
References:
1.
Spencer MR, Garnett MF, Miniño AM. Drug overdose deaths in the United States,
2002–2022. NCHS data brief, no 491. Hyattsville, MD: National Center for Health
Statistics. 2024.
Specific Area of Interest/Project Goal(s): CDC is interested in research that aims to develop innovative
technologies that will leverage data to inform overdose prevention, develop or implement digital overdose
prevention materials, link people who use substances to effective substance use treatment or harm reduction
services, and develop and implement strategies to monitor the provision of care and harm reduction services
and track usage and/or outcomes of these services. The technology should use information relevant in local
communities. For example, the technology could tailor available services options including by location and
current capacity and automate referral processes to local services to provide PWUD a seamless process for
finding, contacting, and accessing services. Other examples include incorporating current characteristics of
the local epidemic into linkage messaging or a menu of locally available recovery support services such as
transportation, employment services, or housing support. Tracking usage and/or outcomes could help
enhance the technology over time by compiling data regarding most used services, gaps in available
services, reason services are sought, and outcomes of specific services.
The technology should reflect feasibility and acceptability for the population it is intended to serve: PWUD.
Therefore, the technology should be accessible for PWUD in the local area.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 186
The expected research outcome is the development of technology that will ultimately better track and
respond to overdoses. Projects will help tailor and deliver prevention and response efforts, connect PWUD to
locally available SUD treatment, harm reduction, and recovery support services, or help service providers
(e.g., SUD treatment providers or harm reduction services) monitor the provision or uptake or services or
track usage and/or outcomes (e.g., linkage and retention in care).
For Phase I awards, the recipient is expected to develop and beta test the new technology(ies) with the
relevant population (e.g., PWUD and at least one type of local service: SUD treatment, hard reduction
services, recovery support services). Recipients must attend to appropriate protections related to
confidentiality, personally identifiable information, health information, and SUD privacy regulations
associated with these technologies, such as those that apply to sharing protected heath data across systems
or any stigma associated with using the technology. The technology must be intuitive and easy to use.
Impact: The availability of technological tools to assist those at risk for drug overdose within
communities can help better implement effective overdose prevention and response, and ensure
that needed referrals, services, and follow-up care are received and that linkages are made in a
timely manner to effectively prevent immediate risks. By improving access to medical and non-
medical support services and near real-time data, the expected public health benefit is reduced
potential for drug overdose, and increased linkage to SUD treatment, harm reduction services, and
recovery support services that can reduce the risk of drug overdose, and a more accurate
understanding of services delivered.
Some populations, communities, and geographic areas are disproportionately impacted by
overdose and can be considered for tailored programs and interventions based on associated risk
for overdose or disparities in access to harm reduction services and SUD treatment. Further,
having a SUD is a chronic health condition, yet individuals with SUD face barriers to accessing the
care they need due to stigma and other factors. Moreover, PWUD may be more likely to have
intersecting characteristics (e.g., experiencing homelessness, being involved in the justice system)
that may be associated with poor health outcomes (such as overdose death), either by having an
increased likelihood of drug use or a reduced likelihood of receiving care if they develop an SUD.
Such characteristics may also impact accessibility to some technologies, and solutions must
incorporate meeting PWUD where they are to have the most reach. The proposed technologies
have the potential to reduce barriers to receiving overdose prevention and care among people who
use drugs, including those who have a SUD, thereby reducing health disparities among this
important population. Research suggests that some populations may have a greater risk for
experiencing disproportionate burden of drug overdose or experiencing adverse outcomes related
to drug use due to social determinants of health, indicating a potential opportunity to intervene and
support such populations. The development of innovative technologies for overdose prevention or
to support linkage to care among PWUD who experience a disproportionate burden of overdose or
experience adverse outcomes related to drug use can help prevent drug overdose and
simultaneously improve health equity.
The availability of technological tools to assist those at risk for drug overdose and those working to
prevent and respond to overdose within communities can better ensure that needed referrals,
services, and follow-up care are received and that information is shared in a timely manner to
effectively prevent immediate risks. By improving access to medical and non-medical support
services and near real-time data, the expected public health benefit is increased linkage to support
services, harm reduction services, and/or data and resources that can reduce the risk of drug
overdose.
Some populations, communities, and geographic areas are disproportionately impacted by overdose
and can be considered for tailored programs and interventions based on associated risk for
overdose or disparities in access to harm reduction services and substance use treatment. Further,
having a substance use disorder (SUD) is a chronic health condition, yet individuals with SUD face
barriers to accessing the care they need due to stigma and other factors. Moreover, PWUD may be
more likely to have intersecting characteristics (e.g., experiencing homelessness, being involved in

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 187
the justice system) that may be associated with poor health outcomes (such as overdose death),
either by having an increased likelihood of drug use or a reduced likelihood of receiving care if they
develop an SUD.
The proposed technologies have the potential to reduce barriers to receiving care among people
who use drugs, including those who have a SUD, thereby reducing health disparities among this
important population. The development of innovative technologies to support linkage to care among
PWUD and population subsets of PWUD (e.g., persons who have previously experienced an
overdose; persons experiencing homelessness; justice-involved populations recently released from
incarceration; those disadvantaged by economic instability, limited education attainment, access,
and quality, and/or limited health care access and quality), can help prevent drug overdose and
simultaneously improve health equity.
Commercialization Potential: Commercial applications include technological tools developed, for
example to link people at risk for drug overdose to treatment or other services. Tools to create
connections and opportunities for data and resource sharing between PWUD and relevant services
also have commercial viability. Commercial applications of this technology may be of interest to those
at risk for overdose and their families, community support programs and staff, health insurance
companies, SUD treatment providers, harm reduction providers and other stakeholders invested in
preventing drug overdose.
(5)
Innovative Technology or Media to Prevent Violence Affecting Children/Youth
Background: Violence is a significant public health problem in the United States. In 2021, more
than 26,000 people died from homicide. Far more people experienced nonfatal violence. For
example, more than 1.4 million people were treated for nonfatal injuries from assaults in U.S.
emergency departments in 2021. Experiencing violence has a profound impact on lifelong health,
opportunity, and well-being. Violence starts early in life. In 2021, there were an estimated 4.0 million
referrals to child protective services for child abuse or neglect involving an estimated 7.2 million
children. In addition to child abuse and neglect, other forms of violence impacting children and youth,
include sexual violence, teen dating violence, youth peer violence, youth/parent suicidal behavior,
and exposure to adult intimate partner violence.
Adverse Childhood Experiences (ACEs) are potentially traumatic events that occur in childhood (0-
17 years). ACEs include experiencing violence, abuse, or neglect; witnessing violence in the home
or community; or having a family member attempt or die by suicide. Also included are aspects of the
child’s environment that can undermine their sense of safety, stability, and bonding such as growing
up in a household with substance use, mental health problems, or instability due to parental
separation or household members being in jail or prison. Common risk factors for ACEs and violence
can also start in early childhood and continue throughout the lifespan. They go beyond individual-
level factors to include family and peer relationships and other influences from schools, the
community, and society. Social determinants of health are the circumstances in which people are
born, grow up, live, work and age, and the systems put in place to deal with illness. These
circumstances are in turn shaped by a wider set of forces: economics, social policies, and politics
(see https://www.who.int/health-topics/social-determinants-of-health#tab=tab_1). Social
determinants of health (e.g., concentrated poverty, structural racism, high rates of unemployment
and community violence, limited access to high-quality education and/or limited access to affordable,
high-quality childcare) are key drivers of health inequities among communities of color and place
them at a greater risk for experiencing violence.
The focus of CDC’s violence prevention work includes the following principles: 1. Advancing
economic, gender, and racial equity, 2. Enhancing positive relationships and environments, 3.
Addressing factors that cut across multiple forms of violence, and 4. Prioritizing efforts that
create societal- and community-level impact.
By focusing on activities that prevent multiple forms of violence, communities can achieve the greatest
impact and increase scalability of their prevention strategies. Additionally, these prevention efforts are ideally

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 188
designed to use resources more effectively and to better address inequities by focusing on the needs of
populations at greatest risk. To help communities make decisions about violence prevention strategies, CDC
has released a series of Resources for Action documents that describe the best available evidence for
violence prevention (see https://www.cdc.gov/violenceprevention/communicationresources/pub/resource-for-
action.html). Many of the strategies in the Resources for Action documents are relevant to multiple forms of
violence, including multiple ACEs (see https://www.cdc.gov/violenceprevention/pdf/ACEs-Prevention-
Resource_508.pdf for strategies drawn from the Resources for Action documents that are relevant to
ACEs).
Broader benefits could be achieved from wider dissemination of effective violence prevention
strategies through innovative media and communication technology (e.g., mobile applications, social
media, games, and Internet-based interventions). Media and communication technology also create
the opportunity for the development of new prevention approaches based on what is known about
violence risk and protective factors and strategies that work in traditional settings. Innovative media
and communication technology can play an important role in effectively reaching populations at
greatest risk and potentially facilitate changes at multiple levels (individual, relationship, community);
however, research is needed to guide the development of technological applications for prevention
strategies.
Project Goal: CDC is interested in research to develop innovative technology or media, such as
applications for mobile devices, social media, games, or Internet-based interventions to prevent
multiple forms of interpersonal violence (e.g., child abuse and neglect, youth violence, sexual
violence, and intimate partner violence) and other ACEs affecting children or youth, particularly
among groups or communities that that are disproportionately impacted by multiple forms of
violence and poor social determinants of health (see
https://www.cdc.gov/injury/researchpriorities/index.html).
This includes, but is not limited to, new media and communication technology to do the following:
increase the accessibility of prevention approaches, modify norms about violence and bystander
behavior, enhance education and support for young children and their families, reduce stigma
and barriers to help seeking, and/or enhance young people’s skills and relationships to reduce
risk for multiple forms of violence. Additionally, CDC is interested in new media and
communication technology that could increase the adoption of community-level violence
prevention strategies and strategies that improve social determinants of health in communities
disproportionately affected by violence (e.g., housing stability, food security, education and
employment opportunities, access to quality and affordable healthcare, etc.).
The widespread use of smartphone applications, social media, and wearable technology also
provides unique opportunities for the broader dissemination, implementation, and evaluation of
evidence-base prevention strategies to significantly reduce violence, such as strategies identified in
the violence prevention technical packages used in real world settings. Applicants are encouraged
to develop technology or media that could help address poor social determinants of health that
contribute to inequities in rates of violence experienced by racial/ethnic and other groups or
communities that are disproportionately impacted by multiple forms of violence, ACEs, and/or
associated risk factors.
The prototype (e.g., developing innovative technology or media) should be informed by prior
research about violence risk and protective factors and/or evidence-based prevention strategies
and through consultation with subject matter experts in the form(s) of violence and the technology
or media selected. The awardee should describe the following: the intended audience and the
type(s) of violence addressed; goals for the product at the individual, family/relationship and/or
community level(s); the process through which the technology or media is expected to work and
the measurements and key performance indicators for tracking progress toward the goals; the
expected impacts on violence and violence-related inequities; the functionality and actions for
users to take; the estimated costs and logistics of scalability; a description of potential barriers to
implementation; and any evidence for the potential benef its from prior research.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 189
Impact: The results from this research will have substantial implications for either the creation of
innovative prevention approaches or for enhanced opportunities to disseminate existing
evidence- based strategies, both of which have the potential to leverage technology to improve
social determinants of health, increase health equity, and substantially reduce multiple forms of
violence (e.g., child abuse and neglect, youth violence, sexual violence, and intimate partner
violence) and other ACEs. Technological or media innovations that show effectiveness in
preventing violence affecting children and youth and communities and groups disproportionately
impacted by violence would have the potential for a range of commercial applications.
Commercialization Potential: Depending on the nature of the strategy, the intended audience, and
the costs/logistics of scalability, the product could be in demand by healthcare systems, school
systems, colleges and universities, youth serving organizations, law enforcement, public health
agencies, community groups and organizations, parents, and their children.
Visit the NCIPC homepage for more information on NCIPC’s research program areas at
http://www.cdc.gov/injury/index.html.
For NCIPC programmatic information, contact:
Tonia Linley
Program Specialist
National Center for Injury Prevention and Control
Centers for Disease Control and Prevention CDC
Phone: 770-488-3593
Email: IMX9@cdc.gov or NCIPC_ERPO@cdc.gov
For grants specific, administrative information contact:
Angie Willard, Team Lead
Office of Grant Services
Office of Financial Resources
Office of the Chief Operating Officer
Centers for Disease Control and Prevention (CDC)
Mail Stop TV-2
2939 Flowers Road NE Atlanta, GA 30341
Phone: 770-498-2596
Email: AEN4@cdc.gov
NATIONAL INSTITUTE FOR OCCUPATIONAL SAFETY AND HEALTH (NIOSH)
Mission and Research Areas of Interest
The National Institute for Occupational Safety and Health (NIOSH) is part of the U.S. Centers for
Disease Control and Prevention (CDC). It has the mandate to assure “every man and woman in the
Nation safe and healthful working conditions and to preserve our human resources.” NIOSH has more
than 1,500 employees from a diverse set of fields including epidemiology, medicine, nursing, industrial
hygiene, safety, psychology, chemistry, statistics, economics, and many branches of engineering.
NIOSH works closely with the Occupational Safety and Health Administration (OSHA) and the Mine
Safety and Health Administration in the U.S. Department of Labor to protect American workers.[=
For additional information about NIOSH, please visit their web site at:
http://www.cdc.gov/niosh/programs. The major research priority areas for NIOSH are listed below.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 190
Investigator initiated applications that have commercial potential that fall outside these topic areas but
fall within the research mission of the NIOSH will also be considered through this Omnibus solicitation.
Research to address emerging health threats, health disparities, and health equity are strongly
encouraged.
NIOSH typically supports Phase I awards at the maximum allowable total cost as stated in the funding
announcement. However, in most cases Phase II applications are limited to a maximum total cost of
$1M for the two-year period of performance.
(1)
Control Technology and Personal Protective Equipment for High-Risk Occupations
Background: Personal protective equipment (PPE) protects workers from death and disabling
injuries and illnesses as well as from the specific threats of exposures to certain airborne biological
particles, chemical agents, nanomaterials, splashes, noise exposures, fall hazards, head hazards,
and fires. It is estimated that 20 million workers use PPE on a regular basis to protect them from job
hazards and a total of 135,000 workers potentially could benefit from the use of PPE (Worker Health
Chartbook 2004).
Engineering controls include substitution of a safe material for a hazardous one, design changes
to equipment, or modification of work methods to eliminate or reduce hazards. Research is needed
to develop and evaluate control strategies and personal protective equipment for specific hazards
and to assure their practicality and usability in workplaces in all of the high-risk industrial sectors.
For additional information about NIOSH PPE and Engineering control programs, please visit
their web site at: http://www.cdc.gov/niosh/programs/ppt/ and
http://www.cdc.gov/niosh/programs/eng/.
Examples of specific research areas of interest include, but are not limited to:
Conduct research on the ability of existing containment and control strategies to prevent releases and
potential human exposures to engineered nanomaterials.
Develop innovative engineering control approaches and technologies for reducing respirable
crystalline silica exposures for workers involved in manufacturing, finishing, and installing natural and
engineered stone countertops.
Research should be conducted to reproduce the original human performance data using modern
advanced technology and a pool of subjects that represents the current American workforce.
Additionally, the scope of the subject pool can be expanded to encompass all workers. The
generated data would be used to confirm or adjust the parameters used for respirator approval
testing, as well as aid in the development of respirator standards used for all workers including
younger (children) workers in high risk occupations.
Conduct research to evaluate the effectiveness of personal protective equipment in protecting
workers against exposure to engineered nanomaterials. Provide data to fill knowledge gaps and
support guidance for the selection and use of gloves and protective garments to prevent exposures.
Respiratory protection research needs to be extended to a broad range of engineered nanomaterials.
Develop a heads-up display coupled with a personal noise exposure monitoring system. Personal
noise alert “badges” and personal noise dosimeters exist, but do not have an effective way to alert
the user immediately when a noise hazard occurs. A system that displays a warning within the user’s
visual field (via lights on protective eyewear, hardhat, etc.) would facilitate hazard recognition.
Develop an inexpensive hand-held earplug test device based on the NIOSH QuickFit concept.
Studies of hearing protector users have shown repeatedly that average protection values are much

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 191
lower than the labeled Noise Reduction Ratings (NRR) determined in laboratories. A QuickFit test
system would help workers determine if their hearing protection is giving them at least 15 decibels of
attenuation.
Develop innovative engineering control approaches and technologies for reducing asphalt exposures
in roofing, and skin exposures and disease in construction workers.
Conduct research to understand PPE integration and interoperability issues. In most cases, individual
PPE are currently used without consideration for their ability to function together. Research is needed
to test interfaces among different PPE and components. Current interfaces do not provide seamless
integration of PPE components resulting in reduced comfort, fit, usability, and protection for the
wearer as well as logistical challenges for safety managers and employers.
Develop innovative educational and professional training materials suitable for today’s diverse
workplace on the role of PPE in occupational safety and health. This is especially critical for high-risk
occupations. Innovative methodologies, including social media, should be explored and evaluated to
demonstrate their effectiveness at improving workplace safety and health. For example, to what
extent can mobile application media be focused on worker safety and health to provide up-to-date
PPE information to a diverse range of employers and employees through portable communication
devices?
Conduct research on the ability of existing containment and control strategies to prevent releases and
potential human exposures to engineered nanomaterials.
Impact and Commercialization Potential: The impact of the proposed research will prevent work-
related injury, illness, and death by advancing the state of knowledge and application of personal
protective equipment. Potential products include technical methods, processes, techniques, tools, and
materials that support the development and use of personal protective equipment worn by individuals to
reduce the effects of their exposure to a hazard.
(2)
Exposure Assessment Methods for High-Risk Occupations
Background: Exposure assessment provides multi-disciplinary strategies and methods to anticipate,
recognize, evaluate, control, and confirm effective management of occupational health stressors,
exposures to those stressors, and resulting health risks. Major gaps in current approaches include:
(1) the lack of practical methods for hazard identification and measurement that can be applied at
reasonable cost in many workplaces where health stressors may exist, (2) the lack of validated,
noninvasive biological methods for monitoring relevant exposure and resulting dose, and (3) the lack
of strategies and methods for epidemiologic studies to demonstrate either a dose-response effect or
a conclusion of no association between the agent and disease in the complex environments of
today's workplaces.
For additional information about NIOSH Exposure Assessment programs, please visit their web site at:
https://www.cdc.gov/niosh/programs/exap/default.html.
Examples of specific research areas of interest include, but are not limited to:
Two areas of research are needed to support effective assessment of worker exposure to engineered
nanomaterials. 1) Real-time sensors capable of reliably detecting nanoparticles and providing
information on size distribution and count, that can be used for personal monitoring; and 2)
Development of methods that can detect and quantify the presence of engineered nanomaterials in
samples collected for the purpose of characterizing exposures. These methods need to be cost-
effective and available to the OS&H practitioner community. Broader application to general public

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 192
health assessments should be factored into the research.
Develop new or improved methods to measure occupational health stressors such as psychological
and ergonomic factors, noise, chemicals, particles and fibers, physical agents, non-ionizing radiation,
or mixtures of stressors in the work environment. Enhanced measurement performance and
functionality can include sensitivity, selectivity, size and weight considerations, ease of use, and
capabilities to measure multiple analyses simultaneously.
Develop or adapt easy-to-use, direct-reading instruments and test kits to rapidly and inexpensively
measure exposures in a variety of workplaces. Critical applications include routine monitoring,
evaluating the success of control technologies, and supporting epidemiological studies.
Develop innovative bioaerosol samplers for collecting airborne infectious viruses onto a medium
without compromising viability or infectivity to allow exposure assessments in occupational settings.
Improve the measurement of low concentrations of chemicals and biomarkers in biological specimens
such as blood, urine, saliva, and sweat so that such concentrations can be linked to internal dose at the
intended organs, work tasks and workers can be categorized according to hazard bands and exposure
bands, and at-risk workers can be identified and protected.
Develop methods to assess mixed exposures such as chemical mixtures containing gas and particulate
matter. Develop improved understanding of dermal exposure’s contribution to exposure assessment.
Develop and apply emerging technologies including Artificial Intelligence (AI) models that support and
enhance exposure assessment data collection and analysis, particularly involving 'smart' exposure
sensors, that could inform control measures.
Develop a computerized system that can be used to predict worker noise exposure from mining
machine noise emissions. The system would include an acoustic model of mining environments and
algorithms to characterize exposure based on noise source characteristics. The main application for
this technology would be for mining machine manufacturers to evaluate the potential effects of noise
controls during the design process. If the impact of design changes on exposure reduction can be
accurately predicted without the need for extensive field measurements, innovative noise controls can
reach implementation much more quickly.
Impact and Commercialization Potential: This research will lead to the development of practical
solutions and prevention activities to address complex problems that cause occupational diseases,
injuries, and fatalities and that will lead to reductions in occupational injuries and illnesses among all
workers. This research will lead to the development and translation of exposure assessment methods
and research findings into prevention practices and products that will be adopted in occupational
settings. Potential products include technical methods, processes, techniques, tools, and materials that
support the assessment of exposure to physical, chemical, and biological hazards in the work
environment.
(3)
Work-related Injuries from Motor Vehicle Crashes
Background: The risk of injury associated with on-the-job operation of motor vehicles affects millions of
U.S. workers who work in all industries and drive all types of vehicles, and for whom driving may be a
primary or incidental job task. Motor vehicle crashes are consistently the leading cause of work-related
fatalities in the United States. From 2011 through 2022, the Bureau of Labor Statistics reported 21,046
work-related fatalities due to motor vehicle crashes, about 35% of all fatalities at work. Over the same
period, workers incurred nearly 515,000 lost-workday injuries due to these crashes. Crash-related fatalities
and serious injuries have a devastating impact on workers and their families, and on the economic health
and productivity of American businesses. Work vehicles such as large trucks also have an impact of the
safety of the motoring public.

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 193
The NIOSH Center for Motor Vehicle Safety coordinates the CDC/NIOSH response to this pressing
worker safety issue. Many NIOSH programs include motor vehicle crashes among their top injury
prevention priorities: Traumatic Injury; Transportation, Warehousing, and Utilities; Wholesale and
Retail Trade; Oil and Gas Extraction; and Public Safety.
Examples of specific research areas of interest include, but are not limited to:
The highest priority is to develop, implement, and evaluate interventions in an effort to build the scientific
evidence base to prevent work-related motor vehicle crashes and resulting injuries. This may be achieved
by:
Developing and testing new design concepts and applications with potential for commercialization and
diffusion to employers and fleet managers.
Developing and testing novel approaches for driver training and assessment to reduce work-related motor
vehicle crashes, including training on the operation of vehicles with Advanced Driver Assistance Systems
(ADAS) or other forms of automation.
Developing and evaluating the effectiveness of technology- or management-based intervention strategies to
reduce the incidence or severity of work-related motor vehicle crashes.
Developing and evaluating engineering controls for preventing work-related crashes and injuries, with
emphasis on specialized work vehicles such as large trucks and fire apparatus.
Developing or evaluating an intervention designed to prevent crashes and injuries for a disproportionally
affected group, such as roadside assistance workers or pedestrian workers engaged in construction or
maintenance of roadways.
Developing and evaluating an easy-to-use computerized system based on readily available technology that
can automate a “fatigue detection” system capable of warning the employee driver and supervisor when the
driver may be at risk for a work-related motor vehicle crash. The system would include a statistical algorithm
capable of using Global Positioning System (GPS) data from cellular phones to characterize potential
number of hours awake within the last 24 hour-cycle. The main application for this technology would be to
allow supervisors and employee drivers to identify and respond to fatigue, thereby reducing the driver’s risk
of a fatigue-related crash.
Developing and evaluating technologies to reduce crash and crash-related injury risk among gig workers
who deliver packages, takeout food, and groceries, whose jobs are linked to a digital online platform.
Impact and Commercialization Potential: Application of evidence-based interventions is
expected to have a large impact on reducing the incidence and severity of work-related motor
vehicle crashes. This will yield substantial public health benefits and will positively affect workers’
compensation and health insurance premiums and costs. CDC/NIOSH has well-established
working relationships with employers, their trade associations, and standards-setting
organizations, and is therefore strongly positioned to communicate findings and guidance to
potential users. CDC/NIOSH also has strong infrastructure to facilitate the transfer of technology-
based interventions to the marketplace. Given the extremely short induction period between
exposure and injury occurrence, CDC can make a measurable difference in a very short period (<
4 years).
Visit the NIOSH homepage for more information on NIOSH’s research program areas
http://www.cdc.gov/niosh/homepage.html.
For NIOSH programmatic information, contact:
Steve Dearwent, PhD

NIH, CDC, and FDA Program Descriptions and Research Topics
CDC 194
National Institute for Occupational Safety and Health (NIOSH)
Centers for Disease Control and Prevention
Mail Stop V24-4
Phone: 404-498-6382
Email: sdearwent@cdc.gov
For grants specific, administrative information, contact:
Brownie Anderson-Rana
Office of Grant Services Office of Financial Resources
Office of the Chief Operating Officer
Centers for Disease Control and Prevention (CDC) Mail Stop K-75
2920 Brandywine Rd.
Atlanta, GA 30341
Phone: 770-488-2771
Email: BAndersonRana@cdc.gov
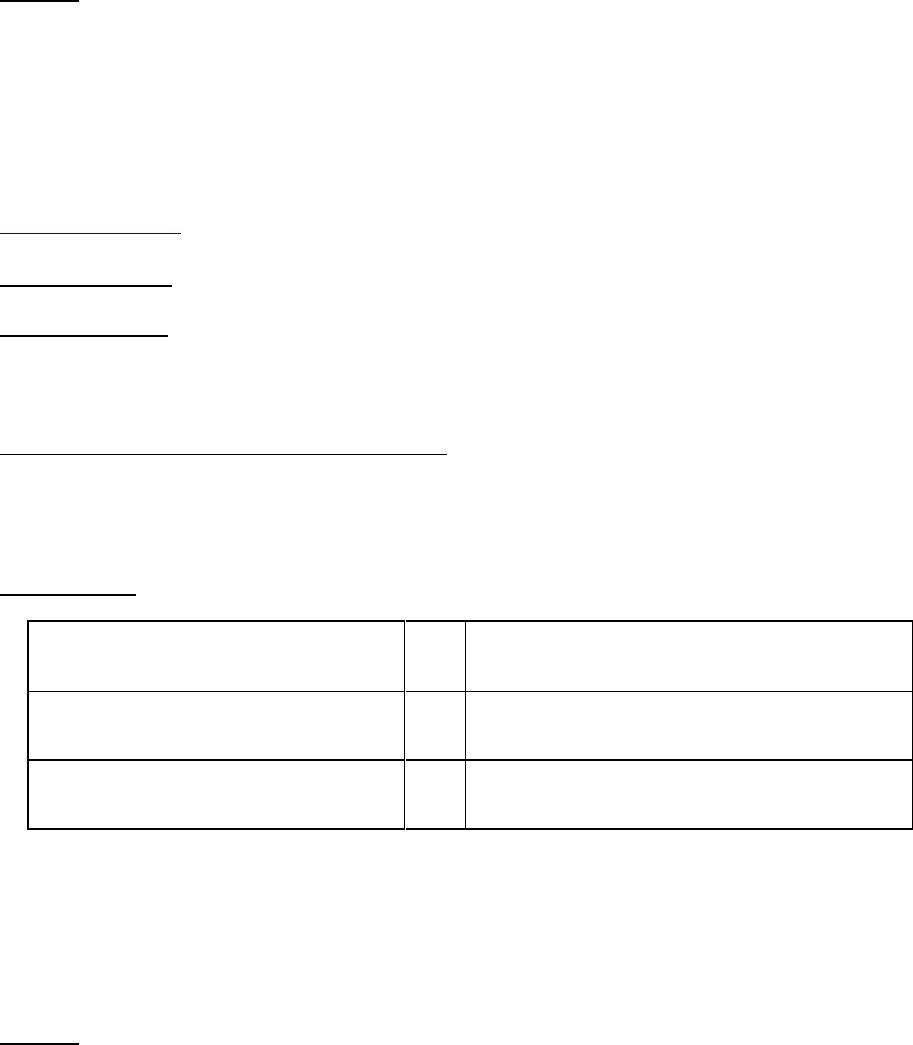
NIH, CDC, and FDA Program Descriptions and Research Topics
FDA 195
FOOD AND DRUG ADMINISTRATION (FDA)
Mission
The FDA is responsible for protecting the public health by assuring the safety, efficacy, and
security of human and veterinary drugs, biological products, medical devices, our nation’s food
supply, cosmetics, and products that emit radiation. The FDA is also responsible for advancing the
public health by helping to speed innovations that make medicines and foods more effective, safer,
and more affordable; and helping the public get accurate, science-based information they need to
use medicines and foods to improve their health.
For additional information about areas of interest to the FDA, please visit our home page at
http://www.fda.gov.
Budget Guidance
FDA will not fund:
•
Phase I applications greater than $200,000
•
Phase II applications greater than $1,500,000
Specific SBIR and STTR Program Information
FDA will not accept SBIR applications that propose clinical trials, and all of the topics listed below must be
for projects that do not propose clinical trials.
Clinical Trials
Does FDA accept Clinical Trials through
the Omnibus/Parent Notices of Funding
Opportunities?
No
Does FDA accept Clinical Trials
through specific Notices of Funding
Opportunities?
No
Does FDA support Clinical Trials
through NON-SBIR/STTR Notices of
Funding Opportunities?
No
CENTER FOR BIOLOGICS EVALUATION AND RESEARCH (CBER)
Mission
CBER is responsible for ensuring the safety, efficacy, potency and purity of biological and related products
intended for use in the treatment, prevention or cure of diseases in humans as well as the safety of the
nation's supply of blood and blood products. The primary responsibility of CBER is to review the

NIH, CDC, and FDA Program Descriptions and Research Topics
FDA 196
quality, safety and efficacy of vaccines, blood products, certain diagnostic products and other
biological and biotechnology-derived human products.
CBER's activities include: evaluating the quality, safety and effectiveness of biological products
before marketing, and monitoring the pre-clinical and clinical testing of new biological products;
licensing biological products and manufacturing establishments, including plasmapheresis centers,
blood banks, vaccine and biotechnology manufacturers; AIDS program and policy activities,
including research on AIDS therapeutic products, diagnostic tests and vaccines; research to
establish product standards, develop improved testing methods and assess the safety of biological
products; compliance, lot release program and post market surveillance; meeting PDUFA goals,
new research programs, and new regulatory initiatives (managed review process for all products).
CENTER FOR DRUG EVALUATION AND RESEARCH (CDER)
Mission
CDER develops FDA policy with regard to the safety, effectiveness, and labeling of all drugs for
human use; evaluates new drug applications and investigational new drug applications; develops
standards for the safety and effectiveness of all over-the-counter drugs; monitors the quality of
marketed drugs through product testing (bioavailability/bioequivalence testing), post marketing
surveillance, and compliance programs; develops guidelines on good manufacturing practices;
conducts research and develops scientific standards on composition, quality, safety, and efficacy of
human drugs.
Drug regulatory research as conducted in CDER is directed at the discovery of new knowledge
relevant to drug development, post marketing drug experience (patterns of drug use and safety), and
drug regulation to enhance FDA regulatory decisions. These drug regulatory decisions impact on
the development of regulations, guidelines and guidance for the regulated industry and provide
clarity and consistency in application of CDER regulatory requirements. These drug regulatory
decisions also impact public health by ensuring that marketing drugs are safe and efficacious and
that their risk: benefit profile remains acceptable during the market life of a drug. Specific areas of
research conducted by the Center include Pharmacology/toxicology, microbiology/virology, clinical
pharmacology, pediatric issues in drug therapy, post marketing drug safety, evaluation of
effectiveness of regulatory actions, patterns of drug use, including off-label, signal detection
methodologies (e.g., data mining techniques), epidemiologic studies of therapeutics using
population-based data, regulatory compliance, product quality, and active surveillance methods.
Research Topics
Research and development opportunities within the FDA that lend themselves to performance by
small businesses include, but are not limited to, the following:
A.
Develop a system for gathering real-time data on physician prescribing behavior,
understanding and compliance with drug product labeling and frequency of off -label
prescribing.
B.
Develop and evaluate the effectiveness of new methods and tools for managing the
known risks of marketed drug products (e.g., communicating newly identified risks to
health care practitioners and patients).
C.
Develop methods for timely active surveillance of newly approved drug products
in large populations to identify both expected and unexpected outcomes.

NIH, CDC, and FDA Program Descriptions and Research Topics
FDA 197
D.
Develop methods for actively collecting information on all cases of classically drug-
associated events (e.g., acute liver failure, blood dyscrasias, severe desquamating
skin disorders) to augment the FDA’s current passive surveillance system.
E.
Develop improved clinical markers and methods with potential for bed-side
application for detection of the early onset of adverse drug events.
F.
Develop surrogate potency methods for biotech drug products to replace
traditional animal testing.
G.
Development of psychochemical and in-vitro biological tests to evaluate
pharmaceutical equivalence of complex drug substances and drug products.
H.
Research into approaches to handle informative missing patient data in clinical trials,
including innovations in study designs and statistical methods of analysis.
I.
Statistical and computational methods and strategies for the design, analysis and
interpretation of microarray, genomic and proteonomic data.
CENTER FOR FOOD SAFETY AND APPLIED NUTRITION (CFSAN)
Mission
The FDA is responsible for the safety of the vast range of food Americans eat; about 80 percent of all
food sold in the United States. This includes everything except for the meat, poultry, and processed egg
products that are regulated by the USDA. Consequently, CFSAN seeks research designed to
complement and accelerate efforts aimed at the detection, prevention, and control of contamination that
may be responsible for illness or injury conveyed by foods, colors, and cosmetics. CFSAN conducts
research, and develops regulations, guidance and standards related to the composition, quality,
nutrition, and safety of food, food additives, colors, and cosmetics. The Center evaluates FDA’s
surveillance and compliance programs relating to foods, colors, and cosmetics; reviews industry
petitions, and develops regulations for food standards to permit the safe use of color and food additives.
Research Topics
CFSAN maintains an active research program that is focused on the following priorities; ensuring the
safety of food, dietary supplements and cosmetics; improving nutrition; and promoting the security
and integrity of the food supply. The Center’s research activities are intended to; support the FDA’s
regulatory activities; reduce the incidence of foodborne illness by improving our ability to detect and
quantify foodborne pathogens, toxins, and chemicals that could jeopardize the safety and security of
the food supply; find new and improved ways to control these agents; and safely produce, process,
and handle food and food products. FDA is committed to reducing the incidence of foodborne illness
to the greatest extent feasible while at the same time protecting the nation's food supply. Mission-
critical knowledge gaps are addressed through translation research focused on the risks associated
with FDA regulated products throughout their life cycles, from production to consumption. Ideally
extramural research is sought that complements the Center’s intramural research efforts, and which
will enhance the Agency’s and the Nation’s ability to reduce the incidence of foodborne illness and
protect the integrity of the nation’s food supply. FDA’s mission-critical needs require that the research
not simply end with the generation of new knowledge and technologies but extend to the validation of
new approaches by using realistic conditions that accurately reflect the diversity of the food industry
and offer potential solutions that can be accept by appropriate sectors of the food industry.

NIH, CDC, and FDA Program Descriptions and Research Topics
FDA 198
CENTER FOR DEVICES AND RADIOLOGICAL HEALTH (CDRH)
Mission
CDRH is responsible for assuring patients and providers have timely and continued access to safe,
effective, and high-quality medical devices and safe radiation-emitting products. Towards this goal,
CDRH develops policy, conducts regulatory science, and evaluates the safety and effectiveness of
medical devices and radiation-emitting products. CDRH classifies medical devices into one of three
classes based on risk and the regulatory controls necessary to provide a reasonable assurance of
safety and effectiveness. CDRH reviews study protocols for investigational devices, applications for
authorization of medical devices, and evaluates exemption requests for investigational devices. CDRH
is integrally involved in developing national and international standards, establishing good
manufacturing practices, and driving postmarket surveillance and compliance programs. The radiation
safety programs at CDRH involve enforcement of mandatory requirements in addition to partnerships
and voluntary programs that promote the safe use of radiation-emitting products. The Center develops
and conducts research and testing programs in the areas of physical, life, and engineering sciences
related to the human health effects of radiation and medical device technologies, provides expertise
and analyses for health-risk assessments, and also develops new or improved measurement methods,
techniques, instruments and analytical procedures for evaluating product performance and reliability.
Research Topics
CDRH welcomes submissions that aid in assessment of safety, effectiveness, and performance of
medical devices and/or promote medical device innovation. Please note, we do not fund development
of any products that may eventually be regulated by the Center. Areas of interest and examples are
listed below.
A. Biocompatibility and Alternative Methods: Develop and advance reliable alternatives to
traditional in vivo biocompatibility testing approaches to characterize biological response
to extracts of medical devices and particulates, including
•
novel in vitro, ex vivo, and in silico models, microphysiological systems,
alternative assays
•
frameworks capable of leveraging clinical, animal, and material information not
obtained through biocompatibility testing to be used as a substitute or to justify
performing more focused safety analyses
•
tools for assessing toxicological risk and chemical characterization in cases
where traditional extraction methods are not feasible and/or reference standards
do not exist.
B. Digital Health and Cybersecurity: Enhance the performance of digital health technologies
and promote medical device cybersecurity, including digital health technologies that
facilitate remote clinical trials and AI/ML algorithm evaluation methodologies.
C. Electrical and electromagnetic compatibility safety evaluation methods for non-clinical
environment use taking into consideration common EM emitters such as radiofrequency
identification (RFID) readers, electronic security systems (e.g., metal detectors, electronic
article surveillance), near-field communications (NFC) systems, wireless power transfer
(WPT), and Cellular 5G.

NIH, CDC, and FDA Program Descriptions and Research Topics
FDA 199
D. Health Equity: Advance the development of knowledge, and safe and effective
technologies, to meet the needs of diverse patient populations and consumers,
including reducing barriers to participate in evidence generation by diverse populations
and the use of medical technologies outside a healthcare setting.
•
Health of Women: Explore unique issues related to the performance of
medical devices in women, improve analysis and communication of sex-
and gender- specific data to better assure the safe and effective use of
medical devices
•
Human Factors: Explore methodologies to test and report on usability or
user experiences with devices in diverse patient populations and
consumers.
•
Pediatric Medical Device Development: Increase and accelerate medical
device development and labelling for the unique and evolving needs of
pediatric and special populations, especially younger sub- populations such
as neonates and children. Optimize or develop infrastructure that supports
safe innovation and development of medical devices designed, evaluated,
and labelled for pediatric and special populations.
E. Sterilization: Encourage the development of new approaches to medical device
sterilization with a focus on identifying alternatives to ethylene oxide (EtO) sterilization
methods, and/or development of strategies to reduce EtO emissions.
CENTER FOR VETERINARY MEDICINE (CVM)
Mission
CVM protects human and animal health by ensuring the safety and effectiveness of animal
drugs, by ensuring the safety of animal food (and food ingredients), and by addressing safety
concerns that may arise with the use of animal devices. The Center makes timely, quality
decisions and takes regulatory actions to ensure that these products are protective of public
health, provide for quality health care of animals, minimize the transmission of diseases, and
increase the efficiency of production of animal- derived food and fiber. The Center, in partnership
with Federal and state agencies and others, ensures animal health and the safety of food derived
from animals. Regulatory decisions are supported by research, the monitoring of product safety,
and efficacy, and continuous process improvement.
Research Topics
Research and development opportunities within the Center for Veterinary Medicine that lend themselves
to performance by small businesses include, but are not limited to, the following areas of interest:
A.
Development, for the specific purpose of obtaining approval or conditional approval, of
products for the treatment, control or prevention of diseases or conditions for which
limited approved therapeutic options are available, particularly those occurring in minor
species or small numbers of major species.
B.
Development and validation of high throughput/screening of quantitative and qualitative
analytical methods for analyzing drugs, additives, and contaminants (chemical and
microbial) in animal tissues and feeds.

NIH, CDC, and FDA Program Descriptions and Research Topics
FDA 200
C.
Development of methods and approaches to determine absorption, distribution, metabolism,
and excretion of drugs, food additives and contaminants (microbial and chemical) in
animals, including minor species. This includes, among other topics, 1) the development of
alternative methods, in support of replacement/reduction/refinement for activities involving
animal research, and 2) methods for the determination/validation of bioequivalence.
D.
Development of new biomarkers and models for determining the safety and effectiveness of
veterinary drugs and food additives in animals, including minor species or development of
alternative methods to animal use.
E.
Development of methods to determine the effects of drugs, food additives, and contaminants
(microbial and chemical) on immunological and physiological functions of animals, including
minor species.
F.
Development/refinement of One Health approaches to monitor and minimize antimicrobial
resistance development and to support antimicrobial stewardship in animals.
G.
Development of methodology for accelerated (short term) shelf-life stability testing for drugs
and food additives for extrapolation to real time stability testing.
OFFICE OF CRITICAL PATH PROGRAMS
Mission
The Office of Critical Path Programs, in FDA’s Office of the Chief Scientist, coordinates the cross-agency
Critical Path Initiative (CPI), FDA's strategy for transforming the way medical products are developed,
evaluated, and manufactured. CPI activities are under way throughout the Agency, from the product
centers to the Office of the Commissioner. For details, see
http://www.fda.gov/ScienceResearch/SpecialTopics/CriticalPathInitiative/default.htm. Collaboration is key
to the CPI initiative because bringing safe, effective, and innovative therapies to the American public
requires FDA to leverage the resources and expertise of all stakeholders, including other Federal
agencies, academia, healthcare professionals, patient and consumer groups, regulated industry, and
health-related organizations. In 2008, CPI collaborations involved 84 government agencies, universities,
industry leaders, and patient groups from 28 states and 5 countries on a raft of groundbreaking research
projects.
Research Topics
Research and development opportunities within FDA that lend themselves to performance by grantees
include, but are not limited to, the following:
A.
Studying the immunological correlates of TB immunity and developing tools to evaluate TB
vaccine efficacy.
B.
Developing study models for testing combination-antimicrobials as a strategy to prevent the
development of drug resistance.
C.
Developing new approaches to preclinical safety testing.
D.
Identifying biomarkers for safety and efficacy evaluation of medical products.

NIH, CDC, and FDA Program Descriptions and Research Topics
FDA 201
OFFICE OF ORPHAN PRODUCTS DEVELOPMENT
Mission
The Office of Orphan Products Development was established to identify and facilitate the
development of orphan products. Orphan products are drugs, biologics, medical devices and
foods for medical purposes, which are indicated for a rare disease or condition (i.e., one affecting
fewer than 200,000 people in the United States). These products may be useful in a rare
disease/disorder but lack commercial sponsorship because they are not considered commercially
attractive for marketing. A subcategory of orphan products are those marketed products in which
there is evidence suggesting usefulness in a rare disease/disorder but which are not labeled for
that disease/disorder because substantial evidence of safety and effectiveness for that use is
lacking.
Research Topics
Research and development opportunities within the FDA that lend themselves to performance by small
businesses include, but are not limited to, the following:
A.
Development of products for the treatment of rare diseases or disorders
including but not limited to neurological, metabolic, genetic, ophthalmologic,
hematologic, and dermatological diseases or disorders for the specific purpose
of obtaining marketing licensure.
B.
Development of products for use in diagnosis of rare diseases for which the
diagnostic tool would be used in fewer than 200,000 persons annually in the
United States.
C.
Development of vaccines for the prevention of rare diseases to be used in
fewer than 200,000 persons annually in the United States.
For additional information on research topics and administrative and business information, contact
Kimberly Pendleton
Director, Division of Grants, Agreements, and Acquisition Support
Email: Kimberly.Pendleton@fda.hhs.gov
Food and Drug Administration
Division of Acquisition Support and Grants


