
Center for Devices and Radiological Health
DESIGN CONTROL GUIDANCEDESIGN CONTROL GUIDANCE
FORFOR
MEDICAL DEVICE MANUFACTURERSMEDICAL DEVICE MANUFACTURERS
This Guidance relates to
FDA 21 CFR 820.30 and Sub-clause 4.4 of ISO 9001
March 11, 1997
i
FOREWORD
To ensure that good quality assurance practices are used for the design of medical devices
and that they are consistent with quality system requirements worldwide, the Food and
Drug Administration revised the Current Good Manufacturing Practice (CGMP)
requirements by incorporating them into the Quality System Regulation, 21 CFR Part 820.
An important component of the revision is the addition of design controls.
Because design controls must apply to a wide variety of devices, the regulation does not
prescribe the practices that must be used. Instead, it establishes a framework that
manufacturers must use when developing and implementing design controls. The
framework provides manufacturers with the flexibility needed to develop design controls
that both comply with the regulation and are most appropriate for their own design and
development processes.
This guidance is intended to assist manufacturers in understanding the intent of the
regulation. Design controls are based upon quality assurance and engineering principles.
This guidance complements the regulation by describing its intent from a technical
perspective using practical terms and examples.
Draft guidance was made publicly available in March, 1996. We appreciate the many
comments, suggestions for improvement, and encouragement we received from industry,
interested parties, and the Global Harmonization Task Force (GHTF) Study Group 3.
The comments were systematically reviewed, and revisions made in response to those
comments and suggestions are incorporated in this version. As experience is gained with
the guidance, FDA will consider the need for additional revisions within the next six to
eighteen months.
The Center publishes the results of its work in scientific journals and in its own technical
reports. Through these reports, CDRH also provides assistance to industry and to the
medical and healthcare professional communities in complying with the laws and
regulations mandated by Congress. These reports are sold by the Government Printing
Office (GPO) and by the National Technical Information Service (NTIS). Many reports,
including this guidance document, are also available via Internet on the World Wide Web
at www.fda.gov.
We welcome your comments and suggestions for future revisions.
D. Bruce Burlington, M.D.
Director
Center for Devices and Radiological Health
ii
iii
PREFACE
Effective implementation of design controls requires that the regulation and its intent be well
understood. The Office of Compliance within CDRH is using several methods to assist
manufacturers in developing this understanding. Methods include the use of presentations,
teleconferences, practice audits, and written guidance.
Those persons in medical device companies charged with responsibility for developing,
implementing, or applying design controls come from a wide variety of technical and non-technical
backgrounds––engineering, business administration, life sciences, computer science, and the arts.
Therefore, it is important that a tool be provided that conveys the intent of the regulation using
practical terminology and examples. That is the purpose of his guidance.
The response of medical device manufacturers and other interested parties to the March, 1996 draft
version of this guidance has significantly influenced this latest version. Most comments centered
on the complaint that the guidance was too prescriptive. Therefore, it has been rewritten to be
more pragmatic, focusing on principles rather than specific practices.
It is noteworthy that many comments offered suggestions for improving the guidance, and that the
authors of the comments often acknowledged the value of design controls and the potential benefit
of good guidance to the medical device industry, the public, and the FDA. Some comments even
included examples of past experiences with the implementation of controls.
Finally, there are several people within CDRH that deserve recognition for their contributions to
the development of this guidance. Al Taylor and Bill Midgette of the Office of Science and
Technology led the development effort and served as co-chairs of the CDRH Design Control
Guidance Team that reviewed the comments received last spring. Team members included Ashley
Boulware, Bob Cangelosi, Andrew Lowrey, Deborah Lumbardo, Jack McCracken, Greg
O'Connell, and Walter Scott. As the lead person within CDRH with responsibility for
implementing the Quality System Regulation, Kim Trautman reviewed the guidance and
coordinated its development with the many other concurrent and related activities. Their
contributions are gratefully acknowledged.
FDA would also like to acknowledge the significant contributions made by the Global
Harmonization Task Force (GHTF) Study Group 3. The Study Group reviewed and revised this
guidance at multiple stages during its development. It is hoped that this cooperative effort will lead
to this guidance being accepted as an internationally recognized guidance document through the
GHTF later this year.
Lillian J. Gill
Director
Office of Compliance
iv
ACKNOWLEDGEMENT
FDA wishes to acknowledge the contributions of the Global Harmonization Task Force
(GHTF) Study Group 3 to the development of this guidance. As has been stated in the
past, FDA is firmly committed to the international harmonization of standards and
regulations governing medical devices. The GHTF was formed in 1992 to further this
effort. The GHTF includes representatives of the Canadian Ministry of Health and
Welfare; the Japanese Ministry of Health and Welfare; FDA; industry members from the
European Union, Australia, Canada, Japan, and the United States; and a few delegates
from observing countries.
Among other efforts, the GHTF Study Group 3 started developing guidance on the
application of design controls to medical devices in the spring of 1995. Study Group 3
has recognized FDA’s need to publish timely guidance on this topic in conjunction with
promulgation of its new Quality System Regulation. The Study Group has therefore
devoted considerable time and effort to combine its draft document with the FDA's efforts
as well as to review and comment on FDA's subsequent revisions. FDA, for its part,
delayed final release of its guidance pending final review by the Study Group. As a result,
it is hoped that this document, with some minor editorial revisions to make the guidance
global to several regulatory schemes, will be recognized through the GHTF as an
international guidance document.
v
TABLE OF CONTENTS
FOREWORD...................................................................................................................i
PREFACE...................................................................................................................... iii
ACKNOWLEDGEMENT.............................................................................................. iv
TABLE OF CONTENTS................................................................................................ v
INTRODUCTION .......................................................................................................... 1
SECTION A. GENERAL............................................................................................... 7
SECTION B. DESIGN AND DEVELOPMENT PLANNING ........................................9
SECTION C. DESIGN INPUT.................................................................................... 13
SECTION D. DESIGN OUTPUT................................................................................ 19
SECTION E. DESIGN REVIEW................................................................................. 23
SECTION F. DESIGN VERIFICATION..................................................................... 29
SECTION G. DESIGN VALIDATION .......................................................................33
SECTION H. DESIGN TRANSFER............................................................................ 37
SECTION I. DESIGN CHANGES............................................................................... 39
SECTION J. DESIGN HISTORY FILE (DHF)............................................................ 43
vi
Introduction 3/11/97 Page 1
INTRODUCTION
I. PURPOSE
This guidance is intended to assist manufacturers in understanding quality system
requirements concerning design controls. Assistance is provided by interpreting the
language of the quality systems requirements and explaining the underlying concepts in
practical terms.
Design controls are an interrelated set of practices and procedures that are incorporated
into the design and development process, i.e., a system of checks and balances. Design
controls make systematic assessment of the design an integral part of development. As a
result, deficiencies in design input requirements, and discrepancies between the proposed
designs and requirements, are made evident and corrected earlier in the development
process. Design controls increase the likelihood that the design transferred to production
will translate into a device that is appropriate for its intended use.
In practice, design controls provide managers and designers with improved visibility of the
design process. With improved visibility, managers are empowered to more effectively
direct the design process—that is, to recognize problems earlier, make corrections, and
adjust resource allocations. Designers benefit both by enhanced understanding of the
degree of conformance of a design to user and patient needs, and by improved
communications and coordination among all participants in the process.
The medical device industry encompasses a wide range of technologies and applications,
ranging from simple hand tools to complex computer-controlled surgical machines, from
implantable screws to artificial organs, from blood-glucose test strips to diagnostic
imaging systems and laboratory test equipment. These devices are manufactured by
companies varying in size and structure, methods of design and development, and methods
of management. These factors significantly influence how design controls are actually
applied. Given this diversity, this guidance does not suggest particular methods of
implementation, and therefore, must not be used to assess compliance with the quality
system requirements. Rather, the intent is to expand upon the distilled language of the
quality system requirements with practical explanations and examples of design control
principles. Armed with this basic knowledge, manufacturers can and should seek out
technology-specific guidance on applying design controls to their particular situation.
When using this guidance, there could be a tendency to focus only on the time and effort
required in developing and incorporating the controls into the design process. However,
readers should keep in mind the intrinsic value of design controls as well. It is a well-
established fact that the cost to correct design errors is lower when errors are detected
early in the design and development process. Large and small companies that have
achieved quality systems certification under ISO 9001 cite improvements in productivity,
product quality, customer satisfaction, and company competitiveness. Additional benefits
Introduction 3/11/97 Page 2
are described in comments received from a quality assurance manager of a medical device
firm regarding the value of a properly documented design control system:
“...there are benefits to an organization and the quality improvement of an
organization by having a written design control system. By defining this system on
paper, a corporation allows all its employees to understand the requirements, the
process, and expectations of design and how the quality of design is assured and
perceived by the system. It also provides a baseline to review the system
periodically for further improvements based on history, problems, and failures of
the system (not the product).”
II. SCOPE
The guidance applies to the design of medical devices as well as the design of the
associated manufacturing processes. The guidance is applicable to new designs as well as
modifications or improvements to existing device designs. The guidance discusses
subjects in the order in which they appear in FDA's Quality System regulation and is cross-
referenced to International Organization for Standards (ISO) 9001:1994, Quality
SystemsModel for Quality Assurance in Design, Development, Production,
Installation, and Servicing, and the ISO draft international standard ISO/DIS 13485,
Quality SystemsMedical DevicesParticular Requirements for the Application of ISO
9001, dated April 1996.
Design controls are a component of a comprehensive quality system that covers the life of
a device. The assurance process is a total systems approach that extends from the
development of device requirements through design, production, distribution, use,
maintenance, and eventually, obsolescence. Design control begins with development and
approval of design inputs, and includes the design of a device and the associated
manufacturing processes.
Design control does not end with the transfer of a design to production. Design control
applies to all changes to the device or manufacturing process design, including those
occurring long after a device has been introduced to the market. This includes
evolutionary changes such as performance enhancements as well as revolutionary changes
such as corrective actions resulting from the analysis of failed product. The changes are
part of a continuous, ongoing effort to design and develop a device that meets the needs
of the user and/or patient. Thus, the design control process is revisited many times during
the life of a product.
Some tools and techniques are described in the guidance. Although aspects of their utility
are sometimes described, they are included in the guidance for illustrative purposes only.
Including them does not mean that they are preferred. There may be alternative ways that
are better suited to a particular manufacturer and design activity. The literature contains an
abundance of information on tools and techniques. Such topics as project management,
design review, process capability, and many others referred to in this guidance are
Introduction 3/11/97 Page 3
available in textbooks, periodicals, and journals. As a manufacturer applies design controls
to a particular task, the appropriate tools and techniques used by competent personnel
should be applied to meet the needs of the unique product or process for that
manufacturer.
III. APPLICATION OF DESIGN CONTROLS
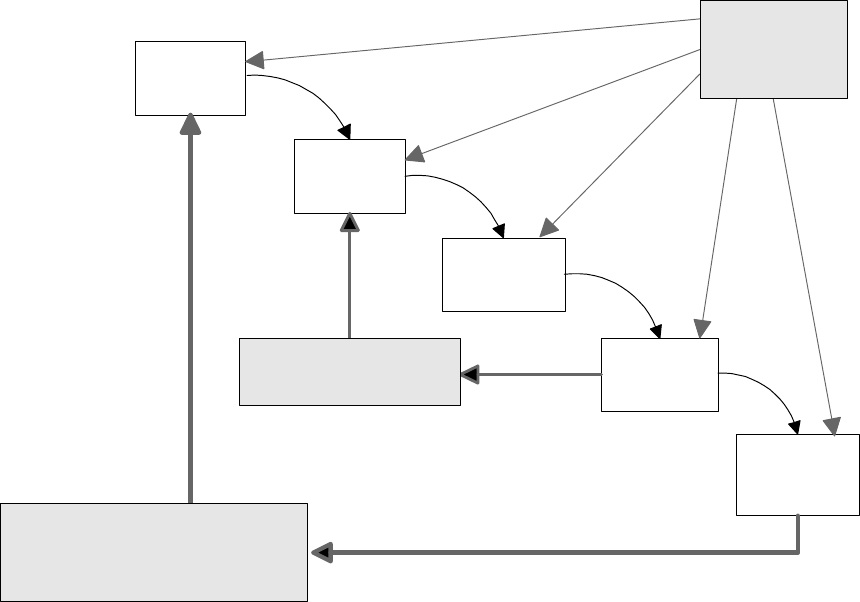
Design controls may be applied to any product development process. The simple example
shown in Figure 1 illustrates the influence of design controls on a design process.
User
Needs
Design
Input
Design
Process
Design
Output
Medical
Device
R
eview
v
erification
V
alidation
Figure 1 – Application of Design Controls to Waterfall Design Process (figure used with
permission of Medical Devices Bureau, Health Canada)
The development process depicted in the example is a traditional waterfall model. The
design proceeds in a logical sequence of phases or stages. Basically, requirements are
developed, and a device is designed to meet those requirements. The design is then
evaluated, transferred to production, and the device is manufactured. In practice,
feedback paths would be required between each phase of the process and previous phases,
representing the iterative nature of product development. However, this detail has been
omitted from the figure to make the influence of the design controls on the design process
more distinct.
Introduction 3/11/97 Page 4
The importance of the design input and verification of design outputs is illustrated by this
example. When the design input has been reviewed and the design input requirements are
determined to be acceptable, an iterative process of translating those requirements into a
device design begins. The first step is conversion of the requirements into system or high-
level specifications. Thus, these specifications are a design output. Upon verification that
the high-level specifications conform to the design input requirements, they become the
design input for the next step in the design process, and so on.
This basic technique is used repeatedly throughout the design process. Each design input
is converted into a new design output; each output is verified as conforming to its input;
and it then becomes the design input for another step in the design process. In this
manner, the design input requirements are translated into a device design conforming to
those requirements.
The importance of design reviews is also illustrated by the example. The design reviews
are conducted at strategic points in the design process. For example, a review is
conducted to assure that the design input requirements are adequate before they are
converted into the design specifications. Another is used to assure that the device design
is adequate before prototypes are produced for simulated use testing and clinical
evaluation. Another, a validation review, is conducted prior to transfer of the design to
production. Generally, they are used to provide assurance that an activity or phase has
been completed in an acceptable manner, and that the next activity or phase can begin.
As the figure illustrates, design validation encompasses verification and extends the
assessment to address whether devices produced in accordance with the design actually
satisfy user needs and intended uses.
An analogy to automobile design and development may help to clarify these concepts.
Fuel efficiency is a common design requirement. This requirement might be expressed as
the number of miles-per-gallon of a particular grade of gasoline for a specified set of
driving conditions. As the design of the car proceeds, the requirements, including the one
for fuel efficiency, are converted into the many layers of system and subsystem
specifications needed for design. As these various systems and subsystems are designed,
design verification methods are used to establish conformance of each design to its own
specifications. Because several specifications directly affect fuel efficiency, many of the
verification activities help to provide confirmation that the overall design will meet the fuel
efficiency requirement. This might include simulated road testing of prototypes or actual
road testing. This is establishing by objective evidence that the design output conforms to
the fuel efficiency requirement. However, these verification activities alone are not
sufficient to validate the design. The design may be validated when a representative
sample of users have driven production vehicles under a specified range of driving
conditions and judged the fuel efficiency to be adequate. This is providing objective
evidence that the particular requirement for a specific intended use can be consistently
fulfilled.
Introduction 3/11/97 Page 5
CONCURRENT ENGINEERING. Although the waterfall model is a useful tool for
introducing design controls, its usefulness in practice is limited. The model does apply to
the development of some simpler devices. However, for more complex devices, a
concurrent engineering model is more representative of the design processes in use in the
industry.
In a traditional waterfall development scenario, the engineering department completes the
product design and formally transfers the design to production. Subsequently, other
departments or organizations develop processes to manufacture and service the product.
Historically, there has frequently been a divergence between the intent of the designer and
the reality of the factory floor, resulting in such undesirable outcomes as low
manufacturing yields, rework or redesign of the product, or unexpectedly high cost to
service the product.
One benefit of concurrent engineering is the involvement of production and service
personnel throughout the design process, assuring the mutual optimization of the
characteristics of a device and its related processes. While the primary motivations of
concurrent engineering are shorter development time and reduced production cost, the
practical result is often improved product quality.
Concurrent engineering encompasses a range of practices and techniques. From a design
control standpoint, it is sufficient to note that concurrent engineering may blur the line
between development and production. On the one hand, the concurrent engineering
model properly emphasizes that the development of production processes is a design
rather than a manufacturing activity. On the other hand, various components of a design
may enter production before the design as a whole has been approved. Thus, concurrent
engineering and other more complex models of development usually require a
comprehensive matrix of reviews and approvals to ensure that each component and
process design is validated prior to entering production, and the product as a whole is
validated prior to design release.
RISK MANAGEMENT AND DESIGN CONTROLS. Risk management is the
systematic application of management policies, procedures, and practices to the tasks of
identifying, analyzing, controlling, and monitoring risk. It is intended to be a framework
within which experience, insight, and judgment are applied to successfully manage risk. It
is included in this guidance because of its effect on the design process.
Risk management begins with the development of the design input requirements. As the
design evolves, new risks may become evident. To systematically identify and, when
necessary, reduce these risks, the risk management process is integrated into the design
process. In this way, unacceptable risks can be identified and managed earlier in the
design process when changes are easier to make and less costly.
An example of this is an exposure control system for a general purpose x-ray system. The
control function was allocated to software. Late in the development process, risk analysis
of the system uncovered several failure modes that could result in overexposure to the
Introduction 3/11/97 Page 6
patient. Because the problem was not identified until the design was near completion, an
expensive, independent, back-up timer had to be added to monitor exposure times.
THE QUALITY SYSTEM AND DESIGN CONTROLS. In addition to procedures
and work instructions necessary for the implementation of design controls, policies and
procedures may also be needed for other determinants of device quality that should be
considered during the design process. The need for policies and procedures for these
factors is dependent upon the types of devices manufactured by a company and the risks
associated with their use. Management with executive responsibility has the responsibility
for determining what is needed.
Example of topics for which policies and procedures may be appropriate are:
• risk management
• device reliability
• device durability
• device maintainability
• device serviceability
• human factors engineering
• software engineering
• use of standards
• configuration management
• compliance with regulatory requirements
• device evaluation (which may include third party product certification or approval)
• clinical evaluations
• document controls
• use of consultants
• use of subcontractors
• use of company historical data

Section A General 3/11/97 Page 7
SECTION A. GENERAL
I. REQUIREMENTS
§ 820.30(a) General.
(1) Each manufacturer of any class III or class II device, and the class I devices listed
in paragraph (a) (2) of this section, shall establish and maintain procedures to
control the design of the device in order to ensure that specified design
requirements are met.
(2) The following class I devices are subject to design controls:
(i) Devices automated with computer software; and
(ii) The devices listed in the chart below.
Section Device
868.6810 Catheter, Tracheobronchial Suction
878.4460 Glove, Surgeon’s
880.6760 Restraint, Protective
892.5650 System, Applicator, Radionuclide,
Manual
892.5740 Source, Radionuclide Teletherapy
II. DEFINITIONS
§ 820.3 (n) Management with executive responsibility means those senior employees of a
manufacturer who have the authority to establish or make changes to the manufacturer’s
quality policy and quality system.
§ 820.3 (s) Quality means the totality of features and characteristics that bear on the
ability of a device to satisfy fitness-for-use, including safety and performance.
§ 820.3 (v) Quality system means the organizational structure, responsibilities,
procedures, processes, and resources for implementing quality management.
Cross reference to ISO 9001:1994 and ISO/DIS 13485 Section 4.4.1 General.
Section A General 3/11/97 Page 8
III. DISCUSSION AND POINTS TO CONSIDER
The essential quality aspects and the regulatory requirements, such as safety, performance,
and dependability of a product (whether hardware, software, services, or processed
materials) are established during the design and development phase. Deficient design can
be a major cause of quality problems.
The context within which product design is to be carried out should be set by the
manufacturer’s senior management. It is their responsibility to establish a design and
development plan which sets the targets to be met. This plan defines the constraints
within which the design is to be implemented.
The quality system requirements do not dictate the types of design process that a
manufacturer must use. Manufacturers should use processes best suited to their needs.
However, whatever the processes may be, it is important that the design controls are
applied in an appropriate manner. This guidance document contains examples of how this
might be achieved in a variety of situations.
It is important to note that the design function may apply to various facets of the operation
having differing styles and time scales. Such facets are related to products, including
services and software, as well as to their manufacturing processes.
Senior management needs to decide how the design function is to be managed and by
whom. Senior management should also ensure that internal policies are established for
design issues such as:
• assessing new product ideas
• training and retraining of design managers and design staff
• use of consultants
• evaluation of the design process
• product evaluation, including third party product certification and approvals
• patenting or other means of design protection
It is for senior management to ensure that adequate resources are available to carry out
the design in the required time. This may involve reinforcing the skills and equipment
available internally and/or obtaining external resources.
Section B Design and Development Planning 3/11/97 Page 9
SECTION B. DESIGN AND DEVELOPMENT PLANNING
I. REQUIREMENTS
§ 820.30(b) Design and development planning.
• Each manufacturer shall establish and maintain plans that describe or reference the
design and development activities and define responsibility for implementation.
• The plans shall identify and describe the interfaces with different groups or
activities that provide, or result in, input to the design and development process.
• The plans shall be reviewed, updated, and approved as design and development
evolves.
Cross-reference to ISO 9001:1994 and ISO/DIS 13485 sections 4.4.2 Design and
development planning and 4.4.3 Organizational and technical interfaces.
II. DISCUSSION AND POINTS TO CONSIDER
Design and development planning is needed to ensure that the design process is
appropriately controlled and that device quality objectives are met. The plans must be
consistent with the remainder of the design control requirements. The following elements
would typically be addressed in the design and development plan or plans:
• Description of the goals and objectives of the design and development program;
i.e., what is to be developed;
• Delineation of organizational responsibilities with respect to assuring quality
during the design and development phase, to include interface with any
contractors;
• Identification of the major tasks to be undertaken, deliverables for each task, and
individual or organizational responsibilities (staff and resources) for completing
each task;
• Scheduling of major tasks to meet overall program time constraints;
• Identification of major reviews and decision points;
• Selection of reviewers, the composition of review teams, and procedures to be
followed by reviewers;
• Controls for design documentation;
• Notification activities.
Planning enables management to exercise greater control over the design and development
process by clearly communicating policies, procedures, and goals to members of the

Section B Design and Development Planning 3/11/97 Page 10
design and development team, and providing a basis for measuring conformance to quality
system objectives.
Design activities should be specified at the level of detail necessary for carrying out the
design process. The extent of design and development planning is dependent on the size
of the developing organization and the size and complexity of the product to be
developed. Some manufacturers may have documented policies and procedures which
apply to all design and development activities. For each specific development program,
such manufacturers may also prepare a plan which spells out the project-dependent
elements in detail, and incorporates the general policies and procedures by reference.
Other manufacturers may develop a comprehensive design and development plan which is
specifically tailored to each individual project.
In summary, the form and organization of the planning documents are less important than
their content. The following paragraphs discuss the key elements of design and
development planning.
ORGANIZATIONAL RESPONSIBILITIES. The management responsibility section of
the quality system requirements
1
requires management to establish a quality policy and
implement an organizational structure to ensure quality. These are typically documented in
a quality manual or similarly named document. In some cases, however, the design and
development plan, rather than the quality manual, is the best vehicle for describing
organizational responsibilities relative to design and development activities. The
importance of defining responsibilities with clarity and without ambiguity should be
recognized. When input to the design is from a variety of sources, their interrelationships
and interfaces (as well as the pertinent responsibilities and authorities) should be defined,
documented, coordinated, and controlled. This might be the case, for example, if a
multidisciplinary product development team is assembled for a specific project, or if the
team includes suppliers, contract manufacturers, users, outside consultants, or independent
auditors.
TASK BREAKDOWN. The plan establishes, to the extent possible:
• The major tasks required to develop the product
• The time involved for each major task
• The resources and personnel required
• The allocation of responsibilities for completing each major task
• The prerequisite information necessary to start each major task and the
interrelationship between tasks
• The form of each task output or deliverable
• Constraints, such as applicable codes, standards, and regulations
1
§ 820.20 of the FDA Quality System Regulation; section 4.1 of ISO 9001 and ISO/DIS 13485.
Section B Design and Development Planning 3/11/97 Page 11
Tasks for all significant design activities, including verification and validation tasks, should
be included in the design and development plan. For example, if clinical trials are
anticipated, there may be tasks associated with appropriate regulatory requirements.
For complex projects, rough estimates may be provided initially, with the details left for
the responsible organizations to develop. As development proceeds, the plan should
evolve to incorporate more and better information.
The relationships between tasks should be presented in such a way that they are easily
understood. It should be clear which tasks depend on others, and which tasks need to be
performed concurrently. Planning should reflect the degree of perceived development risk;
for example, tasks involving new technology or processes should be spelled out in greater
detail, and perhaps be subjected to more reviews and checks, than tasks which are
perceived as routine or straightforward.
The design and development plan may include a schedule showing starting and completion
dates for each major task, project milestone, or key decision points. The method chosen
and the detail will vary depending on the complexity of the project and the level of risk
associated with the device. For small projects, the plan may consist of only a simple flow
diagram or computer spreadsheet. For larger projects, there are a number of project
management tools that are used to develop plans. Three of the most commonly used are
the Program Evaluation and Review Technique (PERT), the Critical Path Method (CPM),
and the Gantt chart. Software is available in many forms for these methods. When
selecting these tools, be careful to choose one that best fits the needs of the project. Some
of the software programs are far more complex than may be necessary.
Unless a manufacturer has experience with the same type of device, the plan will initially
be limited in scope and detail. As work proceeds, the plan is refined. Lack of experience in
planning often leads to optimistic schedules, but slippage may also occur for reasons
beyond the control of planners, for example, personnel turnover, materiel shortage, or
unexpected problems with a design element or process. Sometimes the schedule can be
compressed by using additional resources, such as diverting staff or equipment from
another project, hiring a contractor, or leasing equipment.
It is important that the schedule be updated to reflect current knowledge. At all times, the
plan should be specified at a level of detail enabling management to make informed
decisions, and provide confidence in meeting overall schedule and performance objectives.
This is important because scheduling pressures have historically been a contributing factor
in many design defects which caused injury. To the extent that good planning can prevent
schedule pressures, the potential for design errors is reduced.
However, no amount of planning can eliminate all development risk. There is inherent
conflict between the desire to maximize performance and the need to meet business
objectives, including development deadlines. In some corporate cultures, impending
deadlines create enormous pressure to cut corners. Planning helps to combat this dilemma
by ensuring management awareness of pressure points. With awareness, decisions are
Section B Design and Development Planning 3/11/97 Page 12
more likely to be made with appropriate oversight and consideration of all relevant factors.
Thus, when concessions to the clock must be made, they can be justified and supported.
Section C. Design Input 3/11/97 Page 13
SECTION C. DESIGN INPUT
I. REQUIREMENTS
§ 820.30(c) Design input.
• Each manufacturer shall establish and maintain procedures to ensure that the
design requirements relating to a device are appropriate and address the intended
use of the device, including the needs of the user and patient.
• The procedures shall include a mechanism for addressing incomplete, ambiguous,
or conflicting requirements.
• The design input requirements shall be documented and shall be reviewed and
approved by designated individual(s).
• The approval, including the date and signature of the individual(s) approving the
requirements, shall be documented.
Cross reference to ISO 9001:1994 and ISO/DIS 13485 section 4.4.4 Design input.
II. DEFINITIONS
§ 820.3(f) Design input means the physical and performance requirements of a device
that are used as a basis for device design.
III. DISCUSSION AND POINTS TO CONSIDER
Design input is the starting point for product design. The requirements which form the
design input establish a basis for performing subsequent design tasks and validating the
design. Therefore, development of a solid foundation of requirements is the single most
important design control activity.
Many medical device manufacturers have experience with the adverse effects that
incomplete requirements can have on the design process. A frequent complaint of
developers is that “there’s never time to do it right, but there’s always time to do it over.”
If essential requirements are not identified until validation, expensive redesign and rework
may be necessary before a design can be released to production.
By comparison, the experience of companies that have designed devices using clear-cut,
comprehensive sets of requirements is that rework and redesign are significantly reduced
and product quality is improved. They know that the development of requirements for a
medical device of even moderate complexity is a formidable, time-consuming task. They
accept the investment in time and resources required to develop the requirements because
they know the advantages to be gained in the long run.
Section C. Design Input 3/11/97 Page 14
Unfortunately, there are a number of common misconceptions regarding the meaning and
practical application of the quality system requirements for design input. Many seem to
arise from interpreting the requirements as a literal prescription, rather than a set of
principles to be followed. In this guidance document, the focus is on explaining the
principles and providing examples of how they may be applied in typical situations.
CONCEPT DOCUMENTS VERSUS DESIGN INPUT In some cases, the marketing
staff, who maintain close contact with customers and users, determine a need for a new
product, or enhancements to an existing product. Alternatively, the idea for a new
product may evolve out of a research or clinical activity. In any case, the result is a
concept document specifying some of the desired characteristics of the new product.
Some members of the medical device community view these marketing memoranda, or the
equivalent, as the design input. However, that is not the intent of the quality system
requirements. Such concept documents are rarely comprehensive, and should not be
expected to be so. Rather, the intent of the quality system requirements is that the product
conceptual description be elaborated, expanded, and transformed into a complete set of
design input requirements which are written to an engineering level of detail.
This is an important concept. The use of qualitative terms in a concept document is both
appropriate and practical. This is often not the case for a document to be used as a basis
for design. Even the simplest of terms can have enormous design implications. For
example, the term “must be portable” in a concept document raises questions in the minds
of product developers about issues such as size and weight limitations, resistance to shock
and vibration, the need for protection from moisture and corrosion, the capability of
operating over a wide temperature range, and many others. Thus, a concept document
may be the starting point for development, but it is not the design input requirement. This
is a key principle—the design input requirements are the result of the first stage of the
design control process.
RESEARCH AND DEVELOPMENT. Some manufacturers have difficulty in
determining where research ends and development begins. Research activities may be
undertaken in an effort to determine new business opportunities or basic characteristics for
a new product. It may be reasonable to develop a rapid prototype to explore the
feasibility of an idea or design approach, for example, prior to developing design input
requirements. But manufacturers should avoid falling into the trap of equating the
prototype design with a finished product design. Prototypes at this stage lack safety
features and ancillary functions necessary for a finished product, and are developed under
conditions which preclude adequate consideration of product variability due to
manufacturing.
RESPONSIBILITY FOR DESIGN INPUT DEVELOPMENT. Regardless of who
developed the initial product concept, product developers play a key role in developing the
design input requirements. When presented with a set of important characteristics, it is the
product developers who understand the auxiliary issues that must be addressed, as well as
the level of detail necessary to design a product. Therefore, a second key principle is that
Section C. Design Input 3/11/97 Page 15
the product developer(s) ultimately bear responsibility for translating user and/or patient
needs into a set of requirements which can be validated prior to implementation. While
this is primarily an engineering function, the support or full participation of production and
service personnel, key suppliers, etc., may be required to assure that the design input
requirements are complete.
Care must be exercised in applying this principle. Effective development of design input
requirements encompasses input from both the product developer as well as those
representing the needs of the user, such as marketing. Terminology can be a problem. In
some cases, the product conceptual description may be expressed in medical terms.
Medical terminology is appropriate in requirements when the developers and reviewers are
familiar with the language, but it is often preferable to translate the concepts into
engineering terms at the requirements stage to minimize miscommunication with the
development staff.
Another problem is incorrect assumptions. Product developers make incorrect
assumptions about user needs, and marketing personnel make incorrect assumptions about
the needs of the product designers. Incorrect assumptions can have serious consequences
that may not be detected until late in the development process. Therefore, both product
developers and those representing the user must take responsibility for critically examining
proposed requirements, exploring stated and implied assumptions, and uncovering
problems.
Some examples should clarify this point. A basic principle is that design input
requirements should specify what the design is intended to do while carefully avoiding
specific design solutions at this stage. For example, a concept document might dictate
that the product be housed in a machined aluminum case. It would be prudent for product
developers to explore why this type of housing was specified. Perhaps there is a valid
reason—superior electrical shielding, mechanical strength, or reduced time to market as
compared to a cast housing. Or perhaps machined aluminum was specified because a
competitor’s product is made that way, or simply because the user didn’t think plastic
would be strong enough.
Not all incorrect assumptions are made by users. Incorrect assumptions made by product
developers may be equally damaging. Failure to understand the abuse to which a portable
instrument would be subjected might result in the selection of housing materials
inadequate for the intended use of the product.
There are occasions when it may be appropriate to specify part of the design solution in
the design input requirements. For example, a manufacturer may want to share
components or manufacturing processes across a family of products in order to realize
economies of scale, or simply to help establish a corporate identity. In the case of a
product upgrade, there may be clear consensus regarding the features to be retained.
However, it is important to realize that every such design constraint reduces
implementation flexibility and should therefore be documented and identified as a possible
conflicting requirement for subsequent resolution.
Section C. Design Input 3/11/97 Page 16
SCOPE AND LEVEL OF DETAIL. Design input requirements must be
comprehensive. This may be quite difficult for manufacturers who are implementing a
system of design controls for the first time. Fortunately, the process gets easier with
practice. It may be helpful to realize that design input requirements fall into three
categories. Virtually every product will have requirements of all three types.
• Functional requirements specify what the device does, focusing on the operational
capabilities of the device and processing of inputs and the resultant outputs.
• Performance requirements specify how much or how well the device must
perform, addressing issues such as speed, strength, response times, accuracy, limits
of operation, etc. This includes a quantitative characterization of the use
environment, including, for example, temperature, humidity, shock, vibration, and
electromagnetic compatibility. Requirements concerning device reliability and
safety also fit into this category.
• Interface requirements specify characteristics of the device which are critical to
compatibility with external systems; specifically, those characteristics which are
mandated by external systems and outside the control of the developers. One
interface which is important in every case is the user and/or patient interface.
What is the scope of the design input requirements development process and how much
detail must be provided? The scope is dependent upon the complexity of a device and the
risk associated with its use. For most medical devices, numerous requirements
encompassing functions, performance, safety, and regulatory concerns are implied by the
application. These implied requirements should be explicitly stated, in engineering terms,
in the design input requirements.
Determining the appropriate level of detail requires experience. However, some general
guidance is possible. The marketing literature contains product specifications, but these
are superficial. The operator and service manuals may contain more detailed
specifications and performance limits, but these also fall short of being comprehensive.
Some insight as to what is necessary is provided by examining the requirements for a very
common external interface. For the power requirements for AC-powered equipment, it is
not sufficient to simply say that a unit shall be AC-powered. It is better to say that the
unit shall be operable from AC power in North America, Europe, and Japan, but that is
still insufficient detail to implement or validate the design. If one considers the situation
just in North America, where the line voltage is typically 120 volts, many systems are
specified to operate over the range of 108 to 132 volts. However, to account for the
possibility of brownout, critical devices may be specified to operate from 95 to 132 volts
or even wider ranges. Based on the intended use of the device, the manufacturer must
choose appropriate performance limits.
There are many cases when it is impractical to establish every functional and performance
characteristic at the design input stage. But in most cases, the form of the requirement can
be determined, and the requirement can be stated with a to-be-determined (TBD)
numerical value or a range of possible values. This makes it possible for reviewers to
Section C. Design Input 3/11/97 Page 17
assess whether the requirements completely characterize the intended use of the device,
judge the impact of omissions, and track incomplete requirements to ensure resolution.
For complex designs, it is not uncommon for the design input stage to consume as much
as thirty percent of the total project time. Unfortunately, some managers and developers
have been trained to measure design progress in terms of hardware built, or lines of
software code written. They fail to realize that building a solid foundation saves time
during the implementation. Part of the solution is to structure the requirements
documents and reviews such that tangible measures of progress are provided.
At the other extreme, many medical devices have very simple requirements. For example,
many new devices are simply replacement parts for a product, or are kits of commodity
items. Typically, only the packaging and labeling distinguishes these products from
existing products. In such cases, there is no need to recreate the detailed design input
requirements of the item. It is acceptable to simply cite the predecessor product
documentation, add any new product information, and establish the unique packaging and
labeling requirements.
ASSESSING DESIGN INPUT REQUIREMENTS FOR ADEQUACY. Eventually,
the design input must be reviewed for adequacy. After review and approval, the design
input becomes a controlled document. All future changes will be subject to the change
control procedures, as discussed in Section I (Design Changes).
Any assessment of design input requirements boils down to a matter of judgment. As
discussed in Section E (Design Review), it is important for the review team to be
multidisciplinary and to have the appropriate authority. A number of criteria may be
employed by the review team.
• Design input requirements should be unambiguous. That is, each requirement
should be able to be verified by an objective method of analysis, inspection, or
testing. For example, it is insufficient to state that a catheter must be able to
withstand repeated flexing. A better requirement would state that the catheter
should be formed into a 50 mm diameter coil and straightened out for a total of
fifty times with no evidence of cracking or deformity. A qualified reviewer could
then make a judgment whether this specified test method is representative of the
conditions of use.
• Quantitative limits should be expressed with a measurement tolerance. For
example, a diameter of 3.5 mm is an incomplete specification. If the diameter is
specified as 3.500±0.005 mm, designers have a basis for determining how accurate
the manufacturing processes have to be to produce compliant parts, and reviewers
have a basis for determining whether the parts will be suitable for the intended use.
• The set of design input requirements for a product should be self-consistent. It is
not unusual for requirements to conflict with one another or with a referenced
industry standard due to a simple oversight. Such conflicts should be resolved
early in the development process.
Section C. Design Input 3/11/97 Page 18
• The environment in which the product is intended to be used should be properly
characterized. For example, manufacturers frequently make the mistake of
specifying “laboratory” conditions for devices which are intended for use in the
home. Yet, even within a single country, relative humidity in a home may range
from 20 percent to 100 percent (condensing) due to climactic and seasonal
variations. Household temperatures in many climates routinely exceed 40 °C
during the hot season. Altitudes may exceed 3,000 m, and the resultant low
atmospheric pressure may adversely affect some kinds of medical equipment. If
environmental conditions are fully specified, a qualified reviewer can make a
determination of whether the specified conditions are representative of the
intended use.
• When industry standards are cited, the citations should be reviewed for
completeness and relevance. For example, one medical device manufacturer
claimed compliance with an industry standard covering mechanical shock and
vibration. However, when the referenced standard was examined by a reviewer, it
was found to prescribe only the method of testing, omitting any mention of
pass/fail criteria. It was incumbent on the manufacturer in this case to specify
appropriate performance limits for the device being tested, as well as the test
method.
EVOLUTION OF THE DESIGN INPUT REQUIREMENTS. Large development
projects often are implemented in stages. When this occurs, the design input requirements
at each stage should be developed and reviewed following the principles set forth in this
section. Fortunately, the initial set of requirements, covering the overall product, is by far
the most difficult to develop. As the design proceeds, the output from the early stages
forms the basis for the subsequent stages, and the information available to designers is
inherently more extensive and detailed.
It is almost inevitable that verification activities will uncover discrepancies which result in
changes to the design input requirements. There are two points to be made about this.
One is that the change control process for design input requirements must be carefully
managed. Often, a design change to correct one problem may create a new problem
which must be addressed. Throughout the development process, it is important that any
changes are documented and communicated to developers so that the total impact of the
change can be determined. The change control process is crucial to device quality.
The second point is that extensive rework of the design input requirements suggests that
the design input requirements may not be elaborated to a suitable level of detail, or
insufficient resources are being devoted to defining and reviewing the requirements.
Managers can use this insight to improve the design control process. From a design
control perspective, the number of requirements changes made is less important than the
thoroughness of the change control process.
Section D. Design Output 3/11/97 Page 19
SECTION D. DESIGN OUTPUT
I. REQUIREMENTS
§ 820.30(d) Design output.
• Each manufacturer shall establish and maintain procedures for defining and
documenting design output in terms that allow an adequate evaluation of
conformance to design input requirements.
• Design output procedures shall contain or make reference to acceptance criteria
and shall ensure that those design outputs that are essential for the proper
functioning of the device are identified.
• Design output shall be documented, reviewed, and approved before release.
• The approval, including the date and signature of the individual(s) approving the
output, shall be documented.
Cross-reference to ISO 9001:1994 and ISO/DIS 13485 section 4.4.5 Design output.
II. DEFINITIONS
§ 820.3(g) Design output means the results of a design effort at each design phase and at
the end of the total design effort. The finished design output is the basis for the device
master record. The total finished design output consists of the device, its packaging and
labeling, and the device master record.
§ 820.3(y) Specification means any requirement with which a product, process, service,
or other activity must conform.
III. DISCUSSION AND POINTS TO CONSIDER
The quality system requirements for design output can be separated into two elements:
Design output should be expressed in terms that allow adequate assessment of
conformance to design input requirements and should identify the characteristics of the
design that are crucial to the safety and proper functioning of the device. This raises two
fundamental issues for developers:
• What constitutes design output?
• Are the form and content of the design output suitable?
The first issue is important because the typical development project produces voluminous
records, some of which may not be categorized as design output. On the other hand,
design output must be reasonably comprehensive to be effective. As a general rule, an
item is design output if it is a work product, or deliverable item, of a design task listed in
Section D. Design Output 3/11/97 Page 20
the design and development plan, and the item defines, describes, or elaborates an element
of the design implementation. Examples include block diagrams, flow charts, software
high-level code, and system or subsystem design specifications. The design output in one
stage is often part of the design input in subsequent stages.
Design output includes production specifications as well as descriptive materials which
define and characterize the design.
PRODUCTION SPECIFICATIONS. Production specifications include drawings and
documents used to procure components, fabricate, test, inspect, install, maintain, and
service the device, such as the following:
• assembly drawings
• component and material specifications
• production and process specifications
• software machine code (e.g., diskette or master EPROM)
• work instructions
• quality assurance specifications and procedures
• installation and servicing procedures
• packaging and labeling specifications, including methods and processes used
In addition, as discussed in Section H (Design Transfer), production specifications may
take on other forms. For example, some manufacturers produce assembly instructions on
videotapes rather than written instructions. Similarly, a program diskette, used by a
computer-aided milling machine to fabricate a part, would be considered a production
specification. The videotape and the software on the program diskette are part of the
device master record.
OTHER DESCRIPTIVE MATERIALS. Other design output items might be produced
which are necessary to establish conformance to design input requirements, but are not
used in its production. For example, for each part which is fabricated by computer-aided
machine, there should be an assembly drawing which specifies the dimensions and
characteristics of the part. It is a part of the design output because it establishes the basis
for the machine tool program used to fabricate the part. Other examples of design output
include the following:
• the results of risk analysis
• software source code
• results of verification activities
• biocompatibility test results
• bioburden test results
FORM AND CONTENT. Manufacturers must take steps to assure that the design
output characterizes all important aspects of the design and is expressed in terms which
Section D. Design Output 3/11/97 Page 21
allow adequate verification and validation. Two basic mechanisms are available to
manufacturers to accomplish these objectives.
• First, the manufacturer proactively can specify the form and content of design
output at the planning stage. For some types of design output, form and content
may be codified in a consensus standard which can be referenced. In other cases, a
manufacturer could specify the desired characteristics, or even simply specify that
the form and content of an existing document be followed.
• Second, form and content can be reviewed retroactively as a part of the design
verification process. For example, the verification of design output could include
assessing whether specified documentation standards have been adhered to.
As these examples illustrate, conformance with the quality system requirements
concerning design output generally requires no “extra” effort on the part of the
manufacturer, but simply the application of some common sense procedures during the
planning, execution, and review of design tasks.
Section D. Design Output 3/11/97 Page 22
Section E. Design Review 3/11/97 Page 23
SECTION E. DESIGN REVIEW
I. REQUIREMENTS
§ 820.30(e) Design review.
• Each manufacturer shall establish and maintain procedures to ensure that formal
documented reviews of the design results are planned and conducted at
appropriate stages of the device’s design development.
• The procedures shall ensure that participants at each design review include
representatives of all functions concerned with the design stage being reviewed and
an individual(s) who does not have direct responsibility for the design stage being
reviewed, as well as any specialists needed.
• The results of a design review, including identification of the design, the date, and
the individual(s) performing the review, shall be documented in the design history
file (the DHF).
Cross-reference to ISO 9001:1994 and ISO/DIS 13485 section 4.4.6 Design review.
II. DEFINITIONS
§ 820.3(h) Design review means a documented, comprehensive, systematic examination of
a design to evaluate the adequacy of the design requirements, to evaluate the capability of
the design to meet these requirements, and to identify problems.
III. DISCUSSION AND POINTS TO CONSIDER
In general, formal design reviews are intended to:
• provide a systematic assessment of design results, including the device design and
the associated designs for production and support processes;
• provide feedback to designers on existing or emerging problems;
• assess project progress; and/or
• provide confirmation that the project is ready to move on to the next stage of
development.
Many types of reviews occur during the course of developing a product. Reviews may
have both an internal and external focus. The internal focus is on the feasibility of the
design and the produceability of the design with respect to manufacturing and support
Section E. Design Review 3/11/97 Page 24
capabilities. The external focus is on the user requirements; that is, the device design is
viewed from the perspective of the user.
The nature of reviews changes as the design progresses. During the initial stages, issues
related to design input requirements will predominate. Next, the main function of the
reviews may be to evaluate or confirm the choice of solutions being offered by the design
team. Then, issues such as the choice of materials and the methods of manufacture become
more important. During the final stages, issues related to the verification, validation, and
production may predominate.
The term “review” is commonly used by manufacturers to describe a variety of design
assessment activities. Most, but not all, of these activities meet the definition of formal
design reviews. The following exceptions may help to clarify the distinguishing
characteristics of design reviews.
• Each design document which constitutes the formal output, or deliverable, of a
design task is normally subject to evaluation activities, sometimes referred to as
informal peer review, supervisory review, or technical assessment. These activities,
while they may be called reviews, are often better described as verification
activities, because they are not intended to be comprehensive, definitive, and
multidisciplinary in their scope. Rather, their purpose is to confirm that design
output meets design input. Verification activities affect and add to the design
output, and are themselves subject to subsequent design review.
• Developers may conduct routine or ad hoc meetings to discuss an issue, coordinate
activities, or assess development progress. Decisions from such meetings may not
require formal documentation; however, if a significant issue is resolved, this
should be documented. If the outcome results in change to an approved design
document, then applicable change control procedures should be followed, as
discussed in Section I (Design Changes).
Control of the design review process is achieved by developing and implementing a formal
design review program consistent with quality system requirements. The following issues
should be addressed and documented in the design and development plan(s).
NUMBER AND TYPE OF REVIEWS. It is a well-accepted fact that the cost to correct
design errors increases as the design nears completion, and the flexibility to implement an
optimal solution decreases. When an error is discovered at the end of the development
cycle, difficult decisions have to be made regarding an acceptable corrective action. When
that corrective action is implemented in haste, the result is often an unintended
consequence leading to a new problem. Thus, formal design reviews should be planned to
detect problems early. A corollary is that planners should presume that problems will be
detected, and allocate a reasonable amount of time to implement corrective actions.
Typically, formal reviews are conducted at the end of each phase and at important
milestones in the design process.
Section E. Design Review 3/11/97 Page 25
As discussed in Section C (Design Input), it is beneficial in almost every case to conduct a
formal review of the design input requirements early in the development process. The
number of reviews depends upon the complexity of the device.
• For a simple design, or a minor upgrade to an existing product, it might be
appropriate to conduct a single review at the conclusion of the design process.
• For a product involving multiple subsystems, an early design task is to allocate the
design input requirements among the various subsystems. For example, in a
microprocessor-based system, designers must decide which functions will be
performed by hardware and which by software. In another case, tolerance buildup
from several components may combine to create a clearance problem. System
designers must establish tolerance specifications for each component to meet the
overall dimensional specification. In cases like these, a formal design review is a
prudent step to ensure that all such system-level requirements have been allocated
satisfactorily prior to engaging in detailed design of each subsystem.
• For complex systems, additional reviews are often built into the development plan.
For example, engineering sketches may be developed for prototyping purposes
prior to development of production drawings. Evaluation of the prototype would
typically culminate in a formal design review. Similarly, software development
commonly includes a high-level design phase, during which requirements are
elaborated to a greater level of detail and algorithms are developed to implement
key functions. A formal design review would typically be conducted to review this
work prior to beginning detailed coding.
There are a number of approaches to conducting formal design reviews at the end of the
design process. In some organizations, engineering essentially completes the design, tests
an engineering prototype, and conducts a formal design review prior to turning the design
over to manufacturing. In such cases, an additional review will be needed after the design
has been validated using production devices.
In some instances, components having long lead times may enter production prior to
completion of the overall device design. The primary motivation for early production is to
reduce time to market. The manufacturer runs the business risk that the design review at
the end of the design process will uncover a defect that must be corrected in production
devices before any devices are distributed.
All of these approaches to scheduling formal design reviews are valid. What is important is
that the manufacturer establish a reasonable rationale for the number and type of reviews,
based on sound judgment.
SELECTION OF REVIEWERS. In determining who should participate in a formal
design review, planners should consider the qualifications of reviewers, the types of
expertise required to make an adequate assessment, and the independence of the
reviewers. Each of these concerns is discussed briefly in the following paragraphs.
Section E. Design Review 3/11/97 Page 26
Qualifications. Formal design reviews should be conducted by person(s) having technical
competence and experience at least comparable to the developers. For a small
manufacturer, this may require that an outside consultant be retained to participate in the
evaluation of the design.
A manufacturer will often employ one or more specialists to conduct certain types of
specialized assessments which are beyond the capabilities of the designers. For example, a
mechanical engineer may be retained to perform a structural analysis of a design, and
perhaps conduct vibration testing to verify its performance under stress. Such specialists
may be assigned to participate in the formal design review. Alternatively, they may be
assigned to make an independent assessment and submit observations and
recommendations to the reviewers. Either approach is valid.
Types of expertise required. Many medical device designs involve a number of
technologies, such as electronics, mechanics, software, materials science, or pneumatics.
In addition, a variety of clinical and manufacturing issues may influence the design.
Manufacturers should carefully consider which interests should be represented at formal
design reviews. Subtle distinctions in reviewer perspective may have dramatic impact on
device quality. For example, the marketing department of a small manufacturer shared a
new design with several surgeons on their advisory board. The surgeons all thought the
design was terrific. Subsequently, the manufacturer invited two experienced operating
room nurses to participate in the final design review. During the course of the review, it
became apparent that while surgeons may be the customers, nurses are the primary users
of the device, and no one up to that point had consulted with any nurses. The nurses at the
design review didn’t like some of the features of the design. After some further market
survey, the manufacturer decided to make changes to the design to accommodate these
concerns. It was unfortunate (and expensive) in this case that the user requirements were
not considered until late in the development cycle, but the design review was ultimately
very successful.
Independence. The formal design review should include at least one individual who does
not have direct responsibility for the design stage under review. In a small company,
complete independence is very difficult to obtain. Within the context of formal design
reviews, the practical solution is simply to ensure a fresh perspective, based on the
principle that those who are too close to the design may overlook design errors. Thus,
reviewers will often be from the same organization as the developers, but they should not
have been significantly involved in the activities under review. As discussed in the
following section, the formal design review procedures play a large role in assuring
independent and objective reviews.
DESIGN REVIEW PROCEDURES. The manufacturer should have documented formal
design review procedures addressing the following:
• Evaluation of the design (including identification of concerns, i.e., issues and
potential problems with the design)
• Resolution of concerns
Section E. Design Review 3/11/97 Page 27
• Implementation of corrective actions
Evaluation of the design. Many formal design reviews take the form of a meeting. At this
meeting, the designer(s) may make presentations to explain the design implementation,
and persons responsible for verification activities may present their findings to the
reviewers. Reviewers may ask for clarification or additional information on any topic, and
add their concerns to any raised by the presenters. This portion of the review is focused on
finding problems, not resolving them.
There are many approaches to conducting design review meetings. In simple cases, the
technical assessor and reviewer may be the same person, often a project manager or
engineering supervisor, and the review meeting is a simple affair in the manager’s office.
For more elaborate reviews, detailed written procedures are desirable to ensure that all
pertinent topics are discussed, conclusions accurately recorded, and action items
documented and tracked.
There is a dangerous tendency for design review meetings to become adversarial affairs.
The reputation of the designers tends to be linked to the number of discrepancies found,
causing the designers to become defensive, while the reviewers score points by finding
weaknesses in the design. The resulting contest can be counterproductive. An added
complication is the presence of invited guests, often clinicians, who are expected to
provide the user perspective. These reviewers are often very reluctant to ask probing
questions, especially if they sense that they may become involved in a conflict where all
the rules and relationships are not evident.
These difficulties can be avoided by stating the goals and ground rules for conducting the
formal design review clearly at the outset. While the designers are in the best position to
explain the best features of the design, they are also most likely to be aware of the design’s
weaknesses. If the designers and reviewers are encouraged to work together to
systematically explore problems and find solutions, the resultant design will be improved
and all parties will benefit from the process. Participants must be encouraged to ask
questions, avoid making assumptions, and think critically. The focus must be on the
design, not the participants.
Not all formal design reviews involve meetings. For extremely simple designs or design
changes, it may be appropriate to specify a procedure in which review materials are
distributed or circulated among the reviewers for independent assessment and approval.
However, such a procedure negates the benefits of synergy and teamwork, and should be
considered only in cases where the design issues are limited in scope and well defined.
Resolution of concerns. The reviewers consider concerns raised during the evaluation
portion of the formal design review and decide on an appropriate disposition for each one.
There is wide variation in the way companies implement decision-making processes. In
some cases, the reviewers play an advisory role to the engineering manager or other
company official, who directs the formal design review and ultimately selects a course of
Section E. Design Review 3/11/97 Page 28
action. In other cases, the reviewers are given limited or broad authority to make decisions
and commit resources to resolve problems. The approach used should be documented.
In the real world, reviews often leave unresolved issues. Therefore, review procedures
should include a process for resolving differences, and provide reviewers with enough
leeway to make practical decisions while protecting the integrity of the process.
Implementation of corrective actions. Not all identified concerns result in corrective
actions. The reviewers may decide that the issue is erroneous or immaterial. In most cases,
however, resolution involves a design change, a requirements change, or a combination of
the two. If the solution is evident, the reviewers may specify the appropriate corrective
action; otherwise, an action item will be assigned to study the problem further. In any
case, action items and corrective actions are normally tracked under the manufacturer’s
change control procedures.
RELATIONSHIP OF DESIGN REVIEW TO VERIFICATION AND
VALIDATION. In practice, design review, verification, and validation overlap one
another, and the relationship among them may be confusing. As a general rule, the
sequence is: verification, review, validation, review.
In most cases, verification activities are completed prior to the design review, and the
verification results are submitted to the reviewers along with the other design output to be
reviewed. Alternatively, some verification activities may be treated as components of the
design review, particularly if the verification activity is complex and requires
multidisciplinary review.
Similarly, validation typically involves a variety of activities, including a determination that
the appropriate verifications and reviews have been completed. Thus, at the conclusion of
the validation effort, a review is usually warranted to assure that the validation is complete
and adequate.
Section F. Design Verification 3/11/97 Page 29
SECTION F. DESIGN VERIFICATION
I. REQUIREMENTS
§ 820.30(f) Design verification.
• Each manufacturer shall establish and maintain procedures for verifying the device
design.
• Design verification shall confirm that the design output meets the design input
requirements.
• The results of the design verification, including identification of the design,
method(s), the date, and the individual(s) performing the verification, shall be
documented in the Design History File.
Cross-reference to ISO 9001:1994 and ISO/DIS 13485 section 4.4.7 Design
verification.
I. DEFINITIONS
§820.3(y) Specification means any requirement with which a product, process, service, or
other activity must conform.
§ 820.3(z) Validation means confirmation by examination and provision of objective
evidence that the particular requirements for a specific intended use can be consistently
fulfilled.
(1) Process Validation means establishing by objective evidence that a process
consistently produces a result or product meeting its predetermined specifications.
(2) Design Validation means establishing by objective evidence that device
specifications conform with user needs and intended use(s).
§820.3(aa) Verification means confirmation by examination and provision of objective
evidence that specified requirements have been fulfilled.
III. DISCUSSION AND POINTS TO CONSIDER
Verification and validation are associated concepts with very important differences.
Various organizations have different definitions for these terms. Medical device
Section F. Design Verification 3/11/97 Page 30
manufacturers are encouraged to use the terminology of the quality system requirements in
their internal procedures.
To illustrate the concepts, consider a building design analogy. In a typical scenario, the
senior architect establishes the design input requirements and sketches the general
appearance and construction of the building, but associates or contractors typically
elaborate the details of the various mechanical systems. Verification is the process of
checking at each stage whether the output conforms to requirements for that stage. For
example: does the air conditioning system deliver the specified cooling capacity to each
room? Is the roof rated to withstand so many newtons per square meter of wind loading?
Is a fire alarm located within 50 meters of each location in the building?
At the same time, the architect has to keep in mind the broader question of whether the
results are consistent with the ultimate user requirements. Does the air conditioning
system keep the occupants comfortable throughout the building? Will the roof withstand
weather extremes expected at the building site? Can the fire alarm be heard throughout the
building? Those broader concerns are the essence of validation.
In the initial stages of design, verification is a key quality assurance technique. As the
design effort progresses, verification activities become progressively more comprehensive.
For example, heat or cooling delivery can be calculated and verified by the air conditioning
designer, but the resultant air temperature can only be estimated. Occupant comfort is a
function not only of delivered air temperature, but also humidity, heat radiation to or from
nearby thermal masses, heat gain or loss through adjacent windows, etc. During the latter
design phases, the interaction of these complex factors may be considered during
verification of the design.
Validation follows successful verification, and ensures that each requirement for a
particular use is fulfilled. Validation of user needs is possible only after the building is
built. The air conditioning and fire alarm performance may be validated by testing and
inspection, while the strength of the roof will probably be validated by some sort of
analysis linked to building codes which are accepted as meeting the needs of the user—
subject to possible confirmation during a subsequent severe storm.
Validation is the topic of Section G of this guidance document. The remainder of this
section focuses on verification principles.
TYPES OF VERIFICATION ACTIVITIES. Verification activities are conducted at all
stages and levels of device design. The basis of verification is a three-pronged approach
involving tests, inspections, and analyses. Any approach which establishes conformance
with a design input requirement is an acceptable means of verifying the design with respect
to that requirement. In many cases, a variety of approaches are possible.
Complex designs require more and different types of verification activities. The nature of
verification activities varies according to the type of design output. The intent of this
guidance document is not to suggest or recommend verification techniques which should
Section F. Design Verification 3/11/97 Page 31
be performed by device manufacturers. Rather, the manufacturer should select and apply
appropriate verification techniques based on the generally accepted practices for the
technologies employed in their products. Many of these practices are an integral part of
the development process, and are routinely performed by developers. The objective of
design controls is to ensure adequate oversight by making verification activities explicit
and measuring the thoroughness of their execution. Following are a few examples of
verification methods and activities.
• Worst case analysis of an assembly to verify that components are derated properly
and not subject to overstress during handling and use.
• Thermal analysis of an assembly to assure that internal or surface temperatures do
not exceed specified limits.
• Fault tree analysis of a process or design.
• Failure modes and effects analysis.
• Package integrity tests.
• Biocompatibility testing of materials.
• Bioburden testing of products to be sterilized.
• Comparison of a design to a previous product having an established history of
successful use.
For some technologies, verification methods may be highly standardized. In other cases,
the manufacturer may choose from a variety of applicable methods. In a few cases, the
manufacturer must be creative in devising ways to verify a particular aspect of a design.
Some manufacturers erroneously equate production testing with verification. Whereas
verification testing establishes conformance of design output with design input, the aim of
production testing is to determine whether the unit under test has been correctly
manufactured. In other words, production testing is designed to efficiently screen out
manufacturing process errors and perhaps also to detect infant mortality failures.
Typically, a small subset of functional and performance tests accomplish this objective
with a high degree of accuracy. Therefore, production testing is rarely, if ever,
comprehensive enough to verify the design. For example, a leakage test may be used
during production to ensure that a hermetically-sealed enclosure was properly assembled.
However, the leakage test may not be sensitive enough to detect long-term diffusion of
gas through the packaging material. Permeability of the packaging material is an intrinsic
property of the material rather than an assembly issue, and would likely be verified using a
more specialized test than is used during production.
DOCUMENTATION OF VERIFICATION ACTIVITIES. Some verification methods
result in a document by their nature. For example, a failure modes and effects analysis
produces a table listing each system component, its postulated failure modes, and the
effect of such failures on system operation.
Section F. Design Verification 3/11/97 Page 32
Another self-documenting verification method is the traceability matrix. This method is
particularly useful when the design input and output are both documents; it also has great
utility in software development. In the most common form of the traceability matrix, the
input requirements are enumerated in a table, and references are provided to each section
in the output documents (or software modules) which address or satisfy each input
requirement. The matrix can also be constructed “backwards,” listing each feature in the
design output and tracing which input requirement bears on that feature. This reverse
approach is especially useful for detecting hidden assumptions. Hidden assumptions are
dangerous because they often lead to overdesign, adding unnecessary cost and complexity
to the design. In other cases, hidden assumptions turn out to be undocumented design
input requirements which, once exposed, can be properly tracked and verified.
However, many verification activities are simply some sort of structured assessment of the
design output relative to the design input. When this is the case, manufacturers may
document completion of verification activities by linking these activities with the signoff
procedures for documents. This may be accomplished by establishing a procedure
whereby each design output document must be verified and signed by designated persons.
The presence of the reviewers’ signatures on the document signifies that the design output
has been verified in accordance with the signoff procedure.
Section G. Design Validation 3/11/97 Page 33
SECTION G. DESIGN VALIDATION
I. REQUIREMENTS
§ 820.30(g) Design validation.
• Each manufacturer shall establish and maintain procedures for validating the device
design.
• Design validation shall be performed under defined operating conditions on initial
production units, lots, or batches, or their equivalents.
• Design validation shall ensure that devices conform to defined user needs and
intended uses and shall include testing of production units under actual or
simulated use conditions.
• Design validation shall include software validation and risk analysis, where
appropriate.
• The results of the design validation, including identification of the design,
method(s), the date, and the individual(s) performing the validation, shall be
documented in the Design History File.
Cross-reference to ISO 9001:1994 and ISO/DIS 13485 section 4.4.8 Design
validation.
II. DEFINITIONS
§820.3(y) Specification means any requirement with which a product, process, service, or
other activity must conform.
§ 820.3(z) Validation means confirmation by examination and provision of objective
evidence that the particular requirements for a specific intended use can be consistently
fulfilled.
(1) Process Validation means establishing by objective evidence that a process
consistently produces a result or product meeting its predetermined specifications.
(2) Design Validation means establishing by objective evidence that device
specifications conform with user needs and intended use(s).
§820.3(aa) Verification means confirmation by examination and provision of objective
evidence that specified requirements have been fulfilled.
Section G. Design Validation 3/11/97 Page 34
III. DISCUSSION AND POINTS TO CONSIDER
Whereas verification is a detailed examination of aspects of a design at various stages in
the development, design validation is a cumulative summation of all efforts to assure that
the design will conform with user needs and intended use(s), given expected variations in
components, materials, manufacturing processes, and the use environment.
VALIDATION PLANNING. Planning for validation should begin early in the design
process. The performance characteristics that are to be assessed should be identified, and
validation methods and acceptance criteria should be established. For complex designs, a
schedule of validation activities and organizational or individual responsibilities will
facilitate maintaining control over the process. The validation plan should be reviewed for
appropriateness, completeness, and to ensure that user needs and intended uses are
addressed.
VALIDATION REVIEW. Validation may expose deficiencies in the original
assumptions concerning user needs and intended uses. A formal review process should be
used to resolve any such deficiencies. As with verification, the perception of a deficiency
might be judged insignificant or erroneous, or a corrective action may be required.
VALIDATION METHODS. Many medical devices do not require clinical trials.
However, all devices require clinical evaluation and should be tested in the actual or
simulated use environment as a part of validation. This testing should involve devices
which are manufactured using the same methods and procedures expected to be used for
ongoing production. While testing is always a part of validation, additional validation
methods are often used in conjunction with testing, including analysis and inspection
methods, compilation of relevant scientific literature, provision of historical evidence that
similar designs and/or materials are clinically safe, and full clinical investigations or clinical
trials.
Some manufacturers have historically used their best assembly workers or skilled lab
technicians to fabricate test articles, but this practice can obscure problems in the
manufacturing process. It may be beneficial to ask the best workers to evaluate and
critique the manufacturing process by trying it out, but pilot production should simulate as
closely as possible the actual manufacturing conditions.
Validation should also address product packaging and labeling. These components of the
design may have significant human factors implications, and may affect product
performance in unexpected ways. For example, packaging materials have been known to
cause electrostatic discharge (ESD) failures in electronic devices. If the unit under test is
delivered to the test site in the test engineer’s briefcase, the packaging problem may not
become evident until after release to market.
Validation should include simulation of the expected environmental conditions, such as
temperature, humidity, shock and vibration, corrosive atmospheres, etc. For some classes
Section G. Design Validation 3/11/97 Page 35
of device, the environmental stresses encountered during shipment and installation far
exceed those encountered during actual use, and should be addressed during validation.
Particular care should be taken to distinguish among customers, users, and patients to
ensure that validation addresses the needs of all relevant parties. For a consumer device,
the customer, user, and patient may all be the same person. At the other extreme, the
person who buys the device may be different from the person who routinely uses it on
patients in a clinical setting. Hospital administrators, biomedical engineers, health
insurance underwriters, physicians, nurses, medical technicians, and patients have distinct
and sometimes competing needs with respect to a device design.
VALIDATION DOCUMENTATION. Validation is a compilation of the results of all
validation activities. For a complex design, the detailed results may be contained in a
variety of separate documents and summarized in a validation report. Supporting
information should be explicitly referenced in the validation report and either included as
an appendix or available in the design history file.
Section G. Design Validation 3/11/97 Page 36
Section H. Design Transfer 3/11/97 Page 37
SECTION H. DESIGN TRANSFER
I. REQUIREMENTS
§ 820.30(h) Design transfer.
• Each manufacturer shall establish and maintain procedures to ensure that the
device design is correctly translated into production specifications.
Cross reference to ISO 9001:1994 and ISO/DIS 13485 section 4.2.3(c) Quality
planning.
II. DISCUSSION AND POINTS TO CONSIDER
Production specifications must ensure that manufactured devices are repeatedly and
reliably produced within product and process capabilities. If a manufactured device
deviates outside those capabilities, performance may be compromised. Thus, the process
of encapsulating knowledge about the device into production specifications is critical to
device quality.
The level of detail necessary to accomplish this objective varies widely, based on the type
of device, the relationship between the design and manufacturing organizations, and the
knowledge, experience, and skills of production workers. In some cases, devices are
produced by contract manufacturers who have no involvement in the development and
little or no contact with the designers. At the other extreme, some devices are hand-
crafted by skilled artisans with extensive knowledge about the use of the product.
One normally associates the term “production specifications” with written documents,
such as assembly drawings, component procurement specifications, workmanship
standards, manufacturing instructions, and inspection and test specifications. While these
types of documents are widely employed in medical device production, other equally
acceptable means of conveying design information exist, and manufacturers have the
flexibility to employ these alternate means of communication as appropriate. For example,
each of the following could constitute “production specifications” within the meaning of
the quality system requirements:
• documentation (in electronic format as well as paper)
• training materials, e.g., manufacturing processes, test and inspection methods
• digital data files, e.g., programmable device files, master EPROM, computer-aided
manufacturing (CAM) programming files
• manufacturing jigs and aids, e.g., molds, sample wiring harness to be duplicated
Section H. Design Transfer 3/11/97 Page 38
Historically, shortcomings in the production specifications tend to be manifested late in the
product life cycle. When the design is new, there is often intensive interaction between the
design and production teams, providing ample opportunity for undocumented information
flow. Later, as production experience is gained, some decoupling often occurs between
design and production teams. In addition, key personnel may leave, and their replacements
may lack comparable training, experience, or institutional knowledge.
Particular care should be taken when the product involves new and unproved
manufacturing processes, or established processes which are new to the manufacturer. It
may not be possible to determine the adequacy of full-scale manufacturing on the basis of
successfully building prototypes or models in a laboratory and testing these prototypes or
models. The engineering feasibility and production feasibility may be different because the
equipment, tools, personnel, operating procedures, supervision and motivation could be
different when a manufacturer scales up for routine production.
No design team can anticipate all factors bearing on the success of the design, but
procedures for design transfer should address at least the following basic elements.
• First, the design and development procedures should include a qualitative
assessment of the completeness and adequacy of the production specifications.
• Second, the procedures should ensure that all documents and articles which
constitute the production specifications are reviewed and approved.
• Third, the procedures should ensure that only approved specifications are used to
manufacture production devices.
The first item in the preceding list may be addressed during design transfer. The second
and third elements are among the basic principles of document control and configuration
management. As long as the production specifications are traditional paper documents,
there is ample information available to guide manufacturers in implementing suitable
procedures. When the production specifications include non-traditional means, flexibility
and creativity may be needed to achieve comparable rigor.
Section I. Design Changes 3/11/97 Page 39
SECTION I. DESIGN CHANGES
I. REQUIREMENTS
§ 820.30(i) Design changes.
• Each manufacturer shall establish and maintain procedures for the identification,
documentation, validation or where appropriate verification, review, and approval
of design changes before their implementation.
Cross-reference to ISO 9001:1994 and ISO/DIS 13485 section 4.4.9 Design changes.
II. DISCUSSION AND POINTS TO CONSIDER
There are two principal administrative elements involved in controlling design changes:
• Document control—enumeration of design documents, and tracking their status
and revision history. Throughout this section, the term “document” is used in an
inclusive sense to mean all design documents, drawings, and other items of design
input or output which characterize the design or some aspect of it.
• Change control—enumeration of deficiencies and corrective actions arising from
verification and review of the design, and tracking their resolution prior to design
transfer.
For a small development project, an adequate process for managing change involves little
more than documenting the design change, performing appropriate verification and
validation, and keeping records of reviews. The main objectives are ensuring that:
• corrective actions are tracked to completion;
• changes are implemented in such a manner that the original problem is resolved
and no new problems are created; or if new problems are created, they are also
tracked to resolution; and
• design documentation is updated to accurately reflect the revised design.
For projects involving more than two persons, coordination and communication of design
changes become vitally important. In other words, manufacturers should take steps to
avoid the common situation where, for example, Jon and Marie agree to a make a change
but neglect to inform Pat of their decision.
Medical device manufacturers are usually quite comfortable with the processes of
document control and change control with respect to managing manufacturing documents.
The principles of these processes are reviewed in the following paragraphs. Subsequently,
we will explore how these may be applied to design activities.
Section I. Design Changes 3/11/97 Page 40
DOCUMENT CONTROL. The features of a manufacturing document control system
typically include the following:
• Documents should be identified (i.e., named and numbered) in accordance with
some logical scheme which links the documents to the product or component they
describe or depict and illuminates the drawing hierarchy.
• A master list or index of documents should be maintained which presents a
comprehensive overview of the documentation which collectively defines the
product and/or process.
• Approval procedures should be prescribed which govern entry of documents into
the document control system.
• A history of document revisions should be maintained.
• Procedures for distributing copies of controlled documents and tracking their
location should be prescribed.
• Files of controlled documents should be periodically inventoried to ensure that the
contents are up to date.
• A person or persons should be assigned specific responsibility to oversee and carry
out these procedures. It is desirable that the document control system be
administered by a person who is not directly involved with developing or using the
documents. For a small manufacturer, document control might be a part-time job
for a technician or clerical staff person. More typically, one or more librarians or
full-time clerical or paraprofessional employees are required to administer the
system.
• There should be a procedure for removal and deletion of obsolete documents.
CHANGE CONTROL. Manufacturing change control is usually implemented using a
set of standardized procedures similar to the following:
• A change request might be originated by a developer, manager, reviewer,
marketing representative, user, customer, quality assurance representative, or
production personnel, and identifies a design problem which the requester believes
should be corrected. Change requests are typically reviewed following the
manufacturer’s prescribed review process, and the request might be rejected,
deferred, or accepted.
• If a change request is accepted and corrective action is straightforward, a change
order might be issued on the spot to implement the change. The change order
pertains to an explicitly identified document or group of documents, and specifies
the detailed revision of the document content which will fix the identified problem.
• Often, the change request results in an assignment to developers to further study
the problem and develop a suitable corrective action. If the change is extensive,
wholesale revision of affected documents may be warranted in lieu of issuing
change orders.
• Change requests and change orders should be communicated to all persons whose
work might be impacted by the change.
Section I. Design Changes 3/11/97 Page 41
• It may not be practical to immediately revise documents affected by a change
order. Instead, the common practice is to distribute and attach a copy of the
change order to each controlled copy of the original document.
• Change control procedures should incorporate review and assessment of the
impact of the design change on the design input requirements and intended uses.
• A mechanism should be established to track all change requests and change orders
to ensure proper disposition.
• Change control procedures are usually administered by the document control staff.
APPLICATION OF DOCUMENT AND CHANGE CONTROLS TO DESIGN. The
design control system has to be concerned with the creation and revision of documents, as
well as the management of finished documents. Additional mechanisms are required to
provide needed flexibility while preserving the integrity of design documentation. These
additional mechanisms are embodied in the procedures for review and approval of various
documents.
It is important that the design change procedures always include re-verifying and re-
validating the design. Fortunately, most design changes occur early in the design process,
prior to extensive design validation. Thus, for most design changes, a simple inspection is
all that is required. The later in the development cycle that the change occurs, the more
important the validation review becomes. There are numerous cases when seemingly
innocuous design changes made late in the design phase or following release of the design
to market have had disastrous consequences.
For example, a manufacturer encountered problems in the field with a valve sticking in a
ventilator due to moisture in the breathing circuit. The problem was resolved by slightly
increasing the weight of the disc. Since the change was minor, minimal testing was
performed to verify the change. Subsequently, when the revised valves entered
production, significant numbers of valves began failing. Investigation revealed that the
heavier disc was causing the valve cage to separate due to higher inertia. This failure
mode was more serious than the original sticking problem, and resulted in a safety recall.
Section I. Design Changes 3/11/97 Page 42
Section J. Design History File 3/11/97 Page 43
SECTION J. DESIGN HISTORY FILE (DHF)
I. REQUIREMENTS
§ 820.30(j) Design history file.
• Each manufacturer shall establish and maintain a DHF for each type of device.
• The DHF shall contain or reference the records necessary to demonstrate that the
design was developed in accordance with the approved design plan and the
requirements of this part.
Cross-reference to ISO 9001:1994 and ISO/DIS 13485 section 4.16 Control of
quality records.
II. DEFINITIONS
§ 820.3(e) Design history file (DHF) means a compilation of records which describes the
design history of a finished device.
III. DISCUSSION AND POINTS TO CONSIDER
There is no specific requirement in ISO 9001 or ISO 13485 for a design history file.
However, in order to market a medical device in the United States, a manufacturer must
comply with the U. S. Food and Drug Administration (FDA) quality system regulation,
which requires a design history file. For this reason, some guidance is provided on the
U. S. FDA design history file.
Other national regulations require some form of documentation and records. Product
documentation required by Canada, Europe, and Japan contain certain elements of the
U. S. FDA design history file requirements without requiring all the elements to be
compiled in a file.
Virtually every section of the design control requirements specifies information which
should be recorded. The compilation of these records is sometimes referred to as the
design history file. Throughout this guidance document, suggestions are made when
warranted as to the form and content of documents contained in the design history file.
The primary beneficiary of the device history file is the device manufacturer. For example,
in one case, a microprocessor-controlled enteral feeding pump was reported to be
behaving erratically in the field. Some of the symptoms pointed to software problems. But
the manufacturer admitted that they did not possess a copy of the software source code
for the product. The software had been developed by a contractor who had delivered only
a master EPROM (memory chip) which was duplicated by the manufacturer to install the
Section J. Design History File 3/11/97 Page 44
software in each machine. The contractor had subsequently withdrawn following a
contractual dispute, leaving the manufacturer with no rights to the source code developed
by the contractor, and no practical way to maintain the software. For this and other
reasons, the product was the subject of a mandatory recall and all known units were
collected and destroyed.
This is admittedly an extreme case, but many similar cases have been documented in which
the manufacturer lacked design information necessary to validate a design and maintain it
throughout the product life cycle. This occurs for the most innocent of reasons—
contracts expire, companies reorganize, employees move on to new projects or new jobs.
Even when the designer is available, he or she may forget why a particular decision was
made years, months, or even weeks before. Since design decisions often directly affect the
well-being of device users and patients, it is to the manufacturer’s benefit to maintain the
knowledge base which forms a basis for the product design.
Except for small projects, it is unusual for all design history documents to be filed in a
single location. For example, many design engineers maintain laboratory notebooks which
are typically retained in the engineers’ personal files. In addition, the design history may
include memoranda and electronic mail correspondence which are stored at various
physical locations. Quality system plans applicable to a development project may reside in
the quality assurance department, while the chief engineer may be responsible for
maintaining design and development plans. These diverse records need not be
consolidated at a single location. The intent is simply that manufacturers have access to
the information when it is needed. If a manufacturer has established procedures for
multiple filing systems which together satisfy that intent, there is no need to create
additional procedures or records.
As an example of the level of detail which may be entailed, some manufacturers have
policies covering laboratory notebooks. Manufacturers typically find that without such
written procedures, a breakdown in communications eventually occurs, resulting in a loss
of control. These procedures might address the following points.
• Laboratory notebooks are the property of the manufacturer, not the individual.
• A separate notebook is to be maintained for each project, and surrendered to the
engineering librarian at the conclusion of the engineer’s active participation in the
project.
• Laboratory notebooks are to be surrendered if the employee leaves the company.
• Product development supervisors shall review employees’ laboratory notebooks at
specified intervals to ensure that records are complete, accurate, and legible.
There are no requirements on the location or organization of the design history file. In
some cases, especially for simple designs, the designer will assemble and maintain the
entire design history file. For larger projects, a document control system will likely be
established for design documents, and these files will likely be maintained in some central
location, usually within the product development department.
Section J. Design History File 3/11/97 Page 45
Based on the structure (or lack thereof) of the product development organization, more or
less extensive controls will be required. For example, company policy should state
unequivocally that all design history documentation is the property of the manufacturer,
not the employee or contractor. Design and development contracts should explicitly
specify the manufacturer’s right to design information and establish standards for the form
and content of design documentation. Finally, certain basic design information may be
maintained in a single project file in a specified location. This may include the following:
• Detailed design and development plan specifying design tasks and deliverables.
• Copies of approved design input documents and design output documents.
• Documentation of design reviews.
• Validation documentation.
• When applicable, copies of controlled design documents and change control
records.
END OF DOCUMENT: DCGD.DOC (Microsoft Word 7.0) or DCGD.PDF (Adobe Acrobat format)
