UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
For the fiscal year ended December 31, 2011
For the transition period from to
Commission file number 1-3619
PFIZER INC.
(Exact name of registrant as specified in its charter)
(212) 733-2323
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes No
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes No
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during
the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past
90 days. Yes No
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Website, if any, every Interactive Data File required to be
submitted and posted pursuant to Rule 405 of Regulation S-T (§232-405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant
was required to submit and post such files.) Yes No
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of
registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K.
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer or a smaller reporting company. See the
definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes No
The aggregate market value of the voting stock held by non-
affiliates of the registrant, computed by reference to the closing price as of the last business day of the
registrant’s most recently completed second fiscal quarter, July 1, 2011, was approximately $163 billion. The registrant has no non-voting common stock.
The number of shares outstanding of the registrant’s common stock as of February 21, 2012 was 7,538,520,276 shares of common stock, all of one class.
DOCUMENTS INCORPORATED BY REFERENCE
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
Delaware
13
-
5315170
(State or other jurisdiction of
incorporation or organization)
(I.R.S. Employer
Identification Number)
235 East 42nd Street
10017
-
5755
New York, New York
(Address of principal executive offices)
(Zip Code)
Title of each class
Name of each exchange
on which registered
Common Stock, $.05 par value
New York Stock Exchange
Large accelerated filer
Accelerated filer
Non
-
accelerated filer
Smaller reporting company
Portions of the 2011 Annual Report to Shareholders
Parts
I, II and IV
Portions of the Proxy Statement for the 2012 Annual Meeting of Shareholders
Part III

TABLE OF CONTENTS
Page
PART I
1
ITEM 1. BUSINESS
1
General
1
Pfizer Website
1
Operating Segments
2
Biopharmaceutical Products
4
Other Products
8
Research and Development
10
International Operations
11
Marketing
12
Patents and Intellectual Property Rights
13
Competition
15
Raw Materials
18
Government Regulation and Price Constraints
18
Environmental Law Compliance
26
Tax Matters
26
Employees
26
ITEM 1A. RISK FACTORS
27
ITEM 1B. UNRESOLVED STAFF COMMENTS
37
ITEM 2. PROPERTIES
37
ITEM 3. LEGAL PROCEEDINGS
38
ITEM 4. MINE SAFETY DISCLOSURES
38
EXECUTIVE OFFICERS OF THE COMPANY
39
PART II
42
ITEM 5. MARKET FOR THE COMPANY
’
S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES
OF EQUITY SECURITIES
42
ITEM 6. SELECTED FINANCIAL DATA
42
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
42
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
43
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
43
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
43
ITEM 9A. CONTROLS AND PROCEDURES
43
ITEM 9B. OTHER INFORMATION
43
PART III
44
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
44
ITEM 11. EXECUTIVE COMPENSATION
44
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER
MATTERS
44
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
44
ITEM 14. PRINCIPAL ACCOUNTING FEES AND SERVICES
44
PART IV
45
ITEM 15. EXHIBITS, FINANCIAL STATEMENT SCHEDULES
45
15(a)(1) Financial Statements
45
15(a)(2) Financial Statement Schedules
45
15(a)(3) Exhibits
45

PART I
General
Pfizer Inc. (which may be referred to as Pfizer, the Company, we, us or our ) is a research-based, global biopharmaceutical company. We apply science and our
global resources to improve health and well-being at every stage of life. We strive to set the standard for quality, safety and value in the discovery, development and
manufacturing of medicines for people and animals. Our diversified global healthcare portfolio includes human and animal biologic and small molecule medicines and
vaccines, as well as nutritional products and many of the world’s best-known consumer healthcare products. Every day, we work across developed and emerging
markets to advance wellness, prevention, treatments and cures that challenge the most feared diseases of our time. We also collaborate with other biopharmaceutical
companies, healthcare providers, governments and local communities to support and expand access to reliable, affordable healthcare around the world.
The Company was incorporated under the laws of the State of Delaware on June 2, 1942.
On October 15, 2009, we completed our acquisition of Wyeth. The acquisition was a cash-and-stock transaction valued at $50.40 per share of Wyeth common
stock, or a total of approximately $68.2 billion, based on the closing market price of Pfizer common stock on the acquisition date.
On January 31, 2011, we completed a tender offer for the outstanding shares of common stock of King Pharmaceuticals, Inc. (King) and acquired approximately
92.5% of the outstanding shares for approximately $3.3 billion in cash. On February 28, 2011, we acquired the remaining outstanding shares of King for approximately
$300 million in cash. Commencing from January 31, 2011, our financial statements include the assets, liabilities, operating results and cash flows of King. Therefore, in
accordance with our domestic and international reporting periods, our consolidated financial statements for the fiscal year ended December 31, 2011 reflect
approximately 11 months of King’s U.S. operations and 10 months of King’s international operations.
In July 2011, we announced our decision to explore strategic alternatives for our Animal Health and Nutrition businesses, which may include, among other things,
a full or partial separation of each of these businesses from Pfizer through a spin-off, sale or other transaction. We believe these potential actions may create greater
shareholder value, enable us to become a more focused organization and optimize capital allocation. Given the separate and distinct nature of Animal Health and
Nutrition, we may pursue a different strategic alternative for each of these businesses. Although the timeline for each evaluation may differ, we expect to announce our
strategic decision for each of these businesses in 2012 and to complete any separation of these businesses between July 2012 and July 2013. For additional information,
see the Overview of Our Performance, Operating Environment, Strategy and Outlook – Our Business Development Initiatives section of Management’s Discussion and
Analysis of Financial Condition and Results of Operations
(MD&A) in our 2011 Financial Report.
On August 1, 2011, we completed the sale of our Capsugel business for approximately $2.4 billion in cash. For additional information, see the Notes to
Consolidated Financial Statements – Note 2D. Acquisitions, Divestitures, Collaborative Arrangements and Equity Method Arrangements – Divestitures in our 2011
Financial Report , as well as Other Products – Capsugel below.
Pfizer Website
Our Annual Report on Form 10-K for the fiscal year ended December 31, 2011 (2011 Form 10-K), Quarterly Reports on Form 10-Q, Current Reports on Form 8-
K and amendments to those reports filed or
1
ITEM 1. BUSINESS

furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended (Exchange Act), are available on our website (www.pfizer.com)
, in text
format and, where applicable, in interactive data file format , as soon as reasonably practicable after we electronically file such material with, or furnish it to, the
Securities and Exchange Commission (SEC).
Throughout this 2011 Form 10-K, we “incorporate by reference” certain information from other documents filed or to be filed with the SEC, including our Proxy
Statement for the 2012 Annual Meeting of Shareholders (2012 Proxy Statement) and the 2011 Financial Report, portions of which are filed as Exhibit 13 to this 2011
Form 10-
K, and which also will be contained in Appendix A to our 2012 Proxy Statement (2011 Financial Report). The SEC allows us to disclose important information
by referring to it in that manner. Please refer to such information. Our 2011 Annual Report to Shareholders consists of the 2011 Financial Report and the Corporate and
Shareholder Information attached to the 2012 Proxy Statement. Our 2011 Financial Report will be available on our website (www.pfizer.com) on or about February 28,
2012. Our 2012 Proxy Statement will be available on our website (www.pfizer.com) on or about March 15, 2012.
Information relating to corporate governance at Pfizer, including our Corporate Governance Principles; Director Qualification Standards; Pfizer Policies on
Business Conduct (for all of our employees, including our Chief Executive Officer, Chief Financial Officer and Principal Accounting Officer); Code of Business
Conduct and Ethics for our Directors; information concerning our Directors; ways to communicate by e-mail with our Directors; Board Committees; Committee
Charters; the Lead Independent Director Charter; and transactions in Pfizer securities by Directors and Officers; as well as Chief Executive Officer and Chief Financial
Officer certifications, are available on our website (www.pfizer.com)
. We will provide any of the foregoing information without charge upon written request to Matthew
Lepore, Vice President and Corporate Secretary, Chief Counsel-Corporate Governance, Pfizer Inc., 235 East 42nd Street, New York, NY 10017-5755. Information
relating to shareholder services, including our Shareholder Investment Program, book-entry share ownership and direct deposit of dividends, is also available on our
website (www.pfizer.com) .
The information contained in our website does not constitute a part of this 2011 Form 10-K.
Operating Segments
We manage our operations through five operating segments—Primary Care; Specialty Care and Oncology; Established Products and Emerging Markets; Animal
Health and Consumer Healthcare; and Nutrition. Each operating segment has responsibility for its commercial activities and for certain research and development
activities related to in-line products and in-process research and development (IPR&D) projects that generally have achieved proof-of-concept. Previously, we managed
our operations through two operating segments—Biopharmaceutical and Diversified.
We regularly review our segments and the approach used by management to evaluate performance and allocate resources.
A description of each of our five operating segments follows:
Through the end of 2011, sales of Lipitor in the U.S. are reported in our Primary Care business unit. Beginning in 2012, sales of Lipitor in the U.S. will be
reported in our Established Products business unit.
2
•
Primary Care operating segment—includes revenues from human pharmaceutical products primarily prescribed by primary-care physicians, and may
include products in the following therapeutic and disease areas: Alzheimer’s disease, cardiovascular (excluding pulmonary arterial hypertension), erectile
dysfunction, genitourinary, major depressive disorder, pain, respiratory and smoking cessation. Examples of products in this segment include Celebrex ,
Chantix/Champix , Lipitor , Lyrica , Premarin , Pristiq and Viagra . All revenues for such products are allocated to the Primary Care business unit, except
those generated in emerging markets and those that are managed by the Established Products business unit.

For a further discussion of our operating segments, including certain costs that are not allocated to our operating segment results, as well as comparative segment
information for 2011, 2010 and 2009, see the Notes to Consolidated Financial Statements – Note 18. Segment, Geographic and Other Revenue Information – Segment
3
•
Specialty Care and Oncology operating segment
—
comprises the Specialty Care business unit and the Oncology business unit.
Specialty Care—includes revenues from most human pharmaceutical products primarily prescribed by physicians who are specialists, and may
include products in the following therapeutic and disease areas: anti-infectives, endocrine disorders, hemophilia, inflammation, multiple sclerosis,
ophthalmology, pulmonary arterial hypertension, specialty neuroscience and vaccines. Examples of products in this business unit include
BeneFIX
, Enbrel , Genotropin , Geodon , the Prevnar/Prevenar franchise, Rebif , ReFacto AF , Revatio , Xalatan , Xyntha and Zyvox . All
revenues for such products are allocated to the Specialty Care business unit, except those generated in emerging markets and those that are
managed by the Established Products business unit.
Oncology—includes revenues from human prescription pharmaceutical products addressing oncology and oncology-
related illnesses. Examples of
products in this business unit include Aromasin , Sutent , Torisel and Xalkori . All revenues for such products are allocated to the Oncology
business unit, except those generated in emerging markets and those that are managed by the Established Products business unit.
•
Established Products and Emerging Markets operating segment—comprises the Established Products business unit and the Emerging Markets business
unit.
Established Products—
generally includes revenues from human prescription pharmaceutical products that have lost patent protection or marketing
exclusivity in certain countries and/or regions. Typically, products are transferred to this business unit in the beginning of the fiscal year following
loss of patent protection or marketing exclusivity. In certain situations, products may be transferred to this business unit at a different point than
the beginning of the fiscal year following loss of patent protection or marketing exclusivity in order to maximize their value. This business unit
also excludes revenues generated in emerging markets. Examples of products in this business unit include Arthrotec , Effexor , Medrol , Norvasc ,
Protonix
,
Relpax
and
Zosyn/Tazocin
.
Emerging Markets––includes revenues from all human prescription pharmaceutical products sold in emerging markets, including Asia (excluding
Japan and South Korea), Latin America, Middle East, Africa, Central and Eastern Europe and Turkey.
•
Animal Health and Consumer Healthcare operating segment
—
comprises the Animal Health business unit and the Consumer Healthcare business unit.
Animal Health––includes worldwide revenues from products and services to prevent and treat disease in livestock and companion animals,
including vaccines, parasiticides and anti
-
infectives.
Consumer Healthcare––generally includes worldwide revenues from non-prescription products in the following therapeutic categories: dietary
supplements, pain management, respiratory and personal care. Products marketed by Consumer Healthcare include Advil , Caltrate , Centrum ,
ChapStick
,
Preparation H
and
Robitussin
.
•
Nutrition operating segment
––generally includes revenues from a full line of infant and toddler nutritional products sold outside the U.S. and Canada.
Examples of products in this segment include the
S
-
26
and
SMA
product lines, as well as formula for infants with special nutritional needs.

Information,
including the tables therein captioned Selected Income Statement Information, Geographic Information and Significant Product Revenues in our 2011
Financial Report and the table captioned Revenues by Segment and Geographic Area in the MD&A in our 2011 Financial Report. The information from those tables in
our 2011 Financial Report is incorporated by reference into this 2011 Form 10-K.
Our businesses are heavily regulated in most of the countries in which we operate. In the U.S., the principal authority regulating our operations is the U.S. Food
and Drug Administration (FDA). The FDA regulates the safety and efficacy of the products we offer and our research quality, manufacturing processes, product
promotion, advertising and product labeling. Similar regulations exist in most other countries, and in many countries the government also regulates our prices. See
Government Regulation and Price Constraints below.
Biopharmaceutical Products
Revenues from biopharmaceutical products contributed approximately 86% of our total revenues in 2011, 87% of our total revenues in 2010 and 92% of our total
revenues in 2009.
We recorded direct product sales of more than $1 billion for each of 12 biopharmaceutical products in 2011, each of 15 biopharmaceutical products in 2010 and
each of nine legacy Pfizer biopharmaceutical products in 2009. These products represented 56% of our revenues from biopharmaceutical products in 2011, 60% of our
revenues from biopharmaceutical products in 2010, and 56% of our revenues from biopharmaceutical products in 2009. See Item 1A. Risk Factors –
Dependence on Key
In
-Line Products below.
Worldwide revenues from biopharmaceutical products in 2011 were $57.7 billion, a decrease of 1% compared to 2010, primarily due to the decrease of $4.7
billion in operational revenues from Lipitor , Effexor , Protonix , Xalatan , Caduet , Vfend , Aromasin and Zosyn , and lower Alliance revenues for Aricept , all due to
loss of exclusivity in certain markets, and a reduction in revenues of $359 million due to the Patient Protection and Affordable Care Act, as amended by the Health Care
and Education Reconciliation Act (commonly referred to as the Affordable Care Act, or ACA). This decrease was partially offset by the solid performance of Lyrica ,
the Prevnar/Prevenar franchise and Enbrel , the inclusion of operational revenues from legacy King products of approximately $950 million, which favorably impacted
biopharmaceutical revenues by 2%, and the favorable impact of foreign exchange of $1.7 billion, or 3%.
Geographically, in the U.S., revenues from biopharmaceutical products decreased 9% in 2011, compared to 2010, reflecting lower revenues from Lipitor ,
Protonix
, Effexor , Zosyn , Xalatan , Vfend , Caduet and Aromasin , all due to loss of exclusivity, lower Alliance revenues due to loss of exclusivity of Aricept 5mg and
10mg tablets in November 2010 and lower revenues from Detrol/Detrol LA
, as well as the reduction in revenues of $359 million in 2011 due to the ACA. The impact of
these adverse factors was partially offset by the strong performance of certain other biopharmaceutical products and the addition of U.S. revenues from legacy King
products of approximately $904 million in 2011.
For additional information regarding the impact of the ACA on our revenues, see the Overview of our Performance, Operating Environment, Strategy and
Outlook – Our Operating Environment – U.S. Healthcare Legislation section of the MD&A in our 2011 Financial Report.
In our international markets, revenues from biopharmaceutical products increased 5% in 2011, compared to 2010, reflecting the favorable impact of foreign
exchange of 6% in 2011, partially offset by a net operational decrease. Operationally, revenues were favorably impacted by increases in the Prevenar franchise, Lyrica ,
Enbrel
, Celebrex and Alliance revenues and unfavorably impacted by declines in Lipitor , Effexor , Norvasc and Xalatan/Xalacom . International revenues from legacy
King products were not significant to our international revenues in 2011. During 2011, international revenues from biopharmaceutical products represented 59% of total
revenues from biopharmaceutical products, compared to 56% in 2010.
For additional information, see the Analysis of the Consolidated Statements of Income – Biopharmaceutical Revenues
section of the MD&A in our 2011 Financial
Report.
4

Biopharmaceutical—Selected Product Descriptions:
We currently are conducting the Community-Acquired Pneumonia Immunization Trial in Adults (CAPiTA) to fulfill requirements in connection with the FDA’s
approval of the Prevnar 13 adult indication under its accelerated approval program. CAPiTA is an efficacy trial involving subjects 65 years of age and older that
is designed to evaluate whether Prevnar 13 is effective in preventing the first episode of community-
acquired pneumonia caused by the serotypes contained in the
vaccine. We estimate that this event-driven trial will be completed in 2013. At its regular meeting held on February 22, 2012, the U.S. Centers for Disease
Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) indicated that it will defer voting on a recommendation for the routine use of
Prevnar 13 in adults 50 years of age and older until the results of CAPiTA, as well as data on the impact of pediatric use of Prevnar 13 on the disease burden and
serotype distribution among adults, are available. We expect that the rate of uptake for the use of Prevnar 13 in adults 50 years of age and older will be impacted
by ACIP’s decision to defer voting on a recommendation for the routine use of Prevnar 13 by that population.
5
•
Lipitor
, for the treatment of elevated LDL-cholesterol levels in the blood, lost U.S. exclusivity on November 30, 2011, and faces generic competition in the U.S.
Lipitor
lost exclusivity in Australia in February 2012; in Japan in 2011; and in Brazil, Canada, Spain and Mexico in 2010; and it has lost exclusivity in nearly all
emerging market countries. We do not expect that Lipitor revenues in emerging markets will be materially impacted over the next several years by the loss of
exclusivity. Lipitor will have lost exclusivity in the majority of European markets by May 2012. See Patents and Intellectual Property Rights below for further
information on
Lipitor
.
•
Lyrica
is indicated for the management of post-herpetic neuralgia, neuropathic pain associated with diabetic peripheral neuropathy, the management of
fibromyalgia, and as adjunctive therapy for adult patients with partial onset seizures in the U.S., and for neuropathic pain (peripheral and central), adjunctive
treatment of epilepsy and general anxiety disorder in certain countries outside the U.S.
•
Prevnar 13/Prevenar 13
is our 13-valent pneumococcal conjugate vaccine for the prevention of various syndromes of pneumococcal disease in infants and young
children and in adults 50 years of age and older. Prevnar 13/Prevenar 13
for use in infants and young children has been launched in the U.S. for the prevention of
invasive pneumococcal disease caused by the 13 serotypes in Prevnar 13 and otitis media caused by the seven serotypes in Prevnar , and in the European Union
(EU) and many other international markets for the prevention of invasive pneumococcal disease, otitis media and pneumococcal pneumonia caused by the
vaccine serotypes. The launch of the Prevnar 13/Prevenar 13 pediatric indication has reduced our Prevnar/Prevenar (7-valent) revenues (see discussion below),
and we expect this trend to continue. In addition, in 2011, we received approval of Prevnar 13/Prevenar 13 for use in adults 50 years of age and older in the U.S.
for the prevention of pneumococcal pneumonia and invasive pneumococcal disease caused by the 13 serotypes in Prevnar 13
, and in the EU for the prevention of
invasive pneumococcal disease caused by the vaccine serotypes. Prevenar 13 for use in adults 50 years of age and older also has been approved in many other
international markets. We expect to commence commercial launches for the adult indication in 2012.
•
Enbrel
is our treatment for moderate-to-severe rheumatoid arthritis, polyarticular juvenile rheumatoid arthritis, psoriatic arthritis, plaque psoriasis and ankylosing
spondylitis, a type of arthritis affecting the spine. Under our co-promotion agreement with Amgen Inc. (Amgen), we and Amgen co-promote Enbrel in the U.S.
and Canada and share in the profits from Enbrel sales in those countries, which we include in Alliance revenues. Our co-promotion agreement with Amgen will
expire in October 2013, and, subject to the terms of the agreement, we are entitled to a royalty stream for 36 months thereafter, which we expect will be
significantly less than our current share of Enbrel profits from U.S. and Canadian sales. Following the end of the royalty period, we will not be entitled to any
further revenues from Enbrel sales in the U.S. and Canada. Our exclusive rights to Enbrel outside the U.S. and Canada will not be affected by the expiration of
the co
-
promotion agreement with Amgen.

6
•
Celebrex
is for the treatment of the signs and symptoms of osteoarthritis and rheumatoid arthritis worldwide and for the management of acute pain in adults in the
U.S. and certain markets in the EU. Celebrex is supported by continued educational and promotional efforts highlighting its efficacy and safety profile for
appropriate patients.
•
Viagra remains the leading treatment for erectile dysfunction and one of the world’s most recognized pharmaceutical brands after more than a decade. Viagra
began facing generic competition in certain markets, including Spain and Finland, in December 2009.
•
Norvasc
, for treating hypertension, lost exclusivity in the U.S. and other major markets in 2007 and in Canada in 2009.
•
Zyvox
is the world
’
s best selling agent among those used to treat serious Gram
-
positive pathogens, including methicillin
-
resistant staphylococcus
-
aureus.
•
Xalabrands
consists of Xalatan , a prostaglandin, which is a branded agent used to reduce elevated eye pressure in patients with open-angle glaucoma or ocular
hypertension, and Xalacom , a fixed combination of prostaglandin ( Xalatan ) and beta blocker (timolol), available outside the U.S. Xalatan lost exclusivity in the
U.S. in March 2011.
Xalatan
and
Xalacom
lost exclusivity in 15 major European markets in January 2012.
•
Sutent is for the treatment of advanced renal cell carcinoma, including metastatic renal cell carcinoma (mRCC) and gastrointestinal stromal tumors (GIST) after
disease progression on, or intolerance to, imatinib mesylate. In May 2011, the FDA approved Sutent for the treatment of progressive, well-differentiated
pancreatic neuroendocrine tumors (pNET) in patients with unresectable locally advanced or metastatic disease. In the U.S., Sutent is the most prescribed oral
mRCC therapy, and more than 100,000 patients have been treated with
Sutent
worldwide.
•
Geodon/Zeldox, an atypical antipsychotic, is indicated for the treatment of schizophrenia, as monotherapy for the acute treatment of bipolar manic or mixed
episodes, and as an adjunct to lithium or valproate for the maintenance treatment of bipolar disorder. Geodon/Zeldox is expected to lose exclusivity in the U.S. in
March 2012.
•
Our
Premarin
family of products remains the leading therapy to help women address moderate to severe menopausal symptoms.
•
Genotropin , one of the world’s leading human growth hormones, is used in children for the treatment of short stature with growth hormone deficiency, Prader-
Willi Syndrome, Turner Syndrome, Small for Gestational Age Syndrome, Idiopathic Short Stature (in the U.S. only) and Chronic Renal Insufficiency (outside the
U.S. only), as well as in adults with growth hormone deficiency. Genotropin is supported by a broad platform of innovative injection-delivery devices and patient
support programs.
•
Detrol/Detrol LA
, a muscarinic receptor antagonist, is one of the most prescribed branded medicines worldwide for overactive bladder . Detrol LA is an
extended
-
release formulation taken once a day.
Detrol
immediate release (
Detrol IR
) will lose exclusivity in the U.S. in September 2012.
•
Vfend
is a broad
-
spectrum agent for treating yeast and molds.
Vfend
tablets lost exclusivity in the U.S. in February 2011.
•
Chantix/Champix is an aid to smoking-cessation treatment in adults 18 years of age and older. We are continuing our educational and promotional efforts, which
are focused on addressing the significant health consequences of smoking, highlighting the Chantix benefit-risk proposition and emphasizing the importance of
the physician
-
patient dialogue in helping patients quit smoking.

In July 2011, the U.S. prescribing information was revised to include clinical data showing that Chantix is an effective aid to smoking-cessation treatment for
smokers with stable cardiovascular disease (CVD) and mild-to-moderate chronic obstructive pulmonary disease (COPD). The revised label also includes a
warning/precaution advising smokers with CVD to inform their physician of any new or worsening symptoms of cardiovascular disease, and to seek emergency
medical help if they experience any symptoms of a heart attack. This safety information was added at the FDA’s request following an observation of a small
numeric increase in certain cardiovascular events in patients treated with Chantix versus those taking a placebo in a study of 700 smokers with stable
cardiovascular disease. Approval of the EU labeling, revised at the European Medicines Agency’s (EMA’s) request to include a similar cardiovascular-related
warning/precaution, was received in late December 2011, with regulators reaffirming the positive benefit/risk profile of the medication. Approval of the Japan
labeling, which includes a similar precaution, occurred in late October 2011. In December 2011, Pfizer received a positive opinion from the EMA’s Committee
for Medical Products for Human Use for changes to the Champix EU label regarding schizophrenia data.
7
•
BeneFIX
and ReFacto AF / Xyntha are hemophilia products using state of the art manufacturing that assist patients with a lifelong bleeding disorder. BeneFIX is
the only available recombinant factor IX product for the treatment of hemophilia B, while ReFacto AF/Xyntha are recombinant factor VIII products for the
treatment of hemophilia A. Both products are indicated for the control and prevention of bleeding in patients with these disorders and in some countries also are
indicated for prophylaxis in certain situations, such as surgery.
•
Effexor
is an antidepressant for treating adult patients with major depressive disorder, generalized anxiety disorder, social anxiety disorder and panic disorder.
Effexor
and Effexor XR , an extended-release formulation, face generic competition in most markets, including the U.S., where Effexor XR lost exclusivity on
July 1, 2010. This generic competition has had a significant adverse impact on our revenues for
Effexor
and
Effexor XR
.
•
Zosyn/Tazocin
, our broad
-
spectrum intravenous antibiotic, faces generic global competition. U.S. exclusivity was lost in September 2009.
•
Pristiq
is approved for the treatment of major depressive disorder in the U.S. and in various other countries. Pristiq has also been approved for treatment of
moderate
-
to
-
severe vasomotor symptoms associated with menopause in Thailand, Mexico, the Philippines and Ecuador.
•
Caduet is a single pill therapy combining Lipitor and Norvasc for the prevention of cardiovascular events. Caduet lost U.S. exclusivity on November 30, 2011
and faces generic competition.
•
Revatio
is for the treatment of pulmonary arterial hypertension. In the U.S., Revatio tablet will lose exclusivity in September 2012, and Revatio IV injection will
lose exclusivity in May 2013.
•
Prevnar/Prevenar
( 7-valent ) is our 7-valent pneumococcal conjugate vaccine for preventing invasive, and, in certain international markets, non-invasive
pneumococcal disease in infants and young children. Many markets have transitioned from the use of Prevnar/Prevenar ( 7-valent ) to Prevnar 13/Prevenar 13
(see discussion above).
•
Aricept
, discovered and developed by Eisai Co., Ltd. (Eisai), is the most commonly dispensed medicine to treat symptoms of Alzheimer’s disease. We co-
promote Aricept with Eisai in the U.S. and several other countries and have an exclusive license to sell Aricept in certain other countries. Revenues associated
with this co-promotion are included in Alliance revenues. We lost exclusivity for Aricept
5mg and 10mg tablets in the U.S. in November 2010. We expect that the
Aricept
23mg tablet will have exclusivity in the U.S. until July 2013. Aricept lost exclusivity in many of the major European markets in February 2012, and our
Established Products business unit is introducing a second brand of donepezil HCl (the active ingredient in Aricept ) in Europe. Aricept will have exclusivity in
Canada until December 2013, and our rights to
Aricept
in Japan will return to Eisai in December 2012.

Embeda
– On February 23, 2011, we stopped distribution of our Embeda product due to failed specification tolerances related to naltrexone degradation identified in
post-manufacturing testing. On March 10, 2011, we initiated a voluntary recall to wholesale and retail customers of all Embeda
products. We are committed to returning
this important product to the market as quickly as possible, once the stability issue is resolved.
Other Products
Animal Health
Our Animal Health business unit is the largest animal health business in the world. We discover, develop and sell products for the prevention and treatment of
diseases in livestock and companion animals. Revenues from Animal Health products were approximately $4.2 billion in 2011, an increase of 17% compared to 2010,
reflecting higher operational revenues of 14% and the favorable impact of foreign exchange of 3%. Operational revenues from Animal Health products were favorably
impacted by approximately $329 million, or 9%, due to the addition of revenues from legacy King animal health products. Legacy Pfizer products grew 7% primarily
driven by improving market conditions and resulting increased demand for products across the livestock business, as well as deeper market penetration in emerging
markets. This was partially offset by the adverse impact of required product divestitures in 2010 related to the acquisition of Wyeth.
We market vaccines, anti-infectives, anti-inflammatories, antiemetics and parasiticides, including the following products:
8
•
Spiriva is indicated in the U.S. for the long-term, once-daily, maintenance treatment of bronchospasm associated with COPD, including chronic bronchitis and
emphysema, and for reducing COPD exacerbations. We co-promote Spiriva with Boehringer Ingelheim (BI) in the U.S. and selected countries on a worldwide
basis. Revenues associated with this co-promotion are included in Alliance revenues. Our collaboration with BI for Spiriva will expire on a country-by-country
basis between 2012 and 2016. As a result, we expect to experience a graduated decline in revenues from Spiriva during that period. Our collaboration with BI for
Spiriva
will expire in the EU from 2012 and 2016, in 2014 in the U.S. and Japan, and by 2016 in all other countries where the collaboration exists.
•
Xalkori,
the first-ever therapy targeting anaplastic lymphoma kinase (ALK), for the treatment of patients with locally advanced or metastatic non-small cell lung
cancer that is ALK
-
positive as detected by an FDA
-
approved test, was approved by the FDA in August 2011.
•
Inlyta
was approved by the FDA in January 2012 for the treatment of patients with advanced renal cell carcinoma after failure of one prior systemic therapy.
•
Startect
is a novel dual
-
active parasiticide that delivers a broad spectrum control of parasitic worm infestation in sheep.
•
Improvac/Improvest
is a novel gonadotropin releasing factor (GnRF) product for swine that prevents boar taint.
•
Fostera PCV
is a vaccine that protects pigs against porcine circovirus.
•
Palladia
is a treatment of mast cell tumors, a common form of cancer that affects dogs; it works by killing tumor cells and by cutting off the blood supply to the
tumor.
•
Convenia
is an anti
-
infective for dogs and cats that delivers an assured full course of therapy from a single injection.
•
Cerenia
is a selective NK
-
1 receptor antagonist for the treatment and prevention of vomiting in dogs and for the prevention of motion sickness.
•
Revolution/Stronghold
is a topically administered parasiticide for dogs and cats that controls a number of different parasites such as fleas and heartworm.

The Company is exploring strategic alternatives for Animal Health, which may include, among other things, a full or partial separation from Pfizer through a
spin-off, sale or other transaction. See General above.
Consumer Healthcare
Our Consumer Healthcare business unit is the fifth-largest over-the-counter healthcare products business in the world and sells two of the ten largest selling over-
the-counter healthcare brands ( Centrum and Advil ) in the world. Consumer Healthcare revenues totaled $3.1 billion for 2011, an increase of 10% compared to 2010,
reflecting higher operational revenues of 8% and the favorable impact of foreign exchange of 2%. The operational revenue increase in 2011 was primarily driven by
increased sales of core brands including Advil , Caltrate and Robitussin , as well as the temporary voluntary withdrawal of Centrum in Europe in the third quarter of
2010. The Consumer Healthcare business unit holds strong positions in various geographic markets, with its highest revenue volume in the U.S., Canada, China,
Germany, Italy, Brazil and Australia.
Major categories and product lines include:
In December 2011 (which falls in the first fiscal quarter of 2012 for our international operations), we completed our acquisition of the consumer healthcare
business of Ferrosan Holding A/S, a Danish company engaged in the sale of science-based consumer healthcare products, including dietary supplements and lifestyle
products, primarily in the Nordic region and the emerging markets of Russia and Central and Eastern Europe.
Nutrition
Pfizer Nutrition is a leader in infant nutritionals in the markets in which we operate. We have a targeted geographic presence in key markets throughout Asia, the
Middle East, Europe and Latin America. Our largest markets include China, the Philippines, the U.K., Mexico and Australia, and more than 80% of our revenues are in
emerging markets. Since it became part of Pfizer in 2009, the Nutrition business has grown in new and existing markets through innovation and developing and
launching a number of new products. Nutrition’s revenues totaled $2.1 billion in 2011, an increase of 15% compared to 2010, reflecting higher operational revenues of
11% and the favorable impact of foreign exchange of 4%. The operational revenue increase was primarily due to increased demand for premium products, launches of
new products and strength in China and the Middle East.
Nutrition products include infant milk formula brands for newborns and toddlers: our Gold line includes brands S-26 and/or SMA
(brand names vary slightly from
country to country), and in 2011 we launched our super-premium Illuma brand. We also commercialize specialty formulas such as S-26 Gold Hypoallergenic , S-26
Gold Anti-Regurgitation , S-26 Gold Lactose-Free , and S-26 Picky Eater.
9
•
Rimadyl
relieves pain and inflammation associated with canine osteoarthritis and soft tissue orthopedic surgery.
•
Draxxin
is an effective and convenient single dose anti
-
infective used to treat infections in cattle and swine.
•
Excede
is an effective and convenient single-dose anti-infective used to treat infections in cattle and swine. Excede offers a convenient two-dose regimen for
horses.
•
Zulvac
is a vaccine that protects cattle and sheep against bluetongue disease.
•
Bopriva
is a novel GnRF vaccine which temporarily reduces undesirable bull behaviors such as fighting.
•
Dietary Supplements: Centrum brands (including Centrum , Centrum Silver , Centrum Men’s and Women’s , Centrum Performance , Centrum Specialists ,
Centrum Cardio
,
and
Centrum Kids
),
Caltrate
, and
ProNutrient
brands (including
Probiotic
,
Omega
-
3
, and
Fruit and Veggie
);
•
Pain Management:
Advil
brands (including
Advil
,
Advil PM
,
Advil Liqui
-
Gels
,
Children
’
s Advil
,
Infant
’
s Advil
,
and Advil Migraine)
,
and
ThermaCare
;
•
Respiratory:
Robitussin
,
Advil Cold & Sinus
,
Advil Congestion Relief
,
and
Dimetapp
;
•
Personal Care:
ChapStick
and
Preparation H.

The Company is exploring strategic alternatives for Nutrition, which may include, among other things, a full or partial separation from Pfizer through a spin-off,
sale or other transaction. See General above.
For additional information regarding the revenues of our Animal Health, Consumer Healthcare and Nutrition business units, see the Analysis of the Consolidated
Statements of Income – Other Product Revenues section of the MD&A in our 2011 Financial Report.
Capsugel
On August 1, 2011, we sold our Capsugel business for approximately $2.4 billion in cash. Results of Capsugel, as well as the gain on its sale, are reflected in
discontinued operations through the date of sale. Capsugel was a business that had a diverse product line of hard gelatin capsules, and liquid, softgel, non-animal, and
fish gelatin capsules, all for use in pharmaceutical and dietary supplement dosage delivery. For additional information, see the Notes to Consolidated Financial
Statements – Note 2D. Acquisitions, Divestitures, Collaborative Arrangements and Equity Method Arrangements – Divestitures in our 2011 Financial Report.
Research and Development
Innovation by our research and development operations is very important to our success. As a result, and also because we are predominantly a human health
company, the vast majority of our research and development spending is associated with human health products, compounds and activities. Our goal is to discover,
develop and bring to market innovative products that address major unmet medical needs. We spent $9.1 billion in 2011, $9.4 billion in 2010 and $7.8 billion in 2009 on
research and development.
Biopharmaceutical R&D
We conduct research internally and also through contracts with third parties, through collaborations with universities and biotechnology companies and in
cooperation with other pharmaceutical firms. We also seek out promising compounds and innovative technologies developed by third parties to incorporate into our
discovery and development processes or projects, as well as our product lines, through collaborations, alliance and license agreements, acquisitions and other
arrangements.
Drug discovery and development is time-consuming, expensive and unpredictable. According to the Pharmaceutical Research and Manufacturers of America
(PhRMA), out of 5,000-10,000 screened compounds, only 250 enter preclinical testing, five enter human clinical trials and one is approved by the FDA. The process
from early discovery or design to development to regulatory approval can take more than ten years. Drug candidates can fail at any stage of the process. Candidates may
not receive regulatory approval even after many years of research.
As of year-
end 2011, we had 262 projects in research and development, ranging from discovery through registration, of which 95 programs are in Phase 1 through
registration, with the remainder of the projects in pre-clinical development. At year-end 2011, our Phase III portfolio contained 22 programs. Development of a single
compound is often pursued as part of multiple different programs. While these new candidates may or may not eventually receive regulatory approval, new drug
candidates entering clinical development phases are the foundation for future products.
In addition to discovering and developing new products, our research operations seek to add value to our existing products by improving their effectiveness and
by discovering new uses or indications for them.
Information concerning several of our drug candidates in development, as well as supplemental filings for existing products, is set forth in the Analysis of the
Consolidated Statements of Income – Product Developments – Biopharmaceutical section of the MD&A in our 2011 Financial Report. That information is incorporated
by reference.
10

Our competitors also devote substantial funds and resources to research and development. We also compete against numerous small biotechnology companies in
developing potential drug candidates. The extent to which our competitors are successful in their research could result in erosion of the sales of our existing products and
products in development, as well as unanticipated product obsolescence.
We continue to closely evaluate our global research and development function and pursue strategies to improve innovation and overall productivity by prioritizing
areas with the greatest scientific and commercial promise, utilizing appropriate risk/return profiles, and focusing on areas with the highest potential to deliver value in
the near term and over time. To that end, our research primarily focuses on five high-priority areas that have a mix of small and large molecules – immunology and
inflammation; oncology; cardiovascular, metabolic and endocrine diseases; neuroscience and pain; and vaccines. In addition to reducing the number of disease areas of
focus, we are realigning and reducing our research and development footprint, and outsourcing certain functions that do not drive competitive advantage for Pfizer. As a
result of these actions, we expect significant reductions in our annual research and development expenses, which are reflected in our 2012 financial guidance, and we
expect to incur significant costs, which are also reflected in our 2012 financial guidance. For additional information, see the Overview of Our Performance, Operating
Environment, Strategy and Outlook
– Our Strategy and – Our Financial Guidance for 2012 and Costs and Expenses – Restructuring Charges and Other Costs
Associated with Acquisitions and Cost
-Reduction/Productivity Initiatives sections of the MD&A in our 2011 Financial Report.
For additional information regarding our R&D operations, see the Analysis of the Consolidated Statements of Income – Research and Development Operations
section of the MD&A in our 2011 Financial Report.
International Operations
We have significant operations outside the United States. They are managed through the same segments as our U.S. operations, with our operations in emerging
markets for human pharmaceutical products managed through the Established Products and Emerging Markets segment.
Revenues from operations outside the U.S. of $40.5 billion accounted for 60% of our total revenues in 2011. Revenues exceeded $500 million in each of 18
countries outside the U.S. in 2011. The U.S. is our largest national market, comprising 40% of total revenues in 2011, 43% of total revenues in 2010 and 44% of total
revenues in 2009. Japan is our second-largest national market, with 9% of total revenues in 2011, 7.5% of total revenues in 2010 and 8.7% of total revenues in 2009.
For a geographic breakdown of revenues and changes in revenues, see the table captioned Geographic Information, in the Notes to Consolidated Financial
Statements – Note 18. Segment, Geographic and Other Revenue Information in our 2011 Financial Report, and the table captioned Revenues by Segment and
Geographic Area in the MD&A in our 2011 Financial Report. Those tables are incorporated by reference.
Our international businesses are subject, in varying degrees, to a number of risks inherent in carrying on business in other countries. These include currency
fluctuations, capital and exchange control regulations, expropriation and other restrictive government actions. Our international businesses are also subject to
government-imposed constraints, including laws and regulations on pricing, reimbursement, and access to our products. See Government Regulation and Price
Constraints below for a discussion of these matters.
Depending on the direction of change relative to the U.S. dollar, foreign currency values can increase or decrease the reported dollar value of our net assets and
results of operations. In 2011, both revenues and net income were favorably impacted by foreign exchange in general, as foreign currency movements relative to the
U.S. dollar increased our revenues and net income in many countries. While we cannot predict with certainty future changes in foreign exchange rates or the effect they
will have on us, we attempt to mitigate their impact through operational means and by using various financial instruments, depending upon market conditions. See the
discussions in the Notes to Consolidated Financial Statements – Note 7E. Financial Instruments – Derivative Financial Instruments and Hedging Activities in our 2011
Financial Report.
11

Marketing
In our global biopharmaceutical businesses, we promote our products to healthcare providers and patients. Through our marketing organizations, we explain the
approved uses, benefits and risks of our products to healthcare providers, such as doctors, nurse practitioners, physician assistants, pharmacists, hospitals, Pharmacy
Benefit Managers (PBMs), Managed Care Organizations (MCOs), employers and government agencies. We also market directly to consumers in the U.S. through
direct-to-consumer advertising that communicates the approved uses, benefits and risks of our products while motivating people to have meaningful conversations with
their doctors. In addition, we sponsor general advertising to educate the public on disease awareness, prevention and wellness, important public health issues, and our
patient assistance programs.
Our biopharmaceutical businesses include five human health, customer-
focused business units: Primary Care, Specialty Care, Oncology, Established Products and
Emerging Markets. Our Specialty Care customer-focused business unit includes vaccines. We operate in customer-focused business units within our biopharmaceutical
businesses to better meet the diverse needs of physicians, patients and our customers while maximizing value for our company and our shareholders.
Our U.S. Primary Care operations are structured into regional units in order to create a more flexible organization empowered to identify and address local market
dynamics and customer needs. Our structure aligns the sales, marketing, and medical functions to work closely to meet the needs of key customer groups while ensuring
common coordination, focus and accountability across the organizations.
Our prescription pharmaceutical products are sold principally to wholesalers, but we also sell directly to retailers, hospitals, clinics, government agencies and
pharmacies, and in the case of Prevnar 13
, directly to individual provider offices in the U.S. We seek to gain access to healthcare authority, PBM and MCO formularies
(lists of recommended, approved, and/or reimbursed medicines and other products). We also work with MCOs, PBMs, employers and other appropriate healthcare
providers to assist them with disease management, patient education and other tools that help their medical treatment routines.
During 2011, Pfizer revenues from our three largest biopharmaceutical wholesalers were as follows:
Sales to these wholesalers were concentrated in the biopharmaceutical businesses. Apart from these instances, none of our operating segments is dependent on
any one customer or group of related customers.
Each of our global Animal Health, Consumer Healthcare and Nutrition business units utilizes its own sales and marketing organizations to promote its products,
and each occasionally uses distributors in smaller markets.
Our Animal Health business unit’s advertising and promotions are generally targeted to veterinary healthcare professionals. Animal Health products are sold
directly to veterinarians and livestock producers, as well as through distributors and retail outlets.
Our Consumer Healthcare business unit’s advertising and promotions are generally disseminated to consumers through television, print, digital and other media
advertising, as well as through in-store promotion. Consumer Healthcare products are sold through a wide variety of channels, including distributors, pharmacies, retail
chains and grocery and convenience stores.
Our Nutrition business unit fully supports and adheres to the World Health Organization Code and national codes on the marketing of breast milk substitutes.
Pfizer Nutrition encourages breastfeeding as the best nutrition
12
•
McKesson, Inc.
—
13% of our total revenues (and 30% of our total U.S. revenues);
•
Cardinal Health, Inc.
—
10% of our total revenues (and 26% of our total U.S. revenues); and
•
AmerisourceBergen Corporation
—
9% of our total revenues (and 21% of our total U.S. revenues).

for infants and provides important products for infants who are not exclusively breastfed. Advertising and promotion of our Nutrition products adhere to the aim and
principles of the World Health Organization Code as a minimum standard, consistent with national legislation. Consequently, we do not advertise breast milk
substitutes, but rather products intended for older children and adults, where permitted. Such activities are aimed at healthcare professionals and consumers through
media advertising (print, television, Internet). Our Nutrition products are available to consumers in a wide variety of channels, including pharmacies, hospitals and food
retailers (supermarkets, grocery stores and convenience stores).
Patents and Intellectual Property Rights
Our products are sold around the world under brand-name, logo and certain product design trademarks that we consider in the aggregate to be of material
importance. Trademark protection continues in some countries for as long as the mark is used and, in other countries, for as long as it is registered. Registrations
generally are for fixed, but renewable, terms.
We own or license a number of U.S. and foreign patents. These patents cover pharmaceutical and other products and their uses, pharmaceutical formulations,
product manufacturing processes and intermediate chemical compounds used in manufacturing.
Patents for individual products extend for varying periods according to the date of patent filing or grant and the legal term of patents in the various countries
where patent protection is obtained. The actual protection afforded by a patent, which can vary from country to country, depends upon the type of patent, the scope of its
coverage and the availability of legal remedies in the country. Further, patent term extension may be available in many major countries to compensate for a regulatory
delay in approval of the product.
In the aggregate, our patent and related rights are of material importance to our businesses in the U.S. and most other countries. Based on current product sales,
and considering the vigorous competition with products sold by others, the patent rights we consider most significant in relation to our business as a whole, together with
the year in which the U.S. basic product patent expires (including, where applicable, the additional six-month pediatric exclusivity period and/or the granted patent term
extension), are those for the drugs set forth in the table below. The basic patents for these products in other large markets may expire in the same, earlier or later years.
(1) With respect to the products in the table above, the corresponding European and Japanese patent expiration dates are generally within one year before or
after the U.S. dates indicated, except as follows:
13
Drug
U.S. Basic Product Patent
Expiration Year
Geodon
2012
Viagra
2012
Detrol
2012
Celebrex
2014
Zyvox
2015
Lyrica
2018
Chantix
2020
Sutent
2021
Xalkori
2029
•
With respect to Japan, the patent expiration year for Celebrex is 2019, for Zyvox is 2019, for Lyrica is 2022, for Champix is 2022, for Sutent is
2024 and for
Xalkori
(not currently approved) is 2025. For
Detrol
, post
-
marketing surveillance in Japan extends until 2014.
(1)

In some instances, there are later-expiring patents relating to our products directed to particular forms or compositions, to methods of manufacturing, or to use of
the drug in the treatment of particular diseases or conditions. For example, in addition to the basic product patent covering Viagra , it is also covered by a U.S. method
of treatment patent which, including the six-month pediatric exclusivity period associated with Revatio , which has the same active ingredient as Viagra , expires in
2020. However, in some cases, such patents may not protect our drug from generic competition after the expiration of the basic patent.
We co-promote Aricept with Eisai, Enbrel with Amgen and Spiriva with BI. See Biopharmaceutical Products – Biopharmaceutical – Selected Product
Descriptions
for a further discussion.
We lost exclusivity for Lipitor in the U.S. on November 30, 2011. Pfizer announced in June 2008 that we entered into an agreement providing a license to
Ranbaxy to sell generic versions of Lipitor and Caduet in the U.S effective November 30, 2011. In addition, the agreement provides a license for Ranbaxy to sell a
generic version of Lipitor beginning on varying dates in several additional countries. (See the Notes to Consolidated Financial Statements – Note 17. Commitments and
Contingencies for a discussion of certain litigation relating to this agreement.) We also granted Watson Pharmaceuticals, Inc. (Watson) the exclusive right to sell the
authorized generic version of Lipitor in the U.S. for a period of five years, which commenced on November 30, 2011. As Watson’s exclusive supplier, we manufacture
and sell generic atorvastatin tablets to Watson. We expect the entry of multi-source generic competition in the U.S., with attendant increased competitive pressures,
following the end of Ranbaxy’s 180-day generic exclusivity period in late May 2012. In markets outside the U.S., Lipitor has lost exclusivity in certain countries and
will lose exclusivity at various times in other countries. In Europe, although the Lipitor compound patent expired in November 2011, we have obtained pediatric
extensions in the EU. Accordingly, exclusivity in the majority of the major European markets has been extended for six months to May 2012. See the Notes to
Consolidated Financial Statements – Note 17. Commitments and Contingencies in our 2011 Financial Report regarding pending legal challenges to our Lipitor patents.
Xalatan lost exclusivity in the U.S. in March 2011. Xalatan and Xalacom lost exclusivity in 15 major European markets in January 2012.
Effexor/Effexor XR , Zosyn , Protonix , Xalatan , Norvasc and Vfend tablets are examples of other Pfizer products that currently face generic competition in the
U.S.
Companies have filed applications with the FDA seeking approval of products that we believe infringe our patents covering, among other products, Lipitor ,
Viagra , Detrol/Detrol LA , Lyrica , Tygacil , Sutent , Rapamune , Zyvox , Avinza , EpiPen , Torisel and Embeda. Wyeth, and a subsidiary of Wyeth, are defendants in a
lawsuit alleging that their ReFacto AF and Xyntha products infringe the patents of another company.
We also have other patent rights covering additional products that have lesser revenues than most of the products set forth in the table above. Of these, we also
lost exclusivity in the U.S. for Caduet in 2011 and in the U.S. and the EU for Aromasin in 2011.
The expiration of a basic product patent or loss of patent protection resulting from a legal challenge normally results in significant competition from generic
products against the originally patented product and can result in a significant reduction in sales of that product in a very short period. In some cases, however, we can
continue to obtain commercial benefits from product manufacturing trade secrets; patents on uses for products; patents on processes and intermediates for the
economical manufacture of the active ingredients; patents for special formulations of the product or delivery mechanisms; and conversion of the active ingredient to
over-the-counter products.
14
•
With respect to major European markets, the patent expiration year for Xalkori (not currently approved) is 2025. For Lyrica , regulatory
exclusivity in Europe extends until 2014.

Our biotechnology products, including Enbrel and the Prevnar family, may face competition from biosimilars (also referred to as “follow-on biologics”). Such
biosimilars would reference biotechnology products already approved under the U.S. Public Health Service Act. Abbreviated legal pathways for the approval of
biosimilars exist in certain international markets and since the passage of the ACA, a framework for such approval exists in the U.S. See Government Regulation and
Price Constraints
– Biosimilars below. Specifically, the ACA provided innovator biologics with 12 years of exclusivity, with a potential six-month pediatric extension.
After the exclusivity period expires, the FDA could approve biosimilar versions of innovator biologics. The regulatory implementation of these provisions is ongoing
and expected to take several years. However, the FDA has begun to clarify its expectations for approval via the biosimilar pathway with the recent issuance of three
draft guidance documents. Among other things, these draft guidance documents confirm that the FDA will allow biosimilar applicants to use a non-U.S. licensed
comparator in certain studies to support a demonstration of biosimilarity to a U.S.-licensed reference product. If competitors are able to obtain marketing approval for
biosimilars referencing our biotechnology products, our biotechnology products may become subject to competition from biosimilars, with the attendant competitive
pressures. Concomitantly, a better-defined biosimilars approval pathway will assist us in pursuing approval of our own biosimilar products in the U.S. See Item 1A. Risk
Factors
– Biotechnology Products below.
In Europe, the European Commission has granted marketing authorizations for several biosimilars pursuant to a set of general and product class-specific
guidelines for biosimilar approvals issued over the past few years, and in Japan, the regulatory authority has granted marketing authorizations for certain biosimilars,
including somatropin (the recombinant human growth hormone in our Genotropin product) pursuant to a guideline for biosimilar approvals issued in 2009. If
competitors are able to obtain marketing approval for biosimilars referencing our biotechnology products, our biotechnology products may become subject to
competition from biosimilars, with the attendant competitive pressure, and price reductions could follow. Expiration or successful challenge of applicable patent rights
could generally trigger this competition, assuming any relevant exclusivity period has expired.
We may face litigation with respect to the validity and/or scope of patents relating to our biotechnology products with substantial revenue. Likewise, as we enter
the biosimilars area and seek to launch products, patents may be asserted against us.
One of the main limitations on our operations in some countries outside the U.S. is the lack of effective intellectual property protection for our products. Under
international and U.S. free trade agreements in recent years, global protection of intellectual property rights has been improving. The World Trade Organization
Agreement on Trade Related Aspects of Intellectual Property (WTO-TRIPs) required participant countries to amend their intellectual property laws to provide patent
protection for pharmaceutical products by 2005 with an extension until 2016 for least-developed nations. A number of countries have made improvements. We have
experienced significant growth in our businesses in some of those nations, and our continued business expansion in other participant countries depends to a large degree
on further patent protection improvement.
Competition
Our businesses are conducted in intensely competitive and often highly regulated markets. Many of our human prescription pharmaceutical products face
competition in the form of branded drugs or generic drugs that treat similar diseases or indications. The principal forms of competition include efficacy, safety, ease of
use, and cost effectiveness. Though the means of competition vary among product categories and business groups, demonstrating the value of our products is a critical
factor for success in all of our principal businesses.
Our acquisition of Wyeth in October 2009 created a broader, more diverse portfolio and pipeline with industry-leading positions in potential high-growth areas,
further strengthened by new capabilities in biotechnology and vaccines. The addition of Wyeth not only strengthened our presence in the United States and Europe, but
also enhances our abilities to provide emerging markets in Asia, Latin America, Africa, the Middle East and Eastern Europe/Russia with high-quality, innovative
medicines.
15

Our competitors include other worldwide research-based drug companies, smaller research companies with more limited therapeutic focus, and generic drug and
consumer healthcare manufacturers. We compete with other companies that manufacture and sell products that treat similar diseases or indications as our major
products.
Such competition affects our core product business, which is focused on applying innovative science to discover and market products that satisfy unmet medical
needs and provide therapeutic improvements. Our emphasis on innovation is underscored by our multi-billion-dollar investment in research and development, as well as
our business development transactions over the past decade, resulting in a strong product pipeline. Our investment in research does not stop with drug approval; we
continue to invest in further understanding the value of our products for the conditions they treat, as well as potential new applications. We seek to protect the health and
well-being of patients by striving to ensure that medically sound knowledge of the benefits and risks of our medicines is understood and communicated to patients,
physicians and global health authorities. We also continue to enhance the organizational effectiveness of all of our biopharmaceutical functions, including coordinating
support for our salespersons’ efforts to accurately and ethically launch and promote our products to our customers.
Operating conditions have become more challenging under the mounting global pressures of competition, industry regulation and cost containment. We continue
to take measures to evaluate, adapt and improve our organization and business practices to better meet customer and public needs. For instance, our U.S. Primary Care
operations are structured into regional units in order to create a more flexible organization empowered to identify and address local market dynamics and customer
needs. We have taken an industry-leading role in evolving our approaches to U.S. direct-to-consumer advertising, interactions with, and payments to, healthcare
professionals and medical education grants. We also continue to sponsor programs to address patient affordability and access barriers, as we strive to advance
fundamental health system change through support for better healthcare solutions.
While our Animal Health business unit is the leading animal health company with market positions throughout the world, many other companies offer competing
products. Altogether, there are hundreds of producers of animal health products throughout the world. The principal methods of competition vary somewhat depending
on the particular product. They include product innovation, quality, price, service and effective promotion to veterinary professionals and livestock producers.
Our Consumer Healthcare business unit faces competition from over-the-counter business units in other major pharmaceutical and consumer packaged goods
companies, as well as retailers who carry their own private label brands. Our competitive position is affected by several factors, including the amount and effectiveness
of our and our competitors’ promotional resources; customer acceptance; product quality; our and our competitors’ introduction of new products, ingredients, claims,
dosage forms, or other forms of innovation; and pricing, regulatory and legislative matters (e.g., product labeling, patient access, prescription to over-the-counter
switches, etc.).
Our Nutrition business unit competes with multinational companies in the pharmaceutical and food industries, as well as numerous smaller local and regional
companies manufacturing infant nutrition products. Our competitive position is affected by several factors, particularly the amount of resources deployed to develop,
enhance and promote products; the rapid pace of product and packaging innovation in the category, including scientific and technological advances; overall product
quality; the effectiveness of our promotional efforts; and product launch campaigns. All of these factors drive brand recommendations from healthcare professionals and
overall customer preference for our product brands.
Managed Care Organizations
The growth of MCOs in the U.S. has been a major factor in the competitive makeup of the healthcare marketplace. Approximately 250 million people in the U.S.
now participate in some form of managed care.
16

Because of the size of the patient population covered by MCOs, the marketing of prescription drugs to them and the PBMs that serve many of those organizations
continues to grow in importance.
MCOs can include medical insurance companies, medical plan administrators, health maintenance organizations, alliances of hospitals and physicians and other
physician organizations. The purchasing power of MCOs has increased in recent years due to the growing numbers of patients enrolled in MCOs. At the same time,
those organizations have been consolidating into fewer, even larger entities. This consolidation enhances both their purchasing strength and importance to us.
The growth of MCOs has increased pressure on drug prices. One objective of MCOs is to contain and, where possible, reduce healthcare expenditures. MCOs
typically use formularies, volume purchases and long-term contracts to negotiate discounts from pharmaceutical providers. Also, MCOs use their purchasing power and
their ability to influence market share and volume of prescription drugs to bargain for lower supplier prices. They also emphasize primary and preventive care, out-
patient treatment and procedures performed at doctors’ offices and clinics. Hospitalization and surgery, typically the most expensive forms of treatment, are carefully
managed. Since the use of certain drugs can reduce the need for hospitalization, professional therapy, or even surgery, such drugs can become favored first-line
treatments for certain diseases.
As discussed above in Marketing , MCOs and PBMs typically develop formularies, which are selections of medicines available to members that are tiered
according to co-pay amounts. Formularies are typically based on the prices and therapeutic benefits, or a combination of the two, of the available products. Due to their
generally lower cost, generic medicines typically are placed in lowest cost tiers. The breadth of the products covered by formularies can vary considerably from one
MCO to another and many formularies include alternative and competitive products for treatment of particular medical problems. MCOs use a variety of means to
encourage patients’ use of products listed on their formularies.
Exclusion of a product from a formulary or other MCO-implemented restriction can significantly impact drug usage in the MCO patient population.
Consequently, pharmaceutical companies compete aggressively to gain access to formularies for their products. Unique product features, such as greater efficacy, better
patient ease of use, or fewer side effects, are generally beneficial to achieving access to formularies. However, lower overall cost of therapy is also an important factor.
We have been generally, although not universally, successful in having our major products included on most MCO formularies.
The impact of MCOs on drug prices and volumes has increased as a result of their role in negotiating on behalf of Medicare beneficiaries in connection with the
Medicare Outpatient Prescription Drug Benefit, Medicare Part D, which took effect January 1, 2006. MCOs and PBMs negotiate on behalf of the federal government as
Prescription Drug Plans (PDPs). We have been generally, although not universally, successful in having our major products that are used by the senior population
included on the formularies of the Medicare PDPs in 2011.
Generic Products
One of the biggest competitive challenges that we face is from generic pharmaceutical manufacturers. Upon the expiration or loss of patent protection for a
product, especially a small molecule product, we can lose the major portion of sales of that product in a very short period. Several such competitors make a regular
practice of challenging our product patents before their expiration. Generic competitors operate without large research and development expenses, as well as without
costs of conveying medical information about products to the medical community, all of which we are required to do. In addition, the FDA approval process exempts
generics from costly and time-
consuming clinical trials to demonstrate their safety and efficacy, allowing generic manufacturers to rely on the safety and efficacy data of
the innovator product. Generic products need only demonstrate a level of availability in the body equivalent to that of the innovator product. This means that generic
competitors can market a competing version of our product after the expiration or loss of our patent and often charge much less.
17

In addition, our patent-protected products can face competition in the form of generic versions of competitors’
branded products that lose their market exclusivity.
As noted above, MCOs that focus primarily on the immediate cost of drugs often favor generics over brand-name drugs. Many governments also encourage the
use of generics as alternatives to brand-name drugs in their healthcare programs, including Medicaid in the U.S. Laws in the U.S. generally allow, and in some cases
require, pharmacists to substitute generic drugs that have been rated under government procedures to be therapeutically equivalent to brand-
name drugs. The substitution
must be made unless the prescribing physician expressly forbids it. In the U.S., Pfizer’s Greenstone subsidiary and Pfizer Injectables sell generic versions of Pfizer’s, as
well as certain competitors’, solid oral dose and sterile injectable pharmaceutical products, respectively, upon loss of exclusivity, as appropriate.
Raw Materials
Raw materials essential to our businesses are purchased worldwide in the ordinary course of business from numerous suppliers. In general, these materials are
available from multiple sources. No serious shortages or delays were encountered in 2011, and none are expected in 2012. However, select materials have, from time to
time, increased in price due to short-term imbalances between supply and demand. We have successfully secured the materials necessary to meet our requirements in
these circumstances but generally at higher prices than those historically paid.
Government Regulation and Price Constraints
In the United States
General. Pharmaceutical companies are subject to extensive regulation by national, state and local agencies in the countries in which they do business. Of
particular importance in the U.S. is the FDA. It has jurisdiction over our biopharmaceutical products and administers requirements covering the testing, safety,
effectiveness, manufacturing, labeling, marketing, advertising and post-marketing surveillance of these products. The FDA also regulates our Consumer Healthcare and
Animal Health products. The U.S. Department of Agriculture and the U.S. Environmental Protection Agency also regulate some of our products.
In addition, many of our activities are subject to the jurisdiction of various other federal regulatory and enforcement departments and agencies, such as the
Department of Health and Human Services (HHS), the Federal Trade Commission (FTC) (which also has the authority to regulate the advertising of consumer
healthcare products, including over-the-counter drugs and dietary supplements), the Department of Justice (DOJ) and the SEC. Individual states, acting through their
attorneys general, have become active as well, seeking to regulate the marketing of prescription drugs under state consumer protection and false advertising laws.
We are subject to possible administrative and legal proceedings and actions by these various regulatory bodies. See the Notes to Consolidated Financial
Statements – Note 17. Commitments and Contingencies in our 2011 Financial Report. Such actions may involve product recalls, seizures and other civil and criminal
sanctions.
Healthcare Reform. In March 2010, the ACA was enacted in the U.S. The provisions of the ACA are effective on various dates. The principal provisions
affecting the biopharmaceutical industry include:
18
•
an increase, from 15.1% to 23.1%, in the minimum rebate on branded prescription drugs sold to Medicaid beneficiaries (effective January 1, 2010);
•
extension of Medicaid prescription drug rebates to drugs dispensed to enrollees in certain Medicaid managed care organizations (effective March 23, 2010);
•
expansion of the types of institutions eligible for the “Section 340B discounts” for outpatient drugs provided to hospitals meeting the qualification criteria under
Section 340B of the Public Health Service Act of 1944 (effective January 1, 2010);
•
discounts on branded prescription drug sales to Medicare Part D participants who are in the Medicare “coverage gap,” also known as the “doughnut
hole
”
(
effective January 1, 2011); and

The ACA is estimated to result in the coverage of 32 million previously uninsured individuals. Approximately half of this will occur through an expansion of the
Medicaid program. Effective in 2014, individuals with incomes below 133% of the federal poverty level (FPL) will be eligible for Medicaid. The remainder will be
covered with private sector coverage either through their employers or new state-based Health Insurance Exchanges. With limited exceptions, individuals who fail to
purchase health insurance will pay a penalty. Individuals with incomes between 100%-400% of the FPL will be eligible for subsidies to help pay for coverage.
Expanding insurance coverage and other costs are expected to represent a relatively modest gain to overall pharmaceutical sales as the newly insured are
principally young and relatively healthy. At the same time, the rebates, discounts, taxes and other costs associated with the ACA are a significant cost to the industry.
The ACA created the Independent Payment Advisory Board (IPAB), a 15-member panel appointed by the President with the advice and consent of the Senate.
The IPAB is charged with developing proposals to “reduce the per capita rate of growth in Medicare spending” in the event that the actual Medicare per capita growth
rate exceeds a specified target. Unless Congress acts to alter the proposals, the proposals will be automatically implemented. However, the IPAB cannot directly ration
care, raise premiums, increase cost sharing, or otherwise restrict benefits or modify eligibility. If the IPAB fails to act, the Secretary of HHS is directed to prepare such
proposals. The IPAB is prohibited by statute from making payment reductions to certain sectors such as hospitals and home health agencies, which increases the risk
that the IPAB will propose to limit access to pharmaceutical treatments or mandate price controls for our products.
The ACA also established the Patient Centered Outcomes Research Institute (PCORI), a federally-funded, private, non-profit corporation empowered to fund and
disseminate comparative effectiveness research (CER) and build infrastructure for improved outcomes analysis. PCORI has no authority to impose formulary changes
directly in government-funded health programs. We expect that due to the PCORI, as well as the underlying market demand for data-driven differentiation, CER studies
will have growing influence on access. Overseeing and managing the PCORI is an advisory board drawn from multiple and varied stakeholder organizations, including
the pharmaceutical industry. Pfizer’s Chief Medical Officer currently serves as the industry representative on the advisory board.
The ACA specifies certain benefits and services that must be covered for health insurers to qualify to participate in the state insurance exchanges. Prescription
drugs are among these essential benefits in the law. Regulators are expected to provide guidance to states on the types of benefits and services that must be covered.
In 2012, the U.S. Supreme Court is to review certain provisions of the ACA, including: (1) the constitutionality of the ACA’s individual mandate that requires
most Americans to buy health insurance starting in 2014; (2) the “severability” of the individual mandate from the other provisions of the ACA, if the individual
mandate is struck down; (3) the ACA’s Medicaid expansion, which would require each state to expand its program or lose all federal Medicaid funds; and (4) the
applicability of a federal tax law, the Anti-Injunction Act, to the lawsuits brought on the individual mandate, which could bar review of that ACA provision until at least
2014. The Court is scheduled to hear oral arguments in late March and a decision is likely in June.
Changes in Marketing Activity Disclosure . The ACA expands the government’s investigative and enforcement authority and increases the penalties for fraud and
abuse, including amendments to both the False Claims Act and the Anti-Kickback Statute to make it easier to bring suit under these statutes. The ACA also allocates
additional resources and tools for the government to police healthcare fraud, with expanded subpoena power for HHS, additional funding to investigate fraud and abuse,
and expanded use of Recovery Audit Contractors for enforcement.
19
•
a fee payable to the federal government (which is not deductible for U.S. income tax purposes) based on our prior-calendar-year share relative to other companies
of branded prescription drug sales to specified government programs (effective January 1, 2011, with the total fee to be paid each year by the pharmaceutical
industry increasing annually through 2018).

Starting in 2012, pharmaceutical manufacturers will be required to record any transfers of value made to doctors and teaching hospitals and to disclose such data
to HHS, with the initial disclosure to HHS due in 2013. In addition to civil penalties for failure to report transfers of value to physicians or teaching hospitals, there will
be criminal penalties if a manufacturer intentionally makes false statements in such reports. Further, the increased access to such data by fraud and abuse investigators
could potentially raise the risk of liability for improper payments under the False Claims Act. Pfizer already records and makes public these types of transfers and
expects to be prepared to report the information in the format requested by HHS. This national payment transparency effort and industry commitment to uphold
voluntary codes of conduct (the updated PhRMA Code on Interactions with Healthcare Professionals , PhRMA Guiding Principles Direct to Consumer Advertisements
About Prescription Medicines,
etc.) will complement existing laws and regulations to help ensure ethical collaboration and truthful product communications. These
efforts are in place to help ensure responsible marketing approaches and to address concerns.
Medicare. Medicare Part D went into effect on January 1, 2006. Elderly and disabled beneficiaries have access to the Medicare drug benefit through private plans
approved by the federal government. Beneficiaries with low incomes and modest assets are eligible for assistance with Medicare Part D plan premiums and cost sharing.
Nationally, the share of such beneficiaries with comprehensive drug coverage increased from 59% in 2005 to over 90% in 2010. Medicare beneficiaries report high
levels of satisfaction, with an overwhelming majority saying the program works well. In addition, the program costs less than originally expected.
The ACA made some important changes to the drug benefit—in particular, phasing out the coverage gap by 2020. Prior to the ACA, beneficiaries who reached a
certain level of spending on prescription medications (the Medicare Part D coverage gap or “doughnut hole”) had to pay 100% of the cost of their drugs until personal
out-of-pocket spending reached a level qualifying them for catastrophic coverage. The Medicare Part D Coverage Gap Discount Program uses public and private
funding to relieve the financial burden facing beneficiaries who fall into this coverage gap. Beginning in 2011, branded pharmaceutical companies paid 50% of the cost
of the branded drugs in the gap and the government paid 7% of the cost of the generic drugs in the gap. As a result, rather than paying 100% of the total cost of their
drugs when they reach the coverage gap, enrollees paid 50% of the total cost of branded drugs and 93% of the total cost of generic drugs. In 2012, the branded discount
will remain 50%, but the generic discount will increase to 14%. By 2020, enrollees will pay only 25% of the cost of their branded and generic drugs in the gap as the
share covered by the government will increase.
Biosimilars. The ACA also created a framework for the approval of follow-on biologics, or biosimilars, following the expiration of 12 years of exclusivity for the
innovator biologic, with a potential six-month pediatric extension. The FDA is responsible for implementation of the legislation, which will require the FDA to address
such key topics as the type and extent of data needed to establish biosimilarity; the data required to achieve interchangeability compared to biosimilarity; the naming of
biosimilars; the implications of having or not having unique names; the tracking and tracing of adverse events; and the acceptability of data from an ex-U.S. licensed
reference product comparator to demonstrate biosimilarity and/or interchangeability. The FDA has begun to address some of these issues with its recent release of three
draft guidance documents. Specifically, the FDA has clarified that biosimilar applicants may use a non-U.S. licensed comparator in certain studies to support a
demonstration of biosimilarity to a U.S.-licensed reference product.
Pfizer is developing biosimilar medicines. Leveraging our expertise in biologics, and our regulatory, commercial, and manufacturing strengths, we will seek to
provide high-quality, safe and effective biosimilars that provide patients and prescribers with additional treatment options and expand access by offering high-quality,
more affordable alternatives for biologic medicines.
The budget proposal submitted to Congress by President Obama in February 2012 includes a provision that would reduce the base exclusivity period for a
biologics product from 12 years to seven years. There is no corresponding pending bill designed to amend the ACA to alter the biologics provisions.
20

Medicaid and Related Matters. Federal law requires branded pharmaceutical companies to provide rebates to state Medicaid agencies. The ACA brought about
major changes in the Medicaid program. Collectively, the measures (i) increased federal rebates paid by manufacturers on branded drugs within the traditional Medicaid
program from 15.1% to 23.1%, and for generic drugs from 11% to 13% of Average Manufacturer Price (AMP); (ii) expanded Medicaid drug rebates to cover drugs
provided through managed Medicaid plans; and (iii) changed the rebate rates for line extensions or new formulations of solid oral dosage form drugs. Post-
implementation of ACA, the Centers for Medicare and Medicaid Services (CMS) withdrew its former, detailed AMP-calculation rules, and new CMS AMP guidance is
still pending publication (now expected in 2012). The law also creates a federal upper limit under the Medicaid program for generic drugs at 175% of AMP. In addition,
the law expanded the types of entities eligible for the “Section 340B discounts” for outpatient drugs that began in 2010.
The majority of states use preferred drug lists to restrict access to certain medicines to Medicaid beneficiaries. Restrictions exist for some Pfizer products in
certain states. Access in the Medicaid managed care program is typically determined by the health plans providing coverage for Medicaid recipients contracting for the
provision of services in the state. Given states’ current and potential ongoing fiscal crises, a growing number of states are considering a variety of cost-
control strategies,
including capitated managed care plans that typically contain cost by restricting access to certain treatments.
The ACA expands Medicaid coverage in 2014. It is expected that 16 million additional people will be enrolled in Medicaid by 2019.
We also must give discounts or rebates on purchases or reimbursements of pharmaceutical products by certain other federal and state agencies and programs. See
the discussion regarding rebates in the Analysis of the Consolidated Statement of Income – Revenues – Overview
section of the MD&A in our 2011 Financial Report and
in the Notes to Consolidated Financial Statements – Note 1G. Significant Accounting Policies – Revenues in our 2011 Financial Report, which discussions are
incorporated by reference.
PDUFA Reauthorization. The Prescription Drug User Fee Act (PDUFA) was first enacted in 1992 to provide the FDA with additional resources to speed the
review of important new medicines. Prior to PDUFA, inadequate funding of the FDA drug review process led to a backlog of application reviews and lengthy review
times. PDUFA revolutionized the review process for new drugs and biologics without compromising high approval standards for demonstration of product safety,
quality and efficacy. PDUFA sunsets every five years and must be reauthorized by Congress. PDUFA IV will expire in September 2012. The proposed PDUFA V
reauthorization is the product of months of stakeholder discussion between the FDA, industry, patient groups and consumers. The PDUFA V agreement focuses on three
areas: Modernizing Regulatory Science, Review Process Enhancements, and Strengthening Post-Market Safety Surveillance, with the goal of supporting agency
scientific capabilities and improving process efficiencies. Pfizer supports the PDUFA V reauthorization in order to foster an innovation-supportive regulatory
environment and to ensure that the FDA is properly equipped to evaluate important new medicines in a timely and predictable fashion.
Importation of Drugs. There continue to be legislative proposals to amend U.S. law to allow the importation into the U.S. of prescription drugs from outside the
U.S., which can be sold at prices that are regulated by the governments of various foreign countries. In addition to well-documented safety concerns, such as the
increased risk of counterfeit products entering the supply chain, such importation could impact pharmaceutical prices in the U.S. While the 2003 Medicare
Modernization Act (2003 MMA) maintains a prohibition on such imports, it would allow importation from Canada if the Secretary of HHS certifies that such
importation is safe and would result in savings to consumers. Before the 2003 MMA, federal law would have permitted importation of medicines into the U.S. from a
considerably larger group of developed countries, provided the Secretary of HHS made the same safety and cost-savings certifications. As part of the debate on the
ACA, two Senate proposals that would have restored the broader number of countries from which importation would be permissible were introduced but were ultimately
defeated.
21

The Secretaries of HHS in the Clinton, George W. Bush, and Obama administrations have all declined to certify that importation of medicines is safe and saves
money. If the Secretary of HHS changes its position, an increase in cross-border trade in medicines subject to foreign price controls in other countries could occur,
which could have a material adverse effect on our results of operations.
In December 2004, HHS and the Department of Commerce issued reports on drug importation and foreign price controls. The HHS report noted that it would be
“extraordinarily difficult to ensure that drugs personally imported by individual consumers” could meet the standards of safety that would support certifying such
importation as safe. While the report also concluded that the U.S. could establish a feasible basis for commercial drug importation, such a change in the law would
require “new legal authorities, substantial additional resources and significant restrictions on the types of drugs that could be imported.” The report also noted that the
total savings to be expected from such a commercial importation regime would be relatively small—1% or 2% of total drug spending in the U.S.
Budget Control Act of 2011 . In August 2011, the federal Budget Control Act of 2011 (the Act) was enacted in the U.S. The Act includes provisions to raise the
U.S. Treasury Department’s borrowing limit, known as the debt ceiling, and provisions to reduce the federal deficit by $2.4 trillion between 2012 and 2021. Deficit-
reduction targets include $900 billion of discretionary spending reductions associated with HHS and various agencies charged with national security, but those
discretionary spending reductions do not include programs such as Medicare and Medicaid or direct changes to pharmaceutical pricing, rebates or discounts. A Joint
Select Committee of Congress (the Committee) was appointed to identify the remaining $1.5 trillion of deficit reductions by November 23, 2011, but no
recommendations were made by the Committee prior to the deadline. As a result, the Office of Management and Budget (OMB) is now responsible for identifying the
remaining $1.5 trillion of deficit reductions, which will be divided evenly between defense and non-defense spending. Under this OMB fallback review process, Social
Security, Medicaid, Veteran Benefits and certain other spending categories are excluded from consideration, but reductions in payments to Medicare providers may be
made, although any such reductions are prohibited by law from exceeding 2%. Additionally, certain payments to Medicare Part D plans, such as low-income subsidy
payments, are exempt from reduction. While we do not know the specific nature of the spending reductions under the Act that will affect Medicare, we do not expect
that those reductions will have a material adverse impact on our results of operations. However, any significant spending reductions affecting Medicare, Medicaid or
other publicly funded or subsidized health programs that may be implemented, and/or any significant additional taxes or fees that may be imposed on us, as part of any
broader deficit-reduction effort, could have an adverse impact on our results of operations.
Outside the United States
We encounter similar regulatory and legislative issues in most other countries. In Europe, Canada and some other international markets, governments provide
healthcare at low direct cost to consumers and regulate pharmaceutical prices or patient reimbursement levels to control costs for the government-sponsored healthcare
system. This international patchwork of price regulation has led to different prices and some third-party trade in our products between countries.
Europe. The approval of new drugs across the EU may be achieved using the Mutual Recognition Procedure/Decentralized Procedure or EU Commission/EMA
Centralized Procedure. These procedures apply in the EU member states, plus the European Economic Area countries, Norway and Iceland. The use of these procedures
generally provides a more rapid and consistent approval process across the member states than was the case when the approval processes were operating independently
within each country.
Since the EU does not have jurisdiction over patient reimbursement or pricing matters in its member states, we continue to work with individual countries on such
matters across the region.
22

The world economy in 2011 faced renewed challenges as a result of recent financial conditions and, in particular, increased uncertainty around the solvency of
governments. As a result, global growth has slowed. One of the consequences of the economic downturn for almost all world economies has been an increase in public
debt as a proportion of gross domestic product arising from increased government spending and reduced tax receipts. For many developed economies, particularly in
Europe, this exacerbated existing fiscal imbalances and has created doubt in investment markets about the sustainability of public debt levels in a number of European
countries, further raising the cost of borrowing, with the result that financial support from the EU and the International Monetary Fund has been necessary in the cases of
Greece, Portugal and Ireland. Stringent austerity measures have been implemented in most European countries with the aim of closing the fiscal gap, in particular in
Spain and Italy.
Under these macroeconomic conditions, Pfizer continues to face widespread downward pressures on international pricing and reimbursement, particularly in
developed European markets with a large government share of pharmaceutical spending. Specific pricing pressures in 2011 included measures to reduce pharmaceutical
prices and expenditure in Italy, Spain, Greece, Ireland and Portugal.
Formal processes of international reference pricing (IRP) between EU countries add to the regional impact of price cuts in individual countries. Price variations
have also arisen from exchange rate fluctuations between the euro and other European currencies, and these are also exacerbated by international reference pricing
systems. The downward pricing pressure resulting from this dynamic can be expected to intensify as a result of reforms to IRP policies, emergency measures targeting
pharmaceuticals in some European countries and ongoing exchange rate turbulence.
On January 26, 2007, the new EU Regulation on Medicines for Pediatric Use became effective. This introduced new obligations on pharmaceutical companies to
conduct research on their medicines for children and, subject to various conditions, offered the possibility of incentives for so doing, including exclusivity extensions.
The aim of this regulation is to improve the health of children in the EU through high quality research, stimulating the development of new medicines, creating
infrastructure to enable authorized use and improving the information on medicines for children. A Pediatric Committee (PDCO) was created within the EMA to provide
scientific opinions and input on development plans for medicines for use with children. In line with this regulation, Pfizer is conducting many pediatric research
programs for its in-line and development products, and completed its first EMA-approved pediatric investigation plan (for Lipitor ) in 2009.
On December 31, 2010, the EU published new pharmacovigilance legislation which had been adopted by the Council and Parliament of the EU. The legislation is
required to be implemented by mid-2012 and entails many new and revised requirements for conducting pharmacovigilance, as well as the codification of various
existing requirements previously set out in guidance. Key changes include the possibility for regulators to require pharmaceutical companies to conduct post-
authorization efficacy studies, both at the time of approval and at any time afterwards in light of scientific developments. There are also additional requirements to
include statements in product labeling with regard to adverse drug reaction reporting and additional monitoring of products. There also is expected to be significantly
greater transparency of the safety review process.
The new legislation forms part of a three-part “pharmaceutical package” to amend the existing EU pharmaceutical legislation. The second part, adopted on
May 27, 2011, is a Directive aimed at preventing falsified medicines from entering into the legal supply chain. Notably, the Directive imposes new obligations on all
parties in the distribution chain, including importers, traders, manufacturers, distributors, and any operator who repackages a product. This is required to be implemented
by January 2013. The third part of the package concerns the provision of information on prescription medicines to patients, which has reached a reasonably advanced
stage, but has proved controversial to date and the outcome for which is more uncertain.
At the end of the third quarter of 2010, the Commissioner for Industry and Entrepreneurship of the European Commission announced the launch of a process on
corporate responsibility in the pharmaceutical
23

industry, the impact of which is yet to be determined through ongoing working committees. The process includes three independent platforms: (i) transparency and
ethics in the sector; (ii) access to medicines in Africa; and (iii) access to medicines in Europe in the context of pricing and reimbursement. The platform on access to
medicines in Europe will focus on enhancing collaboration among EU member states and relevant stakeholders, in order to find common non-regulatory approaches to
ensure “timely and equitable access to medicines”.
Canada . Health Canada (HC) is the government agency that provides regulatory and marketing approval for drugs and therapeutic products in Canada. The
upcoming Legislative and Regulatory Modernization (LRM) is the most significant drug regulatory system reform in Canada in over 50 years and is expected to
overhaul Canada’s Food and Drugs Act and Regulations. The LRM supports a ‘lifecycle’ regulatory approach and is focused on strengthening evidence-based decision
making, good regulatory planning, licensing, post-licensing, accountability, authority and enforcement. Through this framework, HC intends to improve the market
authorization process and implement necessary regulatory frameworks. In October 2010, HC accelerated its modernization efforts. This included the proposed
regulatory pathways for Orphan Drugs (harmonized with U.S./EU regulations) and for biosimilars referred to as “Subsequent Entry Biologics” (SEBs). This would
formalize into regulations HC’s Guidance Document, which provided for approval of SEBs in 2009. Furthermore, LRM is to entail increased regulatory oversight
throughout the drug lifecycle to continually assess the benefit-risk balance. Following an extensive technical consultation process, HC intends to propose a first set of
regulatory amendments around the third quarter of 2012. The whole LRM process is expected to take place over a period of three years.
Introductory “non-excessive” prices and price increases are controlled by the federal Patented Medicines Prices Review Board. However, reimbursement is under
provincial jurisdiction. As provinces continue to face budget pressure from growing healthcare expenditures, many provincial governments have developed pricing and
purchasing strategies (including product listing agreements and a pan-Canadian purchasing alliance initiative), to obtain better drug prices. The private sector is also
attempting to exert its negotiating power on drug manufacturers. The 2004 Federal-Provincial-Territorial (FPT) Health Accord that sets out the Canada Health Transfers
payment to provinces plus commitments on health policy initiatives expires on March 31, 2014. Renegotiation will be influenced by the challenging fiscal positions of
all FPT governments, which could increase hurdles for access to newer innovative therapies.
Canada’s intellectual property regime for drugs, which provides some level of patent protection and data exclusivity, can be further enhanced to encourage
innovation through the Canada/EU Comprehensive Economic & Trade Agreement. The federal government also has jurisdiction over international trade and therefore
over the issue of cross-border trade in pharmaceuticals and internet pharmacies.
Asia. The regulatory environment in Asia presents multiple issues for companies trying to achieve simultaneous global development and registration (i.e.,
marketing products at the same time as in the U.S., Europe, Canada and elsewhere). While each country in Asia has its unique regulatory concerns, there are a number of
regulatory issues that are common among the majority of countries in Asia. For example, with the exception of Japan, health authorities in Asia generally require
marketing approval by a recognized regulatory authority (e.g., the U.S. FDA) before they begin to conduct their application review process and/or issue their final
approval. Proof of reference country approval is usually satisfied by submitting a Certificate of Pharmaceutical Product, a legal document that is issued by the competent
health authority certifying that the company’s product has satisfied its country’s registration requirements and manufacturing standards. Often, this requirement delays
marketing authorization in Asia by 12-15 months following market authorization in the U.S. and Europe.
Another common regulatory issue in Asia is the requirement for local clinical data in the country’s population in order to receive final marketing approval. Each
of Japan, China, Korea, Taiwan and India has regulations in some form that require clinical studies in the country (e.g., China requires a prescribed number of Chinese
patients regardless of the product, therapeutic area or disease population). Although some agencies have
24

shown flexibility based on scientific rationale related to ethnicity assessments, it is not uncommon for companies to be required to duplicate costly clinical trials in Asia
pursuant to these regulations. This can further add to marketing approval delays compared to the U.S. and Europe.
In Japan, the government is aiming to reduce the drug lag (i.e., drugs are launched in Japan years after the EU and U.S. markets) in a two-pronged approach:
reducing regulatory agency review times and establishing a new pilot pricing premium. The pilot pricing premium provides a financial incentive for drug development
in Japan. While economic conditions and government debt levels continue to put pressure on healthcare costs resulting in cost containment (particularly in the off-patent
sector) the recent extension of the pilot pricing premium for innovative products is encouraging.
In Korea, the national health insurance deficit prompted the government to make significant price cuts in the off-patent sector at the end of 2011. We continue to
work with a committee established by the government to improve the pricing system for innovative new drugs.
The controlling regulatory agency in China is the State Food and Drug Administration (SFDA). SFDA’s scope of responsibilities is similar to that of the FDA or
EMA. Two key agencies within SFDA are the Center for Drug Evaluation (CDE) and the National Institute for the Control of Pharmaceutical and Biological Products
(NICPBP). The CDE, which is analogous to the FDA’s Center for Drug Evaluation and Research, is primarily responsible for the technical review of product
applications, including clinical trial applications and new drug applications, and drafting technical guidance documents. NICPBP is the quality testing arm of SFDA,
legally responsible for the testing of pharmaceuticals, biologics and medical devices nationwide.
China’s regulatory system is unique in many ways, and its drug development and registration requirements are not always consistent with international standards.
As a result, it is not uncommon to see treatments entering the market in China two to four years after first marketing in the U.S. and Europe.
Intellectual Property . While the global intellectual property environment has improved following WTO-TRIPS, our future business growth depends on further
progress in intellectual property protection. In emerging market countries in particular, governments have used intellectual property policies as a tool for reducing the
price of imported medicines, as well as to protect their national pharmaceutical industries. There is considerable political pressure to weaken existing intellectual
property protection and resist implementation of any further protection, which has led to policies such as higher standards and more difficult procedures for patenting
biopharmaceutical inventions, restrictions on patenting certain types of inventions (e.g., new medical treatment methods) and failure to implement effective regulatory
data protection. Our industry advocacy efforts focus on seeking a more balanced business environment for foreign manufacturers.
In China, the intellectual property environment has improved, although effective enforcement and adequate legal remedies remain areas of concern. The
government has taken steps to protect intellectual property rights in conformity with World Trade Organization (WTO) provisions, and several companies, including
Pfizer, have established research and development centers in China due to increased confidence in China’s intellectual property environment. Despite this, China
remained on the U.S. Department of Commerce Priority Watch List for 2011. A framework exists for protecting patents for 20 years, but enforcement mechanisms are
often lacking or inconsistent, such as the absence of effective patent linkage mechanisms and preliminary injunctions, impractical evidentiary burdens, and heightened
sufficiency standards used to invalidate patents at the enforcement stage.
Additionally, true regulatory data protection remains elusive in China. The Center for Drug Evaluation provides protection against reliance on data by generic
applicants for a fixed period of time. Following its WTO accession in 2001, China revised its laws to incorporate concepts from the WTO-TRIPS, and China’s relevant
laws establish a six-year period of protection against unfair commercial use of undisclosed test and other data of products containing a new chemical ingredient.
However, the current regulations are ambiguous as to how data protection is implemented in practice in China. For example, certain key concepts such as “
new chemical
ingredient” and “unfair commercial use” are undefined.
25

In Brazil and other Latin American countries, backlogs at patent agencies have presented challenges for the protection of certain products. The lack of regulatory
data protection and difficulties in protecting certain types of inventions, such as new medical uses of drug products, may limit the commercial lifespan of some
pharmaceutical products.
In India, limited standards for patentability of pharmaceutical products have made it difficult to protect many of our inventions. India and other countries such as
Israel maintain a system of pre-grant patent oppositions that delay the granting of patents and add an additional challenge in our ability to protect our products through
patents.
In Korea, the laws and regulations for the patent-regulatory approval linkage system have now been finalized, and we are waiting for the effective date of
implementation as part of the United States-Korea Free Trade Agreement. The Korean patent-regulatory approval linkage system includes biologics.
Environmental Law Compliance
Most of our operations are affected by national, state and/or local environmental laws. We have made, and intend to continue to make, necessary expenditures for
compliance with applicable laws. We also are cleaning up environmental contamination from past industrial activity at certain sites. See the Notes to Consolidated
Financial Statements – Note 17. Commitments and Contingencies in our 2011 Financial Report. As a result, we incurred capital and operational expenditures in 2011 for
environmental compliance purposes and for the clean-up of certain past industrial activity as follows:
While we cannot predict with certainty future capital expenditures or operating costs for environmental compliance, including compliance with pending and
potential legislation and potential regulation related to climate change, we have no reason to believe they will have a material effect on our capital expenditures or
competitive position.
We have reviewed the potential for physical risks to our facilities and supply chain that may be exacerbated by climate change and have concluded that, because
of our facility locations and our existing distribution networks, we do not believe these risks are material in the near term.
Tax Matters
The discussion of tax-related matters in the Notes to Consolidated Financial Statements – Note 5. Taxes on Income in our 2011 Financial Report, is incorporated
by reference.
Employees
In our innovation-intensive business, our employees are vital to our success. We believe we have good relationships with our employees. As of December 31,
2011, we employed approximately 103,700 people in our operations throughout the world.
26
•
environment
-
related capital expenditures
—
$
34 million; and
•
other environment
-
related expenses
—
$
145 million.

The statements in this Section describe the major risks to our business and should be considered carefully. In addition, these statements constitute our cautionary
statements under the Private Securities Litigation Reform Act of 1995.
The SEC encourages companies to disclose forward-looking information so that investors can better understand a company’s future prospects and make
informed investment decisions. Our disclosure and analysis in the 2011 Form 10-K and in our 2011 Annual Report to Shareholders contain some forward-looking
statements that set forth anticipated results based on management’s plans and assumptions. From time to time, we also provide forward-looking statements in other
materials we release to the public, as well as oral forward-looking statements. Such forward-looking statements involve substantial risks and uncertainties. We have
tried, wherever possible, to identify such statements by using words such as “anticipate,” “estimate,” “expect,” “project,” “intend,” “plan,” “believe,” “will,”
“target,” “forecast”, “goal”, “objective” and other words or terms of similar meaning, or by using future dates in connection with any discussion of future operating
or financial performance, business plans or prospects, in-line products and product candidates, strategic review, capital allocation, and share-repurchase and dividend-
rate plans. In particular, these include statements relating to future actions, business plans and prospects, prospective products or product approvals, future
performance or results of current and anticipated products, sales efforts, expenses, interest rates, foreign exchange rates, the outcome of contingencies, such as legal
proceedings, share
-repurchase and dividend-rate plans, financial results and government regulation.
We cannot guarantee that any forward-looking statement will be realized, although we believe we have been prudent in our plans and assumptions. Achievement
of future results is subject to substantial risks, uncertainties and potentially inaccurate assumptions. Should known or unknown risks or uncertainties materialize, or
should underlying assumptions prove inaccurate, actual results could differ materially from past results and those anticipated, estimated or projected. You should bear
this in mind as you consider forward-looking statements, and you are cautioned not to put undue reliance on forward-looking statements.
We undertake no obligation to publicly update forward-looking statements, whether as a result of new information, future events or otherwise. You are advised,
however, to consult any further disclosures we make on related subjects in our Form 10-Q and 8-
K reports and our other filings with the SEC. Also note that we provide
the following cautionary discussion of risks, uncertainties and possibly inaccurate assumptions relevant to our businesses. These are factors that, individually or in the
aggregate, may cause our actual results to differ materially from expected and historical results. We note these factors for investors as permitted by the Private
Securities Litigation Reform Act of 1995. You should understand that it is not possible to predict or identify all such factors. Consequently, you should not consider the
following to be a complete discussion of all potential risks or uncertainties.
U.S. Healthcare Reform/Healthcare Legislation
As mentioned above, the ACA was enacted by Congress in March 2010 and its provisions are effective on various dates. We expect that the rebates, discounts,
taxes and other costs over time will have a significant effect on our expenses and profitability in the future. See the discussion under the Overview of our Performance,
Operating Environment, Strategy and Outlook – Our Operating Environment – U.S. Healthcare Legislation section of the MD&A in our 2011 Financial Report and in
Item 1. Business
under the caption Government Regulation and Price Constraints
. Furthermore, the IPAB created by the ACA, to reduce the per capita rate of growth in
Medicare spending, could potentially limit access to certain treatments or mandate price controls for our products. Moreover, expanded government investigative
authority may increase the costs of compliance with new regulations and programs. We also face the uncertainties that might result from any modification, repeal or
invalidation of any of the provisions of the ACA.
27
ITEM 1A.
RISK FACTORS

Pricing Pressures, Government Regulation and Managed Care Trends
U.S. and foreign governmental regulations mandating price controls and limitations on patient access to our products impact our business, and our future results
could be adversely affected by changes in such regulations or policies. In the U.S., many of our biopharmaceutical products are subject to increasing pricing pressures.
Such pressures have increased as a result of the 2003 MMA due to the enhanced purchasing power of the private sector plans that negotiate on behalf of Medicare
beneficiaries. In addition, if the 2003 MMA or the ACA were amended to impose direct governmental price controls and access restrictions, it would have a significant
adverse impact on our business. Furthermore, MCOs, as well as Medicaid and other government agencies, continue to seek price discounts. Some states have
implemented, and other states are considering, price controls or patient access constraints under the Medicaid program, and some states are considering price-control
regimes that would apply to broader segments of their populations that are not Medicaid-eligible. Other matters also could be the subject of U.S. federal or state
legislative or regulatory action that could adversely affect our business, including changes in patent laws, the importation of prescription drugs from outside the U.S. at
prices that are regulated by the governments of various foreign countries, restrictions on U.S. direct-to-consumer advertising, limitations on interactions with healthcare
professionals, or the use of comparative effectiveness methodologies that could be implemented in a manner that focuses primarily on cost differences and minimizes
the therapeutic differences among pharmaceutical products and restricts access to innovative medicines.
The prohibition on the use of federal funds for reimbursement of erectile dysfunction medications by the Medicaid program, which became effective January 1,
2006, and the similar federal funding prohibition for the Medicare Part D program, which became effective January 1, 2007, has had an adverse effect on our business.
Any prohibitions on the use of federal funds for reimbursement of other classes of drugs in the future may also have an adverse effect.
We encounter similar regulatory and legislative issues in most other countries. In Europe and some other international markets, governments provide healthcare at
low direct cost to consumers and regulate pharmaceutical prices or patient reimbursement levels to control costs for government-sponsored healthcare systems. In
particular, there were government-
mandated price reductions for certain biopharmaceutical products in certain European and emerging market countries in 2011, and we
anticipate continuing pricing pressures in Europe and emerging markets in 2012. This international patchwork of price regulation has led to different prices and some
third-party trade in our products between countries. As a result, it is expected that pressures on the pricing component of operating results will continue. The adoption of
restrictive price controls in new jurisdictions or more restrictive ones in existing jurisdictions, failure to obtain government-approved pricing or formulary placement
where required for our products or obtaining such pricing or placement at unfavorable pricing could also adversely impact revenue. In our vaccines business, we
participate in a tender process in many countries for participation in national immunization programs. Failure to secure participation in national immunization programs
or to obtain acceptable pricing in the tender process could adversely affect our business.
MCOs and other private insurers frequently adopt their own payment or reimbursement reductions. Consolidation among MCOs has increased the negotiating
power of these entities. Private third-
party payers, as well as governments, increasingly employ formularies to control costs by negotiating discounted prices in exchange
for formulary inclusion. Private health insurance companies and self-insured employers have been raising co-payments required from beneficiaries, particularly for
branded pharmaceuticals and biotechnology products. Private health insurance companies also are increasingly imposing utilization management tools, such as requiring
prior authorization for a branded product if a generic product is available or requiring that patient treatment first fail on a generic product before permitting access to a
branded medicine. As the U.S. payer market concentrates further and more drugs become available in generic form, biopharmaceutical companies may face greater
pricing pressure from private third-party payers, who will continue to drive more of their patients to use lower cost generic alternatives.
28

Generic Competition
Competition from manufacturers of generic drugs is a major challenge for us around the world. Upon the expiration or loss of patent protection for one of our
products, or upon the “at-risk” launch (despite pending patent infringement litigation against the generic product) by a generic manufacturer of a generic version of one
of our patented products, we can lose the major portion of sales of that product in a very short period, which can adversely affect our business.
Also, the patents covering several of our medicines, including Lipitor , Viagra , Detrol/Detrol LA , Lyrica , Sutent , Tygacil , Rapamune , Zyvox , Avinza ,
EpiPen
, Torisel and Embeda are being challenged by generic manufacturers. In addition, our patent-protected products may face competition in the form of generic
versions of competitors’ branded products that lose their market exclusivity.
Competitive Products
We cannot predict with accuracy the timing or impact of the introduction of competitive products or their possible effect on our sales. Products that compete with
ours, including some of our best-selling medicines, are launched from time to time. Competitive product launches have occurred in recent years, and certain potentially
competitive products are in various stages of development, some of which have been filed for approval with the FDA and with regulatory authorities in other countries.
Dependence on Key In-Line Products
We recorded direct product revenues of more than $1 billion for each of 12 biopharmaceutical products in 2011: Lipitor , Lyrica , Prevnar 13/Prevenar 13 ,
Enbrel
, Celebrex , Viagra , Norvasc , Zyvox , Xalatan/Xalacom , Sutent , Geodon/Zeldox and the Premarin family. Those products accounted for 56% of our total
biopharmaceutical revenues in 2011. Lipitor sales in 2011 were approximately $9.6 billion, accounting for approximately 14% of our total 2011 revenues. Among other
losses of exclusivity for Lipitor , we lost exclusivity for Lipitor in the U.S. on November 30, 2011, and Lipitor will have lost exclusivity in the majority of European
markets by May 2012. As a result, we expect that our revenues for Lipitor in 2012 will be substantially less than our 2011 revenues for Lipitor . If the products
referenced above or any of our other major products were to become subject to problems such as loss of patent protection, changes in prescription growth rates, material
product liability litigation, unexpected side effects, regulatory proceedings, publicity affecting doctor or patient confidence, pressure from existing competitive products,
changes in labeling or, if a new, more effective treatment should be introduced, the adverse impact on our revenues could be significant. As noted, patents covering
several of our best-selling medicines have recently expired or will expire in the next few years (including some of our billion-dollar products such as Lipitor
as discussed
above, Xalatan in the U.S. in March 2011 and Xalatan/Xalacom in 15 major European markets in January 2012 and Geodon in the U.S. in March 2012), and patents
covering a number of our best-selling medicines are the subject of pending legal challenges. In addition, our revenues could be significantly impacted by the timing and
rate of commercial acceptance of key new products.
Specialty Pharmaceuticals
Specialty pharmaceuticals are medicines that treat rare or life-threatening conditions that have smaller patient populations, such as certain types of cancer and
multiple sclerosis. The growing availability and use of innovative specialty pharmaceuticals, combined with their relative higher cost as compared to other types of
pharmaceutical products, is beginning to generate significant payer interest in developing cost-containment strategies targeted to this sector. While the impact on us of
payers’ efforts to control access to and pricing of specialty pharmaceuticals has been limited to date, our growing portfolio of specialty products, combined with the
increasing use of health technology assessment in markets around the world and the deteriorating finances of governments, may lead to a more significant adverse
business impact in the future.
29

Research and Development Investment
The discovery and development of safe, effective new products, and the development of additional uses for existing products, are very important to our success.
Our growth potential depends in large part on our ability to identify and develop new products or new indications for existing products that address unmet medical needs
and receive reimbursement from payers, either through internal research and development or through collaborations, acquisitions, joint ventures or licensing or other
arrangements with third parties. However, balancing current growth and investment for the future remains a major challenge. Our ongoing investments in new product
introductions and in research and development for new products and existing product extensions could exceed corresponding sales growth. This could produce higher
costs without a proportional increase in revenues.
Additionally, our research and development investment plans and resources may not be correctly matched between science and markets, and failure to invest in
the right technology platforms, therapeutic segments, product classes, geographic markets and/or in-licensing and out-licensing opportunities in order to deliver a more
robust pipeline could adversely impact the productivity of our pipeline. Additionally, even if the areas with the greatest market attractiveness are identified, the science
may not work for any given program despite the significant investment required for research and development.
We recently announced a focus on fewer disease areas where we believe we can deliver the greatest medical and commercial success, as well as the
implementation of our R&D footprint reduction by moving forward on our productivity initiatives. As a result of these actions, we expect significant reductions in our
annual research and development expenses, which are reflected in our 2012 financial guidance. There can be no assurance that this strategy will deliver the desired result
in the targeted timeframe or at all, which could affect profitability in the future.
Development, Regulatory Approval and Marketing of Products
Risks and uncertainties apply particularly with respect to product-related, forward-looking statements. The outcome of the lengthy and complex process of
identifying new compounds and developing new products is inherently uncertain. Drug discovery and development is time-consuming, expensive and unpredictable.
The process from early discovery or design to development to regulatory approval can take many years. Drug candidates can fail at any stage of the process. There can
be no assurance as to whether or when we will receive regulatory approval for new products or for new indications or dosage forms for existing products. Decisions by
regulatory authorities regarding labeling, ingredients and other matters could adversely affect the availability or commercial potential of our products. As examples,
there is no assurance that our late stage pipeline products, such as tofacitinib, bosutinib and Eliquis (apixaban) for prevention of stroke in patients with atrial fibrillation,
will receive regulatory approval and/or be commercially successful or that recently approved products, such as Prevnar 13/Prevenar 13 for use in adults 50 years of age
and older, Xalkori (crizotinib) and Inlyta (axitinib) will be approved in other markets and/or be commercially successful. There is also a risk that we may not adequately
address existing regulatory agency findings concerning the adequacy of our regulatory compliance processes and systems or implement sustainable processes and
procedures to maintain regulatory compliance and to address future regulatory agency findings, should they occur.
There are many considerations that can affect marketing of our products around the world. Regulatory delays, delays in or the inability to successfully complete
or adequately design and implement clinical trials, claims and concerns about safety and efficacy, new discoveries, patent disputes and claims about adverse side effects
are a few of the factors that can adversely affect the realization of research and development and product-related, forward-looking statements. Further, claims and
concerns about safety and efficacy can result in a negative impact on product sales, product recalls or withdrawals, and/or consumer fraud, product liability and other
litigation and claims. Also, increasing regulatory scrutiny of drug safety and efficacy, with regulatory authorities increasingly focused on product safety and the
risk/benefit profile of products as they relate to already-approved products, has resulted in a more challenging, expensive and lengthy regulatory approval process due to
requests for, among other things, additional clinical trials prior to granting approval or increased post-approval requirements, such as risk evaluation and mitigation
strategies (see Post-Approval Data below).
30

Post-Approval Data
As a condition to granting marketing approval of a product, the FDA may require a company to conduct additional clinical trials. The results generated in these
Phase IV trials could result in loss of marketing approval, changes in product labeling, and/or new or increased concerns about side effects or efficacy of a product. The
Food and Drug Administration Amendments Act of 2007 (the FDAAA) gives the FDA enhanced post-market authority, including the explicit authority to require post-
market studies and clinical trials, labeling changes based on new safety information and compliance with FDA-approved risk evaluation and mitigation strategies. The
FDA’s exercise of its authority under the FDAAA could result in delays or increased costs during product development, clinical trials and regulatory review, increased
costs to comply with additional post-approval regulatory requirements and potential restrictions on sales of approved products. Foreign regulatory agencies often have
similar authority and may impose comparable costs. Post-marketing studies, whether conducted by us or by others and whether mandated by regulatory agencies or
voluntary, and other emerging data about marketed products, such as adverse event reports, may also adversely affect sales of our products. Further, the discovery of
significant problems with a product similar to one of our products that implicate (or are perceived to implicate) an entire class of products could have an adverse effect
on sales of the affected products. Accordingly, new data about our products, or products similar to our products, could negatively impact demand for our products due to
real or perceived side effects or uncertainty regarding efficacy and, in some cases, could result in product withdrawal or recall. Furthermore, new data and information,
including information about product misuse, may lead government agencies, professional societies, practice management groups or organizations involved with various
diseases to publish guidelines or recommendations related to the use of our products or the use of related therapies or place restrictions on sales. Such guidelines or
recommendations may lead to lower sales of our products.
Patent Protection
Our long-term success largely depends on our ability to market technologically competitive products. If we fail to obtain and maintain adequate intellectual
property protection, we may not be able to prevent third parties from using our proprietary technologies. Our currently pending or future patent applications and/or
extensions may not result in issued patents or be approved on a timely basis or at all. In addition, our issued patents may not contain claims sufficiently broad to protect
us against third parties with similar technologies or products or provide us with any competitive advantage. The scope of our patent claims also may vary between
countries, as individual countries have their own patent laws, some of which create legal uncertainty or unpredictability with respect to the requirements and standards
for patent protection. Our ability to enforce our patents also depends on the laws of individual countries and each country’s practice with respect to enforcement of
intellectual property rights, and the extent to which certain sovereigns may engage in compulsory licensing of pharmaceutical intellectual property as a result of local
political pressure. In addition, mechanisms exist in much of the world permitting some form of challenge by generic drug marketers to our patents prior to, or
immediately following, the expiration of any regulatory exclusivity, and generic companies are increasingly employing aggressive strategies, such as “at risk” launches
to challenge our patent rights.
Our business also may rely on unpatented proprietary technology, know-how and trade secrets. If the confidentiality of this intellectual property is breached, it
could adversely impact our business.
Biotechnology Products
The ACA has created a framework for the approval of biosimilars in the U.S. following the expiration of 12 years of exclusivity for the innovator biologic, with a
potential six-month pediatric extension. Such biosimilars could reference biotechnology products already approved under the U.S. Public Health Service Act.
Additionally, the FDA has approved a biosimilar recombinant human growth hormone that referenced our biotechnology product, Genotropin , which was approved
under the U.S. Federal Food, Drug, and Cosmetic Act. In Europe, the European Commission has granted marketing authorizations for several biosimilars pursuant to a
set of general
31

and product class-specific guidelines for biosimilar approvals issued over the past few years, and in Japan, the regulatory authority has granted marketing authorizations
for certain biosimilars, including somatropin (the recombinant human growth hormone in our Genotropin product) pursuant to a guideline for biosimilar approvals
issued in 2009. If competitors are able to obtain marketing approval for biosimilars referencing our biotechnology products, our biotechnology products may become
subject to competition from biosimilars, with the attendant competitive pressure. Expiration or successful challenge of applicable patent rights could generally trigger
this competition, assuming any relevant exclusivity period has expired. We may face more litigation with respect to the validity and/or scope of patents relating to our
biotechnology products with substantial revenue.
Pfizer is developing biosimilar medicines. The developing pathway for registration and approval of biosimilar products in the U.S. could diminish the value of
investments in biosimilars that Pfizer has made, and may continue to make going forward. Other risks include the potential for steeper than anticipated price erosion due
to increased competitive intensity, coupled with high costs associated with clinical development, secondary patents or intellectual property challenges that may preclude
timely commercialization, and lower prescriptions of biosimilars due to concerns over protein comparability over innovator medicines.
Research Studies
Decisions about research studies made early in the development process of a drug candidate can have a substantial impact on the marketing strategy and payer
reimbursement possibilities once the drug receives approval. More detailed studies may demonstrate additional benefits that can help in the marketing and payer
reimbursement process, but they consume time and resources and can delay submitting the drug candidate for initial approval. We try to plan clinical trials prudently,
but there is no guarantee that a proper balance of speed and testing will be made in each case. The quality of our decisions in this area could affect our future results.
Foreign Exchange and Interest Rate Risk
Significant portions of our revenues and earnings, as well as our substantial international net assets, are exposed to changes in foreign exchange rates. 60% of our
total 2011 revenues were derived from international operations, including 27% from the Europe region and 20% from the Japan and the rest of Asia region. As we
operate in multiple foreign currencies, including the euro, the U.K. pound, the Japanese yen, the Canadian dollar and approximately 100 other currencies, changes in
those currencies relative to the U.S. dollar will impact our revenues and expenses. If the U.S. dollar weakens against a specific foreign currency, our revenues will
increase, having a positive impact, and our overall expenses will increase, having a negative impact, on net income. Likewise, if the U.S. dollar strengthens against a
specific foreign currency, our revenues will decrease, having a negative impact, and our overall expenses will decrease, having a positive impact on net income.
Therefore, significant changes in foreign exchange rates can impact our results and our financial guidance.
In addition, our interest-bearing investments, loans and borrowings are subject to risk from changes in interest rates and foreign exchange rates. These risks and
the measures we have taken to help contain them are discussed in the Financial Risk Management section of the MD&A in our 2011 Financial Report. For additional
details, see the Notes to Consolidated Financial Statements – Note 7E. Financial Instruments – Derivative Financial Instruments and Hedging Activities in our 2011
Financial Report. Those sections of our 2011 Financial Report are incorporated by reference.
Notwithstanding our efforts to foresee and mitigate the effects of changes in fiscal circumstances, we cannot predict with certainty changes in currency and
interest rates, inflation or other related factors affecting our businesses.
Risks Affecting International Operations
Our international operations also could be affected by capital and exchange controls, expropriation and other restrictive government actions, changes in
intellectual property legal protections and remedies, trade regulations
32

and procedures and actions affecting approval, production, pricing, and marketing of, reimbursement for and access to, our products, as well as by political unrest,
unstable governments and legal systems and inter-governmental disputes. Any of these changes could adversely affect our business.
Many emerging markets have experienced growth rates in excess of the world’s largest markets, leading to an increased contribution to the industry’s global
performance. As a result, we have been employing strategies to grow in emerging markets. However, there is no assurance that our strategies in emerging markets will
be successful or that these countries will continue to sustain these growth rates. In addition, some emerging market countries may be particularly vulnerable to periods of
financial instability or significant currency fluctuations or may have limited resources for healthcare spending, which, as discussed above, can adversely affect our
results.
Animal Health
Our Animal Health unit may be impacted by challenging global economic conditions, resulting in high unemployment rates and tight credit conditions. A high
unemployment rate typically results in reduced traffic in veterinary clinics, negatively impacting our companion animal business. Tight credit conditions limit the
borrowing power of livestock producers, causing some to switch to lower-priced alternatives. See Global Economic Conditions below.
Pfizer Nutrition
Pfizer Nutrition may be impacted by challenging global economic conditions and the resulting effect on consumer spending, both in developed and emerging
markets. The Nutrition business may also experience significant financial impact associated with changes in national, regional, and international laws, rules and
guidelines and their enforcement. Our infant and young child nutrition products are subject to an array of rules and regulations enforced by local government entities, as
well as treaties, conventions and guidelines from international authorities. Changes to these requirements can significantly impact costs relating to taxes, tariffs, trade,
labeling, marketing, manufacturing, and the overall availability of our products. See Global Economic Conditions below.
Consumer Healthcare
The Consumer Healthcare unit may be impacted by economic volatility and generic or store brand competition affecting consumer spending patterns and market
share gains of competitors’ branded products or generic store brands. In addition, regulatory and legislative outcomes regarding the safety, efficacy or unintended uses
of specific ingredients in our Consumer Healthcare products may require withdrawal and/or reformulation of certain products (e.g. cough/cold products). See Global
Economic Conditions
below.
Global Economic Conditions
In addition to industry-specific factors, we, like other businesses, continue to face the effects of the challenging economic environment, which have impacted our
biopharmaceutical operations in the U.S. and Europe, affecting the performance of products such as Lipitor , Celebrex and Lyrica
. We believe that patients experiencing
the effects of the challenging economic environment, including high unemployment levels and increases in co-pays, sometimes switch to generic products, delay
treatments, skip doses or use less effective treatments to reduce their costs. Challenging economic conditions in the U.S. also have increased the number of patients in
the Medicaid program, under which sales of pharmaceuticals are subject to substantial rebates and, in many states, to formulary restrictions limiting access to brand-
name drugs, including ours. In addition, we experienced continued pricing pressure in various economic environments around the world, including in Europe and in a
number of emerging markets, resulting in government-mandated reductions in prices for certain biopharmaceutical products, as well as government-imposed access
restrictions in certain countries.
33

The global economic downturn and the challenging global economic environment have not had, nor do we anticipate they will have, a material impact on our
liquidity. Due to our significant operating cash flows, financial assets, access to capital markets and available lines of credit and revolving credit agreements, we
continue to believe that we have the ability to meet our liquidity needs for the foreseeable future. As market conditions change, we continue to monitor our liquidity
position. However, there can be no assurance that our liquidity will not be affected by possible future changes in global financial markets and global economic
conditions.
Other potential impacts of these challenging economic conditions include declining sales; increased costs; changes in foreign exchange rates; a decline in the
value of, or a lower rate of return on, our financial assets and pension plan investments, which may require us to increase our pension funding obligations; adverse
government actions; delays or failures in the performance of customers, suppliers, and other third parties on whom we may depend for the performance of our business;
and the risk that our allowance for doubtful accounts may not be adequate.
Outsourcing
Outsourcing to third parties in areas including transaction processing, accounting, information technology, manufacturing, clinical trial execution, non-clinical
research, safety services and other areas could expose us to sub-optimal quality, missed deadlines, supply disruptions, non-compliance or reputational harm, all with
potential negative implications for our results. For example, we are transitioning our clinical trial execution model from multiple functional service providers to two
strategic partners. Any issues with one or both of these partners or during the transition could adversely impact our business.
Interactions with Healthcare Professionals
Risks and uncertainties apply where we provide something of value to a healthcare professional and/or government official, which, if found to be improper, could
potentially result in government enforcement actions and penalties. These risks may increase as non-U.S. jurisdictions adopt new anti-bribery laws and regulations.
Difficulties of Our Wholesale Distributors
In 2011, our largest wholesale distributor accounted for approximately 13% of our total revenue (and 30% of our total U.S. revenue), and our top three wholesale
distributors accounted for approximately 32% of our total revenue (and 77% of our total U.S. revenue). If one of our significant wholesale distributors encounters
financial or other difficulties, such distributor may decrease the amount of business that it does with us, and we may be unable to collect all the amounts that the
distributor owes us on a timely basis or at all, which could negatively impact our results of operations.
Product Manufacturing and Marketing Risks
Difficulties or delays in product manufacturing or marketing could affect future results through regulatory actions, shut-downs, approval delays, withdrawals,
recalls, penalties, supply disruptions or shortages, reputational harm, product liability, unanticipated costs or otherwise. Examples of such difficulties or delays include,
but are not limited to, the inability to increase production capacity commensurate with demand; the failure to predict market demand for, or to gain market acceptance
of, approved products; the possibility that the supply of incoming materials may be delayed or become unavailable and that the quality of incoming materials may be
substandard and not detected; or that we may fail to maintain appropriate quality standards throughout the internal and external supply network and/or comply with
current Good Manufacturing Practices and other applicable regulations.
Counterfeit Products
A counterfeit medicine is one that has been deliberately and fraudulently mislabeled as to its identity and source. A counterfeit Pfizer medicine, therefore, is one
manufactured by someone other than Pfizer, but which
34

appears to be the same as an authentic Pfizer medicine. Counterfeit medicines pose a risk to patient health and safety because of the conditions under which they are
manufactured – in unregulated, unlicensed, uninspected and often unsanitary sites – as well as the lack of regulation of their contents. Failure to mitigate the threat of
counterfeit medicines could adversely impact our business, by, among other things, causing the loss of patient confidence in the Pfizer name and in the integrity of our
medicines.
Cost and Expense Control/Unusual Events/Intangible Assets and Goodwill
Growth in costs and expenses, changes in product, segment and geographic mix and the impact of acquisitions, divestitures, restructurings, product withdrawals,
recalls and other unusual events that could result from evolving business strategies, evaluation of asset realization and organizational restructuring could adversely affect
future results. Such risks and uncertainties include, in particular, our ability to successfully implement our plans, announced February 1, 2011, regarding our research
and development function (see Research and Development Investment above), as well as our ability to realize the projected benefits of our cost-reduction and
productivity initiatives, including those related to our research and development function.
In addition, our consolidated balance sheet contains significant amounts of intangible assets, including goodwill. For IPR&D assets, the risk of failure is
significant, and there can be no certainty that these assets will ultimately yield successful products. The nature of the biopharmaceutical business is high-risk and
requires that we invest in a large number of projects in an effort to achieve a successful portfolio of approved products. Our ability to realize value on these significant
investments is often contingent upon, among other things, regulatory approvals and market acceptance. As such, we expect that many of these IPR&D assets will
become impaired and be written off at some time in the future.
Further, as discussed above in Item 1. Business under the caption Operating Segments , Pfizer used to report all of its biopharmaceutical businesses in a single
segment (Biopharmaceutical) and now reports these businesses in three operating segments (Primary Care; Specialty Care and Oncology; and Emerging Markets and
Established Products). As a result of this change, the goodwill previously associated with the single biopharmaceutical operating segment has been allocated among the
three new biopharmaceutical operating segments. While all reporting units can confront events and circumstances that can lead to impairments (such as, among other
things, unanticipated competition, an adverse action or assessment by a regulator, a significant adverse change in legal matters or in the business climate and/or a failure
to replace the contributions of products that lose exclusivity), in general, our increased number of biopharmaceutical reporting units significantly increases our risk of
goodwill impairment charges as smaller reporting units are inherently less able to absorb negative developments that might affect certain operating assets but not others.
Changes in Laws and Accounting Standards
Our future results could be adversely affected by changes in laws and regulations, including changes in accounting standards, taxation requirements (including
tax-rate changes, new tax laws and revised tax law and regulatory interpretations including changes affecting the taxation by the U.S. of income earned outside the U.S.
that may result from pending and possible future proposals), competition laws and environmental laws in the U.S. and other countries.
Terrorist Activity
Our future results could be adversely affected by changes in business, political and economic conditions, including the cost and availability of insurance, due to
the threat of terrorist activity in the U.S. and other parts of the world and related U.S. military action overseas.
Legal Proceedings
We and certain of our subsidiaries are involved in various patent, product liability, consumer, commercial, securities, antitrust, environmental, employment and
tax litigations and claims, government investigations and
35

other legal proceedings that arise from time to time in the ordinary course of our business. Litigation is inherently unpredictable, and excessive verdicts do occur.
Although we believe we have substantial defenses in these matters, we could in the future incur judgments, enter into settlements of claims or revise our expectations
regarding the outcome of certain matters, and such developments could have a material adverse effect on our results of operations or cash flows in the period in which
the amounts are paid or accrued.
Our activities relating to the sale and marketing and the pricing of our products are subject to extensive regulation under the U.S. Federal Food, Drug, and
Cosmetic Act, the Medicaid Drug Rebate Program, the U.S. Foreign Corrupt Practices Act and other federal and state statutes, including anti-kickback and false claims
laws, as well as similar laws in foreign jurisdictions. Like many companies in our industry, we have from time to time received inquiries and subpoenas and other types
of information demands from government authorities, and been subject to claims and other actions related to our business activities brought by governmental authorities,
as well as by consumers and private payers. In some instances, we have incurred significant expense and other adverse consequences as a result of these claims, actions
and inquiries. For example, these claims, actions and inquiries may relate to alleged failures to accurately interpret or identify or prevent non-compliance with the laws
and regulations associated with the dissemination of product information (approved and unapproved), potentially resulting in government enforcement and damage to
our reputation.
Patent claims include challenges to the coverage and/or validity of our patents on various products or processes. Although we believe we have substantial
defenses to these challenges with respect to all our material patents, there can be no assurance as to the outcome of these matters, and a loss in any of these cases could
result in a loss of patent protection for the drug at issue, which could lead to a significant loss of sales of that drug and could materially affect future results of
operations.
Business Development Activities
We expect to continue to enhance our in-line products and product pipeline through acquisitions, licensing and alliances. See the Overview of Our Performance,
Operating Environment, Strategy and Outlook – Our Business Development Initiatives section of the MD&A in our 2011 Financial Report, which is incorporated by
reference. However, these enhancement plans are subject to the availability and cost of appropriate opportunities and competition from other pharmaceutical companies
that are seeking similar opportunities and our ability to successfully identify and execute transactions.
Information Technology
We rely to a large extent upon sophisticated information technology systems and infrastructure. The size and complexity of our computer systems make them
potentially vulnerable to service interruption, malicious intrusion and random attack. Likewise, data privacy or security breaches by employees and others with
permitted access to our systems, including in some cases third-party service providers to which we may outsource certain business functions, may pose a risk that
sensitive data, including intellectual property or personal information, may be exposed to unauthorized persons or to the public. While we have invested heavily in the
protection of data and information technology, there can be no assurance that our efforts will prevent service interruptions, or identify breaches in our systems, that
could adversely affect our business and/or result in the loss of critical or sensitive information, which could result in financial, legal, business or reputational harm to us.
Failure to Realize the Anticipated Benefits of Strategic Initiatives and Acquisitions
Our future results may be affected by (i) the impact of, and our ability to successfully execute, any strategic alternatives we decide to pursue for our Animal
Health and Nutrition businesses, as well as any other corporate strategic initiatives we may pursue in the future, and (ii) our ability to realize the projected benefits of our
acquisition of King Pharmaceuticals, Inc., as well as any other acquisitions we may pursue in the future.
36

Not applicable.
In 2011, Pfizer continued to consolidate its operations to achieve efficiencies and to dispose of excess space. Presently, we have 521 leased properties and 400
owned properties, amounting to approximately 8 million and 51 million square feet, respectively, down from a total of approximately 67 million square feet of leased
and owned properties at the end of 2010. Our goal is to continue with further consolidation in 2012.
Pfizer corporate headquarters are in New York City. With the exception of the Specialty Care customer-focused unit (which is headquartered in Collegeville,
Pennsylvania), our biopharmaceutical units also are headquartered in New York City. Our other business units are headquartered in Madison, New Jersey.
In 2011, we successfully disposed of surplus space, exiting or reducing our real estate space in certain locations in the United States, Europe, Australia and Puerto
Rico. Further, active marketing is continuing in a number of locations.
Our biopharmaceutical and other businesses expect to continue to own and lease space around the world for sales and marketing, customer service and
administrative support functions. In many locations these businesses will be co-located to achieve synergies and operational efficiencies.
Our Worldwide R&D facilities support our R&D organizations around the world, with heavy concentration in North America. We are continuing with
implementation of our previously announced R&D footprint reduction, which includes a substantial reduction at our Sandwich, U.K. site, and a shift of our
Cardiovascular, Metabolic and Endocrine Disease (CVMED) and Neuroscience research units from our site in Groton, CT to Cambridge, MA, where we signed a lease
with Massachusetts Institute of Technology in the Kendall Square area, with occupancy anticipated in early 2014. We are presently marketing for sale, lease, or sale and
lease-back, either a portion or all of certain of our R&D campuses. Further, we disposed of our toxicology site in Catania, Italy; exited our R&D site in Aberdeen, U.K;
disposed of a vacant site in St. Louis, MO; and sold our Gosport, U.K. R&D site. Additionally, we opened Centers for Therapeutic Innovation laboratories in Boston,
MA; New York, NY; and South San Francisco, CA.
We have veterinary medicine research and development operations in owned or leased facilities in Kalamazoo and Richland Township, MI; Durham, NC; San
Diego, CA; Exton, PA; Thane, India; Wavre, Belgium; Brisbane, Australia; and British Columbia, Canada.
Our Pfizer Global Supply (PGS) Division is headquartered in various locations, with leadership primarily in New York, NY and in Peapack, NJ. PGS operates 90
plants around the world, which manufacture products for our commercial divisions. Locations with major manufacturing facilities include Belgium, China, Germany,
Ireland, Italy, Japan, Philippines, Puerto Rico, Singapore and the U.S. Our Global Supply Division’
s plant network strategy is expected to result in the exit of 10 of these
sites over the next several years. PGS also operates multiple distribution facilities around the world.
In general, we believe that our properties are well-maintained, adequate and suitable for their purposes. See the Notes to Consolidated Financial Statements –
Note 9. Property, Plant and Equipment
in our 2011 Financial Report, which provides amounts invested in land, buildings and equipment and which is incorporated by
reference. See also the discussion in the Notes to Consolidated Financial Statements – Note 15. Lease Commitments in our 2011 Financial Report, which is also
incorporated by reference.
37
ITEM 1B.
UNRESOLVED STAFF COMMENTS
ITEM 2.
PROPERTIES

Certain legal proceedings in which we are involved are discussed in the Notes to Consolidated Financial Statements – Note 17. Commitments and Contingencies
in our 2011 Financial Report, which is incorporated by reference.
Not applicable.
38
ITEM 3.
LEGAL PROCEEDINGS
ITEM 4.
MINE SAFETY DISCLOSURE

EXECUTIVE OFFICERS OF THE COMPANY
The executive officers of the Company are set forth in this table. Each holds the office or offices indicated until his or her successor is chosen and qualified at the
regular meeting of the Board of Directors to be held on the date of the 2012 Annual Meeting of Shareholders. Each of the executive officers is a member of the Pfizer
Executive Leadership Team.
39
Name
Age
Position
Ian C. Read
58
Chairman and Chief Executive Officer since December 2011. President and Chief Executive Officer from
December 2010 until December 2011. Senior Vice President, Group President of the Worldwide
Biopharmaceutical Businesses (Primary Care, Specialty Care, Oncology, Established Products and Emerging
Markets), from 2006 through December 2010. Since joining Pfizer in 1978 as an operational auditor,
Mr. Read has held various positions of increasing responsibility in pharmaceutical operations. He worked in Latin
America through 1995, holding positions including Chief Financial Officer, Pfizer Mexico, and Country Manager,
Pfizer Brazil. In 1996, Mr. Read was appointed President of Pfizer’s International Pharmaceuticals Group, with
responsibility for Latin America and Canada. He became Executive Vice President, Europe in 2000, was named a
Corporate Vice President in 2001, and assumed responsibility for Canada, in addition to Europe, in 2002.
Mr. Read later became accountable for operations in both the Africa/Middle East region and Latin America as
well. Currently a Director of Kimberly-Clark Corporation. Serves on the Boards of Pharmaceutical Research and
Manufacturers of America (PhRMA), the European Federation of Pharmaceutical Industries and Associations, and
the Partnership for New York City. Our Director since December 2010 and Chair of our Board’s Executive
Committee.
Olivier Brandicourt
56
President and General Manager of Pfizer Primary Care since 2009. In early 2009, served as President and General
Manager of Pfizer Specialty Care. Senior Vice President and General Manager of U.S. Pratt Business Unit from
2007 until 2008. Managing Director of the United Kingdom/Ireland Pfizer subsidiary from 2004 to 2007.
Frank A. D’Amelio
54
Executive Vice President, Business Operations and Chief Financial Officer since December 2010. Senior Vice
President and Chief Financial Officer from September 2007 until December 2010. Prior to joining Pfizer he was
Senior Executive Vice President of Integration and Chief Administrative Officer of Alcatel-Lucent from
November 2006 until August 2007. Chief Operating Officer of Lucent Technologies from January 2006 until
November 2006. Director of Humana, Inc. and Chair of the Humana Audit Committee. He is a Director of the
Independent College Fund of New Jersey.
Mikael Dolsten
53
President of Worldwide Research and Development since December 2010. Senior Vice President; President of
Worldwide Research and Development from May 2010 until December 2010. Senior Vice President; President of
Pfizer BioTherapeutics Research & Development Group from October 2009 until May 2010. He was Senior Vice
President of Wyeth and President, Wyeth Research from June 2008 until October 2009. He was a Private Equity
Partner at Orbimed Advisors, LLC from January 2008 until June 2008. Dr. Dolsten was Global Head, Corporate
Division Pharma Research and Discovery, of Boehringer Ingelheim Corporation from 2003 to 2007.

40
Name
Age
Position
Geno J. Germano
51
President and General Manager, Pfizer Specialty Care and Oncology since December 2010. President and General
Manager, Specialty Care from October 2009 until December 2010. President, U.S. Pharmaceuticals and Women’s
Health Care Unit, Wyeth Pharmaceuticals from 2008 through October 2009. President and General Manager, U.S.
Pharmaceutical Business Unit, Wyeth Pharmaceuticals from 2007 through 2008. Executive Vice President and
General Manager, Pharmaceutical Business Unit, Wyeth Pharmaceuticals from 2004 through 2007. Member of the
Board of Trustees for Albany College of Pharmacy and Member of the Board of Directors of BIO –
Biotechnology
Industry Organization.
Charles H. Hill III
56
Executive Vice President, Worldwide Human Resources since December 2010. Senior Vice President, Human
Resources for Worldwide Biopharmaceuticals Businesses from 2008 through December 2010. Vice President,
Human Resources, Worldwide Pharmaceutical Operations from 2004 through 2008.
Douglas M. Lankler
46
Executive Vice President, Chief Compliance and Risk Officer since February 2011. Executive Vice President,
Chief Compliance Officer from December 2010 until February 2011. Senior Vice President and Chief Compliance
Officer from January 2010 until December 2010. Senior Vice President, Deputy General Counsel and Chief
Compliance Officer from August 2009 until January 2010. Senior Vice President, Associate General Counsel and
Chief Compliance Officer from October 2006 until August 2009. Prior to October 2006, Mr. Lankler held various
positions of increasing responsibility within the Pfizer Legal Division.
Freda C. Lewis-Hall
57
Executive Vice President, Chief Medical Officer since December 2010. Senior Vice President, Chief Medical
Officer from May 2009 until December 2010. Previously, she was Chief Medical Officer and Executive Vice
President, Medicines Development at Vertex Pharmaceuticals from June 2008 until May 2009. Dr. Lewis-Hall
was Senior Vice President, U.S. Pharmaceuticals, Medical Affairs for Bristol-Myers Squibb Company from 2003
until May 2008.
Kristin C. Peck
40
Executive Vice President, Worldwide Business Development and Innovation since December 2010. Senior Vice
President, Worldwide Business Development, Strategy and Innovation from April 2010 until December 2010.
Senior Vice President, Worldwide Strategy and Innovation from 2008 until April 2010. Vice President, Strategic
Planning, from 2007 to 2008. Chief of Staff to the Vice Chairman from 2006 to 2007 and Senior Director,
Strategic Planning from 2004 to 2006.
Cavan M. Redmond
51
Group President, Animal Health, Consumer Healthcare and Corporate Strategy since August 2011. Group
President, Animal Health, Consumer Healthcare, Capsugel and Corporate Strategy from December 2010 until
August 2011. Senior Vice President; Group President, Pfizer Diversified Businesses from October 2009 until
December 2010. President, Wyeth Consumer Healthcare and Animal Health Business from May 2009 until
October 2009. President, Wyeth Consumer Healthcare from December 2007 until May 2009. Executive Vice
President and General Manager, BioPharma, Wyeth Pharmaceuticals from 2003 until December 2007.

41
Name
Age
Position
Amy W. Schulman
51
Executive Vice President and General Counsel; President and General Manager, Nutrition since December 2010.
Senior Vice President and General Counsel from June 2008 until December 2010. Ms. Schulman was a partner at
the law firm of DLA Piper from 1997 until joining Pfizer in June 2008. Member of the Board of Directors of
Wesleyan University and the Brooklyn Academy of Music.
David S. Simmons
47
President and General Manager, Emerging Markets and Established Products units since December 2010.
President and General Manager, Established Products from 2008 until December 2010. Since joining Pfizer in
1996, Mr. Simmons has held various positions of increasing responsibility in pharmaceutical operations, including
Regional President, Central Southern Europe; Vice President of Marketing, Pfizer Canada; and Country Manager,
Pfizer Greece. He is a member of the U.S.
-
Russia Business Council and a Trustee of The Linsly School.
Sally Susman
50
Executive Vice President, Policy, External Affairs and Communications of Pfizer since December 2010. Senior
Vice President, Policy, External Affairs and Communications from December 2009 until December 2010. Senior
Vice President and Chief Communications Officer from February 2008 until December 2009. Prior to joining
Pfizer, Ms. Susman held senior level positions at The Estee Lauder Companies, including Executive Vice
President from 2004 to January 2008.

PART II
The principal market for our Common Stock is the New York Stock Exchange. Our stock is also listed on the London Stock Exchange and the SIX Swiss Stock
Exchange and is traded on various United States regional stock exchanges. Additional information required by this item is incorporated by reference from the table
captioned Quarterly Consolidated Financial Data (Unaudited) in our 2011 Financial Report.
The following table provides certain information with respect to our purchases of shares of the Company’s Common Stock during the fiscal fourth quarter of
2011:
Issuer Purchases of Equity Securities (a)
Information required by this item is incorporated by reference from the discussion under the heading Financial Summary in our 2011 Financial Report.
Information required by this item is incorporated by reference from the discussion under the heading Financial Review in our 2011 Financial Report.
42
ITEM 5. MARKET FOR THE COMPANY’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF
EQUITY SECURITIES
Period
Total Number of
Shares Purchased
Average
Price
Paid per
Share
Total Number of
Shares Purchased as
Part of Publicly
Announced Plan
Approximate
Dollar Value of
Shares that May
Yet Be Purchased
Under the Plan
October 3, 2011
Through
October 30, 2011
34,244,258
$
18.44
34,153,990
$
2,614,785,325
October 31, 2011
Through
November 30, 2011
65,765,061
$
19.63
65,705,854
$
1,324,856,629
December 1, 2011
Through
December 31, 2011
63,210,330
$
20.45
63,124,603
$
10,034,160,705
Total
163,219,649
$
19.70
162,984,447
On February 1, 2011, Pfizer announced that the Board of Directors had authorized a new $5 billion share-
purchase plan (the February 2011 Stock Purchase Plan).
On December 12, 2011, Pfizer announced that the Board of Directors had authorized an additional $10 billion share-purchase plan (the December 2011 Stock
Purchase Plan). Pfizer currently expects to repurchase approximately $5 billion of its common stock in 2012, with the remaining authorized amount available in
2013 and beyond.
In addition to amounts purchased under the February and December 2011 Stock Purchase Plans, these columns reflect the following transactions during the fourth
quarter of 2011: (i) the surrender to Pfizer of 191,816 shares of common stock to satisfy tax withholding obligations in connection with the vesting of restricted
stock and restricted stock units issued to employees; (ii) the open market purchase by the trustee of 40,445 shares of common stock in connection with the
reinvestment of dividends paid on common stock held in trust for employees who were granted performance share awards and who deferred receipt of such
awards and (iii) the surrender to Pfizer of 2,941 shares of common stock to satisfy tax withholding obligations in connection with the vesting of performance
share awards issued to employees.
ITEM 6.
SELECTED FINANCIAL DATA
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
(b) (b) (a) (a)
(a)
(b)

Information required by this item is incorporated by reference from the discussion under the Financial Risk Management section of the MD&A in our 2011
Financial Report.
Information required by this item is incorporated by reference from the Report of Independent Registered Public Accounting Firm on the Consolidated Financial
Statements in our 2011 Financial Report and from the consolidated financial statements, related notes and supplementary data in our 2011 Financial Report.
None.
Disclosure Controls
As of the end of the period covered by this 2011 Form 10-K, we carried out an evaluation, under the supervision and with the participation of our principal
executive officer and principal financial officer, of the effectiveness of the design and operation of our disclosure controls and procedures (as such term is defined in
Rules 13a-15(e) and 15d-15(e) under the Exchange Act). Based on this evaluation, our principal executive officer and principal financial officer concluded that our
disclosure controls and procedures are effective in alerting them in a timely manner to material information required to be disclosed in our periodic reports filed with the
SEC.
Internal Control over Financial Reporting
Management’s report on the Company’s internal control over financial reporting (as such term is defined in Rules 13a-15(f) and 15d-15(f) under the Exchange
Act), and the related report of our independent registered public accounting firm, are included in our 2011 Financial Report under the headings Management’
s Report on
Internal Control Over Financial Reporting
and Report of Independent Registered Public Accounting Firm on Internal Control Over Financial Reporting , respectively,
and are incorporated by reference.
Changes in Internal Controls
During our most recent fiscal quarter, there has not been any change in our internal control over financial reporting (as such term is defined in Rules 13a-15(f)
and 15d-15(f) under the Exchange Act) that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting. However,
we do wish to highlight some changes which, taken together, are expected to have a favorable impact on our controls over a multi-year period. We continue to pursue a
multi-year initiative to outsource some transaction-processing activities within certain accounting processes and are migrating to a consistent enterprise resource
planning system across the organization. These are enhancements of ongoing activities to support the growth of our financial shared service capabilities and standardize
our financial systems. None of these initiatives is in response to any identified deficiency or weakness in our internal control over financial reporting.
Not applicable.
43
ITEM 7A.
QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
ITEM 8.
FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
ITEM 9.
CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
ITEM 9A.
CONTROLS AND PROCEDURES
ITEM 9B. OTHER INFORMATION

PART III
Information about our Directors is incorporated by reference from the discussion under the heading Proposals Requiring Your Vote – Item 1– Election of
Directors
in our 2012 Proxy Statement. Information about compliance with Section 16(a) of the Exchange Act is incorporated by reference from the discussion under
the heading Section 16(a) Beneficial Ownership Reporting Compliance, Related Person Transactions, and Indemnification – Section 16(a) Beneficial Ownership
Reporting Compliance
in our 2012 Proxy Statement. Information about the Pfizer Policies on Business Conduct governing our employees, including our Chief
Executive Officer, Chief Financial Officer and Principal Accounting Officer, and the Code of Business Conduct and Ethics governing our Directors, is incorporated by
reference from the discussions under the headings Governance of the Company – Governance Information – Pfizer Policies on Business Ethics and Conduct and – Code
of Conduct for Directors in our 2012 Proxy Statement. Information regarding the procedures by which our stockholders may recommend nominees to our Board of
Directors is incorporated by reference from the discussion under the headings Governance of the Company – Governance Information – Criteria for Board Membership
and Requirements, Including Deadlines, for Submission of Proxy Proposals and Nomination of Directors in our 2012 Proxy Statement. Information about our Audit
Committee, including the members of the Committee, and our Audit Committee financial experts, is incorporated by reference from the discussion under the heading
Governance of the Company – Board and Committee Information – The Audit Committee in our 2012 Proxy Statement. The balance of the information required by this
item is contained in the discussion entitled Executive Officers of the Company in Part I of this 2011 Form 10-K.
Information about Director and executive compensation is incorporated by reference from the discussion under the headings Governance of the Company –
Compensation of Non-Employee Directors, Executive Compensation, and Governance of the Company – Board and Committee Information – Compensation Committee
–
Compensation Committee Interlocks and Insider Participation in our 2012 Proxy Statement.
Information required by this item is incorporated by reference from the discussion under the headings Executive Compensation – Equity Compensation Plan
Information
and Securities Ownership in our 2012 Proxy Statement.
Information about certain relationships and transactions with related parties is incorporated by reference from the discussion under the headings Section 16(a)
Beneficial Ownership Reporting Compliance, Related Person Transactions, and Indemnification
– Review of Related Person Transactions and – Transactions with
Related Persons
in our 2012 Proxy Statement. Information about director independence is incorporated by reference from the discussion under the heading Governance
of the Company – Governance Information – Director Independence in our 2012 Proxy Statement.
Information about the fees for professional services rendered by our independent auditors in 2011 and 2010 is incorporated by reference from the discussion
under the heading Proposals Requiring Your Vote – Item 2 – Ratification of Independent Registered Public Accounting Firm – Audit and Non-Audit Fees in our 2012
Proxy Statement. Our Audit Committee’s policy on pre-approval of audit and permissible non-audit services of our independent auditors is incorporated by reference
from the discussion under the heading Proposals Requiring Your Vote – Item 2 – Ratification of Independent Registered Public Accounting Firm – Policy on Audit
Committee Pre-Approval of Audit and Permissible Non-Audit Services of Independent Registered Public Accounting Firm in our 2012 Proxy Statement.
44
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
ITEM 11. EXECUTIVE COMPENSATION
ITEM 12.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
ITEM 13.
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
ITEM 14.
PRINCIPAL ACCOUNTING FEES AND SERVICES

PART IV
15(a)(1) Financial Statements. The following consolidated financial statements, related notes, report of independent registered public accounting firm and
supplementary data from our 2011 Financial Report are incorporated by reference into Item 8 of Part II of this 2011 Form 10-K:
15(a)(2) Financial Statement Schedules. Schedules are omitted because they are not required or because the information is provided elsewhere in the financial
statements. The financial statements of unconsolidated subsidiaries are omitted because, considered in the aggregate, they would not constitute a significant subsidiary.
15(a)(3) Exhibits. These exhibits are available upon request. Requests should be directed to Matthew Lepore, Vice President and Corporate Secretary, Chief
Counsel-Corporate Governance, Pfizer Inc., 235 East 42nd Street, New York, NY 10017-5755. The exhibit numbers preceded by an asterisk (*) indicate exhibits filed
with this 2011 Form 10-K. All other exhibit numbers indicate exhibits filed by incorporation by reference. Exhibit numbers 10(1) through 10(24) are management
contracts or compensatory plans or arrangements.
45
ITEM 15. EXHIBITS, FINANCIAL STATEMENT SCHEDULES
•
Report of Independent Registered Public Accounting Firm on the Consolidated Financial Statements
•
Consolidated Statements of Income
•
Consolidated Balance Sheets
•
Consolidated Statements of Shareholders
’
Equity
•
Consolidated Statements of Cash Flows
•
Notes to Consolidated Financial Statements
•
Quarterly Consolidated Financial Data (Unaudited)
3(1)
Our Restated Certificate of Incorporation dated April 12, 2004, is incorporated by reference from our 10-Q report for the period ended March 28, 2004 (File
No. 001
-
03619).
3(2)
Amendment dated May 1, 2006 to Restated Certificate of Incorporation dated April 12, 2004, is incorporated by reference from our 10-Q report for the period
ended July 2, 2006 (File No. 001
-
03619).
3(3)
Our By
-
laws, as amended April 22, 2010, are incorporated by reference from our 10
-
Q report for the period ended April 4, 2010 (File No. 001
-
03619).
4(1)
Indenture, dated as of January 30, 2001, between us and The Chase Manhattan Bank, is incorporated by reference from our 8-
K report filed on January 30, 2001
(File No. 001
-
03619).
4(2)
First Supplemental Indenture, dated as of March 24, 2009, between us and The Bank of New York Mellon (successor to JPMorgan Chase Bank, N.A. (formerly
JPMorgan Chase Bank, formerly The Chase Manhattan Bank)), as Trustee, to Indenture dated as of January 30, 2001, is incorporated by reference from our 10-
Q report for the period ended June 28, 2009 (File No. 001
-
03619).
4(3)
Second Supplemental Indenture, dated as of June 2, 2009, between us and The Bank of New York Mellon (successor to JPMorgan Chase Bank, N.A. (formerly
JPMorgan Chase Bank, formerly The Chase Manhattan Bank)), as Trustee, to Indenture dated as of January 30, 2001, is incorporated by reference from our 8-
K
report filed on June 3, 2009 (File No. 001
-
03619).
We agree to furnish to the SEC, upon request, a copy of each instrument with respect to issuances of long-term debt of the Company and its subsidiaries.
46
4(4)
Indenture, dated as of April 10, 1992, between Wyeth and The Bank of New York Mellon (as successor to JPMorgan Chase Bank, N.A.), as Trustee, is
incorporated by reference from Wyeth
’
s Registration Statement on Form S
-
3 (File No. 33
-
57339), filed on January 18, 1995.
4(5)
Supplemental Indenture, dated as of October 13, 1992, between Wyeth and The Bank of New York Mellon (as successor to JPMorgan Chase Bank, N.A.),
as Trustee, is incorporated by reference from Wyeth
’
s Registration Statement on Form S
-
3 (File No. 33
-
57339), filed on January 18, 1995.
4(6)
Third Supplemental Indenture, dated as of February 14, 2003, between Wyeth and The Bank of New York Mellon (as successor to JPMorgan Chase Bank,
N.A.), as Trustee, is incorporated by reference from Wyeth
’
s 2002 10
-
K report (File No. 001
-
01225).
4(7)
Fifth Supplemental Indenture, dated as of December 16, 2003, between Wyeth and The Bank of New York Mellon (as successor to JPMorgan Chase Bank,
N.A.), as Trustee, is incorporated by reference from Wyeth
’
s 2003 10
-
K report (File No. 001
-
01225).
4(8)
Sixth Supplemental Indenture, dated as of November 14, 2005, between Wyeth and The Bank of New York Mellon (as successor to JPMorgan Chase
Bank, N.A.), as Trustee, is incorporated by reference from Wyeth
’
s 8
-
K report filed on November 15, 2005 (File No. 001
-
01225).
4(9)
Seventh Supplemental Indenture, dated as of March 27, 2007, between Wyeth and The Bank of New York Mellon (as successor to JPMorgan Chase Bank,
N.A.), as Trustee, is incorporated by reference from Wyeth
’
s 8
-
K report filed on March 28, 2007 (File No. 001
-
01225).
4(10)
Eighth Supplemental Indenture, dated as of October 30, 2009, between Wyeth, us and The Bank of New York Mellon (as successor to JPMorgan Chase
Bank, formerly The Chase Manhattan Bank), as Trustee, to Indenture dated as of April 10, 1992 (as amended on October 13, 1992), is incorporated by
reference from our 8
-
K report filed on November 3, 2009 (File No. 001
-
03619).
4(11)
Except as set forth in Exhibits 4(1) – (10) above, the instruments defining the rights of holders of long-term debt securities of the Company and its
subsidiaries have been omitted.
10(1)
2001 Stock and Incentive Plan is incorporated by reference from our Proxy Statement for the 2001 Annual Meeting of Shareholders (File No. 001
-
03619).
*10(2)
Pfizer Inc. 2004 Stock Plan, as Amended and Restated.
10(3)
Form of Stock Option Grant Notice and Summary of Key Terms is incorporated by reference from our 10-Q report for the period ended September 26,
2004 (File No. 001
-
03619).
10(4)
Form of Performance-Contingent Share Award Grant Notice is incorporated by reference from our 10-Q report for the period ended September 26, 2004
(File No. 001
-
03619).
*10(5)
Amended and Restated Nonfunded Supplemental Retirement Plan, together with all material Amendments.
*10(6)
Amended and Restated Nonfunded Deferred Compensation and Supplemental Savings Plan.
1
1

47
10(7)
Executive Annual Incentive Plan is incorporated by reference from our Proxy Statement for the 1997 Annual Meeting of Shareholders (File No. 001-
03619).
10(8)
Deferred Compensation Plan is incorporated by reference from our 1997 10
-
K report (File No. 001
-
03619).
10(9)
Non
-
Employee Directors
’
Retirement Plan (frozen as of October 1996) is incorporated by reference from our 1996 10
-
K report (File No. 001
-
03619).
10(10)
Restricted Stock Plan for Non
-
Employee Directors is incorporated by reference from our 1996 10
-
K report (File No. 001
-
03619).
*10(11)
Amended and Restated Wyeth Supplemental Employee Savings Plan (effective as of January 1, 2005), together with all material Amendments.
*10(12)
Amended and Restated Wyeth Supplemental Executive Retirement Plan (effective as of January 1, 2005), together with all material Amendments.
10(13)
Warner
-
Lambert Company 1996 Stock Plan, as amended, is incorporated by reference from Warner
-
Lambert
’
s 1999 10
-
K report (File No. 001
-
03608).
10(14)
Wyeth Directors’ Deferral Plan (as amended through December 15, 2007) is incorporated by reference from Wyeth’s 2007 10-K report (File No. 001-
01225).
10(15)
The form of Indemnification Agreement with each of our non-employee Directors is incorporated by reference from our 1996 10-K report (File No. 001-
03619).
10(16)
The form of Indemnification Agreement with each of the Named Executive Officers identified in our 2012 Proxy Statement is incorporated by reference
from our 1997 10
-
K report (File No. 001
-
03619).
10(17)
Post-Retirement Consulting Agreement, dated as of April 20, 2000, between us and William C. Steere, Jr., is incorporated by reference from our 10-Q
report for the period ended April 2, 2000 (File No. 001
-
03619).
10(18)
Letter to Frank A. D’Amelio regarding replacement pension benefit dated August 22, 2007 is incorporated by reference from our 8-K report filed on
August 22, 2007 (File No. 001
-
03619).
10(19)
Executive Severance Plan is incorporated by referenced from our 8
-
K report filed on February 20, 2009 (File No. 001
-
03619).
10(20)
Annual Retainer Unit Award Plan (for Non-Employee Directors) (frozen as of March 1, 2006) as amended, is incorporated by reference from our 2008
10
-
K report (File No. 001
-
03619).
10(21)
Nonfunded Deferred Compensation and Unit Award Plan for Non
-Employee Directors, as amended, is incorporated by reference from our 10-Q report
for the period ended July 3, 2011 (File No. 001
-
03619).
10(22)
Form of Special Award Letter Agreement is incorporated by reference from our 8
-
K report filed on October 28, 2009 (File No. 001
-
03619).

48
10(23)
Offer Letter to G. Mikael Dolsten, dated April 6, 2009, is incorporated by reference from our 10-
Q report for the period ended April 3, 2011 (File No.
001
-
03619).
10(24)
Offer Letter to Geno J. Germano, dated April 6, 2009, is incorporated by reference from our 10-Q report for the period ended April 3, 2011 (File No.
001
-
03619).
*12
Computation of Ratio of Earnings to Fixed Charges.
*13
Portions of the 2011 Financial Report, which, except for those sections incorporated by reference, are furnished solely for the information of the SEC
and are not to be deemed
“
filed.
”
*21
Subsidiaries of the Company.
*23
Consent of KPMG LLP, Independent Registered Public Accounting Firm.
*24
Power of Attorney (included as part of signature page).
*31.1
Certification by the Chief Executive Officer Pursuant to Section 302 of the Sarbanes
-
Oxley Act of 2002.
*31.2
Certification by the Chief Financial Officer Pursuant to Section 302 of the Sarbanes
-
Oxley Act of 2002.
*32.1
Certification by the Chief Executive Officer Pursuant to 18 U.S.C. Section 1350, as Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of
2002.
*32.2
Certification by the Chief Financial Officer Pursuant to 18 U.S.C. Section 1350, as Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of
2002.
*101.INS
XBRL Instance Document
*101.SCH
XBRL Taxonomy Extension Schema
*101.CAL
XBRL Taxonomy Extension Calculation Linkbase
*101.LAB
XBRL Taxonomy Extension Label Linkbase
*101.PRE
XBRL Taxonomy Extension Presentation Linkbase
*101.DEF
XBRL Taxonomy Extension Definition Document

SIGNATURES
Under the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, this report was signed on behalf of the Registrant by the authorized person
named below.
We, the undersigned directors and officers of Pfizer Inc., hereby severally constitute Amy W. Schulman and Matthew Lepore, and each of them singly, our true
and lawful attorneys with full power to them and each of them to sign for us, in our names in the capacities indicated below, any and all amendments to this Annual
Report on Form 10-K filed with the Securities and Exchange Commission.
Under the requirements of the Securities Exchange Act of 1934, this report was signed by the following persons on behalf of the Registrant and in the capacities
and on the date indicated.
49
Pfizer Inc.
Dated: February 28, 2012
By:
/s/ M
ATTHEW
L
EPORE
Matthew Lepore
Vice President and Corporate Secretary,
Chief Counsel – Corporate Governance
Signature
Title
Date
/
S
/ I
AN
C. R
EAD
Ian C. Read
Chairman, Chief Executive Officer and Director
(Principal Executive Officer)
February 28, 2012
/
S
/ F
RANK
A. D’A
MELIO
Frank A. D’Amelio
Executive Vice President, Business
Operations and Chief Financial Officer (Principal
Financial Officer)
February 28, 2012
/
S
/ L
ORETTA
V. C
ANGIALOSI
Loretta V. Cangialosi
Senior Vice President—Controller
(Principal Accounting Officer)
February 28, 2012
/
S
/ D
ENNIS
A. A
USIELLO
Dennis A. Ausiello
Director
February 28, 2012
/
S
/ M
ICHAEL
S. B
ROWN
Michael S. Brown
Director
February 28, 2012
/
S
/ M. A
NTHONY
B
URNS
M. Anthony Burns
Director
February 28, 2012
/
S
/ W. D
ON
C
ORNWELL
W. Don Cornwell
Director
February 28, 2012
/
S
/ F
RANCES
D. F
ERGUSSON
Frances D. Fergusson
Director
February 28, 2012
/
S
/ W
ILLIAM
H. G
RAY
III
William H. Gray III
Director
February 28, 2012
/
S
/ H
ELEN
H. H
OBBS
Helen H. Hobbs
Director
February 28, 2012

50
Signature
Title
Date
/
S
/ C
ONSTANCE
J. H
ORNER
Constance J. Horner
Director
February 28, 2012
/
S
/ S
UZANNE
N
ORA
J
OHNSON
Suzanne Nora Johnson
Director
February 28, 2012
/
S
/ J
AMES
M. K
ILTS
James M. Kilts
Director
February 28, 2012
/
S
/ G
EORGE
A. L
ORCH
George A. Lorch
Director
February 28, 2012
/
S
/ J
OHN
P. M
ASCOTTE
John P. Mascotte
Director
February 28, 2012
/
S
/ S
TEPHEN
W. S
ANGER
Stephen W. Sanger
Director
February 28, 2012
/
S
/ M
ARC
T
ESSIER
-L
AVIGNE
Marc Tessier
-
Lavigne
Director
February 28, 2012

Exhibit 10(2)
Pfizer Inc. 2004 Stock Plan,
As Amended and Restated
(through February 23, 2012)
SECTION 1. PURPOSE
The purpose of the Pfizer Inc. 2004 Stock Plan (“the Plan”) is to furnish a material incentive to employees and non-employee Directors of
the Company and its Affiliates by making available to them the benefits of a larger common stock ownership in the Company through stock
options and awards. It is believed that these increased incentives stimulate the efforts of employees and non-employee Directors towards the
continued success of the Company and its Affiliates, as well as assist in the recruitment of new employees and non-employee Directors. The
Plan was amended and restated as of January 1, 2008, to reflect the intended exemption from Section 409A (as defined below) for certain
Awards, as well as continued compliance with Section 409A for certain other Awards, and further amended and restated, as of April 23,
2009, to make certain other changes designed to promote the effective operation of the Plan and to replenish the number of Shares available
for grant.
SECTION 2. DEFINITIONS
As used in the Plan, the following terms shall have the meanings set forth below:
1
(a) “Affiliate” shall mean (i) any Person that directly, or through one or more intermediaries, controls, or is controlled by, or is under
common control with, the Company or (ii) any entity in which the Company has a significant equity interest, as determined by the
Committee.
(b) “Award” shall mean any Option, Stock Appreciation Right, Restricted Stock Award, Performance Share, dividend equivalent,
Other Stock Unit Award or any other right, interest or option relating to Shares issued and delivered pursuant to the provisions of
the Plan.
(c) “Award Agreement” shall mean any written agreement, contract or other instrument or document evidencing any Award granted
by the Committee hereunder, which in the sole and absolute discretion of the Committee may, but need not, be signed or
acknowledged by the Company or the Participant.
(d)
“
Board
”
shall mean the Board of Directors of the Company.
(e) “Cause” shall mean a willful breach of duty in the course of employment. No act or failure to act shall be deemed “willful” unless
done, or omitted to be done, not in good faith and without reasonable belief that the action or omission was in the best interest of
the Company and its Affiliates.
(f) “Change in Control” shall mean the occurrence of any of the following events: (i) at any time during the initial two-year period
following the Effective Date or during each subsequent Renewal Term, as the case may be, at least a majority of the Board shall
cease to consist of “Continuing Directors” (meaning directors of the Company who either were directors at the beginning of such
initial two-year period or subsequent Renewal Term, as the case may be, or who subsequently became directors and whose
election, or nomination for election by the Company’s stockholders, was approved by a majority of the then Continuing
Directors); or (ii) any “person” or “group” (as determined for purposes of Section 13(d)(3) of the Exchange Act, except any
majority-owned subsidiary of the Company or any employee benefit plan of the Company or any trust thereunder, shall have
acquired “beneficial ownership” (as determined for purposes of Securities and Exchange Commission (“SEC”) Regulation 13d-3)
of Shares having 20% or more of the voting power of all

2
outstanding Shares, unless such acquisition is approved by a majority of the directors of the Company in office immediately
preceding such acquisition; or (iii) a merger or consolidation occurs to which the Company is a party, in which outstanding Shares
are converted into shares of another company (other than a conversion into shares of voting common stock of the successor
corporation or a holding company thereof representing 80% of the voting power of all capital stock thereof outstanding
immediately after the merger or consolidation) or other securities (of either the Company or another company) or cash or other
property; or (iv) the sale of all, or substantially all, of the Company’s assets occurs; or (v) the stockholders of the Company
approve a plan of complete liquidation of the Company.
(g) “Change in Control Price” means, with respect to a Share, the higher of (A) the highest reported sales price, regular way, of such
Share in any transaction reported on the New York Stock Exchange Composite Tape or other national exchange on which such
Shares are listed or on the Nasdaq National Market during the 60-
day period prior to and including the date of a Change in Control
or Change in Control Event or (B) if the Change in Control or Change in Control Event is the result of a tender or exchange offer
or a corporate transaction, the highest price per such Share paid in such tender or exchange offer or corporate transaction;
provided, however, that in the case of an Award exempt from Section 409A, the Change in Control Price shall be the Fair Market
Value of such Share on the date such Award is exercised or deemed exercised pursuant to Section 11. To the extent the
consideration paid in any such transaction described above consists in full or in part of securities or other noncash consideration,
the value of such securities or other noncash consideration shall be determined in the sole discretion of the Board.
(h)
“
Code
”
shall mean the Internal Revenue Code of 1986, as amended from time to time, and any successor thereto.
(i) “Committee” shall mean the Compensation Committee of the Board or such other persons or committee to whom it has delegated
any authority, as may be appropriate. A person may serve on the Compensation Committee only if he or she (i) is a “Non-
employee Director” for purposes of Rule 16b-3 under the Exchange Act, and (ii) satisfies the requirements of an “outside director”
for purposes of Section 162(m) of the Code.
(j)
“
Company
”
shall mean Pfizer Inc., a Delaware corporation.
(k) “Covered Employee” shall mean a “covered employee” within the meaning of Section 162(m)(3) of the Code, or any successor
provision thereto.
(l)
“
Director
”
shall mean a member of the Board.
(m)
“
Effective Date
”
shall mean the date the Plan was last approved by the stockholders of the Company.
(n)
“
Employee
”
shall mean any employee of the Company or any Affiliate. For any and all purposes under this Plan, the term
“Employee” shall not include a person hired as an independent contractor, leased employee, consultant or a person otherwise
designated by the Committee, the Company or an Affiliate at the time of hire as not eligible to participate in or receive benefits
under the Plan or not on the payroll, even if such ineligible person is subsequently determined to be a common law employee of
the Company or an Affiliate or otherwise an employee by any governmental or judicial authority. Unless otherwise determined by
the Committee in its sole discretion, for purposes of the Plan, an Employee shall be considered to have terminated employment or
services and to have ceased to be an Employee if his or her employer ceases to be an Affiliate, even if he or she continues to be
employed by such employer.
(o)
“
Exchange Act
”
shall mean the Securities Exchange Act of 1934, as amended.
(p) “Fair Market Value” shall mean, with respect to Shares, as of any date, the closing price for the Shares as reported on the New
York Stock Exchange for that date or, if no such price is reported for that date, the closing price on the next preceding date for
which such price was reported, unless otherwise determined by the Committee, in a manner consistent with Section 409A.

3
(q) “Grandfathered Benefits” shall mean any Awards that were earned and vested as of December 31, 2004, within the meaning of
Section 409A. Grandfathered Benefits are subject to the distribution rules under the Plan that were in effect on October 3, 2004
and are summarized on Appendix A.
(r)
“
Grant Date
”
shall mean the date on which an Award is granted.
(s) “Incentive Stock Option”
shall mean an Option granted under Section 6 that is intended to meet the requirements of Section 422 of
the Code or any successor provision thereto.
(t) “Key Employee” means an Employee treated as a “specified employee” as of his or her Separation from Service under Code
Section 409A(a)(2)(B)(i), i.e., a key employee (as defined in Code Section 416(i) without regard to paragraph (5) thereof) of the
Company or its Affiliates if the Company’s stock is publicly traded on an established securities market or otherwise. Key
Employees shall be determined under rules adopted by the Company in accordance with Section 409A. Key Employees shall also
include those key employees who are eligible for the Company’s Executive Long-Term Incentive Program as “specified
employees” for the 12 month period following the specified employee effective date, if not already included pursuant to the
foregoing. Key Employees shall be determined in accordance with Section 409A using a December 31 identification date and the
listing of Key Employees as of any such identification date shall be effective for the 12-month period beginning on the effective
date following the identification date. Notwithstanding the foregoing, the Committee may, under the alternative permissible
methods allowable under Section 409A, adopt an alternative identification and effective date for purposes of determining which
employees are Key Employees.
(u) “Nonqualified Stock Option” shall mean either an Option granted under Section 6 that is not intended to be an Incentive Stock
Option or an Incentive Stock Option that has been disqualified.
(v) “Option” shall mean any right granted to a Participant under the Plan allowing such Participant to purchase Shares at such price or
prices and during such period or periods as the Committee shall determine.
(w)
“
Other Stock Unit Award
”
shall mean any right granted to a Participant by the Committee pursuant to Section 10.
(x) “Participant” shall mean an Employee or a non-employee member of the Board who is selected by the Committee or the Board
from time to time in their sole discretion to receive an Award under the Plan.
(y)
“
Performance Award
”
shall mean any Award of Performance Shares granted pursuant to Section 9.
(z) “Performance Period” shall mean a period of not less than one year, as established by the Committee at the time any Performance
Award is granted or at any time thereafter, during which any performance goals specified by the Committee with respect to such
Award are to be measured.
(aa) “Performance Share” shall mean any grant pursuant to Section 9 of a unit valued by reference to a designated number of Shares,
which value may be paid to the Participant by delivery of such property as the Committee shall determine, including, without
limitation, cash, Shares, other property, or any combination thereof, upon achievement of such performance goals during the
Performance Period as the Committee shall establish at the time of such grant or earlier (but no later than ninety (90) days after the
commencement of the Performance Period).
(bb)
“
Person
”
shall mean any individual, corporation, partnership, association, limited liability company, joint
-
stock company, trust,
unincorporated organization or government or political subdivision thereof.
(cc) “Renewal Term” shall mean the two-year period beginning on the second anniversary of the Effective Date and each successive
two
-
year period thereafter.
(dd) “Restricted Stock” shall mean any Share issued with the restriction that the holder may not sell, transfer, pledge or assign such
Share and with such other restrictions as the Committee, in its sole discretion, may impose (including, without limitation, any
restriction on the right to vote such Share, and the right to receive any cash dividends), which restrictions may lapse separately or
in combination at such time or times, in installments or otherwise, as the Committee may deem appropriate.

SECTION 3. ADMINISTRATION
The Plan shall be administered by the Committee. The Committee shall have full power and authority, subject to such orders or resolutions
not inconsistent with the provisions of the Plan as may from time to time be adopted by the Board, to (a) select the Employees of the
Company and its Affiliates to whom Awards may from time to time be granted hereunder; (b) determine the type or types of Award to be
granted to each Participant hereunder; (c) determine the number of Shares to be covered by or relating to each Award granted hereunder;
(d) determine the terms and conditions, not inconsistent with the provisions of the Plan, of any Award granted hereunder; (e) determine
whether, to what extent and under what circumstances Awards may be settled in cash, Shares or other property or canceled or suspended,
consistent with the terms of the Plan; (f) determine whether, to what extent, and under what circumstances shares or cash paid to or gain
realized by the Participant based on an Award shall be returned to the Company, consistent with the terms of the Plan; (g) determine
whether, to what extent, and under what circumstances a Participant may be ineligible to retain an Award; (h) determine whether, to what
extent, and under what circumstances payment of cash, Shares, other property and other amounts payable with respect to an Award made
under the Plan shall be deferred either automatically or at the election of the Participant, consistent with the terms of the Plan; (i) interpret
and administer the Plan and any instrument or agreement entered into under the Plan; (j) establish such rules and regulations and appoint
such agents as it shall deem appropriate for the proper administration of the Plan; and (k) make any other determination and take any other
action that the Committee deems necessary or desirable for administration of the Plan. The Committee may, in its sole and absolute
discretion, and subject to the provisions of the Plan, from time to time delegate any or all of its authority to administer the Plan to any other
persons or committee as it deems necessary or appropriate for
4
(ee)
“
Restricted Stock Award
”
shall mean an award of Restricted Stock under Section 8.
(ff) “Section 409A” shall mean Section 409A of the Code and the regulations and other guidance issued thereunder by the U.S.
Treasury or Internal Revenue Service.
(gg) “Retirement” shall mean having attained a minimum age of 55 and a minimum of 10 years of service at the time of a Participant’s
separation from the Company, unless determined otherwise by the Committee, and which shall also constitute a Separation from
Service.
(hh)
“
Separation from Service
”
means a
“
separation from service
”
within the meaning of Section 409A.
(ii)
“
Shares
”
shall mean the shares of common stock of the Company.
(jj)
“
Stock Appreciation Right
”
shall mean any right granted to a Participant pursuant to Section 7 to receive, upon exercise by the
Participant, the excess of (i) the Fair Market Value of one Share on the date of exercise over (ii) the grant price of the right on the
date of grant, or if granted in connection with an outstanding Option on the date of grant of the related Option, as specified by the
Committee in its sole discretion, which, except in connection with an adjustment provided in Section 4(c), shall not be less than
the Fair Market Value of one Share on such date of grant of the right or the related Option, as the case may be. Any payment by
the Company in respect of such right may be made in cash, Shares, other property, or any combination thereof, as the Committee,
in its sole discretion, shall determine.
(kk) “Substitute Awards”
shall mean Awards granted or Shares issued by the Company in assumption of, or in substitution or exchange
for, awards previously granted, or the right or obligation to make future awards, by a company acquired by the Company or an
Affiliate or with which the Company or an Affiliate combines.
(ll) “Total and Permanent Disability” shall mean total and permanent disability as determined in accordance with rules established by
the Committee, and in compliance with Section 409A.
(mm)
“
Vesting Period
”
shall mean the period of time before unrestricted shares become non
-
forfeitable and issuable to a Participant
within the meaning of Section 10.

the proper administration of the Plan, except that no such delegation shall be made in the case of Awards intended to be qualified under
Section 162(m) of the Code. The decisions of the Committee shall be final, conclusive and binding with respect to the interpretation and
administration of the Plan and any grant made under it. The Committee shall make, in its sole discretion, all determinations arising in the
administration, construction or interpretation of the Plan and Awards under the Plan, including the right to construe disputed or doubtful
Plan or Award terms and provisions, and any such determination shall be conclusive and binding on all persons, except as otherwise
provided by law. A majority of the members of the Committee may determine its actions and fix the time and place of its meetings.
Except as provided in Section 12, the Committee shall be authorized to make adjustments in Performance Award criteria or in the terms and
conditions of other Awards in recognition of unusual or nonrecurring events affecting the Company or its financial statements or changes in
applicable laws, regulations or accounting principles. The Committee may correct any defect, supply any omission or reconcile any
inconsistency in the Plan or any Award in the manner and to the extent it shall deem desirable to carry it into effect. In the event that the
Company shall assume outstanding employee benefit awards or the right or obligation to make future such awards in connection with the
acquisition of or combination with another corporation or business entity, the Committee may, in its discretion, make such adjustments in
the terms of Awards under the Plan as it shall deem appropriate.
SECTION 4. SHARES SUBJECT TO THE PLAN
(a) Subject to adjustment as provided in Section 4(c), a total of four hundred twenty-five million (425,000,000) Shares shall be authorized
for grant pursuant to Awards under the Plan, plus any shares remaining available for grant under the Plan as of the Effective Date, provided
that no more than four hundred twenty-five million (425,000,000) Shares may be granted as Incentive Stock Options. Any Shares granted in
connection with Options and Stock Appreciation Rights shall be counted against this limit as one (1) Share for every one (1) Option or Stock
Appreciation Right awarded. Any Shares granted in connection with Awards other than Options and Stock Appreciation Rights shall be
counted against this limit as two (2) Shares for every one (1) Share granted in connection with such Award or by which the Award is valued
by reference. No Participant under this Plan shall be granted Options, Stock Appreciation Rights or other Awards (counted, as described
above, as two (2) Shares awarded for every one Share issued in connection with such Award or by which the Award is valued by reference)
in any consecutive 36-month period covering more than eight million (8,000,000) Shares. The grant limit under the preceding sentence shall
apply to an Award other than an Option or Stock Appreciation Right only if the Award is intended to be “performance-based”
as that term is
used in Section 162(m) of the Code. No Award will be granted to any Participant who owns more than ten percent of the stock of the
Company within the meaning of Section 422 of the Code.
(b) Any Shares issued hereunder may consist, in whole or in part, of authorized and unissued Shares, treasury Shares or Shares purchased in
the open market or otherwise.
(c) In the event of any merger, reorganization, consolidation, recapitalization, stock dividend, extraordinary cash dividend, stock split,
reverse stock split, spin-off or similar transaction or other change in corporate structure affecting the Shares, such adjustments and other
substitutions shall be made to the Plan and to Awards as the Committee, in its sole discretion, deems equitable or appropriate, including,
without limitation, such adjustments in the aggregate number, class and kind of securities that may be delivered under the Plan, in the
aggregate or to any one Participant, in the number, class, kind and option or exercise price of securities subject to outstanding Awards
granted under the Plan (including, if the Committee deems appropriate, the substitution of similar options to purchase the shares of, or other
awards denominated in the shares of, another company) as the Committee may determine to be appropriate in its sole discretion; provided,
however, that the number of Shares subject to any Award shall always be a whole number and further provided that in no event may any
change be made to an Incentive Stock Option which would constitute a modification within the meaning of Section 424(h)(3) of the Code.
Moreover, notwithstanding
5

anything herein to the contrary, an adjustment to an Award under this Section 4(c) may not be made in a manner that would result in the
grant of a new Option or Stock Appreciation Right under Section 409A, unless the Committee specifically determines that such adjustment
is desirable and will not cause the modified award to create adverse tax consequences under Section 409A.
(d) Any Shares subject to Awards that terminate, expire, or are forfeited, cancelled or settled in cash, either in whole or in part, may be used
for the further grant of Awards to the extent of such termination, forfeiture, cancellation or settlement. Any Shares that again become
available for future grants pursuant to the preceding sentence shall be added back as one (1) Share if such Shares were subject to Options or
Stock Appreciation Rights, and as two (2) Shares if such Shares were subject to Awards other than Options or Stock Appreciation Rights. In
addition, in the case of any Substitute Award, Shares delivered or deliverable in connection with such assumed or Substitute Award shall not
reduce the number of Shares authorized for grant in Section 4(a) above. Notwithstanding the foregoing, Shares subject to an Award under
the Plan may not again be made available for issuance or delivery under the Plan if such Shares are (i) Shares that were subject to a stock-
settled Stock Appreciation Right and were not issued upon the net settlement or net exercise of such Stock Appreciation Right, (ii) Shares
delivered or withheld by the Company to pay the exercise price of an Option, (iii) Shares delivered to or withheld by the Company to pay
the withholding taxes related to an Award, or (iv) Shares repurchased on the open market with the proceeds of an Option exercise.
SECTION 5. ELIGIBILITY
Any Employee or non-employee Director shall be eligible to be selected as a Participant; provided, however, that Incentive Stock Options
shall only be awarded to Employees of the Company, or a parent or Affiliate, within the meaning of Section 422 of the Code.
Notwithstanding any provision in this Plan to the contrary, the non
-employee Directors, including a designated committee of the Board
composed solely of non-employee Directors, shall have the authority, in their sole and absolute discretion, to select non-employee Directors
as Participants who are eligible to receive Awards other than Incentive Stock Options under the Plan. The non-employee Directors shall set
the terms of any such Awards in their sole and absolute discretion, and the non-employee Directors shall be responsible for administering
and construing such Awards in substantially the same manner that the Committee administers and construes Awards to Employees.
SECTION 6. STOCK OPTIONS
Options may be granted hereunder to any Participant, either alone or in addition to other Awards granted under the Plan and shall be subject
to the following terms and conditions:
(a) Option Price. The option price per Share shall be not less than the Fair Market Value of the Shares on the date the Option is granted.
(b) Number of Shares. The Option shall state the number of Shares covered thereby.
(c) Exercise of Option. Unless otherwise determined by the Committee, an Option will be deemed exercised by the optionee, or in the event
of death, an option shall be deemed exercised by the estate of the optionee, or by a person who acquired the right to exercise such option by
bequest or inheritance or by reason of the death of the optionee, upon delivery of (i) a notice of exercise to the Company or its
representative, or by using other methods of notice as the Committee shall adopt, and (ii) accompanying payment of the option price or other
methods of satisfying the option exercise prices as approved by the Committee and in accordance with any restrictions as the Committee
shall adopt. The notice of exercise, once delivered, shall be irrevocable. Notwithstanding the above, and unless the Committee determines
otherwise, in the event that (i) an optionee dies, (ii) his representative has a right to exercise an Option, (iii) the Option is not exercised
6

by the last day on which it is exercisable, and (iv) the option price per share is below the Fair Market Value of a Share on such date, the
Option shall be deemed exercised on such date via a cashless exercise procedure and the resulting proceeds net of the option price and any
required tax withholding shall be paid to the representative.
(d) Term of Option. The Committee shall determine the option exercise period of each Option. The period for Incentive Stock Options shall
not exceed ten years from the grant date. A Nonqualified Stock Option may be exercisable for a period of up to ten years and six months so
as to conform with or take advantage of governmental requirements, statutes or regulations.
(e) First Exercisable Date. Except in the case of death, Total and Permanent Disability, Change in Control or the sale or restructuring of a
business or plant closing, no Option may be exercised during the first year of its term or such longer period as may be specified in the
Option.
(f) Termination of Option. All Options shall terminate upon their expiration, their surrender, upon breach by the optionee of any provisions
of the Option, or in accordance with any other rules and procedures incorporated into the terms and conditions governing the Options as the
Committee shall deem advisable or appropriate.
(g) Incorporation by Reference. The Option shall contain a provision that all the applicable terms and conditions of this Plan are
incorporated by reference therein.
(h) Other Provisions. The Option shall also be subject to such other terms and conditions as the Committee shall deem advisable or
appropriate, consistent with the provisions of the Plan as herein set forth. In addition, Incentive Stock Options shall contain such other
provisions as may be necessary to meet the requirements of the Code and the Treasury Department rulings and regulations issued thereunder
with respect to Incentive Stock Options.
(i) Exemption from Section 409A. It is intended that all Options granted under this Plan will be exempt from Section 409A. Nevertheless,
the Company does not represent, covenant or guarantee that any particular Award made under the Plan will qualify for favorable tax
treatment (e.g., as in incentive stock options) or will avoid unfavorable tax consequences to the Participant (e.g., Section 409A penalties).
SECTION 7. STOCK APPRECIATION RIGHTS
(a) Grant of a Stock Appreciation Right. Stock Appreciation Rights may be granted hereunder to any Participant, either alone
(“freestanding”) or in addition to other Awards granted under the Plan and may, but need not, relate to a specific Option granted under
Section 6. The provisions of Stock Appreciation Rights need not be the same with respect to each recipient. Any Stock Appreciation Right
related to a Nonqualified Stock Option may be granted at the same time such Option is granted or at any time thereafter before exercise or
expiration of such Option. Any Stock Appreciation Right related to an Incentive Stock Option must be granted at the same time such Option
is granted. In the case of any Stock Appreciation Right related to any Option, the Stock Appreciation Right or applicable portion thereof
shall terminate and no longer be exercisable upon the termination or exercise of the related Option, except that a Stock Appreciation Right
granted with respect to less than the full number of Shares covered by a related Option shall not be reduced until the exercise or termination
of the related Option exceeds the number of Shares not covered by the Stock Appreciation Right. Any Option related to any Stock
Appreciation Right shall no longer be exercisable to the extent the related Stock Appreciation Right has been exercised.
(b) Terms. The Committee may impose such terms and conditions or restrictions on the exercise of any Stock Appreciation Right, as it shall
deem advisable or appropriate; provided that a Stock Appreciation Right shall not have an exercise price less than Fair Market Value of a
Share on the date of grant or a term of greater than ten years.
(c) Section 409A. Stock Appreciation Rights may be granted hereunder by the Committee either (i) in a manner consistent with
Section 409A such that the Stock Appreciation Right will not provide for a deferral of compensation under Section 409A, or (ii) in a manner
that is intended from grant to subject the Stock Appreciation Right to Section 409A. In the event Stock Appreciation Rights are granted to
be so subject to
7

Section 409A, then the Stock Appreciation Right shall be settled and paid in a single lump sum (i) as of a specified date, (ii) upon the
Participant’s Separation from Service, or (iii) the earlier of (i) or (ii) hereof, as specified and set forth by the Committee in an Award
Agreement at the time of grant, and shall otherwise be granted, administered, settled and paid in accordance with Section 409A.
Notwithstanding the foregoing, any such settlement and payment may not be made to a Key Employee upon a Separation from Service
before the date which is 6 months after the date of the Key Employee’s Separation from Service (or, if earlier, the date of death of the Key
Employee).
SECTION 8. RESTRICTED STOCK
(a) Grant of Restricted Stock. A Restricted Stock Award shall be subject to restrictions imposed by the Committee at the time of grant for a
period of time specified by the Committee (the “Restriction Period”). Restricted Stock Awards may be issued hereunder to Participants for
no cash consideration or for such minimum consideration as may be required by applicable law, either alone or in addition to other Awards
granted under the Plan. Any Award of Restricted Stock shall also be subject to such other terms and conditions as the Committee shall deem
advisable or appropriate, consistent with the provisions of the Plan as herein set forth. Except in the event of a termination of employment
due to death, Retirement, Total and Permanent Disability, Change in Control or the sale or restructuring of a business or plant closing,
Restricted Stock Awards shall have a Restriction Period of not less than three (3) years from the date of grant, which may include pro-rata
lapsing of restrictions thereon. In the event of a termination of employment due to death, Total and Permanent Disability or a Change in
Control, awards of Restricted Stock immediately and fully vest. In the event of a termination of employment due to Retirement or a sale or
restructuring of a business or plant closing, awards of Restricted Stock vest pro-
rata. Notwithstanding the above, Awards covering up to five
(5) percent of the total number of Shares that may be issued or delivered under the Plan (other than as Awards of Options or Stock
Appreciation Rights) may contain no restrictions or be subject to a Restriction Period of less than three (3) years.
(b) Registration. Any Restricted Stock issued hereunder may be evidenced in such manner, as the Committee, in its sole discretion, shall
deem appropriate, including, without limitation, book entry registration or issuance of a stock certificate or certificates. In the event any
stock certificates are issued in respect of Shares of Restricted Stock awarded under the Plan, such certificates shall be registered in the name
of the Participant and shall bear an appropriate legend referring to the terms, conditions and restrictions applicable to such Award.
(c) Section 409A. Restricted Stock Awards may be granted hereunder by the Committee either (i) in a manner consistent with Section 409A
such that the Restricted Stock Award not provide for a deferral of compensation under Section 409A, or (ii) in a manner that is intended
from grant to subject the Restricted Stock Award to Section 409A. In the event Restricted Stock Awards are subject to Section 409A, then
the Restricted Stock Award shall be settled and paid in a single lump sum (i) as of a specified date, (ii) upon the Participant’s Separation
from Service, or (iii) the earlier of (i) or (ii) hereof, as specified and set forth by the Committee in an Award Agreement at the time of grant,
and shall otherwise be granted, administered, settled and paid in accordance with Section 409A. Notwithstanding the foregoing, any such
settlement and payment may not be made to a Key Employee upon a Separation from Service before the date which is 6 months after the
date of the Key Employee’s Separation from Service (or, if earlier, the date of death of the Key Employee).
SECTION 9. PERFORMANCE AWARDS
Performance Awards may be paid in cash, Shares, other property, or any combination thereof, and may be subject to such other terms and
conditions as the Committee shall deem advisable or appropriate, consistent
8

with the provisions of the Plan as set forth, in the sole discretion of the Committee at the time of payment. The performance levels to be
achieved for each Performance Period and the amount of the Award to be distributed shall be conclusively determined by the Committee.
Performance Awards will be paid in a lump sum prior to the 15 day of the third month of the year immediately following the year in which
the close of the Performance Period occurs in accordance with the applicable short-term deferral exception provisions of Section 409A, or,
in accordance with procedures established by the Committee and the applicable provisions of Section 409A, on a deferred basis pursuant to
Section 15 hereof, if applicable. All Performance Awards must satisfy the definition of “performance-based compensation” of Treasury
Regulation Section 1.409A-1(e), but the Committee may designate whether any Performance Award, either alone or in addition to other
Awards granted under the Plan, being granted to any Employee is intended to be “performance-based compensation” as that term is used in
Section 162(m) of the Code. Any such awards designated to be “performance-based compensation” within the meaning of Code Section 162
(m) shall be conditioned on the achievement of one or more performance measures, to the extent required by Code Section 162(m), and shall
be issued in accordance with Section 12. Except in the event of a Change in Control or Change in Control Event described in Section 11, in
the event Performance Awards are subject to Section 409A, then the Performance Award shall be settled and paid in a single lump sum (i) as
of a specified date, (ii) upon the Participant’s Separation from Service, or (iii) the earlier of (i) or (ii) hereof, in accordance with rules
established by the Committee at the time of grant, and shall otherwise be granted, administered, settled and paid in accordance with
Section 409A. Notwithstanding the foregoing, any such settlement and payment may not be made to a Key Employee upon a Separation
from Service before the date which is 6 months after the date of the Key Employee’
s Separation from Service (or, if earlier, the date of death
of the Key Employee).
SECTION 10. OTHER STOCK UNIT AWARDS
(a) Stock and Administration. Awards that are valued by reference to, or are otherwise based on, Shares (“Other Stock Unit Awards”) may
be granted hereunder to Participants, either alone or in addition to other Awards granted under the Plan, and such Other Stock Unit Awards
shall also be available as a form of payment in the settlement of other Awards granted under the Plan. Other Stock Unit Awards may be paid
in Shares, cash or any other form of property, as the Committee shall determine. Subject to the provisions of the Plan, the Committee shall
have sole and complete authority to determine the Employees to whom and the time or times at which such Awards shall be made, the
number of Shares to be issued or delivered pursuant to such Awards, and all other conditions of the Awards. Any Other Stock Unit Awards
shall be subject to such other terms and conditions as the Committee shall deem advisable or appropriate, consistent with the provisions of
the Plan as herein set forth. Except in the event of a termination of employment due to death, Retirement, Total and Permanent Disability,
Change in Control or the sale or restructuring of a business or plant closing, Other Stock Unit Awards shall have a Vesting Period of not less
than three (3) years, which may include pro-rata lapsing of restrictions thereon. In the event of a termination of employment due to death,
Total and Permanent Disability or a Change in Control, Other Stock Awards immediately and fully vest. In the event of a termination of
employment due to Retirement or a sale or restructuring of a business or plant closing, Other Stock Awards vest pro-rata. Notwithstanding
the above, Awards covering up to five (5) percent of the total number of Shares that may be issued or delivered under the Plan (other than as
Awards of Options or Stock Appreciation Rights) may contain no restrictions or be subject to a Vesting Period of less than three (3) years.
(b) Other Provisions. Shares (including securities convertible into Shares) subject to Awards granted under this Section 10 may be issued
for no cash consideration or for such minimum consideration as may be required by applicable law.
(c) Section 409A. Other Stock Unit Awards may be granted hereunder by the Committee (i) in a manner consistent with Section 409A such
that the Other Stock Unit Awards will not provide for a deferral of compensation under Section 409A, or (ii) in a manner that will subject
the Other Stock Unit Award to
9
th

Section 409A. In the event Other Stock Unit Awards are granted to be so subject to Section 409A, then the Other Stock Unit Awards shall
be settled and paid in a single lump sum (i) as of a specified date, (ii) upon the Participant’s Separation from Service, or (iii) the earlier of
(i) or (ii) hereof, as specified by the Committee at the time of grant or otherwise in a fashion which is compliant with Section 409A, and
shall otherwise be granted, administered, settled and paid in compliance with Section 409A. Notwithstanding the foregoing, any such
settlement and payment may not be made to a Key Employee upon a Separation from Service before the date which is 6 months after the
date of the Key Employee’s Separation from Service (or, if earlier, the date of death of the Key Employee).
(d) Section 162(m) Deferrals. Except for Other Stock Unit Awards which are subject to the satisfaction of performance goals, any
outstanding Other Stock Unit Awards that are scheduled to be settled or otherwise paid to a Participant during a taxable year in which such
Participant is, or is likely to be, a Covered Employee, shall automatically be deferred into the Pfizer Inc Deferred Compensation Plan, as
Amended and Restated, effective January 1, 2008, in accordance with the terms of such plan and in compliance with the applicable
provisions of Section 409A until the earlier of (i) the first day of the Participant’s first taxable year in which the Company reasonably
anticipates that if the payment is made during such year, the deduction of such payment by the Company will not be barred by the
application of Code Section 162(m), or (ii) the Participant’
s Separation from Service. Notwithstanding the foregoing, any such payment may
not be made to a Key Employee upon a Separation from Service before the date which is 6 months after the date of the Key Employee’s
Separation from Service (or, if earlier, the date of death of the Key Employee).
SECTION 11. CHANGE IN CONTROL PROVISIONS
(a) Unless the Committee or Board shall determine otherwise at the time of grant with respect to a particular Award, and notwithstanding
any other provision of the Plan to the contrary, in the event a Participant’s employment or service is involuntarily terminated without Cause
(as determined by the Committee or Board in its sole discretion) during the 24-month period following a Change in Control, and provided
that, with respect to any Awards that are considered deferred compensation under Section 409A, the Participant’
s involuntary termination of
employment or service also constitutes a Separation from Service:
(i) notwithstanding a provision in any Award Agreement to the contrary, any Options and Stock Appreciation Rights outstanding and
which are not then exercisable and vested shall upon such involuntary termination fully vest and become exercisable for their full term, and
shall remain in effect for the respective terms of such award as set forth in the grant documents at the time of grant notwithstanding such
involuntary termination.
(ii) any vested Options and Stock Appreciation Rights outstanding shall remain in effect and be
exercisable for the respective terms of such
award as set forth in the grant documents at the time of grant notwithstanding such involuntary termination;
(iii) the restrictions and deferral limitations applicable to any Restricted Stock shall lapse, and such Restricted Stock shall immediately
become free of all restrictions and limitations and become fully vested and transferable to the full extent of the original grant;
(iv) all Performance Awards shall be considered to be earned and payable in full, based on the applicable performance criteria or, if not
determinable, at the target level and any deferral or other restriction shall lapse and such Performance Awards shall be immediately settled
and paid upon the Participant’s Separation from Service (and the Participant shall have no discretion to choose the date of settlement and
payment) provided, however, that any such settlement and payment may not be made to a Key Employee upon a Separation from Service
before the date which is 6 months after the date of the Key Employee’s Separation from Service (or, if earlier, the date of death of the Key
Employee); and
(v) the restrictions and deferral limitations and other conditions applicable to any Other Stock Unit Awards or any other Awards shall
immediately lapse, and any such Other Stock Unit Awards or such other Awards shall become free of all restrictions, limitations or
conditions and become fully vested and transferable to the full extent of the original grant;
10

(vi) notwithstanding any other provision of this Section 11(a), the proceeds, from exercise or otherwise, of any Options, Stock
Appreciation Rights, Restricted Stock, Performance Shares or Other Stock Unit Awards that are considered deferred compensation under
Section 409A shall be paid (and if not exercised prior to the date of the Participant’
s Separation from Service, shall be deemed exercised and
settled and paid) upon the Participant’s Separation from Service (and the Participant shall have no discretion to choose the date of payment)
provided, however, that any such payment may not be made to a Key Employee upon a Separation from Service before the date which is 6
months after the date of the Key Employee’s Separation from Service (or, if earlier, the date of death of the Key Employee).
(b) Change in Control Cash Out. Notwithstanding any other provision of the Plan, in the event of a Change in Control, or, with respect to
Options, Stock Appreciation Rights, Restricted Stock, Performance Shares or Other Stock Unit Awards that are considered deferred
compensation under Section 409A, in the event of a Change in Control that is also a “Change in Control Event” described in Section 409A
(a)(2)(A)(v) or otherwise under Section 409A, (i) the Committee or Board may, in its discretion, provide in the terms of the Option, Stock
Appreciation Right, Restricted Stock, Performance Share or Other Stock Unit Award that is intended to be exempt from Section 409A, that
such Option, Stock Appreciation Right, Restricted Stock, Performance Share or Other Stock Unit Award shall, upon the occurrence of a
Change in Control, be cancelled in exchange for a cash payment to be made within 60 days of the Change in Control (and the Participant
shall have no discretion to choose the date of payment) in an amount equal to the amount by which the Fair Market Value per Share on the
date of the payment exceeds the purchase price per Share under the Option or Stock Appreciation Right multiplied by the number of Shares
issued and delivered under the Option or Stock Appreciation Right, or in an amount equal to the Fair Market Value per Share on the date of
the payment for the Restricted Stock, Performance Share, or Other Stock Unit Award, or (ii) the Committee or Board may, in its discretion,
provide in the terms of an Option, Stock Appreciation Right, Restricted Stock, Performance Share, or Other Stock Unit Award that is
deferred compensation under Section 409A, that such Option, Stock Appreciation Right, Restricted Stock, Performance Share or Other
Stock Unit shall, upon the occurrence of a Change in Control Event, be cancelled in exchange for a cash payment to be made within 60 days
of the Change in Control Event (and the Participant shall have no discretion to choose the date of payment) in an amount equal to the
amount by which the Change in Control Price per Share exceeds the purchase price per Share under the Option or Stock Appreciation Right,
multiplied by the number of Shares issued and delivered under the Option or Stock Appreciation Right, or in an amount equal to the Change
in Control Price per Share for the Restricted Stock, Performance Share or Other Stock Unit Award.
(c) Notwithstanding the above, if the Change in Control is the result of a transaction pursuant to Section 2(e)(iii) and the surviving entity
does not assume, substitute or replace Awards, such Awards shall become fully vested and immediately exercisable or transferable to the
full extent of the original grant upon the Change in Control and shall be distributed, settled or paid in full within 90 days of the Change in
Control as provided in Section 11(b) above.
SECTION 12. CODE SECTION 162(m) PROVISIONS
(a) Notwithstanding any other provision of the Plan if the Committee determines at the time, a Performance Award is granted to a
Participant who is then an officer that such Participant is, or is likely to be as of the end of the tax year in which the Company would
ordinarily claim a tax deduction in connection with such Award, a Covered Employee, then the Committee may provide that this Section 12
is applicable to such Award.
11

(b) If a Performance Award is subject to this Section 12, then the lapsing of restrictions thereon and the distribution of cash, Shares or other
property pursuant thereto, as applicable, shall be subject to the achievement of one or more objective performance goals established by the
Committee, which shall be based on the attainment of specified levels of one or any combination of the following: revenues, cost reductions,
operating income, income before taxes, net income, adjusted net income, earnings per share, adjusted earnings per share, operating margins,
working capital measures, return on assets, return on equity, return on invested capital, cash flow measures, market share, shareholder return
or economic value added of the Company or the Affiliate or division of the Company for or within which the Participant is primarily
employed. Such performance goals also may be based on the achievement of specified levels of Company performance (or performance of
an applicable Affiliate or division of the Company) under one or more of the measures described above relative to the performance of other
corporations. Such performance goals shall be set by the Committee within the time period prescribed by, and shall otherwise comply with
the requirements of, Section 162(m) of the Code, or any successor provision thereto, and the regulations thereunder.
(c) In setting performance goals, the Committee may provide in any such Award Agreement that resulting from the following items shall be
included or excluded: (i) asset write-downs; (ii) litigation or claim judgments or settlements; (iii) the effect of changes in tax laws,
accounting principles or other laws or provisions affecting reported results, (iv) charges for any reorganization and restructuring programs;
(v) extraordinary nonrecurring charges or losses as described in Accounting Principles Board Opinion No. 30 and/or in Management’s
Discussion and Analysis of Financial Condition and Results of Operations appearing in the Company’s annual report to stockholders for the
applicable year, (vi) the impact of acquisitions or divestitures; (vii) foreign exchange gains and losses and (viii) gains or losses on asset
sales. To the extent such inclusions or exclusions affect Awards to a Covered Employee, they shall be prescribed in a form that satisfies the
requirements for “performance-based compensation” within the meaning of Section 162(m) of the Code.
(d) Notwithstanding any provision of the Plan other than Section 11, with respect to any Performance Award that is subject to this
Section 12, the Committee may adjust downwards, but not upwards, the amount payable pursuant to such Award, and the Committee may
not waive the achievement of the applicable performance goals except in the case of the death or Total and Permanent Disability of the
Participant, or under such other conditions where such waiver will not jeopardize the treatment of other Awards under this Section as
“performance-based compensation” under Section 162(m) of the Code.
(e) The Committee shall have the power to impose such other restrictions on Awards subject to this Section 12 as it may deem necessary or
appropriate to ensure that such Awards satisfy all requirements for “performance-based compensation” within the meaning of Section 162
(m)(4)(C) of the Code, or any successor provision thereto.
12

SECTION 13. AMENDMENTS AND TERMINATION
The Board may amend, alter, suspend, discontinue or terminate the Plan or any portion thereof at any time; provided, however, that no such
amendment, alteration, suspension, discontinuation or termination shall be made without (a) stockholder approval if such approval is
necessary to qualify for or comply with any tax or regulatory requirement for which or with which the Board deems it necessary or desirable
to qualify or comply, (b) the consent of the affected Participant, if such action would materially impair the rights of such Participant under
any outstanding Award or (c) approval of the holders of a majority of the outstanding Common Stock present or represented by proxy and
entitled to vote at a meeting of the Company’s stockholders with respect to any alteration or amendment to the Plan which increases the
maximum number of shares of Common Stock which may be issued under the Plan or the number of shares of such stock which may be
issued to any one Participant, extends the term of the Plan or of options granted thereunder, changes the eligibility criteria in Section 5, or
reduces the option price below that now provided for in the Plan. In addition, notwithstanding the above, any termination of the Plan shall
comply with all requirements under Section 409A that are necessary to be met to avoid adverse tax consequences to Participants under
Section 409A.
The Committee may delegate to another committee, as it may appoint, the authority to take any action consistent with the terms of the Plan,
either before or after an Award has been granted, which such other committee deems necessary or advisable to comply with any government
laws or regulatory requirements of a foreign country, including but not limited to, modifying or amending the terms and conditions
governing any Awards, or establishing any local country plans as sub-plans to this Plan. In addition, under all circumstances, the Committee
may make non-substantive administrative changes to the Plan as to conform with or take advantage of governmental requirements, statutes
or regulations.
The Committee may amend the terms of any Award theretofore granted, prospectively or retroactively, but no such amendment shall
(a) materially impair the rights of any Participant without his or her consent, (b) except for adjustments made pursuant to Section 4(c) or in
connection with Substitute Awards, reduce the exercise price of outstanding Options or Stock Appreciation Rights or cancel or amend
outstanding Options or Stock Appreciation Rights for the purpose of repricing, replacing or regranting such Options or Stock Appreciation
Rights with an exercise price that is less than the exercise price of the original Options or Stock Appreciation Rights or cancel or amend
outstanding Options or Stock Appreciation Rights with an exercise price that is greater than the Fair Market Value of a Share for the purpose
of exchanging such Options or Stock Appreciation Rights for cash or any other Awards without stockholder approval or (c) cause any
Award intended to be exempt from Section 409A to become subject to Section 409A. Notwithstanding the foregoing, the Committee may
amend the terms of any award heretofore granted, prospectively or retroactively, in order to cure any potential defects under Section 409A,
in a manner deemed appropriate by the Committee in its sole discretion, without the consent of the Participant. Any change or adjustment to
an outstanding Incentive Stock Option shall not, without the consent of the Participant, be made in a manner so as to constitute a
“modification” that would cause such Incentive Stock Option to fail to continue to qualify as an Incentive Stock Option. Notwithstanding
the foregoing, any adjustments made pursuant to Section 4(c) shall not be subject to these restrictions.
Notwithstanding the foregoing, no amendment of the Plan shall apply to amounts that were earned and vested (within the meaning of
Section 409A) under the Plan prior to 2005, unless the amendment specifically provides that it applies to such amounts. The purpose of this
restriction is to prevent a Plan amendment from resulting in an inadvertent “material modification” to amounts that are Grandfathered
Benefits.
13

SECTION 14. DIVIDENDS
Subject to the provisions of the Plan and any Award Agreement, the recipient of an Award (including, without limitation, any deferred
Award) may, if so determined by the Committee, be entitled to receive, currently or on a deferred basis, cash or stock dividends, or cash
payments in amounts equivalent to cash or stock dividends on Shares (“dividend equivalents”) with respect to the number of Shares covered
by the Award, as determined by the Committee, in its sole discretion, and the Committee may provide that such amounts (if any) shall be
deemed to have been reinvested in additional Shares or otherwise reinvested. Provided however, that if the receipt of any such dividend
equivalents granted with respect to Options, Restricted Stock, Other Stock Unit Awards and Stock Appreciation Rights is contingent upon
the exercise of the Options or Stock Appreciation Rights, or the vesting of the Restricted Stock or Other Stock Unit Awards, then the
Options, Restricted Stock, Other Stock Unit Awards, or Stock Appreciation Rights shall be granted and administered in accordance with all
applicable provisions of Section 409A.
SECTION 15. DEFERRAL OF AWARDS UNDER THE COMPANY’S DEFERRED COMPENSATION PLAN
Except as otherwise provided in this Plan, the Committee may provide upon the granting of an Award hereunder, other than an Award that is
intended to be a stock right which does not constitute a deferral of compensation within the meaning of Treasury Regulations
Section 1.409A-1(a)(5) so that it is subject to the requirement that it not include any feature for the deferral of compensation until an event
enumerated in such provision, that it is eligible to be deferred under, and pursuant to the terms and conditions of, the Pfizer Inc Deferred
Compensation Plan, as Amended and Restated, effective January 1, 2008. Any such deferral shall be in accordance with the terms of such
plan and in compliance with the applicable provisions of Section 409A.
SECTION 16. GENERAL PROVISIONS
(a) An Award may not be sold, pledged, assigned, hypothecated, transferred, or disposed of in any manner other than by will or by the laws
of descent or distribution and may be exercised, during the lifetime of the Awardee, only by the Awardee; provided that the Committee, in
its sole discretion, may permit additional transferability, on a general or specific basis, other than to a third party for consideration, and may
impose conditions and limitations on any permitted transferability.
(b) No Employee shall have the right to be selected to receive an Option or other Award under this Plan or, having been so selected, to be
selected to receive a future Award grant or Option. Neither the Award nor any benefits arising out of this Plan shall constitute part of a
Participant’s employment or service contract with the Company or any Affiliate and, accordingly, this Plan and the benefits hereunder may
be terminated at any time in the sole and exclusive discretion of the Company without giving rise to liability on the part of the Company or
any Affiliate for severance payments. The Awards under this Plan are not intended to be treated as compensation for any purpose under any
other Company plan.
(c) No Employee shall have any claim to be granted any Award under the Plan, and there is no obligation for uniformity of treatment of
Employees or Participants under the Plan.
(d) The prospective recipient of any Award under the Plan shall not, with respect to such Award, be deemed to have become a Participant, or
to have any rights with respect to such Award until and unless such recipient shall have accepted any Award Agreement or other instrument
evidencing the Award.
(e) Nothing in the Plan or any Award granted under the Plan shall be deemed to constitute an employment or service contract or confer or be
deemed to confer on any Employee or Participant any right to continue in the employ or service of, or to continue any other relationship
with, the Company or any Affiliate or limit in any way the right of the Company or any Affiliate to terminate an Employee’
s employment or
Participant’s service at any time, with or without Cause.
14

(f) All Shares delivered under the Plan pursuant to any Award shall be subject to such stock transfer orders and other restrictions as the
Committee may deem advisable under the rules, regulations and other requirements of the Securities and Exchange Commission, any stock
exchange upon which the Shares are then listed, and any applicable federal or state securities law, and the Committee may cause a legend or
legends to be put on any such certificates to make appropriate reference to such restrictions.
(g) In appropriate circumstances, the Committee in its sole discretion may determine that an Award shall be cancelled, or the shares or cash
paid or gain realized from an Award shall be returned to the Company.
(h) No Award granted hereunder shall be construed as an offer to sell securities of the Company, and no such offer shall be outstanding,
unless and until the Committee in its sole discretion has determined that any such offer, if made, would comply with all applicable
requirements of the U.S. federal securities laws and any other laws to which such offer, if made, would be subject.
(i) Except as otherwise required in any applicable Award Agreement or by the terms of the Plan, recipients of Awards under the Plan shall
not be required to make any payment or provide consideration other than the rendering of services.
(j) The Company and its Affiliates shall be authorized to withhold from any Award granted or payment due under the Plan, and/or to
withhold from wages or other cash compensation paid to the Participant, the minimum statutory amount of withholding taxes due in respect
of an Award or payment hereunder and to take such other action as may be necessary in the opinion of the Company or Affiliate to satisfy
all obligations for the payment of such taxes. Such other actions may include, without limitation, the requirement that the Participant execute
a market sale of Shares or other consideration received pursuant to the Award. The Committee shall be authorized to establish procedures
for elections by Participants to satisfy such obligation for the payment of such taxes by delivery of or transfer of Shares to the Company (in
a manner limited so as to avoid adverse accounting treatment for the Company), or by directing the Company to retain Shares (up to the
employee’s minimum statutory required tax withholding rate) otherwise deliverable in connection with the Award.
(k) Nothing contained in the Plan shall prevent the Board from adopting other or additional compensation arrangements, subject to
stockholder approval if such approval is required; and such arrangements may be either generally applicable or applicable only in specific
cases.
(l) Any Award shall contain a provision that it may not be exercised at a time when the exercise thereof or the issuance of shares thereunder
would constitute a violation of any federal or state law or listing requirements of the New York Stock Exchange for such shares or a
violation of any foreign jurisdiction where Awards are or will be granted under the Plan. The provisions of the Plan shall be construed,
regulated and administered according to the laws of the State of New York without giving effect to principles of conflicts of law, except to
the extent superseded by any controlling Federal statute. Notwithstanding anything herein to the contrary, the terms of the Plan are intended
to, and shall be interpreted and applied so as to, comply in all respects with Section 409A. The Committee may amend the terms of any
award heretofore granted, prospectively or retroactively, in order to cure any potential defects under Section 409A, in a manner deemed
appropriate by the Committee in its sole discretion, without the consent of the Participant. Nothing in this Section 16(l) shall be construed as
an admission that any of the compensation and/or benefits payable under this Plan constitutes “deferred compensation” subject to
Section 409A.
(m) If any provision of the Plan is or becomes or is deemed invalid, illegal or unenforceable in any jurisdiction, or would disqualify the Plan
or any Award under any law deemed applicable by the Committee, such provision shall be construed or deemed amended to conform to
applicable laws or if it cannot be construed or deemed amended without, in the determination of the Committee, materially altering the
intent of the Plan, it shall be stricken and the remainder of the Plan shall remain in full force and effect.
(n) Awards may be granted to Participants who are foreign nationals or employed outside the United States, or both, on such terms and
conditions different from those applicable to Awards to Employees employed in the United States as may, in the judgment of the
Committee, be necessary or desirable in order to recognize
15

differences in local law or tax policy. The Committee also may impose conditions on the exercise or vesting of Awards in order to minimize
the Company’s obligation with respect to tax equalization for Employees on assignments outside their home country.
(o) If approved by the Committee in its sole discretion, an Employee’s absence or leave because of military or governmental service, Total
and Permanent Disability or other reason shall not be considered an interruption of employment for any purpose under the Plan; provided,
however, that to the extent an Award under this Plan is subject to Section 409A, such absence or leave shall be considered a Separation from
Service to the extent provided by Section 409A.
SECTION 17. TERM OF PLAN
The Plan shall terminate on the tenth anniversary of the Effective Date, unless sooner terminated by the Board pursuant to Section 13.
SECTION 18. COMPLIANCE WITH SECTION 16
With respect to Participants subject to Section 16 of the Exchange Act (“Members”), transactions under the Plan are intended to comply
with all applicable conditions of Rule 16b-
3 or its successors under the Exchange Act. To the extent that compliance with any Plan provision
applicable solely to such Members that is included solely for purposes of complying with Rule 16b-3 is not required in order to bring a
transaction by such Member in compliance with Rule 16b-3, it shall be deemed null and void as to such transaction, to the extent permitted
by law and deemed advisable by the Committee. To the extent any provision in the Plan or action by the Committee involving such
Members is deemed not to comply with an applicable condition of Rule 16b-3, it shall be deemed null and void as to such Members, to the
extent permitted by law and deemed advisable by the Committee.
16

APPENDIX A
GRANDFATHERED BENEFITS
Distribution, settlement or payment of amounts that were earned and vested (within the meaning of Section 409A) under
the Plan prior to 2005 (and earnings thereon) and are exempt from the requirements of Section 409A shall be made in accordance
with certain Plan terms as in effect on December 31, 2004, and as set forth in this Appendix A. Unless otherwise specified in this
Appendix A, Grandfathered Benefits shall be governed by the terms of the Plan.
SECTION 2. DEFINITIONS
As used in this Appendix A, all terms have the same meaning as defined in Section 2 of the Plan, except as set forth below:
(a) “Fair Market Value” shall mean, with respect to Shares, as of any date, the average of the high and low trading prices for the Shares as
reported on the New York Stock Exchange for that date or, if no such prices are reported for that date, the average of the high and low
trading prices on the next preceding date for which such prices were reported, unless otherwise determined by the Committee.
SECTION 6. STOCK OPTIONS
Options may be granted hereunder to any Participant, either alone or in addition to other Awards granted under the Plan and shall be subject
to the following terms and conditions:
(a) Option Price. The option price per Share shall be not less than the Fair Market Value of the Shares on the date the Option is granted.
(b) Number of Shares. The Option shall state the number of Shares covered thereby.
(c) Exercise of Option. Unless otherwise determined by the Committee, an Option will be deemed exercised by the optionee, or in the event
of death, an option shall be deemed exercised by the estate of the optionee, or by a person who acquired the right to exercise such option by
bequest or inheritance or by reason of the death of the optionee, upon delivery of (i) a notice of exercise to the Company or its
representative, or by using other methods of notice as the Committee shall adopt, and (ii) accompanying payment of the option price in
accordance with any restrictions as the Committee shall adopt. The notice of exercise, once delivered, shall be irrevocable. Notwithstanding
the above, and unless the Committee determines otherwise, in the event that (i) an optionee dies, (ii) his representative has a right to exercise
an Option, (iii) the Option is not exercised by the last day on which it is exercisable, and (iv) the option price per share is below the Fair
Market Value of a Share on such date, the Option shall be deemed exercised on such date via a cashless exercise procedure and the resulting
proceeds net of any required tax withholding shall be paid to the representative.
(d) Other Provisions. The Option shall also be subject to such other terms and conditions as the Committee shall deem advisable or
appropriate, consistent with the provisions of the Plan as herein set forth. In addition, Incentive Stock Options shall contain such other
provisions as may be necessary to meet the requirements of the Code and the Treasury Department rulings and regulations issued thereunder
with respect to Incentive Stock Options.
17

SECTION 9. PERFORMANCE AWARDS
Performance Awards may be paid in cash, Shares, other property, or any combination thereof, and may be subject to such other terms and
conditions as the Committee shall deem advisable or appropriate, consistent with the provisions of the Plan as set forth, in the sole discretion
of the Committee at the time of payment. The performance levels to be achieved for each Performance Period and the amount of the Award
to be distributed shall be conclusively determined by the Committee. Performance Awards may be paid in a lump sum or in installments
following the close of the Performance Period or, in accordance with procedures established by the Committee, on a deferred basis.
SECTION 10. OTHER STOCK UNIT AWARDS
(a) Stock and Administration. Awards that are valued by reference to, or are otherwise based on, Shares (“Other Stock Unit Awards”) may
be granted hereunder to Participants, either alone or in addition to other Awards granted under the Plan, and such Other Stock Unit Awards
shall also be available as a form of payment in the settlement of other Awards granted under the Plan. Other Stock Unit Awards may be paid
in Shares, cash or any other form of property, as the Committee shall determine. Subject to the provisions of the Plan, the Committee shall
have sole and complete authority to determine the Employees to whom and the time or times at which such Awards shall be made, the
number of Shares to be granted pursuant to such Awards, and all other conditions of the Awards. Any Other Stock Unit Awards shall be
subject to such other terms and conditions as the Committee shall deem advisable or appropriate, consistent with the provisions of the Plan
as herein set forth. Unless the Committee determines otherwise to address specific considerations, Other Stock Unit Awards granted to
Employees shall have a vesting period of not less than one year.
(b) Other Provisions. Shares (including securities convertible into Shares) subject to Awards granted under this Section 10 may be issued
for no cash consideration or for such minimum consideration as may be required by applicable law.
SECTION 11. CHANGE IN CONTROL PROVISIONS
(a) Unless the Committee or Board shall determine otherwise at the time of grant with respect to a particular Award, and notwithstanding
any other provision of the Plan to the contrary, in the event a Participant’s employment or service is involuntarily terminated without cause
(as determined by the Committee or Board in its sole discretion) during the 24-month period following a Change in Control:
(i) any Options and Stock Appreciation Rights outstanding, and which are not then exercisable and vested, shall become immediately fully
vested and exercisable;
(ii) the restrictions and deferral limitations applicable to any Restricted Stock shall lapse, and such Restricted Stock shall immediately
become free of all restrictions and limitations and become fully vested and transferable to the full extent of the original grant;
(iii) all Performance Awards shall be considered to be earned and payable in full, based on the applicable performance criteria or, if not
determinable, at the target level and any deferral or other restriction shall lapse and such Performance Awards shall be immediately settled
or distributed; and
(iv) the restrictions and deferral limitations and other conditions applicable to any other Stock Unit Awards or any other Awards shall
immediately lapse, and any such Other Stock Unit Awards or such other Awards shall become free of all restrictions, limitations or
conditions and become fully vested and transferable to the full extent of the original grant.
(b) Change in Control Cash Out. Notwithstanding any other provision of the Plan, in the event of a Change in Control the Committee or
Board may, in its discretion, provide that each Option or Stock Appreciation
18

Right shall, upon the occurrence of a Change in Control, be cancelled in exchange for a cash payment to be made within 60 days of the
Change in Control in an amount equal to the amount by which the Change in Control Price per Share exceeds the purchase price per Share
under the Option or Stock Appreciation Right (the “spread”) multiplied by the number of Shares granted under the Option or Stock
Appreciation Right.
19

Exhibit 10(5)
PFIZER INC
NONFUNDED SUPPLEMENTAL
RETIREMENT PLAN

The Pfizer Inc Nonfunded Supplemental Retirement Plan (the “Supplemental Plan”) is hereby amended and restated
effective January 1, 2005 by Pfizer Inc (the “Company”) to provide supplemental retirement benefits to eligible employees
pursuant to the terms and provisions set forth below.
This Supplemental Plan is intended (1) to comply with section 409A of the Internal Revenue Code of 1986, as
amended (the “Code”),
and official guidance issued thereunder (except for Grandfathered Benefit amounts described in
Section 3(A)), and (2), for purposes of the Employee Retirement Income Security Act of 1974 (“ERISA”),
as amended, to be
treated as two separate, unfunded plans. One plan shall be an “excess benefit plan”
within the meaning of Section 3(36) of
ERISA, and shall be comprised of accruals under the Supplemental Plan that are made solely because of the applicable
limitations under Code section 415, plus earnings thereon. All other accruals under the Supplemental Plan, plus earnings
thereon, shall be treated as made under a separate “top-hat”
plan maintained by the Company primarily for the purpose of
providing deferred compensation to a select group of management or highly compensated employees, within the meanings of
Sections 201(a)(2), 301(a)(3) and 401(a)(1) of ERISA.
1.
Through this Supplemental Plan, the Company shall make payments supplementing the amounts payable under the
Pfizer Retirement Annuity Plan (the “Annuity Plan”)
to retiring employees whose benefits under the Annuity Plan are limited by
reason of Code section 415 and, on and after January 1, 1989, Code section 401(a)(17), to amounts less than would be
payable under the provisions of the Annuity Plan if calculated without reference to the limitations imposed by Code section 415
and, on and after January 1, 1989, Code section 401(a)(17).
2
Such supplemental payments by the Company shall, in the case of each such employee, be equal to the difference
between the benefits payable under the Annuity Plan and the benefits that would be payable under the provisions of the
Annuity Plan if calculated without reference to the limitations imposed by Code section 415 and,
-
2
-

on and after January 1, 1989, Code section 401(a)(17), and further the Company shall make payments supplementing the
amounts payable under the Annuity Plan for employees who elect to defer income under the “
Pfizer Inc Nonfunded Deferred
Compensation and Supplemental Savings Plan”
or its successor by treating such deferred amounts as though they were a part
of the employee’s “Creditable Earnings” under the Annuity Plan.
2A.
Notwithstanding Section 2 or any other provision of this Supplemental Plan, the amount of supplemental payments
by the Company may, to the extent provided in separate written agreements with an employee, be increased by calculating the
benefits payable under the provisions of the Plan which are to be calculated without reference to the limitations imposed by
Code section 415 and Code section 401(a)(17) as adjusted in any of the following manners by: (1) imputing additional credited
service, which may or may not be taken into account for vesting and participation purposes as determined in the written
agreement, (2) imputing additional earnings, and/or (3) offsetting amounts relating to benefits actually paid or payable under
qualified or nonqualified plans of prior employers. No such adjustment to the amount of any benefit pursuant to this Section 2A
shall affect the time or form of payment of any benefit payable under this Supplemental Plan.
3. (A) Grandfathered Benefits
. With respect to Supplemental Plan benefits that were earned and vested as of
December 31, 2004 within the meaning of Code section 409A and regulations thereunder (the “Grandfathered Benefits”),
at
least six (6) months before an employee ceases to be an employee of the Company, the employee may elect, or may modify an
election that the employee had previously made, to receive payment of such supplemental payments by the Company in a lump
sum or in annual installments, and provided that in the absence of an election, such supplemental payments by the Company
shall be made in ten annual installments (10-year Certain). Calculation of present value shall be made using the Annuity Plan’
s
actuarial assumptions for payment of lump sums at the time of the benefit commencement date of the Annuity Plan benefit. The
lump sum payment or first annual installment payment
-
3
-

shall be made in the January coincident with or following the commencement of the employee’s (or spouse’
s in the case of the
employee’
s death prior to commencement) benefit under the Annuity Plan. This Section 3(A) is intended to reflect the
requirements of the Supplemental Plan as in effect on October 3, 2004, without any subsequent material modification and shall
be interpreted to that effect.
(B) NonGrandfathered Benefits
. With respect to Supplemental Plan benefits that are earned or vested on or after
January 1, 2005 (within the meaning of Code section 409A and regulations thereunder (the “NonGrandfathered Benefits”
),
except as provided in Section 4(B)(ii) and (iii) and Section 7, the employee will receive payment of such supplemental
payments by the Company in a lump sum in the January coincident with or following the later of (i) such employee’
s
“Separation from Service” (
within the meaning of Code section 409A) or (ii) attainment of the earliest of the following:
(a) attainment of age fifty five (55), or (b), such employee’
s age added to years of Creditable Service as determined under the
Annuity Plan equaling or exceeding ninety (90). Except in the case of death or a re-
deferral under Section 7, when the
supplemental annuity payments under this Section are converted to a lump sum form of payment, such lump sum shall be
calculated using the actuarial assumptions for calculations of lump sum benefits under the Annuity Plan at the first of the month
coincident with or following the later of (i) such employee’s “Separation from Service” (
within the meaning of Code section
409A) or (ii) attainment of the earliest of the following: (a) attainment of age fifty five (55), or (b), such employee’
s age added to
years of Creditable Service as determined under the Annuity Plan equaling or exceeding ninety (90). Notwithstanding the
foregoing, payments may not be made to a Key Employee upon Separation from Service before the date which is six
(6) months after the date of the Key Employee’
s Separation from Service (within the meaning of Code section 409A). Any
payments that would otherwise be made during this period of delay shall be accumulated and paid on the first day of the
seventh month following the Key Employee’
s Separation from Service (within the meaning of Code section 409A), or, if earlier,
the first day of the month after such employee’
s death. Key Employees are those employees who are (i) eligible for the
Company’s Executive Long-Term Incentive
-
4
-

Program (or successor program) on January 1 of the relevant year, or (ii) treated as a “specified employee”
as of his or her
Separation from Service under Code section 409A(a)(2)(B)(i), i.e., a key employee (as defined in Code section 416(i) without
regard to paragraph (5) thereof) of the Company or its affiliates, determined in accordance with Code section 409A using a
January 1 identification date, and effective for the 12-month period.
(i) Transition Elections. With respect to employees with NonGrandfathered Benefits that were earned or vested prior to
December 31, 2007, transition distribution elections allowing for the election of optional forms of payment other than the lump
sum form for NonGrandfathered Benefits, were filed by certain employees with NonGrandfathered Benefits, and such elections
shall be enforced and irrevocable except to the extent any NonGrandfathered Benefits are subsequently re-
deferred as allowed
under Section 7.
(ii) Death. Notwithstanding any elections under, or provisions of, this Supplemental Plan to the contrary, with respect to
NonGrandfathered Benefits, upon the employee’s death, NonGrandfathered Benefits shall be paid to the employee’
s
beneficiary (to the extent payable), in a lump sum in the January following the later of (i) the employee’
s date of death; or (ii) at
the time when the employee would have attained the earliest of the following: (a) attainment of age fifty five (55), or (b), such
employee’
s age added to years of Creditable Service as determined under the Annuity Plan equaling or exceeding ninety (90).
Such payment shall be made regardless of any re-
deferral by the employee under Section 7, and irrespective of whether the
employee was a Key Employee. When the supplemental annuity payments under this Section are converted to a lump sum
form of payment, such lump sum shall be calculated using the actuarial assumptions for calculations of lump sum benefits
under the Annuity Plan at the first of the month coincident with or following the later of (i) the employee’
s death, or (ii) at the
time when the employee would have attained the earliest of the following: (a) attainment of age fifty five (55), or (b), such
employee’s age added to years of Creditable Service as determined under the Annuity Plan equaling or exceeding ninety (90).
-
5
-

(
iii) Disability. Notwithstanding any elections under, or provisions of, this Supplemental Plan to the contrary, with
respect to NonGrandfathered Benefits, such payments shall be paid in a lump sum in the January coincident with or following
the latest to occur of: (i) the employee’s cessation of entitlement to benefits under the Company’s long-
term disability program;
(ii) the employee’s “Separation from Service” (as defined in Code section 409A); or (iii) the employee’
s attainment of age 65. If
the employee subsequently recovers from Disability and resumes work with the Company, NonGrandfathered Benefits accrued
to such date of return to work shall continue to be paid in accordance with the foregoing sentence. Any NonGrandfathered
Benefits accrued thereafter shall be governed under Section 3(B).
(C) Automatic Cash Out. If the lump sum amount of the entire supplemental payment (including both Grandfathered
and NonGrandfathered Benefits) is $10,000 or less, such payment of both Grandfathered and NonGrandfathered Benefits shall
be made in a lump sum at such time. The calculation shall be performed only once at the first distribution date to occur under
Section 3A or 3B.
4. An employee’
s right to supplemental payments hereunder may not be assigned. If an employee does assign such
right, the Company may disregard such assignment and discharge its obligation by making payment as though no such
assignment had been made.
5. The Committee may make non-
substantive administrative changes to this Supplemental Plan so as to conform with or
take advantage of governmental requirements, statutes or regulations.
6.
In addition to the benefit payable under the first sentence of Section 2, if any, the Company shall make a lump sum
cash payment to those employees who (i) have
-
6
-

attained age fifty (50) on the date of their termination, (ii) accepted the pension enhancement offered to them under the Pfizer
Enhanced Employee Separation Program implemented in connection with the April 2003 acquisition of Pharmacia Corporation
(the “Enhancement”), (
iii) after giving effect to the Enhancement, are eligible for early retirement, normal retirement, or the rule
of 90 described under Section 4.2 of the Annuity Plan (the “Rule of 90”),
and (iv) were credited with pensionable earnings within
the meaning of the Annuity Plan in 2002 of between $103,000 and $200,000. The amount of the lump sum cash payment shall
be equal to the difference between (i) and (ii) where: (i) is the present value of the accrued benefit of the employee under the
Annuity Plan determined as of the employee’
s termination date if calculated (a) by giving effect to a five (5) point enhancement
in age and/or service solely for purposes of determining early retirement or normal retirement eligibility under the Annuity Plan
and the Rule of 90, but not for purposes of actuarial reduction on account of age under Schedules B or C of the Annuity Plan if
the employee has not attained normal retirement age or met the Rule of 90 after taking into account the five (5) point
enhancement (but no more than a combined total of five (5) points in the combination of age and service which provides the
employee with the greater benefit), and (b) without reference to the limitations of Code sections 415 and 401(a)(17); and (ii) is
the present value of the sum of (a) the accrued benefit of the employee under the Annuity Plan determined as of the
employee’
s termination date and (b) the payments, if any, by the Company to the employee under the first sentence of
Section 2 above. Calculation of present value shall be made using the Annuity Plan’
s actuarial assumptions for payment of
lump sums. Such lump sum payment shall be made as soon as practicable following the employee’
s Separation from Service
(within the meaning of Code section 409A and regulations thereunder), but in no event more than ninety (90) days following
such Separation from Service.
-
7
-

7. Re-
Deferrals. Notwithstanding any election or provision of this Supplemental Plan to the contrary, an employee may
make one or more subsequent elections to change the time and form of a payment for a NonGrandfathered Benefit, provided
that, except in the case of changing among annuity forms, such an election shall be effective only if the following conditions are
satisfied:
(i) An election may not take effect until at least twelve (12) months after the date on which the election is made;
(ii) In the case of an election related to a payment, other than a payment described in Section 3(B)(ii), a distribution
may not be made earlier than at least five (5) years from the date the distribution would have otherwise have been paid; and
(iii) In the case of an election to change the time or form of a distribution related to a payment at a specified time or
pursuant to a fixed schedule, the election must be made at least twelve (12) months before the date the distribution is
scheduled to be paid.
When the supplemental annuity payments under this Section are converted to a lump sum form of payment, such lump sum
shall be calculated using the actuarial assumptions for calculations of lump sum benefits under the Annuity Plan at the first of
the month following the later of (i) such employee’s “Separation from Service” (
within the meaning of Code section 409(A) or
(ii) attainment of the earliest of the following: (a) attainment of age fifty five (55), or (b) such employee’
s age added to years of
Creditable Service as determined under the Annuity Plan equaling or exceeding ninety (90), and such lump sum amount shall
be carried forward with interest until the date of distribution under this Section.
8.
Permitted Delays. Notwithstanding any other provision of this Supplemental Plan, any payment on account of an
employee under the Supplemental Plan shall be delayed upon the Company’
s reasonable anticipation of one or more of the
following events:
(a) The Company’s deduction with respect to such payment would be eliminated by application of Code section
162(m); or
-
8
-

(b) The making of the payment would violate Federal securities laws or other applicable law;
provided, that (i) the delay rule shall be applied consistently to similarly situated employees, (ii) the employee shall not have a
choice as to the timing of the payment, and (iii) any payment delayed pursuant to this Section 8 shall be paid in accordance
with Code section 409A.
9. FICA Taxes. The payment of NonGrandfathered Benefits shall be accelerated as necessary to pay FICA taxes
(and any corresponding income taxes and/or to satisfy any withholding requirements related thereto), in a timely manner.
10. Amendment and Termination. The Board of Directors of the Company or its authorized designee shall have the
rights to amend, suspend, or terminate the Supplemental Plan at any time. Upon termination of the Supplemental Plan,
payment of benefits shall be made to employees and beneficiaries in the manner and at the time described in the Supplemental
Plan unless the Board of Directors of the Company or its authorized designee determines in its sole and absolute discretion that
all such amounts shall be distributed upon termination and in accordance with the requirements under Code section 409A.
Upon termination of the Supplemental Plan, no further accruals of benefits shall be permitted. In the event the Supplemental
Plan is terminated, the Company shall continue to administer the Supplemental Plan in accordance with the relevant provisions
thereof until the employees’ benefits have been paid hereunder. Notwithstanding the foregoing, no amendment of the
Supplemental Plan shall apply to Grandfathered Benefits unless the amendment specifically provides that it applies to such
amounts. The purpose of this restriction is to prevent a Supplemental Plan amendment from resulting in an inadvertent
“material modification” to Grandfathered Benefits.
* * *
-
9
-

Form of Amendments to the
PFIZER INC NONFUNDED SUPPLEMENTAL RETIREMENT
PLAN (“SPRAP”)
Any amounts which may not be paid under Appendix B of Part B of the PCPP due to such Member having more than $132,000 in
pensionable earnings in 2004 (as adjusted in accordance with tax laws and regulations) shall be payable hereunder in accordance with
the terms of such Appendix B.
11. Errors in Calculating Lump-Sum Option Payments. Whenever due to (1) a bona fide mathematical or actuarial error, or
(2) additional compensation for purposes of the Plan for the taxable year of the Participant in which the Participant has a Separation
from Service which has been administratively impracticable to take into account at the time of such Separation from Service, the amount
of such 409A Benefit is determined after such payment to have been less than if such error had not been made or such compensation
taken into account, then a supplemental corrective lump sum payment correcting such error or taking into account such additional
compensation may be made by the Company prior to December 31st of the year in which the lump sum payment was made, After such
December 31 , no further corrective payment shall be made.
12. Claims Procedures. Any request by a participant or any other person for any benefit alleged to be due under the Plan shall
be known as a “Claim” and the participant or other person making a Claim, or the authorized representative of either, shall be known as
a “Claimant.” The Retirement Committee or its delegate reviewer has sole discretion to determine whether a communication from an
individual shall be
1.
Section 6 of the SPRAP is amended by adding to the end thereof to read as follows:
2.
Section 11 is added to the end of the SPRAP to read as follows:
3.
Section 12 is added to the end of the SPRAP to read as follows:
st

a Claim. To the extent of their responsibility to review benefit claims or to review the denial of benefit claims, the Committee and the
reviewer shall have full authority to interpret and apply, in their discretion, the provisions of the Plan. The decisions of the Committee
and reviewer shall be final and binding upon any and all Claimants, including, but not limited to, participants and their beneficiaries,
and any other individuals making a Claim or requesting review of a Claim through or under them, and shall be afforded the maximum
deference permitted by law. A participant may not maintain a court action over a disputed claim until he or she has exhausted the Plan’s
claims procedures.
Claimant may submit a written application to the Committee or its delegate reviewer for payment of any benefit that he believes
may be due him under the Plan, in accordance with Plan procedures. Such application shall include a general description of the benefit
which the Claimant believes is due, the reasons the Claimant believes such benefit is due and any information as the Committee or its
delegate reviewer may reasonably request. The Committee or its delegate reviewer will process the Claimant’
s application within ninety
(90) days of the receipt of the Claim by the Committee or its delegate reviewer unless special circumstances require an extension of time
for processing the Claim. In such event, written notice of the extension shall be furnished to the Claimant prior to the termination of the
initial ninety (90) day period but in no event shall the extension exceed a period of ninety (90) days from the end of such initial period.
The notice shall indicate the special circumstances requiring an extension of time and the date by which the Plan expects to render the
final decision. If the Committee or its delegate reviewer has not determined the Claimant’s eligibility for a Plan benefit within this
ninety (90) day period (one hundred eighty (180) day period if circumstances require an extension of time), the Claim is deemed denied.
A Claim is considered approved only if such approval is memorialized by the Committee or its delegate reviewer in writing.
If a Claim is denied in whole or in part, the notice of denial shall set forth (i) the specific reason or reasons for the denial,
(ii) specific reference to the pertinent Plan provisions on which the denial is based, (iii) a description of any additional material or
information necessary for the Claimant to perfect the Claim and an explanation of why such material or information is necessary, if
applicable, and (iv) an explanation that, if an adverse determination is made on review, the Claimant may have a right to bring civil
action under Section 502(a) of ERISA. Within sixty (60) days of the receipt of a notice of denial of a Claim in whole or in part or a
deemed denial, a Claimant (i) may request a review upon written application to the Committee, (ii) may review documents pertinent to
the Claim, and (iii) may submit issues

and comments in writing to the Committee. The Claimant shall be provided upon request and free of charge, reasonable access to all
documents, records and other information relevant to the Claimant’s Claim for benefits.
The Committee will review a Claim for which a request for review has been made and render a decision not later than sixty
(60) days after receipt of a request for review; provided, however, that if special circumstances require extension of a time for
processing, a decision shall be rendered no later than one hundred and twenty (120) days after receipt of the request for review. Written
notice of any such extension shall be furnished to the Claimant within sixty (60) days after receipt of request for review. The
Committee’s decision shall be in writing and shall set forth (i) the specific reason or reasons for the denial on review, (ii) specific
reference to the pertinent Plan provisions on which the denial on review is based, (iii) an explanation that the Claimant is entitled to
receive, upon request and free of charge, reasonable access to, and copies of, all documents, records, and other information relevant to
the Claimant’s Claim for benefits, and (iv) an explanation that if an adverse determination is made on review, the Claimant may have
the right to bring a civil action under Section 502(a) of ERISA. If the decision on review is not furnished within the applicable time, the
Claim shall be deemed denied on review.

Exhibit 10(6)
PFIZER INC NONFUNDED DEFERRED COMPENSATION AND
SUPPLEMENTAL SAVINGS PLAN
Amended and Restated as of January 1, 2008

1.1 Continuation
. There is hereby continued for the benefit of Members an unfunded plan of deferred compensation known as the
“Pfizer Inc Nonfunded Deferred Compensation and Supplemental Savings Plan.”
1.2 Purpose . The purpose of this Plan is to provide a means by which an Eligible Employee may, in certain circumstances, elect
to defer receipt of a portion of his “Regular Earnings,” and such other deferrals as determined by the Company in accordance with
Section 4.2.
1.3 Description of the Plan . The Plan became effective July 1, 1983, was amended and restated effective February 1, 2002, and
was again amended and restated effective January 1, 2008, except as otherwise provided herein, to reflect: (i) the merger of Pharmacia
Savings Plus Plan into the Plan, and (ii) the enactment of Code Section 409A and corresponding regulations, and (iii) certain other
administrative design changes. The provisions of this restated and amended Plan shall govern Accounts on and after January 1, 2008 except
with respect to Grandfathered Amounts. Except as specifically otherwise provided herein, the Grandfathered Amounts for Members who
were Participants in the Pharmacia Savings Plus Plan on December 31, 2004 shall be governed by the provisions of the Pharmacia Savings
Plus Plan as amended and restated effective July 1, 2002; the Grandfathered Amounts with respect to Members in this Plan on December 31,
2004, shall be governed by the provisions of this Plan as amended and restated effective February 1, 2002; and, for the period from
January 1, 2005 through December 31, 2007 the provisions of this amended and restated Plan shall govern, except to the extent the
provisions of this Plan are inconsistent with the administrative practices, policies, election forms and participant communications designed
for reasonable good faith compliance with Code Section 409A during that interim period, which are incorporated herein by reference. For
purposes of the Employee Retirement Income Security Act of 1974 (“ERISA”), as amended, the Plan shall be treated as two separate,
unfunded plans. One plan shall be an “excess benefit plan” within the meaning of Section 3(36) of ERISA, and shall be comprised of
accruals under the Plan that are made solely because of the applicable limitations under Section 415 of the Code, plus earnings thereon. All
other accruals under the Plan, plus earnings thereon, shall be treated as made under a separate “top-hat” plan maintained by the Company
primarily for the purpose of providing deferred compensation to a select group of management or highly compensated employees, within the
meanings of Sections 201(a)(2), 301(a)(3) and 401(a)(1) of ERISA. The Company shall be able to separately account for excess benefit plan
accruals and earnings thereon, top-hat plan accruals and earnings thereon, and Special Accruals and earnings thereon.
The following words and phrases as used in this Plan have the following means:
2.1 Account . The term “Account” shall mean a Member’s individual account(s), as described in Section 5.1 of the Plan.
-
2
-
SECTION 1
.
CONTINUATION AND PURPOSE OF THE PLAN
.
SECTION 2
.
DEFINITIONS
.

2.2 Annual Enrollment . The term “Annual Enrollment” shall mean the time period, as determined by the Committee in its sole
and absolute discretion, prior to the beginning of a Plan Year in which Eligible Employees can elect to enroll or change their deferral
elections under the Plan with respect to Regular Earnings expected to be earned in the next succeeding Plan Year. Notwithstanding the
foregoing, the Annual Enrollment period for any Plan Year shall not extend beyond December 31 of the Plan Year immediately preceding
the Plan Year that the election is with respect to.
2.3 Beneficiary . The term “Beneficiary” means the beneficiary on file for this Plan, or if none is on file, the person or entity who
is the “Beneficiary” under the Qualified Plan, and with respect to Grandfathered Amounts under the Pharmacia Savings Plus Plan, the
person or entity who is the “beneficiary” under the rules of that plan.
2.4 Board of Directors . The term “Board of Directors” means the Board of Directors of the Company.
2.5 Code . The term “Code” means the Internal Revenue Service Code of 1986, as amended.
2.6 Committee . The term “Committee” means the Committee, as described in the Qualified Plan, or any other person or entity
that the Committee has authorized to act on its behalf under the Plan.
2.7 Company . The term “Company” means Pfizer Inc, a Delaware corporation, and any successor corporation.
2.8 Controlled Group . The term “Controlled Group” means the Company and any other entity with which the Company would
be considered a single employer under Code section 414 (b) or (c), provided that, in applying Code sections 1563(a)(1), (2) and (3) and for
purposes of determining a controlled group of corporations under section 414(b), “50 percent” shall be used instead of “80 percent”, and in
applying Treas. Reg. section 1.414(c)-2 for purposes of determining trades or businesses that are under common control for purposes of
Code section 414(c), “50 percent” shall be used instead of “80 percent” each place it appears in Treas. Reg. section 1.414(c)-2. In addition,
solely for purposes of determining a Separation from Service, the foregoing sentence shall be applied by using 30 percent instead of 50
percent.
2.9 Disability . The term “Disability” means a disability where the Employee is unable to engage in any substantial gainful
activity by reason of any medically determinable physical or mental impairment that (i) can be expected to result in death or can be expected
to last for a continuous period of not less than twelve (12) months or is receiving income replacement benefits for a period of not less than
three (3) months under an accident and health plan covering employees of the Company by reason of any medically determinable physical
or mental impairment that can be expected to result in death or can be expected to last for a continuous period of not less than 12 months,
and (ii) also satisfies the requirements of a “Disability” as defined under the Qualified Plan.
-
3
-
st

2.10 Eligible Employee . The term “Eligible Employee” means any “Member” as defined under the Qualified Plan who:
(i) (a) in the year he or she first becomes eligible to participate in the Plan (as determined in accordance with consistent rules
established by the Committee in its sole and absolute discretion and in accordance with Section 409A) in any month of that year: (1) is
projected to receive Compensation (as defined in the Qualified Plan) for that Plan Year in excess of the limitation of Section 401(a)(17) of
the Code or whose account under the Qualified Plan is projected to be credited during that PlanYear with “annual additions,” as defined in
Section 415(c)(2) of the Code equal to the maximum permitted under Section 415(c)(1)(A) of the Code; and (2) who is an employee of an
Employer who: (A) has reached the point in time when he or she has been projected to have Compensation (as defined in the Qualified Plan)
in excess of the Limitation under Section 401(a)(17) of the Code, or (B) has reached the point in time when he or she has Compensation (as
defined in the Qualified Plan) equal to the amount of Compensation at which point he or she was projected to reach the Section 415(c)(2)
Limitation on annual additions under the Qualified Plan; or (b) in any subsequent year an Eligible Employee who in any prior year was an
Eligible Employee under the Plan and who the Company determines in an Annual Enrollment (based on elections in effect and salary
projections on the last business day of the calendar year of the Annual Enrollment (or as otherwise determined based on consistent rules
established by the Committee in its sole and absolute discretion and in accordance with Section 409A) is expected to have annualized
Compensation for the subsequent Plan Year in excess of the limitation of Section 401(a)(17) of the Code, or is expected in the next
succeeding Plan Year to have his or her Account under the Qualified Plan credited with annual additions in excess of the maximum amount
permitted under Section 415(c)(1)(A) of the Code and who is an Employee and has an election to defer Excess Regular Earnings under the
Plan in effect at the time he or she had been projected to reach the limitation under Section 401(a)(17) or Section 415(c)(1)(A) of the Code;
or,
(ii) any other person who is a member of a select group of management or highly compensated employees of the Company and who
is designated by the Committee or an authorized officer of the Company (or his or her delegate) as an Eligible Employee to receive accruals
under the Plan in accordance with Article 4.
2.11 ELT . The term “ELT” means the Chief Executive Officer of the Company and the team composed of his or her direct reports
or any of their properly authorized delegates.
2.12 Employer . The term “Employer” means the Company and any other member of the Controlled Group which is also an
“Associate Company” under the Qualified Plan.
2.13 Employer Accrual . The term “Employer Accrual” means the amounts described in Section 4.1.
2.14 Excess Regular Earnings . The term “Excess Regular Earnings” means:
-
4
-

(i) with respect to the first year that an Eligible Employee is eligible to participate in the Plan (as determined in accordance with
consistent rules established by the Committee in its sole and absolute discretion and in accordance with Section 409A), (a) the portion of an
Eligible Employee’s Regular Earnings earned after the point in time when the Eligible Employee was projected to exceed the Limitation on
compensation taken into account under Section 401(a)(17) of the Code, (b) all Regular Earnings that the Eligible Employee receives after
the point in time that the Eligible Employee was projected to exceed the Limitation on contributions to defined contribution plans under
Section 415(c)(1)(A) of the Code, to the extent not included in (a) above, (c) any bonus elected to be deferred under the Pfizer Inc Deferred
Compensation Plan, in accordance with the rules under that Plan and Section 409A, or (d) any other compensation determined by the
Committee to be Excess Regular Earnings for purposes of this Plan; and,
(ii) for an Eligible Employee who was an Eligible Employee in the immediately preceding prior Plan Year or who is not in his or her
first year of eligibility to participate in the Plan, (a) the portion of Regular Earnings earned during a Plan Year that based on the Eligible
Employee’s Qualified Plan elections and Compensation (as defined in the Qualified Plan) in effect during the last business day of the
calendar year of the Annual Enrollment (or as otherwise determined based on consistent rules established by the Committee in its sole and
absolute discretion and in accordance with Section 409A) were projected to exceed the Limitation on compensation taken into account under
Section 401(a)(17) of the Code, (b) all Regular Earnings that the Member has received after the point in time that the Member was projected
to become subject to the Limitation on contributions to defined contribution plans under Section 415(c)(1)(A) of the Code, to the extent not
included in (a) above, (c) any bonus eligible to be deferred under the Pfizer Inc Deferred Compensation Plan, in accordance with the rules
under that Plan and Section 409A, or (d) any other compensation determined by the Committee to be Excess Regular Earnings for purposes
of this Plan.
2.15 Excess Regular Earnings Deferrals . The term “Excess Regular Earnings Deferrals” means the portion of a Member’s Excess
Regular Earnings that the Member elects to defer under the terms of the Plan.
2.16 Grandfathered Amounts . The term “Grandfathered Amounts” shall mean the portion of the Member’s Account that reflects
the amount that was earned and vested (within the meaning of Section 409A of the Code and regulations thereunder) under the Plan prior to
2005 (and earnings thereon), or with respect to Accounts transferred from the Pharmacia Savings Plus Plan, the portion of the Member’s
Account that reflects the amount that was earned and vested (within the meaning of Section 409A of the Code and regulations thereunder)
under the Pharmacia Savings Plus Plan prior to 2005 (and earnings thereon).
2.17 Key Employee . The term “Key Employee” means an Employee treated as a “specified employee” as of his or her Separation
from Service under Section 409A(a)(2)(B)(i) of the Code, i.e. , a key employee (as defined in Section 416(i) of the
-
5
-

Code without regard to paragraph (5) thereof) of the Company or its affiliates. Key Employees shall be determined in accordance with
Section 409A using a January 1 identification date. A listing of Key Employees as of an identification date shall be effective for the 12-
month period beginning on the April 1 following the identification date and ending on March 31 of the next calendar year.
2.18 Limitation(s) . The term “Limitation(s)” means the limitation on contributions to defined contribution plans under Section 415
(c)(1)(A) of the Code, and on compensation taken into account under Section 401(a)(17) of the Code, and with respect to Eligible
Employees who are a select group of management or highly compensated employees, within the meanings of Sections 201(a)(2) and 401(a)
(1) of ERISA and are eligible to defer their bonuses under the Pfizer Deferred Compensation Plan, such other Code or Qualified Plan limits
that prevent the deferred bonuses as being recognized as Regular Earnings under the Qualified Plan.
2.19 Member . The term Member means an Eligible Employee who has Excess Regular Earnings Deferrals made to the Plan or is
otherwise credited with an Employer Accrual.
2.20 Payment Option . The term “Payment Option”
means the following forms of payment under which an Eligible Employee may
elect to receive amounts credited to his Account upon his Separation from Service with the Controlled Group: (i) single sum payable in the
January following the Member’s Separation from Service with the Controlled Group, or (ii) substantially equal annual installment payments
over a period of two (2) to twenty (20) years, with the first installment to be paid the January following the Member’s Separation from
Service. Where payment of the Account is made in installment payments, the first installment shall be a fraction of the value of the
Member’s Account as of the applicable valuation date, the numerator of which is one (1) and the denominator of which is the total number
of installments remaining to be paid at that time. Each subsequent installment shall be calculated in the same manner, except that the
denominator shall be reduced by the number of installments that have been paid previously. Unless otherwise provided under this Plan,
including in the event of death, Disability and or a payment due to Unforeseeable Emergency, if a payment election is not timely made in
accordance with the requirements of Section 409A the Member shall be deemed to have elected to receive payment of his or her Account in
a single lump sum payment to be paid in the January following the Member’s Separation from Service. Notwithstanding anything in this
Section 2.20 or the Pharmacia Savings Plus Plan to the contrary, effective January 1, 2007, the Account of any Member who has Separated
from Service and is no longer living at the time his benefits commence shall be paid in a single lump sum the January following the
Member’s death, provided, however, that payment of Grandfathered Amounts to Beneficiaries under the Pharmacia Savings Plus Plan shall
be governed under the terms of that plan.
-
6
-

2.21 Pfizer Deferred Compensation Plan . The term “Pfizer Deferred Compensation Plan” shall mean the Pfizer Inc Deferred
Compensation Plan, a bonus deferral program, or its successor.
2.22 Pfizer Match Fund . The term “Pfizer Match Fund”
shall mean the investment fund known as the Pfizer Match Fund under the
Qualified Plan, or its successor.
2.23 Pharmacia Savings Plus Plan . The term Pharmacia Savings Plus Plan means the Pharmacia Savings Plus + Plan, effective
July 1, 1999, as subsequently amended and restated effective July 1, 2002 which was merged into this Plan effective January 1, 2008.
2.24 Plan . The term “Plan” means this “Pfizer Inc Nonfunded Deferred Compensation and Supplemental Savings Plan,” as set
forth herein and as amended from time to time.
2.25 Plan Year . The term “Plan Year” means the calendar year.
2.26 Prior Plan . The term “Prior Plan” means: (i) with respect to Grandfathered Amounts attributable to the Plan, the Plan as in
effect on October 3, 2004, which has not been materially modified (attached hereto as Exhibit A), and (ii) with respect to Grandfathered
Amounts attributable to the Pharmacia Savings Plus Plan, the Pharmacia Savings Plus Plan as in effect on October 3, 2004, which has not
been materially modified (attached hereto as Exhibit B).
2.27 Qualified Plan . The term “Qualified Plan” means the Pfizer Savings Plan, as amended from time to time.
2.28 Regular Earnings . The term “Regular Earnings” shall have the meaning given such term under the Qualified Plan. For
Eligible Employees who are eligible to defer their bonus under the Pfizer Deferred Compensation Plan, the term “Regular Earnings” shall
also include such deferred amounts as determined by the Committee.
2.29 Section 409A . The term “Section 409A” shall mean Section 409A of the Code and the regulations and other guidance issued
thereunder by the U.S. Treasury or Internal Revenue Service.
2.30 Separation from Service . The term “Separation from Service” or “Separates from Service” means a “separation from service”
within the meaning of Section 409A.
2.31 Special Accrual . The term “Special Accrual” means a special lump sum accrual amount made pursuant to a Written
Agreement as provided for in Section 4.2 of the Plan.
-
7
-

2.32 Unforeseeable Emergency . The term “Unforeseeable Emergency” means a severe financial hardship to the Member resulting
from an illness or accident of the Member, the Member’s spouse, or dependent (as defined in Section 152(a) of the Code); the Member’s
loss of property due to casualty; or other similar extraordinary and unforeseeable circumstances arising as a result of events beyond the
Member’s control, within the meaning of Section 409A. Withdrawals for Unforseeable Emergencies are only available to Members who
were Participants in, and with respect to the portion of a Member’s Account that was credited under, the Pharmacia Savings Plus Plan (as
adjusted for earnings and losses) on December 31, 2007 and other than Grandfathered Amounts that are governed under the distribution
rules of that Prior Plan.
2.33 Written Agreement . The term “Written Agreement” shall have the meaning ascribed to it in Section 4.2.
3.1 Designation of Eligible Employees . The Committee in its sole and absolute discretion will designate as Eligible Employees
those employees who satisfy the terms of Section 2.10 and are eligible to participate in the Plan.
3.2 Election to Make Excess Regular Earnings Deferrals .
(a) Initial Election . An Eligible Employee may elect to begin making Excess Regular Earnings Deferrals by filing an election
with the Committee or its authorized designee in accordance with this Section 3.2 and the requirements of Section 409A and any rules
established by the Committee. Deferral elections for Excess Regular Earnings Deferrals in the year in which an employee first becomes
eligible to participate in the Plan (as determined in accordance with consistent rules established by the Committee in its sole and absolute
discretion and in accordance with Section 409A) may be made within 30 days of his or her first becoming eligible to participate in the Plan
or another account balance plan required to be aggregated with this Plan under Section 409A, provided that such elections shall apply only
with respect to Regular Earnings received subsequent to the date of receipt of election by the Committee and with such paycheck as
determined administratively practicable by the Committee. If no such election is made, an Eligible Employee may not make Excess Regular
Earnings Deferrals to the Plan in the year he or she first becomes eligible to participate in the Plan (as determined in accordance with
consistent rules established by the Committee in its sole and absolute discretion and in accordance with Section 409A) but may make Excess
Regular Earnings Deferrals in subsequent Plan Years to the extent he/or she submits a proper and timely election to do so under the Plan
during a subsequent Annual Enrollment and consistent with rules established by the Committee in its sole and absolute discretion and in
accordance with Section 409A.
-
8
-
SECTION 3
.
PARTICIPATION
.

(b) Subsequent Elections . For Plan Years after the first Plan Year in which the Eligible Employee participates in the Plan, an
Eligible Employee may make Excess Regular Earnings Deferrals to the Plan to the extent he/or she submits a proper and timely election to
do so under the Plan during an Annual Enrollment ending prior to such Plan Year (or at such other time as the Committee shall permit for
Employees who are treated (and eligible to be treated) as in their first year of eligibility under uniform rules established by the Committee in
accordance with Section 409A).
3.3 Amendment or Suspension of Election . Except as otherwise provided in this Section 3.3, once made, a Member may not
change his or her existing Excess Regular Earnings Deferrals election under this Plan during the Plan Year until the next Annual
Enrollment. Notwithstanding the foregoing, if a Member receives a hardship withdrawal under the Qualified Plan, incurs a Disability or
obtains a distribution under Section 6.4 on account of an Unforeseeable Emergency during a year, his or her Excess Regular Earnings
Deferral election shall be cancelled.
3.4 Amount of Elections . Each election for Excess Regular Earnings Deferrals to the Plan filed by an Eligible Employee must
specify the amount of Excess Regular Earnings Deferrals in a whole percentage from 1% to 20% of the Member’s Excess Regular Earnings
unless the Committee establishes a lesser percentage for the Plan Year.
4.1 General Rule . An Employer Accrual will be credited to a Member’s Account with respect to the eligible portion of Excess
Regular Earnings Deferrals of such Member at the Member’s applicable percentage rate of “Matching Contributions” with respect to “After-
Tax Contributions,” “Before-Tax Contributions,” and Roth 401(k) Contributions under the Qualified Plan. The Employer Accrual shall be
credited as soon as practicable following the payroll period for which the Excess Regular Earnings Deferrals are made. An Employer
Accrual (based on the Member’s matching contribution formula under the Qualified Plan) also will be credited to the Account of a Member
who elects to defer a percent of his or her bonus that otherwise would have been deferred under the Pfizer Deferred Compensation Plan,
subject to the requirements of Section 409A. Such Employer Accrual shall be credited as soon as practical following the payroll period in
which the bonus is deferred. In no event shall a Special Accrual be subject to Employer Accruals under this Section 4.1. Notwithstanding
anything in this Section 4.1 to the contrary, for purposes of any distribution or withdrawal under the Plan, except as otherwise provided
under Section 5.4, the amounts distributed or withdrawn shall be valued as of the last business day of the calendar quarter preceding the
calendar quarter of the distribution or withdrawal.
-
9
-
SECTION 4
.
EMPLOYER ACCRUALS
.

4.2 Special Accrual for Recruitment Purposes
Effective September 1, 2007, the Company may, in its sole and absolute discretion,
credit an amount to the Account of an Eligible Employee, provided that a member of the ELT approves such credit (but such ELT member
cannot approve such credit for him or herself).
(a) This credit to the Eligible Employee’s Account shall be made at the time, and subject to any restrictions, specified in the
written agreement or agreement that is evidenced in a writing from the ELT member (the “Written Agreement”), with the
Eligible Employee. At such time, the Eligible Employee shall become a Member if he is not already a Member. Such Written Agreement
cannot include any election on the part of the Eligible Employee unless such election satisfies the requirements of Code section 409A and
the election provisions have been approved by the ELT member and the Committee.
(b) Such credit shall be invested as specified in the Written Agreement, or, if no such investment election is specified, as provided
in accordance with Section 5.4 of the Plan except that no portion of this credit shall be considered Employer Accruals that are subject to a
deemed investment in the Pfizer Match Fund. No portion of this credit is eligible for an Employer Accrual under the Plan.
(c) Such credit, as adjusted for any investment gains or losses, shall be paid in accordance with Sections 5.2, 6.1 and 6.2 of the
Plan or as otherwise determined under the Written Agreement; provided that nothing herein (other than if the Written Agreement specifies
to the contrary) shall be interpreted so not as to afford the opportunity for the Member to change his time and form of payment election in
accordance with Section 409A if such right has been so provided by the ELT member and approved by the Committee.
5.1 Creation of Accounts . The Company will maintain an Account under the Plan in the name of each Member. Each Member’s
Account will be credited with the amount of the Member’s Excess Regular Earnings Deferrals, Employer Accruals, Special Accruals and
will be adjusted for earnings and losses thereon.
5.2 Payment Option Election . Except with respect to a Special Accrual and to the extent otherwise provided: (a) in a Written
Agreement or (b) in this Section 5.2, at the time a Member is first eligible to elect to make Excess Regular Earnings Deferrals under the
Plan, the Member shall elect the particular Payment Option that is to apply to amounts credited to the Member’s Account (other than
Grandfathered Amounts). For a Payment Option election to be effective, it must be made (i) within 30 days of the date the Employee is first
eligible to participate in the Plan (as determined in accordance with consistent rules established by the Committee in its sole and absolute
discretion and in
-
10
-
SECTION 5
.
INDIVIDUAL ACCOUNT
.

accordance with Section 409A). Notwithstanding the foregoing: (1) any Member who has a Special Accrual under the Plan who has a right
to elect a form of payment pursuant to the Written Agreement and as approved by an ELT member and the Committee shall only have 30
days from the date of initial eligibility (whether that date is with respect to the Special Accrual or with respect to the Excess Regular
Earnings Deferrals, whichever is earlier) to elect his or her Payment Option for all non-Grandfathered amounts which he or she has the right
to elect a Payment Option for under the Plan; and (2) any Eligible Employee who becomes eligible to participate in an account balance plan
that must be aggregated with the Plan under Section 409A of the Code before he or she otherwise would become eligible to participate in
this Plan has 30 days from the date he or she first becomes eligible to participate in such other plan to elect his or her Payment Option under
the Plan. In the absence of a timely election (except as otherwise may be required pursuant to a Written Agreement), the Member shall be
deemed to have made a Payment Option to receive his or her Account under the Plan in a single lump sum payment in the January after his
or her Separation from Service (other than with respect to Grandfathered Amounts). In addition, unless otherwise provided pursuant to a
Written Agreement (and as approved by an ELT member and the Committee) a Member shall be deemed to have made a Payment Option
election to receive the portion of his or her Account attributable to his or her Special Accrual under the Plan in a single lump sum payment
in the January after his or her Separation from Service. Except as provided in Section 5.3 below, or if payment is subsequently re-
deferred in
accordance with Section 6.8 and subject to the rules on Key Employee payments as provided in Section 6.5, any Payment Option election
made or deemed made under the Plan shall apply with respect to a Member’s entire Account under the Plan (except with respect to
Grandfathered Amounts).
5.3 Exceptions to Binding Payment Option Election . Notwithstanding any Payment Option elected (or deemed elected): (i) if the
value of a Member’s account on the last business day of the calendar year of the Member’s Separation from Service (excluding
Grandfathered Amounts credited to the Member’s Account) is $10,000 or less the Member’s Payment Option election shall be paid in a
lump sum in the January following the Member’s termination; (ii) if a Member incurs a Disability, the Member’s Account (except with
respect to Grandfathered Amounts which shall be payable under the terms of that Prior Plan) shall be paid in a lump sum the January after
the Member has been determined to have incurred a Disability; (iii) if a Member who was a Participant in the Pharmacia Savings Plus Plan
requests a distribution on account of an Unforeseeable Emergency the portion of the Member’s Account attributable to amounts accrued
under the Pharmacia Savings Plus Plan (as adjusted for earnings and losses and other than Grandfathered amounts, which shall be paid in
accordance with the terms of that Prior Plan) to the extent requested on account of the Unforeseeable Emergency shall be paid in a lump
sum the next business day after the Unforeseeable Emergency (in accordance with uniform rules established by the Committee and in
accordance with 409A); (iv) if a Member ceases to be an Eligible Employee and subsequently becomes eligible to participate in the Plan, he
or she may elect a new Payment Option that shall apply with respect to any amounts credited to his or her Accounts under the Plan after the
date of his or her re-eligibility (provided the Employee becomes an Eligible Employee in a different
-
11
-

calendar year than the year in which he or she ceased to be an Eligible Employee), and if no such election the portion of the Account
accrued with respect to the new eligibility period shall be paid in a single lump sum in the January after Separation from Service (or as
otherwise required in this Section 5.3); (v) if a Member has a Special Accrual under the Plan any Payment Option election made or deemed
made by the Member in accordance with Section 5.2, the exceptions to the Payment Option elected as provided for in this Section 5.3 (or
Section 6.8) shall not apply to amounts attributable to the Special Accrual unless otherwise provided in the terms of the Written Agreement
as approved by an ELT member and the Committee; and (vi) if a Member dies before distribution of his or her entire Account, the Payment
Option election shall be cancelled and the entire account (except with respect to Grandfathered Amounts under the Pharmacia Savings Plus
Plan which shall be paid pursuant to the terms of that plan) shall be paid in a single lump sum distribution the January following the
Member’s death. Effective January 1, 2007, the immediately preceding sentence shall also apply with respect to amounts accrued under the
Pharmacia Savings Plus Plan (as adjusted for earnings and losses) other than Grandfathered Amounts under that plan.
5.4 Investments . All Excess Regular Earnings Deferrals will be credited with an amount equal to the amount which would have
been earned had such amounts been actually invested in one or more of the “Funds” (other than the Pfizer Match Fund available for
investment under the Qualified Plan, as the Member may be defaulted into or elect from time to time, in one percent (1%) increments. To
the extent no investment election is provided with respect to a Special Accrual when such Special Accrual is credited to the Plan or
otherwise, the Special Accrual shall be deemed to be invested in the default fund under the Plan. The portion of the Member’s Account
attributable to Employer Accruals shall be deemed to be invested in the Pfizer Match Fund. Rules similar to those which govern the
Qualified Plan shall apply for purposes of determining the value of the deemed investments (but based on this Plan’s valuation dates) and
the timing, frequency and permissibility of investment transfers. No provision of this Plan shall require the Company or any other Employer
to actually invest any amount in any “Fund” or in any other investment vehicle. The Plan is an unfunded plan that is not subject to the
funding requirements of ERISA, meaning that there are no actual investments held in a trust. The Accounts represent unsecured obligations
of the Company, and no funds are set aside from the Company’s general assets to cover such Accounts. The Plan is subject to the full faith
and credit of the Company, and Members would be general creditors in the event of the Company’s insolvency. Except as otherwise
provided in this Section, distributions and withdrawals from the Plan are valued as of the last business day of the calendar quarter preceding
the calendar quarter of the distribution. Withdrawals on account of an Unforeseeable Emergency are valued as of the last business day of the
month preceding the day that the withdrawal request is received. Payments that would otherwise be made but are delayed on account of a
Member being a Key Employee are valued on the distribution date.
With respect to a Member subject to Section 16 of the Securities Exchange Act of 1934, an election to transfer a portion of his
Account into, or out of, the Pfizer company
-
12
-

stock funds, shall be permitted only if the Member has not elected during the immediately preceding six (6) months to transfer out of, or
into, such funds within this Plan, any Pfizer company stock funds under the Qualified Plan or the unit account within the Pfizer Inc
Nonfunded Deferred Compensation and Unit Award Plan for Non
-Employee Directors, the Pfizer Inc Retainer Units Award Plan for Non-
Employee Directors, or the Pfizer Deferred Compensation Plan.
6.1 Distribution of Benefits . Unless otherwise specifically provided for in the Plan, distribution of a Member’s Grandfathered
Amounts shall be paid in accordance with the distribution provisions of the Prior Plans. Except as otherwise provided in this Section and the
Plan, a Member shall be paid the balance of his Account following his or her Separation of Service in accordance with the Payment Option
or Payment Options elected (or deemed elected by the Member) by the Member as permitted under the Plan. A Member may have different
Payment Option elections with respect to the portions of his or her Account, for example, for a Special Accrual or for a Member who was
ineligible or a period of time and subsequently became eligible and was permitted or deemed to have made a new Payment Option election
under the Plan with respect to future accruals under the Plan and in accordance with Section 409A.
6.2. Benefits Subject to Withholding . The benefits payable under this Plan shall be subject to the deduction of any federal, state,
or local income taxes, employment taxes or other taxes which are required to be withheld from such payments by applicable laws and
regulations. Any employment taxes owed by the Member with respect to any deferral, accrual or benefit payable under this Plan may be
withheld from other compensation of the Member in the year in which such tax liability accrues.
6.3 Disability . Notwithstanding any elected Payment Option or deemed elected Payment Option (made or deemed made), if the
Member incurs a Disability under the Plan, the balance of the Member’s Account shall be paid in a single lump sum the January following
the Disability, except with respect to Grandfathered Amounts shall be payable in accordance with the terms of that Prior Plan.
6.4 Distributions on Account of Unforeseeable Emergency . Notwithstanding any elected Payment Option or deemed elected
Payment Option made (or deemed made) by a Member who was a Participant in the Pharmacia Savings Plus Plan, upon the occurrence of an
Unforeseeable Emergency, a Member may withdraw all or any portion of his or her Account balance attributable to amounts accrued under
the Pharmacia Savings Plus Plan (as adjusted for earnings and losses) provided that the amounts distributed with respect to an Unforeseeable
Emergency (including any Grandfathered Amounts distributed under the rules of the Pharmacia Savings Plus Plan) may not exceed the
amounts necessary to satisfy such Unforeseeable Emergency plus amounts necessary to pay taxes reasonably anticipated as a result of the
distribution, after taking into account the extent to which such hardship is or may be relieved through reimbursement or compensation by
insurance or
-
13
-
SECTION 6
.
DISTRIBUTION OF ACCOUNTS
.

otherwise or by liquidation of the Member’s assets (to the extent the liquidation of such assets would not itself cause severe financial
hardship) or by cessation of deferrals under the Plan. Distributions of Grandfathered Amounts for a hardship or unforeseeable emergency
shall be governed by the terms of the Prior Plan.
6.5 Delay for Key Employees . Notwithstanding anything in the Plan to the contrary, distributions (other than distributions of
Grandfathered Amounts) may not be made to a Key Employee upon a Separation from Service before the date which is six (6) months after
the date of the Key Employee’s Separation from Service (or, if earlier, the date of death of the Key Employee). Any payments that would
otherwise be made during this period of delay shall be accumulated and paid on the day that is six (6) months following the Member’s
Separation from Service (or, if earlier, the January following the Member’s death).
6.6 Distributions upon Death
. Notwithstanding any elected Payment Option or deemed elected Payment Option made (or deemed
made) by the Member, if a Member dies before distribution of his or her Account balance has begun any remaining balance shall be
distributed in a lump sum payment to the Member’s Beneficiary the January following the calendar year in which the Member’s death
occurs. Effective January 1, 2007, the immediately preceding sentence also applies with respect to amounts accrued under the Pharmacia
Savings Plus Plan (as adjusted for earnings and losses) other than Grandfathered Amounts under that plan.
6.7 Change in Control . Notwithstanding any provision in the Plan or the Pharmacia Savings Plus Plan to the contrary, a
Member’s Payment Option election or a Member’s deemed Payment Option election, for Member’s who were Participants in the Pharmacia
Savings Plus Plan and with respect to amounts accrued under the Pharmacia Savings Plus Plan on or after January 1, 2005 (as adjusted for
earnings and losses) only, such portion of the Member’s Account under the Plan shall be distributed in an immediate lump sum payment
upon the occurrence of a Change in Control that is a “Change in Control Event.” For these amounts a “Change in Control Event” means an
event described in Code section 409A(a)(2)(A)(v) or otherwise under Section 409A. With respect to Grandfathered amounts under the
Pharmacia Savings Plus Plan, Change in Control shall have the meaning defined in that Prior Plan and such Grandfathered Amounts
distributed in accordance with the terms of that Prior Plan.
6.8 Redeferrals . Notwithstanding any elected Payment Option or deemed elected Payment Option made (or deemed made),
except with respect to Special Accruals for which the Member was not provided with a redeferral option under the Written Agreement, a
Member may make one or more subsequent elections to change form of a distribution for a deferred amount, provided that such an election
shall be effective only if the following conditions are satisfied:
(a) The election may not take effect for at least twelve (12) months;
-
14
-

(b) The election must be made at least twelve (12) months before payments would have otherwise begun; and
(c) In the case of an election to change the form of a distribution upon a Member’s Separation from Service, a
distribution may not be made earlier than at least five (5)
years from the date the distribution (or, with respect to installments, the first
scheduled installment) would have otherwise been made.
Members who have elected to receive their distribution (or portion thereof) in installments may not change the corresponding election once
their installment distributions have begun.
Members who pursuant to a Written Agreement are permitted to specify a Payment Option with respect to a Special Accrual shall also have
the redeferral rights provided in this Section 6.8 except as otherwise provided in the Written Agreement.
6.9 Effect of Taxation . If a portion of the Member’s Account balance is includible in income under Section 409A, such portion
shall be distributed immediately to the Member.
6.10 Permitted Delays
. Notwithstanding the foregoing, any payment on account of a Member under the Plan shall be delayed upon
the Committee’s reasonable anticipation of one or more of the following events:
(a) The Company’s deduction with respect to such payment would be eliminated by application of Code section 162(m);
or
(b) The making of the payment would violate federal securities laws or other applicable law;
provided, that any payment delayed pursuant to this Section 6.10 shall be paid in accordance with Section 409A.
Participation in this Plan will not create, in favor of any Member, any rights or lien in or against any of the assets of the Company or
any Employer, and all amounts of Excess Regular Earnings, Special Accruals and Employer Accruals deferred hereunder shall at all times
remain an unrestricted asset of the Company or the Employer. A Member’s rights to benefits payable under the Plan are not subject in any
manner to anticipation, alienation, sale, transfer, assignment, pledge, or encumbrance. Nothing contained in this Plan, and no action taken
pursuant to its provisions, shall create or be construed to create a trust of any
-
15
-
SECTION 7
.
NATURE OF INTEREST OF MEMBER
.

kind, or a fiduciary relationship, between any Employer and a Member or any person, and the Company’s and each Employer’s promise to
pay benefits hereunder shall at all times remain unfunded as to the Member.
A Member’s beneficiary under this Plan will automatically be the same as such Member’s beneficiary under the Qualified Plan
unless a separate designation of beneficiary form for this Plan has been properly filed with the Committee or its authorized designee in
accordance with any rule established by the Committee and received prior to the death of the Member. In the absence of a designation of
specific beneficiary under either the Qualified Plan or this Plan, which beneficiary survives the Member, upon the Member’s death, except
to the extent as may otherwise be provided with respect to amounts attributable to accruals made under the Pharmacia Savings Plus Plan
prior to January 1, 2007 (including Grandfathered Amounts) which shall be governed by the terms of that Plan, payment of his Account
shall be made to the Member’s estate in a lump sum in the January following the Member’s death.
9.1 Committee . This Plan will be administered by the Committee.
9.2 Powers of the Committee . The Committee’s powers under this Plan are the same as are described in the Qualified Plan and
include, but are not limited to, the power:
(a) to determine who are Eligible Employees for purposes of participation in the Plan;
(b) to interpret the terms and provisions of the Plan and the Prior Plans and to determine any and all questions arising
under the Plan or Prior Plans, including without limitation, the right to remedy possible ambiguities, inconsistencies, or omissions by
a general rule or particular decision; and
(c) to adopt rules consistent with the Plan or Prior Plans.
9.3 Claims Procedure . Effective January 1, 2008, this Section 9.3 and Section 9.4 shall apply with respect to a Member’s entire
Account under the Plan including Grandfathered Amounts under this Plan and the Pharmacia Savings Plus Plan. Any request by a Member
or any other person for any benefit alleged to be due under the Plan shall be known as a “Claim” and the Member or other person making a
Claim, or the authorized representative of either, shall be known as a “Claimant.” The Committee has sole discretion to determine whether a
communication from an individual shall be a Claim for purposes of this Section 9.3 and Section 9.4. To the extent of their responsibility to
review benefit claims or to review the denial of benefit claims, the Committee and the reviewer shall have full authority to interpret and
apply, in their discretion, the provisions of the Plan. The decisions of the Committee and reviewer shall be final and binding upon any
-
16
-
SECTION 8
.
BENEFICIARY DESIGNATION
.
SECTION 9
.
ADMINISTRATION
.

and all Claimants, including, but not limited to, Members and their Beneficiaries, and any other individuals making a Claim or requesting
review of a Claim through or under them, and shall be afforded the maximum deference permitted by law. A Member may not maintain a
court action over a disputed claim until he or she has exhausted the Plan’s claims procedures.
Claimant may submit a written application to the Committee for payment of any benefit that he believes may be due him under the
Plan, in accordance with Plan procedures. Such application shall include a general description of the benefit which the Claimant believes is
due, the reasons the Claimant believes such benefit is due and any information as the Committee may reasonably request. The Committee
will process the Claimant’s application within ninety (90) days of the receipt of the Claim by the Committee unless special circumstances
require an extension of time for processing the Claim. In such event, written notice of the extension shall be furnished to the Claimant prior
to the termination of the initial ninety (90) day period but in no event shall the extension exceed a period of ninety (90) days from the end of
such initial period. The notice shall indicate the special circumstances requiring an extension of time and the date by which the Plan expects
to render the final decision. If the Committee has not determined the Claimant’s eligibility for a Plan benefit within this ninety (90) day
period (one hundred eighty (180) day period if circumstances require an extension of time), the Claim is deemed denied. A Claim is
considered approved only if such approval is memorialized by the Committee in writing.
If a Claim is denied in whole or in part, the notice of denial shall set forth (i) the specific reason or reasons for the denial, (ii) specific
reference to the pertinent Plan provisions on which the denial is based, (iii) a description of any additional material or information necessary
for the Claimant to perfect the Claim and an explanation of why such material or information is necessary, (iv) an explanation of the Plan’s
claim review procedure, and (v) an explanation that, if an adverse determination is made on review, the Claimant may have a right to bring
civil action under Section 502(a) of ERISA. Within sixty (60) days of the receipt of a notice of denial of a Claim in whole or in part or a
deemed denial, a Claimant (i) may request a review upon written application to the Committee, (ii) may review documents pertinent to the
Claim, and (iii) may submit issues and comments in writing to the Committee. The Claimant shall be provided upon request and free of
charge, reasonable access to all documents, records and other information relevant to the Claimant’s Claim for benefits.
-
17
-

The Committee will review a Claim for which a request for review has been made and render a decision not later than
sixty (60) days after receipt of a request for review; provided, however, that if special circumstances require extension of a time for
processing, a decision shall be rendered no later than one hundred and twenty (120) days after receipt of the request for review. Written
notice of any such extension shall be furnished to the Claimant within sixty (60) days after receipt of request for review. The Committee’s
decision shall be in writing and shall set forth (i) the specific reason or reasons for the denial on review, (ii) specific reference to the
pertinent Plan provisions on which the denial on review is based, (iii) an explanation that the Claimant is entitled to receive, upon request
and free of charge, reasonable access to, and copies of, all documents, records, and other information relevant to the Claimant’s Claim for
benefits, and (iv) an explanation that if an adverse determination is made on review, the Claimant may have the right to bring a civil action
under Section 502(a) of ERISA. If the decision on review is not furnished within the applicable time, the Claim shall be deemed denied on
review.
9.4 Limitation on Period for Filing Claims . No claim for benefits based upon a claim that contributions were not properly made
under this Plan shall be approved under this Plan, and no action may be brought for benefits under this Plan pursuant to the denial of such a
claim pursuant to Section 9.3 of this Plan, unless such claim for benefits is duly filed under Section 9.3 of this Plan no later than the last day
of the second Plan Year beginning after the Plan Year in which the claim alleges that the contributions should have been credited.
No provisions of the Plan or any action taken by the Company, the Board of Directors, the Committee, or any of their properly
authorized representatives shall give any person any right to be retained in the employ of any Employer, and the right and power of the
Company or any Employer to dismiss or discharge any Member is specifically reserved.
The Board of Directors or its authorized designee shall have the rights to amend, suspend, or terminate the Plan at any time, except
that the Committee may make non-substantive administrative changes to this Plan so as to conform with or take advantage of governmental
requirements, statutes or regulations. Except as provided in the next sentence, no amendment, modification or termination shall, without the
consent of a Member, adversely affect the amount of the Member’s benefits in his or her Account as of the date of such amendment,
modification or termination. Upon termination of the Plan, distribution of the balances in Accounts shall be made to Members and
Beneficiaries in the manner and at the time described in the Plan (or the Prior Plan) unless the Board of Directors of the Company or its
designee determines in its sole and absolute discretion that all such amounts shall be distributed upon termination and in accordance with the
requirements under Section 409A. Upon termination of the Plan, no further deferrals of
-
18
-
SECTION 10
.
NO EMPLOYMENT RIGHTS
.
SECTION 11
.
AMENDMENT, SUSPENSION, AND TERMINATION
.

eligible compensation shall be permitted; however, earnings, gains and losses shall continue to be credited to Account balances in
accordance with the Plan until the Account balances are fully distributed.
In the event the Plan is terminated, the Committee shall continue to administer the Plan in accordance with the relevant provisions
thereof until the Member’s benefits have been paid hereunder. Notwithstanding the foregoing, no amendment of the Plan shall apply to
Grandfathered Amounts, unless the amendment specifically provides that it applies to such amounts. The purpose of this restriction is to
prevent a Plan amendment from resulting in an inadvertent “material modification” to Grandfathered Amounts.
Notwithstanding anything herein to the contrary, the terms of the Plan are intended to, and shall be interpreted and applied so as to,
comply in all respects with the provisions of Section 409A. Any provision of this Plan governing the timing or form of payment of benefits
hereunder may be modified by the Plan Administrator if, and to the extent deemed necessary or advisable, to comply with Section 409A
including, but not limited to, a 6-
month delay in payment to a Key Employee, which shall be paid as provided in Section 6.5. Nothing in this
Section shall be construed as an admission that any of the benefits payable under this Plan (or any predecessor Plan) constitutes “deferred
compensation” subject to the provisions of Section 409A.
-
19
-
SECTION 12
.
PROVISIONS GOVERNED BY CODE SECTION 409A
.

Appendix A –Pfizer Supplemental Savings Plan as in effect on 12/31/04. Except as otherwise specifically provided for in the Plan, the
provisions of this Plan shall apply to Grandfathered Amounts covered under the Pfizer Supplemental Savings Plan on 12/31/2004.
PFIZER INC
NONFUNDED DEFERRED COMPENSATION AND
SUPPLEMENTAL SAVINGS PLAN
1

1.1 Continuation
. There is hereby continued for the benefit of Members an unfunded plan of deferred compensation known as the
“Pfizer Inc Nonfunded Deferred Compensation and Supplemental Savings Plan.”
1.2 Purpose . The purpose of this Plan is to provide a means by which an Eligible Employee may, in certain circumstances, elect
to defer receipt of a portion of his “Regular Earnings.” The Plan also provides that the Company will, in certain instances, credit the
Account of a Member with Employer Accruals.
1.3 Description of the Plan . The Plan became effective July 1, 1983 and is amended and restated effective February 1, 2002,
except as otherwise provided herein. For purposes of the Employee Retirement Income Security Act of 1974 (“ERISA”), as amended, the
Plan shall be treated as two separate, unfunded plans. One plan shall be an “excess benefit plan” within the meaning of Section 3(36) of
ERISA, and shall be comprised of accruals under the Plan that are made solely because of the applicable limitations under Section 415 of
the Code, plus earnings thereon. All other accruals under the Plan, plus earnings thereon, shall be treated as made under a separate “top-hat”
plan maintained by the Company primarily for the purpose of providing deferred compensation to a select group of management or highly
compensated employees, within the meanings of Sections 201(a)(2) and 401(a)(1) of ERISA. Separate accounts shall be maintained under
the Plan for each Member, as applicable, to account for excess benefit plan accruals and earnings, and top-hat plan accruals and earnings.
The following words and phrases as used in this Plan have the following means:
2.1 Account . The term “Account” shall mean a Member’s individual account(s), as described in Section 5 of the Plan.
2.2 Board of Directors . The term “Board of Directors” means the Board of Directors of the Company.
2.3 Code . The term “Code” means the Internal Revenue Service Code of 1986, as amended.
2.4 Committee . The term “Committee” means the Committee, as described in the Qualified Plan, or any other person or entity
that the Committee has authorized to act on its behalf under the Plan.
2.5 Company . The term “Company” means Pfizer Inc, a Delaware corporation, and any successor corporation.
2.6 Controlled Group . The term “Controlled Group” means the Company and any other entity in which the Company owns
directly or indirectly 30 percent or more of the value or voting power.
2
SECTION 1
.
CONTINUATION AND PURPOSE OF THE PLAN
.
SECTION 2
.
DEFINITIONS
.

2.7 Eligible Employee . The term “Eligible Employee” means any “Member” under the Qualified Plan (i) who receives Regular
Earnings for any Plan Year in excess of the limitation of Section 401(a)(17) of the Code, (ii) whose account under the Qualified Plan is
credited with “annual additions,” as defined in Section 415(c)(2) of the Code, during any Plan Year equal to the maximum permitted under
Section 415(c)(1)(A) of the Code, (iii) who is otherwise credited with Employer Accruals, or (iv) who is a member of a select group of
management or highly compensated employees and is designated as an Eligible Employee by the Committee.
2.8 Employer . The term “Employer” means the Company and any other member of the Controlled Group which is also an
“Associate Company” under the Qualified Plan.
2.9 Employer Accrual . The term “Employer Accrual” means the amounts described in Section 4.
2.10 Excess Regular Earnings . The term “Excess Regular Earnings” means (i) the portion of a Member’
s Regular Earnings earned
during a Plan Year that exceeds the Limitation on compensation taken into account under Section 401(a)(17) of the Code, (ii) all Regular
Earnings earned after the Member becomes subject to the Limitation on contributions to defined contribution plans under Section 415(c)(1)
(A) of the Code, to the extent not included in (i) above, (iii) a bonus deferred under the Pfizer Inc Deferred Compensation Plan, or (iv) any
other compensation determined by the Committee to be compensation for purposes of this Plan .
2.11 Excess Regular Earnings Deferrals . The term “Excess Regular Earnings Deferrals” means the portion of a Member’s Excess
Regular Earnings that the Member elects to defer under the terms of the Plan.
2.12 Limitation(s) . The term “Limitation(s)” means the limitation on contributions to defined contribution plans under Section 415
(c)(1)(A) of the Code, and on compensation taken into account under Section 401(a)(17) of the Code.
2.13 Member . The term Member means an Eligible Employee who elects to have Excess Regular Earnings Deferrals made to the
Plan or is otherwise credited with an Employer Accrual.
2.14 Payment Options . The term “Payment Option” means the following forms of payment under which a Member may elect to
receive amounts credited to his Account upon his termination of employment with the Controlled Group: (i) single sum payable as soon as
practicable following the end of the Plan Year in which the Member terminates employment with the Controlled Group, or (ii) substantially
equal annual installment payments over a period of two to twenty years commencing as soon as practicable following the end of the Plan
Year in which the Member terminates employment with the Controlled Group. Where payment of the Account is made in installment
payments, the first installment shall be a fraction of the value of the Member’s Account as of the applicable valuation date, the numerator of
which is one (1) and the denominator of which is the total number of installments remaining to be paid at that time. Each subsequent
installment shall be calculated in the same manner, except that the denominator shall be reduced by the number of installments that have
been paid previously.
3

2.15 Plan . The term “Plan” means the “Pfizer Inc Nonfunded Deferred Compensation and Supplemental Savings Plan,” as set
forth herein and as amended from time to time.
2.16 Plan Year . The term “Plan Year” means the calendar year.
2.17 Qualified Plan . The term “Qualified Plan” means the Pfizer Savings Plan, as amended from time to time.
2.18 Regular Earnings . The term “Regular Earnings” shall have the meaning given such term under the Qualified Plan.
3.1 Designation of Eligible Employees . The Committee in its sole and absolute discretion will designate as Eligible Employees
those employees who satisfy the terms of Section 2.7 and are eligible to participate in the Plan. The Committee in its sole and absolute
discretion may terminate the designation of an employee as an Eligible Employee at any time.
3.2 Election to Make Excess Regular Earnings Deferrals . An Eligible Employee may elect at any time after becoming eligible to
begin making Excess Regular Earnings Deferrals by filing an election with the Committee or its authorized designee in accordance with this
Section 3 and any rules established by the Committee. Such election will be effective on a prospective basis beginning with the payroll
period that occurs as soon as administratively practicable following receipt of the election by the Committee or its authorized designee.
3.3 Amendment or Suspension of Election . Members may change (including, suspend) their existing Excess Regular Earnings
Deferrals election under this Plan during the Plan Year by filing a new election in accordance with the prescribed administrative guidelines.
Such new election will be effective on a prospective basis beginning with the payroll period that occurs as soon as administratively
practicable following receipt of the election by the Committee or its authorized designee. A Member shall not be permitted to make up
suspended Excess Regular Earnings Deferrals, and during any period in which a Member’s Excess Regular Earnings Deferrals are
suspended, the Employer Accruals under the Plan with respect to Excess Regular Earnings Deferrals shall also be suspended. A Member
who receives a hardship withdrawal under the Qualified Plan shall be suspended from making Excess Regular Earnings Deferrals hereunder
for a period of six (6) months from the date of such withdrawal.
3.4 Amount of Elections . Each election filed by an Eligible Employee must specify the amount of Excess Regular Earnings
Deferrals in a whole percentage from 1% to 20% of the Member’s Excess Regular Earnings unless the Committee establishes a lesser
percentage for the Plan Year; provided, however, that, with respect to an Eligible Employee who is also eligible to participate in the
Qualified Plan, the rate of Excess Regular Earnings Deferrals hereunder for any payroll period shall not exceed the rate at which the
Member was contributing to the Qualified Plan on a combined pre-tax and post-tax basis for the current year.
4
SECTION 3
.
PARTICIPATION
.

4.1 General Rule .
An Employer Accrual will be credited to a Member’s Account with respect to the eligible portion of Excess Regular Earnings
Deferrals of such Member at the applicable rate of “Matching Contributions” with respect to “After-Tax Contributions” and “Before-Tax
Contributions” under the Qualified Plan. The Employer Accrual shall be credited as soon as practicable following the payroll period for
which the Excess Regular Earnings Deferrals are made. The eligible portion of a Member’s Excess Regular Earnings Deferrals shall be
limited to six percent (6%) of such Excess Regular Earnings for each payroll period. In addition, an Employer Accrual will be credited as of
the end of each Plan Year to a Member’s Account equal to the difference between (i) the amount that would have been credited to the
Member’s account under the Qualified Plan as a “Matching Contribution,”
including Additional Contribution, if any, if the Limitations were
not applicable to the Member under the Qualified Plan during such Plan Year and (ii) the “Matching Contributions,” including Additional
Contributions, if any, actually credited to the Member’s account under the Qualified Plan during such Plan Year. Lastly, an Employer
Accrual will be credited to the Account of a Member who elects to defer his bonus under the Warner-Lambert Company Incentive
Compensation Plan (“ICP”). The amount of such Employer Accrual will be equal to the product of: (i) six percent (6%) of the bonus
deferred under the ICP, and (ii) the applicable rate of “Matching Contributions” with respect to “After-Tax Contributions” and “Before-Tax
Contributions” under the Qualified Plan. The Employer Accrual shall be credited as soon as practical following the payroll period in which
the bonus is deferred.
4.2 Special Employer Accrual for Certain Former Warner-Lambert Employees .
In the case of any Eligible Employee who (i) was an “Eligible Participant” under the Warner-Lambert Savings and Stock Plan as in
effect on January 31, 2002 (the “Warner-Lambert Plan”), (ii) is a participant under the Warner-Lambert Enhanced Severance Plan on
May 15, 2003, (iii) has completed at least three years of Plan membership (including Warner-Lambert Plan membership) under the
Qualified Plan as of May 15, 2003, and (iv) was an Eligible Employee on May 15, 2003, an Employer Accrual shall be credited to such
Eligible Employee’s Account in an amount equal to the difference between (a) and (b) below:
(a) the “Matching Contribution” which would have been made to the Eligible Employee’s account under the Qualified
Plan with respect to the period June 1, 2002 through May 15, 2003 if such “Matching Contribution”
had been based on the terms of Article 5
of the Warner-Lambert Plan, assuming an additional matching contribution rate of 65%; and
(b) the “Matching Contribution” actually made to the Eligible Employee’s account under the Qualified Plan with respect
to the period June 1, 2002 through May 15, 2003.
5
SECTION 4
.
EMPLOYER ACCRUALS
.

This Employer Accrual shall be credited as soon as practicable following the payroll period which includes May 15, 2003.
4.3 Special Employer Accrual for Certain Former Agouron Employees . In the case of any Eligible Employee who (i) was an
“Eligible Employee” under the Agouron Pharmaceuticals, Inc. 401(k) Plan as in effect on January 31, 2002 (the “Agouron Plan”), (ii) was a
participant under the Warner-Lambert Enhanced Severance Plan on May 15, 2003, and (iii) was an Eligible Employee on May 15, 2003, an
Employer Accrual shall be credited to such Eligible Employee’s Account in an amount equal to the difference between (a) and (b) below:
(a) the “Matching Contribution” which would have been made to the Eligible Employee’s account under the Qualified
Plan with respect to the period June 1, 2002 through May 15, 2003 if such “Matching Contribution” had been based on the terms of Article
6.4 of the Agouron Plan, assuming an additional matching contribution rate of 35%; and
(b) the “Matching Contribution” actually made to the Eligible Employee’s Account under the Qualified Plan with respect
to the period June 1, 2002 through May 15, 2003.
This Employer Accrual shall be credited as soon as practicable following the payroll period which includes May 15, 2003.
5.1 Creation of Accounts . The Company will maintain an Account under the Plan in the name of each Member. Each Member’s
Account will be credited with the amount of the Member’s Excess Regular Earnings Deferrals, Employer Accruals, and will be adjusted for
earnings and losses thereon. In the case of Members covered under both the “excess benefit” and “top-hat” portions of the Plan, separate
accounts will be maintained to reflect the Members’ interest in each such portion of the Plan.
5.2 Payment Account Option Election . Each Member shall elect the particular Payment Option that is to apply to amounts
credited to the Member’s Account. In order for a Payment Option election to be effective, it must be made (i) no later than ninety (90) days
(one hundred and eighty (180) days for employment terminations on or after January 1, 2003) prior to the date the Member terminates
employment with the Controlled Group, and (ii) in a taxable year preceding the taxable year in which payment would otherwise be made or
commence. In the absence of a timely election, payment of the Member’
s Account will be made in accordance with the most recent Payment
Option election which satisfies the requirements of the immediately preceding sentence or, in the absence of any such Payment Option
election, in five (5) substantially equal annual installments commencing as soon as practicable following the end of the Plan Year in which
the Member terminates employment with the Controlled Group. The foregoing notwithstanding, in any case where the value of the
Member’s Plan Account is less than ten percent (10%) of the value of the Member’s interest in both the Plan and the Qualified Plan,
payment of the Member’s Account shall be made in a lump sum as soon as practicable following the end of the Plan Year in which the
Member terminates
6
SECTION 5
.
INDIVIDUAL ACCOUNT
.

employment with the Controlled Group. Upon a Member becoming “Disabled,” as determined under the Qualified Plan, the balance of the
Member’s Account shall be paid in a lump sum as soon as practicable after such determination is made.
5.3 Investments . All Excess Regular Earnings Deferrals will be credited with an amount equal to the amount which would have
been earned had such amounts been actually invested in one or more of the “Funds” (other than the Pfizer Match Fund) available for
investment under the Qualified Plan, as the Member may elect from time to time, in one percent (1%) increments. The portion of the
Member’
s Account attributable to Employer Accruals shall be deemed to be invested in the Pfizer Match Fund. Rules similar to those which
govern the Qualified Plan shall apply for purposes of determining the value of the deemed investments and the timing, frequency and
permissibility of investment transfers, except that no diversification of Employer Accruals which are deemed to be invested in the Pfizer
Match Fund shall be permitted. No provision of this Plan shall require the Company or any other Employer to actually invest any amount in
any “Fund” or in any other investment vehicle. The Plan is an unfunded plan that is not subject to the funding requirements of ERISA,
meaning that there are no actual investments held in a trust. The Accounts represent unsecured obligations of the Company, and no funds are
set aside from the Company’s general assets to cover such Accounts. The Plan is subject to the full faith and credit of the Company, and
Members would be general creditors in the event of the Company’s insolvency.
With respect to a Member subject to Section 16 of the Securities Exchange Act of 1934, an election to transfer a portion of his
Account into, or out of, the “Pfizer Company Stock Fund” shall be permitted only if the Member has not elected during the immediately
preceding six months to transfer out of, or into, such Fund within this Plan, the Pfizer Company Stock Fund under the Qualified Plan or the
unit account within the Pfizer Inc Nonfunded Deferred Compensation and Unit Award Plan for Non-Employee Directors, the Pfizer Inc
Retainer Units Award Plan for Non-Employee Directors, or the Pfizer Inc Deferred Compensation Plan.
6.1 Payment of Benefits
. A Member shall be paid the balance of his Account following termination of employment in accordance
with the Payment Option elected by the Member. Upon the death of a Member, the Member’s beneficiary shall be paid the balance of the
Member’s Account in a lump sum as soon as practicable after the death of the Member.
6.2. Benefits Subject to Withholding . The benefits payable under this Plan shall be subject to the deduction of any federal, state,
or local income taxes, employment taxes or other taxes which are required to be withheld from such payments by applicable laws and
regulations. Any employment taxes owed by the Member with respect to any deferral, accrual or benefit payable under this Plan may be
withheld from other compensation of the Member in the year in which such tax liability accrues.
Participation in this Plan will not create, in favor of any Member, any rights in or lien against any of the assets of the Company or any
Employer, and all amounts of
7
SECTION 6
.
PAYMENT
.
SECTION 7
.
NATURE OF INTEREST OF MEMBER
.

Excess Regular Earnings deferred hereunder shall at all times remain an unrestricted asset of the Company or the Employer. A Member’s
rights to benefits payable under the Plan are not subject in any manner to anticipation, alienation, sale, transfer, assignment, pledge, or
encumbrance. Nothing contained in this Plan, and no action taken pursuant to its provisions, shall create or be construed to create a trust of
any kind, or a fiduciary relationship, between any Employer and a Member or any person, and the Company’s and each Employer’
s promise
to pay benefits hereunder shall at all times remain unfunded as to the Member.
A Member’s beneficiary under this Plan will automatically be the same as such Member’s beneficiary under the Qualified Plan
unless a separate designation of beneficiary form for this Plan has been properly filed with the Committee or its authorized designee in
accordance with any rule established by the Committee. In the absence of a designation of specific beneficiary under either the Qualified
Plan or this Plan, which beneficiary survives the Member, upon the Member’
s death, payment of his Account shall be made to his estate in a
lump sum as soon as practicable.
9.1 Committee . This Plan will be administered by the Committee.
9.2 Powers of the Committee . The Committee’s powers under this Plan are the same as are described in the Qualified Plan and
include, but are not limited to, the power:
9.3 Claims Procedure . The Committee shall make, in its sole discretion, all determinations arising in the administration,
construction or interpretation of the Plan including the right to construe disputed or doubtful Plan terms and provisions, and any such
determination shall be conclusive and binding on all persons, except as otherwise provided by law. Any claim by a Member or any other
person for any benefit alleged to be due under the Plan shall be made in writing to the Committee. Within 90 days of the filing of such
claim, unless special circumstances require an extension of such period, such person will be given notice in writing of the approval or denial
of the claims. If the claim is denied, the notice will set forth the reason for the denial, the Plan provisions on which the denial is based, an
explanation of what other material or information, if any, is needed to perfect the claim, and an explanation of the claims review procedure.
The claimant may request a review of such denial within 60 days of the date of receipt of such
8
SECTION 8
.
BENEFICIARY DESIGNATION
.
SECTION 9
.
ADMINISTRATION
.
(i)
to determine who are Eligible Employees for purposes of participation in the Plan;
(ii)
to interpret the terms and provisions of the Plan and to determine any and all questions arising under the Plan,
including without limitation, the right to remedy possible ambiguities, inconsistencies, or omissions by a general
rule or particular decision; and
(iii)
to adopt rules consistent with the Plan.

denial by filing notice in writing with the Committee. The claimant will have the right to review pertinent Plan documents and to submit
issues and comments in writing. The Committee will respond in writing to a request for review within 60 days of receiving it, unless special
circumstances require an extension of such period. The Committee, in its discretion, may request a meeting to clarify any matters deemed
appropriate.
9.4 Limitation on Period for Filing Claims . No claim for benefits based upon a claim that contributions were not properly made
under this Plan shall be approved under this Plan, and no action may be brought for benefits under this Plan pursuant to the denial of such a
claim pursuant to Section 9.3 of this Plan, unless such claim for benefits is duly filed under Section 9.3 of this Plan no later than the last day
of the second Plan Year beginning after the Plan Year in which the claim alleges that the contributions should have been credited.
No provisions of the Plan or any action taken by the Company, the Board of Directors, the Committee, or any of their properly
authorized representatives shall give any person any right to be retained in the employ of any Employer, and the right and power of the
Company or any Employer to dismiss or discharge any Member is specifically reserved.
The Board of Directors or its authorized designee shall have the rights to amend, suspend, or terminate the Plan at any time, except
that the Committee may make non-substantive administrative changes to this Plan so as to conform with or take advantage of governmental
requirements, statutes or regulations. No amendment, modification or termination shall, without the consent of a Member, adversely affect
the amount of the Member’s benefits in his or her Account as of the date of such amendment, modification or termination. Any
modification, amendment or termination may accelerate the time at which any Member is entitled to a distribution. In the event the Plan is
terminated, the Committee shall continue to administer the Plan in accordance with the relevant provisions thereof until the Member’s
benefits have been paid hereunder.
9
SECTION 10
.
NO EMPLOYMENT RIGHTS
.
SECTION 11
.
AMENDMENT, SUSPENSION, AND TERMINATION
.

Appendix B – Pharmacia Savings Plus Plan as in effect on 12/31/04. Except as otherwise specifically provided for in the Plan, the
provisions of this Plan shall apply to Grandfathered Amounts transferred from the Pharmacia Savings Plus Plan
Pharmacia
Savings Plus+Plan
Amended & Restated Effective as of July 1, 2002
10

PURPOSE
In recognition of the services provided by certain key employees, the Board of Directors of Pharmacia & Upjohn, Inc. (“P&U”)
adopted a deferred compensation plan (the “Plan”) to make additional retirement benefits and increased financial security, on a tax-deferred
basis, available to those individuals, effective July 1, 1999. Under the Plan, P&U provided a vehicle that will allow additional future
compensation to be paid to key employees so that such employees may be retained and their productive efforts encouraged.
Effective September 22, 1999, the Board of Directors of P&U amended and restated the Plan to permit a new Affiliate, Sugen, Inc.,
to join the Plan as a “Company” and permit its Eligible Employees, as defined below, to make an Incentive Deferral, as defined below, as
well as other forms of Compensation Deferrals, as defined below, when and if eligible.
On March 31, 2000, the Board of Directors of P&U amended and restated the Plan to reflect the transaction by which P&U became a
wholly-owned subsidiary of Pharmacia Corporation (“Pharmacia”). On December 7, 2000, the Board of Directors of Pharmacia amended
the Plan to (i) require deferral under this Plan of any incentive compensation earned under the Pharmacia Corporation Cash Long-Term
Incentive Plan and (ii) permit deferral under this Plan of benefits payable under the Pharmacia Corporation Key Executive Pension Plan or
payment under any other individual contractual pension arrangements for key executives at the Participant’s election. The Plan was
subsequently amended to reflect the Company’s adoption of the Pharmacia Corporation Long-Term Performance Share Unit Incentive Plan,
effective January 1, 2002.
Effective July 1, 2002, the Plan was amended to conform to Pharmacia’s Retirement Choice Program by including a “restoration”
arrangement and a “bonus deferral” arrangement. Also effective July 1, 2002, the Plan was amended to assume the obligations of the
Pharmacia Corporation ERISA Parity Savings and Investment Plan and to make certain other changes relating to a Change in Control of
Pharmacia. Accordingly, the Plan, as amended and restated as of the Amendment Effective Date, as defined below, now reads as follows:
DEFINITIONS
Account . “Account” means, with respect to a Participant, the account established on the books of the Company pursuant to
Section 5.1 and recording the benefit due to the Participant under the Plan.
Affiliate . “Affiliate” means any firm, partnership, or corporation that directly or indirectly through one or more intermediaries,
controls, is controlled by, or is under common control with Pharmacia. “Affiliate” also includes any other organization similarly related to
Pharmacia that is designated as such by the Board.

Base Salary . “Base Salary” means the sum of an Eligible Employee’s W-2 compensation paid by the Company to the Eligible
Employee including any pre-tax deferrals and benefits deducted from gross income including, but not limited to, any amounts that are
excluded from gross income under section 125, 402(e)(3), or 132(f) of the Code and any deferrals under this Plan. “Base Salary” shall
exclude any one-time or other special bonuses, moving and relocation expenses, stock options, or severance payments.
Base Salary Deferral . “Base Salary Deferral” means the portion of a Participant’
s Base Salary that the Participant has elected to defer
pursuant to Article 4.
Bonus . “Bonus” means any bonus or other incentive compensation awarded by the Company to the Eligible Employee under such
plans as specifically refer to this Plan and under such other plans as Pharmacia’s Senior Vice President Human Resources may from time to
time designate.
Bonus Deferral . “Bonus Deferral” means the portion of a Participant’s Bonus that the Participant has elected to defer pursuant to
Article 4.
Beneficiary . “Beneficiary” means the person or persons designated as such in accordance with Section 10.3.
Board . “Board” means the Board of Directors of Pharmacia.
Cause . “Cause”
means, if applicable to the Participant, the definition of that term used in the written employment agreement between
the Participant and the Company or an Affiliate as in effect on the date of the Participant’s termination of employment or in the Company’s
Change in Control Severance Benefit Plan. Otherwise, the term “Cause” shall mean (i) a material breach by the Participant of the
Participant’s duties and responsibilities (other than as a result of incapacity due to physical or mental illness) which is demonstrably willful
and deliberate on the part of the Participant, which is committed in bad faith or without reasonable belief that such breach is in the best
interests of the Company or an Affiliate, and which is not remedied within 30 days after receipt of written notice from the Company or an
Affiliate specifying such breach; or (ii) the Participant’s conviction of a felony which is materially and demonstrably injurious to the
Company or an Affiliate.
Change in Control . “Change in Control” means:
(1) the acquisition by any individual, entity, or group (a “Person”), including any “person” within the meaning of Section 13(d)(3)
or 14(d)(2) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), of beneficiary ownership within the meaning of Rule
13d-3 promulgated under the Exchange Act, of 33% or more of either (i) the then outstanding shares of Common Stock of Pharmacia (the
“Outstanding Company Common Stock”) or (ii) the combined voting power of the then outstanding securities of Pharmacia entitled to vote
generally in the election of directors (the “Outstanding Company Voting Securities”): provided, however, that the following acquisitions of
Outstanding Company Common Stock or Outstanding Company Voting Securities shall not constitute a Change in Control: (A) any
acquisition by Pharmacia, (B)
2

any acquisition by an employee benefit plan (or related trust) sponsored or maintained by Pharmacia or any corporation controlled by
Pharmacia, or (C) any acquisition by any corporation pursuant to a reorganization, merger, or consolidation involving Pharmacia, if,
immediately after such reorganization, merger, or consolidation, each of the conditions described in clauses (i), (ii), and (iii) of subsection
(3) of this Section shall be satisfied; and provided further that, for purposes of clause (A), if any Person (other than Pharmacia or any
employee benefit plan (or related trust) sponsored or maintained by Pharmacia or any corporation controlled by Pharmacia) shall become the
beneficial owner of 33% or more of the Outstanding Company Common Stock or 33% or more of the Outstanding Company Voting
Securities by reason of any acquisition of Outstanding Company Common Stock or Outstanding Company Voting Securities by Pharmacia
and such Person shall, after such acquisition by Pharmacia, becomes the beneficial owner of any additional shares of the Outstanding
Company Stock or any additional Outstanding Voting Securities and such beneficial ownership is publicly announced, such additional
beneficial ownership shall constitute Change in Control;
(2) individuals who, as of the date hereof, constitute the Board (the “Incumbent Board”) cease for any reason to constitute at least
a majority of such Board; provided, however, that any individual who becomes a director of Pharmacia subsequent to the date hereof whose
election, or nomination for election by Pharmacia’s stockholders, was approved by the vote of at least three-quarters of the directors then
comprising the Incumbent Board (either by a specific vote or by approval of the proxy statement of Pharmacia in which such person is
named as a nominee for director, without objection to such nomination) shall be deemed to have been a member of the Incumbent Board;
and provided further, that no individual who was initially elected as a director of Pharmacia as a result of an actual or threatened election
contest, as such terms are used in Rule 14a-11 of Regulation 14A promulgated under the Exchange Act, or any other actual or threatened
solicitation of proxies or consents by or on behalf of any Person other than the Board shall be deemed to have been a member of the
Incumbent Board;
(3) approval by the stockholders of Pharmacia of a reorganization, merger, or consolidation involving Pharmacia unless, in any
such case, immediately after such reorganization, merger, or consolidation, (i) more than 50% of the then outstanding shares of common
stock of the corporation resulting from such reorganization, merger, or consolidation and more than 50% of the combined voting power of
the then outstanding securities of such corporation entitled to vote generally in the election of directors is then beneficially owned, directly
or indirectly, by all or substantially all of the individuals or entities who were the beneficial owners, respectively, of the Outstanding
Company Common Stock and the Outstanding Company Voting Securities immediately prior to such reorganization, merger, or
consolidation and in substantially the same proportions relative to each other as their ownership, immediately prior to such reorganization,
merger, or consolidation, of the Outstanding Company Common Stock and the Outstanding Company Voting Securities, as the case may be,
(ii) no Person (other than Pharmacia, any employee benefit plan (or related trust) sponsored or maintained by Pharmacia or the corporation
resulting from such reorganization, merger, or consolidation (or any corporation controlled by Pharmacia), or any Person which beneficially
owned, immediately prior to such reorganization, merger, or consolidation, directly or indirectly, 33% or more of the
3

Outstanding Company Common Stock or the Outstanding Company Voting Securities, as the case may be) beneficially owns, directly or
indirectly, 33% or more of the then outstanding shares of common stock of such corporation or 33% or more of the combined voting power
of the then outstanding securities of such corporation entitled to vote generally in the election of directors, and (iii) at least a majority of the
members of the board of directors of the corporation resulting from such reorganization, merger, or consolidation were members of the
Incumbent Board at the time of the execution of the initial agreement or action of the Board providing for such reorganization, merger, or
consolidation; or
(4) (i) approval by the stockholders of Pharmacia of a plan of complete liquidation or dissolution of Pharmacia or (ii) the sale or
other disposition of all or substantially all of the assets of Pharmacia other than to a corporation with respect to which, immediately after
such sale or other disposition, (A) more than 50% of the then outstanding shares of common stock thereof and more than 50% of the
combined voting power of the then outstanding securities thereof entitled to vote generally in the election of directors is then beneficially
owned, directly or indirectly, by all or substantially all of the individuals and entities who were the beneficial owners, respectively, of the
Outstanding Company Common Stock and the Outstanding Company Voting Securities immediately prior to such sale or other disposition
and in substantially the same proportions relative to each other as their ownership, immediately prior to such sale or other disposition, of the
Outstanding Company Common Stock and the Outstanding Company Voting Securities, as the case may be, (B) no Person (other than
Pharmacia, any employee benefit plan (or related trust) sponsored or maintained by Pharmacia or such corporation (or any corporation
controlled by Pharmacia), or any Person which beneficially owned, immediately prior to such sale or other disposition, directly or indirectly,
33% or more of the Outstanding Company Common Stock or the Outstanding Company Voting Securities, as the case may be) beneficially
owns, directly or indirectly, 33% or more of the then outstanding shares of common stock thereof entitled to vote generally in the election of
directors, and (C) at least a majority of the members of the board of directors thereof were members of the Incumbent Board at the time of
the execution of the initial agreement or action of the Board providing for such sale or other disposition (or were approved directly or
indirectly by the Incumbent Board).
CIC Consummation . “CIC Consummation” means the consummation of a transaction approved by stockholders as described in
paragraphs (3) or (4) of the definition of Change in Control.
Code . “Code” means the Internal Revenue Code of 1986, as amended from time to time.
Committee . “Committee” means the Compensation Committee appointed by the Board which shall administer the Plan and which
also may act for the Company or the Board in making decisions and performing specified duties under the Plan. The Committee may
delegate any or all of its duties under the Plan in which case the term “Committee” shall apply to the Committee’s delegate to the same
extent as it would have applied to the Committee.
4

Company . “Company” means Pharmacia and any Affiliate that is authorized by the Board to adopt the Plan and to cover its Eligible
Employees and whose designation as such has become effective upon acceptance of such status by the board of directors of the Affiliate. An
Affiliate may revoke its acceptance of such designation at any time, but until such acceptance has been revoked, all the provisions of the
Plan and amendments thereto shall apply to the Eligible Employees of the Affiliate. In the event the designation is revoked by the board of
directors of an Affiliate, the Plan shall be deemed terminated only with respect to such Affiliate.
Company Contributions . “Company Contributions” means those matching contributions credited to the Participant’s Account by the
Company at a rate equal to the matching contribution rate under the Savings Plan applicable to the Participant. “Company Contributions”
shall be deemed to have been made in phantom shares of the Voting Securities valued on the basis of the closing price of the Voting
Securities on the principal exchange on which the Voting Securities are traded on the day the Company Contribution is credited to the
Participant’s Account.
Compensation Deferral . “Compensation Deferral” means that portion of Base Salary and/or Bonus as to which a Participant has
made an irrevocable election to defer receipt until the Plan Year following the Participant’s Termination Date, except as otherwise
specifically provided herein.
Disabled . “Disabled” means a mental or physical condition that qualifies a Participant for total and permanent disability benefits
under a Company sponsored long term disability plan.
Earnings Crediting Options . “Earnings Crediting Options” means the deemed investment options selected by the Participant from
time to time pursuant to which deemed earnings are credited to the Participant’s Account.
Effective Date . “Effective Date” means the effective date of the Plan which is July 1, 1999. “Amendment Effective Date” means
July 1, 2002.
Eligible Employee . “Eligible Employee” means, with respect to the “bonus deferral” portion of the Plan, an Employee who is
eligible to participate in the Savings Plan and whose annualized rate of Base Salary, determined at the time of enrollment, exceeds the
compensation limit of section 401(a)(17) of the Code, as in effect from time to time. “Eligible Employee” means, with respect to the
“restoration” portion of the Plan, either an Employee who meets the definition of Eligible Employee in the preceding sentence or an
Employee whose annualized rate of Base Salary, determined at the time of enrollment, exceeds the compensation limit of section 401(a)(17)
of the Code, as in effect from time to time, when combined with the Employee’s most recently paid Bonus. “Eligible Employee”
means with
respect to the “rollover” portion of the Plan, an Employee who meets the definition in the preceding sentence and who is eligible for a
rollover as described in Section 6.3.
5

Employee . “Employee” means any individual employed by the Company on a regular, full-time basis (in accordance with the
personnel policies and practices of the Employer), including citizens of the United States employed outside of their home country and
resident aliens employed in the United States; provided, however, that to qualify as an “Employee” for purposes of the Plan, the individual
must be a member of a group of “key management or other highly compensated employees” within the meaning of Sections 201, 301, and
401 of the Employee Retirement Income Security Act of 1974, as amended.
Enrollment Agreement . “Enrollment Agreement” means the authorization form, which an Eligible Employee files with the
Committee to participate under Article 4 of the Plan.
Participant . “Participant” means an Eligible Employee who has filed a completed and executed Enrollment Agreement with the
Committee or its designee or is otherwise automatically participating in the Plan in accordance with the provisions of Article 4 or by reason
of a “rollover” pursuant to Section 6.3. In the event of the death or incompetency of a Participant, the term shall mean his personal
representative or guardian. An individual shall remain a Participant until that individual has received full distribution of all amounts credited
to the Participant’s Account.
Pharmacia . “Pharmacia” means Pharmacia Corporation and any successor by merger or otherwise.
Plan . “Plan” means this plan, called the Pharmacia Savings Plus+Plan, as amended from time to time.
Plan Year . “Plan Year” means the 12 month period beginning on each January 1 and ending on the following December 31 except
that the first Plan Year of this amended and restated Plan shall begin on the Amended Effective Date and end on the following December 31.
Retirement Date . “Retirement Date” means the date that the Participant attains age 50.
Rollover Deferral . “Rollover Deferral” means the amount credited to the Participant under this Plan pursuant to Section 6.3.
Savings Plan . “Savings Plan” means the Pharmacia Savings Plan, as it may be amended from time to time, and any successor
thereof.
Termination Date . “Termination Date” means the date of termination of a Participant’s service with the Company and its Affiliates
and shall be determined without reference to any compensation continuation arrangement or severance benefit arrangement that may be
applicable.
Voting Securities . “Voting Securities” means the common securities of Pharmacia, which carry the right to vote generally in the
election of directors.
6

ADMINISTRATION OF THE PLAN AND DISCRETION
3.1 Subject to the terms of the Plan, the Committee shall have full power and authority to interpret the Plan, to prescribe, amend,
and rescind any rules, forms, and procedures as it deems necessary or appropriate for the proper administration of the Plan and to make any
other determinations and to take any other such actions as it deems necessary or advisable in carrying out its duties under the Plan. All
action taken by the Committee arising out of, or in connection with, the administration of the Plan or any rules adopted thereunder, shall, in
each case, lie within its sole discretion, and shall be final, conclusive, and binding upon the Company, the Board, all Employees, all
Beneficiaries of Employees, and all persons and entities having an interest therein.
3.2 The Committee may adjust the phantom shares of Voting Securities to reflect a change in corporate capitalization (such as a
stock split or stock dividend), or a corporate transaction (such as a merger, consolidation, separation, reorganization, or partial or complete
liquidation), or to reflect equitably the occurrence of any extraordinary event, any change in applicable accounting rules or principles, any
change in Pharmacia’s method of accounting, any change in applicable law, any change due to a merger, consolidation, acquisition,
reorganization, combination of shares, or other changes in Pharmacia’s corporate structure or share, or any other change of similar nature.
3.3 The Company shall indemnify and hold harmless each member of the Committee from any and all claims, losses, damages,
expenses (including counsel fees), and liability (including any amounts paid in settlement of any claim or any other matter with the consent
of the Board) arising from any act or omission of such member, except when the same is due to gross negligence or willful misconduct.
3.4 Any decisions, actions, or interpretations to be made under the Plan by the Company, the Board, or the Committee shall be
made in its respective sole discretion, not as a fiduciary and need not be uniformly applied to similarly situated individuals and shall be final,
binding, and conclusive on all persons interested in the Plan.
PARTICIPATION
4.1 Election to Participate
. Each Eligible Employee shall be offered the opportunity to participate in the portion or portions of the
Plan for which he is eligible on an annual basis or as otherwise determined by the Committee. An Eligible Employee may enroll in the Plan
for a Plan Year by filing a completed and fully executed Enrollment Agreement with the Committee or its designee by the last day of
November of the preceding year for which the election is to be effective or such other time as the Committee determines (for the Plan Year
beginning on the Amendment Effective Date, the executed Enrollment Agreement shall be due no later than June 20, 2002 or such other
time as the Committee determines). For an Eligible Employee who begins employment with the Company after the end of the enrollment
period, such Eligible Employee shall have 31
7

days after being hired to file a completed and fully executed Enrollment Agreement with the Committee or its designee. Participation in this
Plan shall be automatic for any Eligible Employee with a Rollover Deferral.
(a) Restoration Portion . With respect to the “restoration” portion of the Plan, each active Participant shall irrevocably
elect on an Enrollment Agreement (i) whether to convert his or her pre-tax deferrals under the Savings Plan to after-tax deferrals under the
Savings Plan upon reaching the annual elective deferral limit under section 402(g) of the Code; (ii) if the Participant does elect to convert
pre-tax deferrals under the Savings Plan to after-tax deferrals under the Savings Plan, whether to convert the after-tax deferrals under the
Savings Plan to pre-tax deferrals under the Plan upon reaching either the annual addition limit under section 415 of the Code or the annual
compensation limit under section 401(a)(17) of the Code; and (iii) if the Participant has otherwise elected to make after-tax deferrals under
the Savings Plan, whether to convert these after-tax deferrals to pre-tax deferrals under the Plan upon reaching either the annual elective
deferral limit under section 402(g) of the Code, the annual addition limit under section 415 of the Code, or the annual compensation limit
under section 401(a)(17) of the Code.
(b) Bonus Deferral Portion . With respect to the “bonus deferral” portion of the Plan, an active Participant shall
irrevocably elect on an Enrollment Agreement (i) whether to defer all or a percentage of his or her Bonus under the Plan, and (ii) whether to
defer an additional percentage of his or her Base Salary under the Plan over and above the amount deferred under the “restoration” portion
of the Plan. Any amount elected to be deferred under the Plan shall be made in whole percentages.
(c) Rollover Portion . With respect to the “rollover” portion of the Plan each Participant shall elect on an Enrollment
Agreement the Earnings Crediting Options for his or her Account if the Participant does not already have an election in effect. Until such
election is provided, the Committee shall determine the Earnings Crediting Options applicable to the Participant’s Account.
The Committee may establish minimum or maximum amounts that may be deferred under this Section and may change such standards from
time to time. Any such limits shall be communicated by the Committee to the Participants prior to the commencement of a Plan Year. Each
Eligible Employee shall also provide in the Enrollment Agreement such other information as the Committee shall require.
4.2 Company Contributions . The Company shall credit to each Participant’s Account a Company Contribution based upon the
amount of the Participant’s Base Salary Deferral and/or Bonus Deferral. Company Contributions shall be credited as frequently as
determined by the Committee acting on behalf of the Company. Unless otherwise determined by the Committee, Participants who
participate in Option 1 under Pharmacia’s Retirement Choice Program shall receive a dollar for dollar matching contribution on a payroll
period basis for each dollar that they defer under the Plan up to 5% of their Compensation and Participants who participate in Option 2
under Pharmacia’s Retirement Choice Program shall receive an age-weighted matching contribution identical to the matching contribution
offered under the Savings Plan up to 5% of their Compensation.
8

ACCOUNTS
5.1 Creation of Accounts . The Committee shall establish and maintain separate Accounts with respect to each Participant. The
amount of the Participant’s Compensation Deferral and Rollover Deferral shall be credited by the Company to the Participant’s Account no
later than the first day of the month following the month in which such Compensation would otherwise have been paid and immediately
upon rollover pursuant to Section 6.3. The Participant’s Account shall be reduced by the amount of payments made by the Company to the
Participant or the Participant’s Beneficiary pursuant to this Plan.
5.2 Compensation Deferrals . A Participant’
s Account shall be credited with Compensation Deferrals in the following amount and
manner:
(a) Restoration Portion . If a Participant elects to convert his or her pre-tax deferrals under the Savings Plan directly to
pre-tax deferrals under the Plan upon reaching the annual elective deferral limit under section 402(g) of the Code, the Participant’s Base
Salary Deferrals shall commence on the date on which the attainment of the annual elective deferral limit under section 402(g) of the Code
would have been reached if the Participant’s before-tax contribution rate under the Savings Plan that was in effect on the last day of the
applicable enrollment period remained in effect throughout the remainder of the Plan Year. The amount of such Base Salary Deferrals shall
be equal to the total percentage of Compensation that the Participant has elected to defer as pre-tax and after-tax deferrals under the Savings
Plan. If a Participant elects to convert his or her pre-tax deferrals under the Savings Plan to after-tax deferrals under the Savings Plan upon
reaching the annual elective deferral limit under section 402(g) of the Code, the Participant’s Base Salary Deferrals shall commence on the
date on which the attainment of the annual addition limit under section 415 of the Code or the annual compensation limit under section 401
(a)(17) of the Code would have been reached if the Participant’s before and after-tax contribution rates under the Savings Plan that were in
effect on the last day of the applicable enrollment period remained in effect throughout the remainder of the Plan Year. The amount of such
Base Salary Deferrals shall be equal to the percentage that the Participant has elected to defer as before-tax and after-tax deferrals under the
Savings Plan.
(b) Bonus Deferral Portion . If a Participant has elected to defer all or a portion of his or her Bonus, the Participant’s
Bonus Deferral shall commence on the date that the Participant would otherwise have received such Bonus in an amount equal to the
percentage elected by the Participant. If a Participant has elected to make additional Base Salary Deferrals under the “bonus deferral”
portion of the Plan, the Participant’s additional Base Salary Deferrals shall commence on the date on which the attainment of the annual
elective deferral limit under section 402(g) of the Code, the annual addition limit under section 415 of the Code, or the annual compensation
limit under section 401(a)(17) of the Code would have been reached if the Participant’s before and after-tax contribution rates under the
Savings Plan that were in effect on the last day of the applicable enrollment period remained in effect throughout the remainder of the Plan
Year in an amount equal to the percentage that the Participant has elected to defer under the “bonus deferral” portion of the Plan. The
combined maximum amount that a Participant may defer under the Plan and the Savings Plan shall be 80% of the Participant’s Base Salary.
9

5.3 Earnings on Accounts . A Participant’s Account shall be credited with earnings in accordance with the Earnings Crediting
Options elected by the Participant from time to time. Participants may allocate the sums credited to their Account among the Earnings
Crediting Options available under the Plan only in whole percentages; provided, however, that the Company Contribution shall be deemed
to be invested, and shall remain, in phantom shares of Voting Securities until the Participant reaches age 50. No Rollover Deferral shall be
required to be deemed invested in phantom shares of Voting Securities. The deemed rate of return, positive or negative, credited under each
Earnings Crediting Option is based upon the actual investment performance of the corresponding investment portfolios of the Savings Plan,
or such other investment fund(s) as the Committee may designate from time to time, and shall equal the total return of such investment fund
net of asset based charges, including, without limitation, money management fees, fund expenses, mortality and expense risk insurance
contract charges, and such other administrative expenses as the Committee determines should be charged to the Participants’ Accounts. The
Committee reserves the right, on a prospective basis, to add or delete Earnings Crediting Options; provided, however, that, following a CIC
Consummation, the Committee (a) may not alter the Earnings Crediting Options (except to the extent that a change is made to the
corresponding investment portfolios of the Savings Plan or, if such Plan is terminated, the comparable plan in which Employees of the
Company are eligible to participate) and (b) may not increase any of the expenses charged to a Participant’s Account except to the extent
such expenses apply to the corresponding investment portfolio maintained under the Savings Plan and are charged to Participants in that
plan.
5.4 Earnings Crediting Options . Notwithstanding that the rates of return credited to Participants’ Account under the Earnings
Crediting Options are based upon the actual performance of the corresponding portfolios of Savings Plan, or such other investment funds as
the Committee may designate, the Company shall not be obligated to invest any Compensation deferred by Participants under this Plan,
Company Contributions, or any other amounts, in such portfolios or in any other investment funds.
5.5 Changes in Earnings Crediting Options . A Participant may change the Earnings Crediting Options to which the Participant’s
Account is deemed to be allocated with whatever frequency is determined by the Committee which shall not be less than four times per Plan
Year. Each such change may include (a) reallocation of the Participant’s existing Account in whole percentages and/or (b) change in
investment allocation of amounts to be credited to the Participant’s Account in the future, as the Participant may elect. Following a CIC
Consummation, the Committee shall not reduce the frequency of permitted changes.
5.6 Valuation of Accounts . The value of a Participant’s Account as of any date shall equal the amounts theretofore credited to
such Account, including any earnings (positive or negative) deemed to be earned on such Account in accordance with Section 5.3 through
the day preceding such date, less the amounts deducted from such Account.
10

5.7 Statement of Account
. The Committee shall provide to each Participant, not less frequently than quarterly, a statement setting
forth in reasonable detail the balance standing to the credit of each Participant in the Participant’s Account and the amount credited to and
debited from such Account since the prior statement.
DISTRIBUTION OF ACCOUNTS
6.1 Distribution . Benefits shall be distributed in the Plan Year following (i) the Participant’s Retirement or (ii) the Plan Year in
which occurs the Participant’s Termination Date, other than on account of death or becoming Disabled, as follows:
(a) Benefits Upon Retirement . In the case of a Participant whose Service with the Employer terminates on or after his
Retirement Date, the Participant’s Account shall be distributed in one of the following methods, as elected by the Participant in writing
either in the Enrollment Agreement or in a separate election made at least three months prior to the beginning of the Plan Year in which
distribution is to occur: (i) in a lump sum; or (ii) in annual installments not in excess of 15, as elected by the Participant. Any lump-sum
benefit payable in accordance with this paragraph shall be paid in, but not later than January 31 of the Plan Year following the Plan Year in
which occurs the Participant’s Retirement Date valued as of the last business day of the Plan Year preceding the date of payment. Annual
installment payments, if any, shall commence not later than January 31 of the Plan Year following the Plan Year in which occurs the
Participant’s Retirement and each remaining installment shall be paid no later than each January 31 thereafter. Each such installment shall
be in an amount equal to (i) the value of the Participant’s Account as of the last business day of the Plan Year preceding the date of
payment, divided by (ii) the number of annual installment payments elected by the Participant in the Enrollment Agreement or election form
that remain to be paid. A Participant may change the election regarding the manner of payment of the Participant’s Account at any time
prior to the October 1 preceding the Plan Year in which occurs the later of the Participant’s Retirement Date and his or her Termination
Date. If a Participant has made a timely election but his Termination Date occurs prior to the end of the Plan Year in which the election was
made, then payment will be made pursuant to his or her prior election unless the Participant’s Termination Date is after a CIC
Consummation (in which case the Participant’
s most recent election shall be given effect). Such election may also specify that payment shall
be made or commence as of the beginning of any later Plan Year (but not beyond the Plan Year in which the Participant attains age 65) in
the event of a CIC Consummation prior to the Participant’s Termination Date.
(b) Benefits Upon Termination of Employment
. In the case of a Participant whose Termination Date occurs prior to the
Participant’s Retirement Date, other than on account of becoming Disabled or by reason of death, the Participant’s Account shall be
distributed in a lump sum by January 31 of the Plan Year following the Participant’s Termination Date valued as of the last business day of
the Plan Year preceding the date of payment; provided, however, that, following a CIC Consummation, if the Participant’
s termination is not
for Cause the Participant’s Account shall be
11

distributed in (i) a lump sum or (ii) annual installments not in excess of 15, as elected by the Participant and such lump sum shall be paid or
such installments shall commence as of the beginning of any later Plan Year (but not later than January 31 of such year) selected by the
Participant (but not beyond the Plan Year in which the Participant attains age 65). Any such election as to method and timing of payments
following a CIC Consummation shall be made at least twelve (12) months prior to the date payment is to be made or commence, otherwise
payments shall be made in one lump sum as specified in the first sentence of this paragraph (b); provided, however, that any such election
made before October 1, 2002, shall be given effect if the Participant’s Termination Date is after a CIC Consummation.
(c) Benefits Upon Change in Control . Within six months after a Change in Control, or if the Committee determines that
a Change in Control is imminent, but in no event after a CIC Consummation, the Committee may determine to distribute a Participant’s
Account in a lump sum by January 31 of the Plan Year following the Change in Control or the date of the Committee’s decision regarding
the imminency of the Change in Control valued as of the last business day of the Plan Year preceding the date of payment.
(d) Post Change in Control Elections . Any election as to the time of payment made by a Participant pursuant to
paragraphs 6.3(a) or (b) above that would take effect after a CIC Consummation may be changed by the Participant subject to the following
limitations: (1) the election must be made at least twelve (12) months prior to the date that payment was scheduled to be made, (2) the
election can be made only to further defer payment to the beginning of a Plan Year that is at least two (2) years later. After a Participant’s
Termination Date, he shall be entitled to no more than three (3) such elections to defer payment.
6.2 Form of Distributions . Any distribution made to or on behalf of a Participant from the Participant’s Account shall be in cash
except that, to the extent the Account is deemed invested in phantom shares of Voting Securities, a Participant may elect, in the manner
prescribed by the Committee, to receive Voting Securities. Where a distribution will be in an amount that is less than the entire balance of
any such Account, the distribution shall be made pro rata from each of the Earnings Crediting Options to which such Account is then
allocated.
6.3 Rollovers . Any distribution that is payable from or amount credited to any Eligible Employee under the Pharmacia
Supplemental Pension Plan, the Pharmacia Key Executive Pension Plan, the Pharmacia Annual Incentive Plan, the Pharmacia Cash Long-
Term Incentive Plan, the Pharmacia Long-Term Performance Share Unit Incentive Plan, any individual contractual pension arrangements,
any arrangement or plans that expressly provide for deferral pursuant to this Plan and such other plans as may be from time to time
designated by Pharmacia’s Senior Vice President Human Resources may be or shall be further deferred under this Plan in the manner set
forth in such plans and in accordance with procedures established from time to time by the Committee or its delegate. All such amounts
shall be credited to the Participant’s Account as Rollover Deferrals.
12

6.4 Pharmacia Corporation ERISA Parity Savings and Investment Plan . Effective July 1, 2002, this Plan shall assume all rights
and obligations of the Pharmacia Corporation ERISA Parity Savings and Investment Plan (the “Parity Plan”).
Any amount accrued under the
Parity Plan shall be payable exclusively under this Plan in accordance with this Plan’s terms and provisions. The amount credited to a
Participant’s account under the Parity Plan as of July 1, 2002 shall be the opening balance of a Participant’
s Account under this Plan on such
date. Any participants under the Parity Plan who have commenced receiving distributions under the Parity Plan prior to July 1, 2002 shall be
entitled to continue receiving the same form of distribution that they have been receiving under the Parity Plan.
DISABILITY
In the event a Participant becomes Disabled, the Participant’s right to make any further deferrals under this Plan shall terminate as of
the date for which the Participant first receives long-term disability benefits from the Company. A Participant who becomes Disabled shall
be entitled to elect, within 90 days after receiving the first long-term disability benefit, but in any event prior to the end of the current Plan
Year, to be treated as a Participant who has attained his or her Retirement Date, in which case the provisions of Section 6.1(a) shall be
applicable, or else shall be treated as not having incurred a Termination Date until the Participant would otherwise have attained his or her
Retirement Date in which case the provisions of Section 6.1(a) shall be applicable when the Participant would otherwise have actually been
eligible under that provision. The Participant’
s Account shall continue to be credited with earnings in accordance with Section 5.3 until such
Account is fully distributed.
OTHER WITHDRAWALS
A Participant may, by written request on a form provided by the Committee, withdraw all or any portion of any of his Accounts as of
the end of any calendar quarter, provided that the Participant shall forfeit 10% of the amount withdrawn as a penalty. Such penalty shall not
apply if (a) the Participant provides the Company with written notice of withdrawal (which shall be irrevocable) at least one year prior to the
year in which payment to the Participant is to be made, (b) such payment is for the Participant’s entire Account and (c) the Participant’s
Base Salary Deferrals and Bonus Deferrals shall be suspended for one calendar year.
SURVIVOR BENEFITS
9.1 Death of Participant Prior to the Commencement of Benefits . In the event of a Participant’
s death prior to the commencement
of benefits in accordance with Article 6, benefits shall be paid to the Participant’s Beneficiary, as determined under Section 10.3, as
provided in Section 8.2 in lieu of any benefits otherwise payable under the Plan to or on behalf of such Participant.
13

9.2 Survivor Benefits . In the case of a Participant who dies prior to the commencement of benefits pursuant to Section 6.1,
distribution of such Account shall be made, as elected by the Participant in the Enrollment Agreement or as may have been changed by the
Participant, (a) in a lump sum as soon as practicable following the Participant’s death (subject to the Beneficiary’s rights to obtain payment
in accordance with Articles 8 and 10), or (b) in the manner and at such time as such Account would otherwise have been distributed in
accordance with Section 6.1(a) had the Participant lived and retired on the earliest possible date. The amount of any lump sum benefit
payable in accordance with this Section shall equal the value of such Account as of the last business day of the calendar month immediately
preceding the date on which such benefit is paid. The amount of any annual installment benefit payable in accordance with this Section shall
equal (a) the value of such Account as of the last business day of the calendar month immediately preceding the date on which such
installment is paid, divided by (b) the number of annual installments remaining to be paid pursuant to the election of the Participant in the
Enrollment Agreement or as may have been changed by the Participant.
9.3 Death of Participant After Benefits Have Commenced . In the event a Participant dies after annual installment benefits
payable under Section 6.1 from the Participant’s Account has commenced, but before the entire balance of such Account has been paid, any
remaining installments shall continue to be paid to the Participant’
s Beneficiary, as determined under Section 10.3, at such times and in such
amounts as they would have been paid to the Participant had he survived, subject to such Beneficiary’s rights to obtain payment in
accordance with Articles 8 and 10.
EMERGENCY BENEFIT
In the event that the Committee, upon written request of a Participant, determines that the Participant has suffered an “unforeseeable
emergency” (or any similar circumstance under which a payment would be permitted, without causing the imposition of federal income
taxes on Participant Accounts that have not been paid, pursuant to Revenue Procedure 92-65 or any successor Revenue Procedure, Revenue
Ruling, regulation or other applicable administrative determination issued by the Internal Revenue Service), the Company shall pay to the
Participant from the Participant’s Account, as soon as practicable following such determination, an amount necessary to meet the
emergency, after deduction of any and all taxes as may be required pursuant to Section 11.8 (the “Emergency Benefit”), based on the value
of the Participant’s Account as of the last business day of the month preceding the date of the distribution.
14

MISCELLANEOUS
11.1 Amendment and Termination . The Plan may be amended, suspended, discontinued, or terminated at any time by action of
the Board or its delegate; provided, however, that no such amendment, suspension, discontinuance, or termination shall reduce or in any
manner adversely affect the rights of any Participant or Beneficiary with respect to benefits that are payable or may become payable under
the Plan based upon the balance of the Participant’s Accounts as of the effective date of such amendment, suspension, discontinuance, or
termination. After a CIC Consummation, no amendment to the Plan may be made to this Section, Sections 5.3, 5.5, 5.7, 6.1, 6.2, 6.3 or
11.11, or Articles 8, 9 or 10; provided, however, that changes may be made to Section 6.1 but only to the extent such changes are necessary,
in the Committee’s reasonable judgment, upon the advice of nationally recognized legal counsel, to fulfill the intent of this Plan to defer
federal income taxation of Participants with respect to their Accounts until such Accounts are paid in accordance with the terms of the Plan.
11.2 Claims Procedure .
(a) Claim . A person who believes that he is being denied a benefit to which he is entitled under the Plan (hereinafter
referred to as a “Claimant”) may file a written request for such benefit with the Committee, setting forth the claim.
(b) Claim Decision . Upon receipt of a claim, the Committee shall advise the Claimant that a reply will be forthcoming
within ninety days and shall, in fact, deliver such reply within such period. The Committee may, however, extend the reply period for an
additional ninety days for reasonable cause.
If the claim is denied in whole or in part, the Claimant shall be provided a written opinion, using language calculated to be
understood by the Claimant, setting forth:
(i) The specific reason or reasons for such denial;
(ii) The specific reference to pertinent provisions of this Agreement on which such denial is based;
(iii) A description of any additional material or information necessary for the Claimant to perfect his claim and
an explanation why such material or such information is necessary;
(iv) Appropriate information as to the steps to be taken if the Claimant wishes to submit the claim for review;
and
(v) The time limits for requesting a review under subsection (c) and for review under subsection (d) hereof,
including a statement of the Claimant’s right to bring a civil action under section 502(a) of the Employee Retirement
Income Security Act of 1974, as amended (“ERISA”) following an adverse benefit determination on review.
15

(c) Request for Review . Within sixty days after the receipt by the Claimant of the written opinion described above, the
Claimant may request in writing that the Committee review the determination of the Committee. The Claimant or his duly authorized
representative may, but need not, review the pertinent documents and submit written documents, records, comments and other information
related to the claim for consideration by the Committee and shall be entitled to review, upon request without charge, copies of documents,
records and all other information relevant to his claim. If the Claimant does not request a review of the initial determination within such
sixty-day period, the Claimant shall be barred and estopped from challenging the determination.
(d) Review of Decision . Within sixty days after the Committee’s receipt of a request for review, the Committee shall
review the initial determination. After considering all materials presented by the Claimant, the Committee shall render a written opinion,
written in a manner calculated to be understood by the Claimant, setting forth the specific reasons for the decision, specific references to the
pertinent provisions of this Agreement on which the decision is based, informing the Claimant of his right to receive, upon request and free
of charge, reasonable access to and copies of all documents, records, and other information relevant to his benefits, and informing the
Claimant of his right to bring a civil action under section 502(a) of ERISA. If special circumstances require that the sixty day time period be
extended, the Committee shall so notify the Claimant and shall render the decision as soon as possible, but no later than one hundred twenty
days after receipt of the request for review.
11.3 Designation of Beneficiary . Each Participant may designate a Beneficiary or Beneficiaries (which Beneficiary may be an
entity other than a natural person) to receive any payments which may be made following the Participant’s death. Such designation may be
changed or canceled at any time without the consent of any such Beneficiary. Any such designation, change or cancellation must be made in
a form approved by the Committee and shall not be effective until received by the Committee, or its designee. If no Beneficiary has been
named, or the designated Beneficiary or Beneficiaries shall have predeceased the Participant, the Beneficiary shall be the Participant’s
estate. If a Participant designates more than one Beneficiary, the interests of such Beneficiaries shall be paid in equal shares, unless the
Participant has specifically designated otherwise.
11.4 Limitation of Participant’s Right . Nothing in this Plan shall be construed as conferring upon any Participant any right to
continue in the employment of the Company, nor shall it interfere with the rights of the Company to terminate the employment of any
Participant and/or to take any personnel action affecting any Participant without regard to the effect which such action may have upon such
Participant as a recipient or prospective recipient of benefits under the Plan. Any amounts payable hereunder shall not be deemed salary or
other compensation to a Participant for the purposes of computing benefits to which the Participant may be entitled under any other
arrangement established by the Employer for the benefit of its employees.
11.5 Obligations to Company
. If a Participant becomes entitled to a distribution of benefits under the Plan, and if at such time the
Participant has outstanding any debt, obligation, or other liability representing an amount owing to the Employer, then the Employer may
offset such amount owed to it against the amount of benefits otherwise distributable. Such determination shall be made by the Committee.
16

11.6 Nonalienation of Benefits . Except as expressly provided herein, no Participant or Beneficiary shall have the power or right
to transfer (otherwise than by will or the laws of descent and distribution), alienate, or otherwise encumber the Participant’s interest under
the Plan. The Company’s obligations under this Plan are not assignable or transferable except to (a) any corporation or partnership which
acquires all or substantially all of the Company’s assets or (b) any corporation or partnership into which the Company may be merged or
consolidated. The provisions of the Plan shall inure to the benefit of each Participant and the Participant’s Beneficiaries, heirs, executors,
administrators, or successors in interest.
11.7 Protective Provisions . Each Participant shall cooperate with the Employer by furnishing any and all information reasonably
requested by the Employer in order to facilitate the payment of benefits hereunder. If a Participant refuses to cooperate, the Employer shall
have no further obligation to the Participant under the Plan, other than payment to such Participant of the then current balance of the
Participant’s Account in accordance with his prior elections.
11.8 Withholding Taxes . The Company may make such provisions and take such action as it may deem necessary or appropriate
for the withholding of any taxes which the Company is required by any law or regulation of any governmental authority, whether federal,
state or local, to withhold in connection with any benefits under the Plan, including, but not limited to, the withholding of appropriate sums
from any amount otherwise payable to the Participant (or his Beneficiary). Each Participant, however, shall be responsible for the payment
of all individual tax liabilities relating to any such benefits.
11.9 Unfunded Status of Plan . The Plan is intended to constitute an “unfunded” plan of deferred compensation for Participants.
Benefits payable hereunder shall be payable out of the general assets of the Company, and no segregation of any assets whatsoever for such
benefits shall be made. Notwithstanding any segregation of assets or transfer to a grantor trust, with respect to any payments not yet made to
a Participant, nothing contained herein shall give any such Participant any rights to assets that are greater than those of a general creditor of
the Company. In the event that the Committee determines that a Change in Control is imminent, the Committee shall cause the Company to
create and fund a grantor trust of the Company that shall serve as the vehicle for paying all benefits due under the Plan and the amount
contributed by the Company shall be the amount the Committee determines would then be due if the Plan were to terminate and all benefits
were then to be paid in lump sum. If a Change in Control shall not have occurred within six months of such contribution by the Company,
the Board may adopt a resolution to the effect that a Change in Control is not imminent and, in that event, up to all amounts contributed to
the Trust and all earnings thereon, shall be repaid to the Company at the direction of the Board.
11.10 Severability . If any provision of this Plan is held unenforceable, the remainder of the Plan shall continue in full force and
effect without regard to such unenforceable provision and shall be applied as though the unenforceable provision were not contained in the
Plan.
17

11.11 Governing Law . The Plan shall be construed in accordance with and governed by the laws of the state of New Jersey,
without reference to the principles of conflict of laws, to the extent not preempted by ERISA; provided, however, that upon a CIC
Consummation any court or tribunal that adjudicates any dispute, controversy or claim arising between a Participant and the Committee, its
delegate, the Company or an Affiliate (or any successor to either), relating to or concerning the provisions of this Plan, will apply a de novo
standard of review to any determinations made by such person. Such de novo standard shall apply notwithstanding the grant of full
discretion hereunder to any such person or characterization of any decision as final, binding or conclusive on any party.
11.12 Headings . Headings are inserted in this Plan for convenience of reference only and are to be ignored in the construction of
the provisions of the Plan.
11.13 Gender, Singular, and Plural . All pronouns and any variations thereof shall be deemed to refer to the masculine, feminine,
or neuter, as the identity of the person or persons may require. As the context may require, the singular may read as the plural and the plural
as the singular.
11.14 Notice . Any notice or filing required or permitted to be given to the Committee under the Plan shall be sufficient if in
writing and hand delivered, or sent by registered or certified mail, to the Human Resources Department, or to such other entity as the
Committee may designate from time to time. Such notice shall be deemed given as to the date of delivery, or, if delivery is made by mail, as
of the date shown on the postmark on the receipt for registration or certification.
18

Exhibit 10(11)
WYETH
SUPPLEMENTAL EMPLOYEE SAVINGS PLAN
(amended and restated effective as of January 1, 2005)
PURPOSE
The purpose of the Plan is to provide an additional savings plan of deferred compensation for a select group of
management and highly compensated employees. Accordingly, the Plan supplements the benefits of Participants whose benefits under the
Savings Plan are limited (i) by the Code Limits or (ii) as a result of their election to defer Base Salary under the DCP or the Prior DCP. The
Plan is intended to be an unfunded deferred compensation plan for a select group of management or highly compensated employees within
the meaning of ERISA, and shall be construed and administered accordingly.
The Plan is an amendment and restatement of the Prior Plan effective as of the Restatement Date.
Capitalized terms not otherwise defined in the text hereof shall have the meanings set forth in Section 1.
SECTION 1
DEFINITIONS
1.1 Rules of Construction . Except where the context indicates otherwise, any masculine terminology used herein
shall also include the feminine gender, and the definition of any term herein in the singular shall also include the plural. All references to
sections and appendices are, unless otherwise indicated, to sections or appendices of the Plan.
1.2 Terms Defined in the Plan
. Whenever used herein, the following terms shall have the meanings set forth below:
(a) “ 409A Account ” means a bookkeeping account (including all sub-accounts) maintained by the Company for each
Participant, to record: (i) the Participant’s Base Salary and/or Excess Compensation deferrals under the Plan; (ii) all Matching Contributions
credited to a Participant, plus or minus (iii) Investment Earnings/Losses on those amounts minus (iv) all distributions or withdrawals made
to a Participant or his Beneficiary, or forfeitures of unvested Matching Contributions that relate to his 409A Account, in each case to the
extent that such amounts are not included in the Participant’s Grandfathered Account. The 409A Account shall be divided into Base Salary
and/or Excess Compensation deferral and Matching Contribution sub-accounts.
1

(b) “ Administrative Procedures ” means the policies and procedures established by the Committee and/or the
Administrative Record Keeper from time to time governing elections to participate in the Plan, maintenance of Deferral Accounts,
Investment Options, calculation of Investment Earnings/Losses, required Election Forms, distributions from the Plan and such other matters
as are necessary for the proper administration of the Plan.
(c) “ Administrative Record Keeper ” means the person or persons designated by the Committee in accordance with
Section 2.
(d) “ Affiliate ” means any corporation which is included in a controlled group of corporations (within the meaning of
Section 414(b) of the Code) which includes Wyeth and any trade or business (whether or not incorporated) which is under common control
with Wyeth (within the meaning of Section 414(c) of the Code); provided , however , that in applying Section 1563(a)(1), (2), and (3) of the
Code for purposes of determining a controlled group of corporations under Section 414(b) of the Code the language “at least 50 percent”
shall be used instead of “at least 80 percent” each place it appears in Section 1563(a)(1), (2) and (3) of the Code, and in applying
Section 1.414(c)-2 of the Treasury Regulations, for purposes of determining trades or businesses (whether or not incorporated) that are
under common control for purposes of Section 414(c) of the Code, “at least 50 percent” shall be used instead of “at least 80 percent” in each
place it appears in Section 1.414(c)-2 of the Treasury Regulations.
(e) “ Base Salary ” means the annual base compensation from all sources ( i.e. , regardless of whether United States
source or foreign source) to be paid during a Plan Year by the Company to an Employee for services rendered to the Company. Base Salary
may only be deferred under the Plan to the extent it would otherwise be payable from the Company’s regular U.S. payroll.
(f) “ Beneficiary ” shall have the meaning ascribed to it in the Savings Plan.
(g) “ Board of Directors ” means the Board of Directors of Wyeth (or any Committee of the Board of Directors to
whom the Board of Directors delegates, from time to time, its authority hereunder).
(h) “ Business Day ” means each day that the New York Stock Exchange is open for business.
(i) “ Claimant ” has the meaning set forth in Section 9.1.
(j) “ Code ” means the Internal Revenue Code of 1986, as amended, and any applicable rulings and regulations
promulgated thereunder.
(k) “ Code Limits ” means Section 401(a)(17) of the Code.
2

(l) “ Committee ” means the committee of three or more officers or employees of the Company designated from time
to time by Wyeth to administer the Plan and any successor thereto.
(m) “ Company ” means Wyeth and its Affiliates.
(n) “ Company Account Plan ” means any arrangement sponsored by the Company, other than the Plan, that (i) is
required to be aggregated with the Plan under Treasury Regulation 1.409A-1(c)(2) and (ii) (A) is an “account balance plan,” as such term is
defined in Treasury Regulation 1.409A-1(c)(2)(i)(A) or (B) provides for the deferral of compensation other than at the election of the
Employee, as described in Treasury Regulation 1.409A-1(c)(2)(i)(B).
(o) “ Company Stock Fund ” means the Investment Option available under the Plan and the Savings Plan that is
designed to track the performance of Wyeth’s Common Stock, par value $0.33 -1/3.
(p) “ Covered Compensation ” shall have the meaning ascribed to it in the Savings Plan.
(q) “ DCP ” means the Wyeth 2005 (409A) Deferred Compensation Plan, (amended and restated effective as of
January 1, 2005), as amended from time to time.
(r) “ Death Payment Date ” shall mean within ninety days after the date of the Participant’s death.
(s) “ Deferral Account ” means a bookkeeping account (including all sub-accounts) maintained by the Company for
each Participant to record (i) the Participant’s Base Salary and/or Excess Compensation deferrals under the Plan; (ii) all Matching
Contributions credited to a Participant, plus or minus (iii) Investment Earnings/Losses on those amounts, minus (iv) all distributions or
withdrawals made to a Participant or his Beneficiary, or forfeitures of unvested Matching Contributions, pursuant to the Plan. The Deferral
Account shall be divided into a 409A Account and a Grandfathered Account.
(t) “ Deferred Compensation Tax Compliance Committee ” means a committee of such officers or employees of the
Company as shall be designated from time to time by the Company.
(u) “ Election Form ” means the form or forms established from time to time by the Administrative Record Keeper
and/or the Committee, that an Eligible Employee completes, signs and returns to the Administrative Record Keeper to make an election
under the Plan.
(v) “ Eligible Employee ” means an Employee who is eligible to participate in the DCP; provided , however
, that in no
event shall an Employee who is a resident of Puerto Rico be an Eligible Employee.
(w) “ Employee ” means an employee of the Company.
3

(x) “ ERISA ” means the Employee Retirement Income Security Act of 1974, as amended from time to time, including
any applicable rulings and regulations promulgated thereunder.
(y) “ Excess Compensation ” means an Eligible Employee’s compensation in excess of Covered Compensation.
(z) “ Grandfathered Account ” means that portion of a Participant’s Deferral Account under the Plan that, for purposes
of Section 409A, was both earned and vested on December 31, 2004, plus or minus Investment Earnings/Losses on those amounts, plus or
minus retained earnings, minus all distributions or withdrawals made to a Participant or his Beneficiary, pursuant to the Plan that relate to
his Grandfathered Account. The Grandfathered Account shall be divided into separate Base Salary and/or Excess Compensation deferral and
Matching Contribution sub-
accounts. For example, the Grandfathered Account of a Participant will equal all amounts deferred and vested as
of December 31, 2004 and all earnings on such amounts until the balance of the Grandfathered Account is distributed.
(aa) “ Investment Earnings/Losses ” means the income, gains and losses that would have been realized had an amount
deferred under the Plan actually been invested in the Investment Option or Options selected by the Participant.
(bb) “ Investment Options ” means the investment options that are selected by the Committee that are used as
hypothetical investment options among which the Participant may allocate all or a portion of his Deferral Account.
(cc) “ Matching Contribution ” has the meaning set forth in Section 5.1.
(dd) “ Participant ” means an Eligible Employee who participates in the Plan.
(ee) “ Payment Date ” means as soon as practicable following the first anniversary of the Participant’s Separation from
Service, but in no event later than the last Business Date of the month following the month that includes such first anniversary of the
Participant’s Separation from Service.
(ff) “ Plan ” means this Wyeth Supplemental Employee Savings Plan, as amended from time to time.
(gg) “ Plan Year ” means the calendar year.
(hh) “ Prior DCP means the terms of the Wyeth Deferred Compensation Plan (as amended and restated as of
November 20, 2003), as set forth in the Company
’s written documentation, rules, practices and procedures applicable to such plan (but
without regard to any amendments thereto after October 3, 2004 that would result in any material modification of such plan, within the
meaning of Section 409A).
(ii) “ Prior Plan ” means the terms of the Plan in effect immediately prior to the Restatement Date, as set forth in the
Company’s written documentation, rules, practices and
4

procedures applicable to the Plan (but without regard to any amendments thereto after October 3, 2004 that would result in any material
modification of the Grandfathered Account, within the meaning of Section 409A).
(jj) “ Restatement Date ” means January 1, 2005.
(kk) “ Retirement Eligible ” means a Participant who, as of the date of his Separation from Service is (i) at least age 55
with at least five Years of Vesting Service or (ii) at least age 65.
(ll) “ Retirement Plan ” means the Wyeth Retirement Plan - United States, as amended from time to time.
(mm) “ Savings Plan ” means the Wyeth Savings Plan, as amended from time to time.
(nn) “ Section 409A ” means Section 409A of the Code and the applicable notices, rulings and regulations promulgated
thereunder.
(oo) “ Section 409A Compliance ” has the meaning set forth in Section 10.1.
(pp) “ Separation from Service ” means a separation from service with the Company for purposes of Section 409A,
determined using the default provisions set forth in Treasury Regulation Section 1.409A-1(h); provided , however , that, for purposes of the
Grandfathered Account, “Separation from Service” shall be determined in accordance with the terms of the Prior Plan. Notwithstanding the
foregoing, if a Participant would otherwise incur a Separation from Service in connection with a sale of assets of the Company, the
Company shall retain the discretion with respect to the 409A Account to determine whether a Separation from Service has occurred in
accordance with Treasury Regulation Section 1.409A-1(h)(4).
(qq) “ Transition Elections ” means contingent distribution elections made by a Participant prior to January 1, 2009 in
accordance with the provisions of Notices 2005-1, 2006-79 and 2007-86 promulgated by the U.S. Treasury Department and the Internal
Revenue Service and the Proposed Regulations under Section 409A, 70 Fed. Reg. 191 (Oct. 4, 2005).
(rr) “ Treasury Regulations ” means the regulations adopted by the Internal Revenue Service under the Code, as they
may be amended from time to time.
(ss) “ Unforeseeable Emergency ” means “unforeseeable emergency” within the meaning of Section 409A.
(tt) “ Valid Notional Rollover ” means a notional rollover of all or a portion of the balance of (i) a Participant’s
Grandfathered Account to the Prior DCP at the time of Separation from Service of a Participant who has an account balance in the
Prior DCP or the DCP and is Retirement Eligible at the time of such Separation from Service or (ii) a Participant’s 409A Account to the
DCP at the time of Separation from Service of a Participant who is Retirement Eligible at the time of such Separation from Service. The
effective date of a Valid Notional Rollover shall be the first of the month following the Participant’s Separation from Service even though
the Payment Date may otherwise have been a later date.
5

(uu) “ Wyeth ” means Wyeth, a Delaware corporation, and any successor thereto.
(vv) “ Year of Vesting Service ” has the meaning ascribed to it in the Retirement Plan as of January 1, 2006, and, prior
to such date, has the meaning ascribed to "Continuous Service," as such term was defined in the Retirement Plan prior to January 1, 2006.
SECTION 2
ADMINISTRATION
2.1 General Authority . The general supervision of the Plan shall be the responsibility of the Committee, which, in
addition to such other powers as it may have as provided herein, shall have the power, subject to the terms of the Plan: (i) to determine
eligibility to participate in, and the amount of benefit to be provided to any Participant under, the Plan; (ii) to make and enforce such rules
and regulations as it shall deem necessary or proper for the efficient administration of the Plan; (iii) to determine all questions arising in
connection with the Plan, to interpret and construe the Plan, to resolve ambiguities, inconsistencies or omissions in the text of the Plan, to
correct any defects in the text of the Plan and to take such other action as may be necessary or advisable for the orderly administration of the
Plan; (iv) to make determinations regarding the valuation of Deferral Accounts; (v) to make any and all legal and factual determinations in
connection with the administration and implementation of the Plan; (vi) to designate the Administrative Record Keeper and review actions
taken by the Administrative Record Keeper or any other person to whom authority is delegated under the Plan; and (vii) to employ and rely
on legal counsel, actuaries, accountants and any other agents as may be deemed to be advisable to assist in the administration of the Plan.
All such actions of the Committee shall be conclusive and binding upon all persons. The Committee shall be entitled to rely conclusively
upon all tables, valuations, certificates, opinions, and reports furnished by any actuary, accountant, controller, counsel, or other person
employed or engaged by the Company with respect to the Plan. If any member of the Committee is a Participant, such member shall not
resolve, or participate in the resolution of, any matter relating specifically to such Committee member’s eligibility to participate in the Plan
or the calculation or determination of such member’s benefits under the Plan.
2.2 Delegation . The Committee shall have the power to delegate to any person or persons the authority to carry out
such administrative duties, powers and authority relative to the administration of the Plan as the Committee may from time to time
determine. Any action taken by any person or persons to whom the Committee makes such a delegation shall, for all purposes of the Plan,
have the same force and effect as if undertaken directly by the Committee. If any individual to whom the Committee delegates authority is a
Participant, such individual shall not resolve, or participate in the resolution of, any matter specifically relating to such individual’s
eligibility to participate in the Plan or the calculation or determination of such individual’s benefits under the Plan.
6

2.3 Administrative Record Keeper . The Administrative Record Keeper shall be responsible for the day-to-day
operation of the Plan, having the power (except to the extent such power is reserved to the Committee) to take all action and to make all
decisions necessary or proper in order to carry out his duties and responsibilities under the provisions of the Plan. If the Administrative
Record Keeper is a Participant, the Administrative Record Keeper shall not resolve, or participate in the resolution of, any question which
relates directly or indirectly to him and which, if applied to him, would significantly vary his eligibility for, or the amount of, any benefit to
him under the Plan. The Administrative Record Keeper shall report to the Committee at such times and in such manner as the Committee
shall request concerning the operation of the Plan.
2.4 Actions; Indemnification . The members of the Board of Directors, the Committee, the Administrative Record
Keeper, the members of the Deferred Compensation Tax Compliance Committee, the members of any other committee and any director,
officer or employee of the Company to whom responsibilities are delegated by the Committee shall not be liable for any actions or failure to
act with respect to the administration or interpretation of the Plan, unless such person acted in bad faith or engaged in fraud or willful
misconduct. The Company shall indemnify and hold harmless, to the fullest extent permitted by law, the Board of Directors (and each
member thereof), the Committee (and each member thereof), the Deferred Compensation Tax Compliance Committee (and each member
thereof), the Administrative Record Keeper, the members of any other committee and any director, officer or employee of the Company to
whom responsibilities are delegated by the Committee from and against any liabilities, damages, costs and expenses (including attorneys’
fees and amounts paid in settlement of any claims approved by the Company) incurred by or asserted against it or him by reason of its or his
duties performed in connection with the administration or interpretation of the Plan, unless such person acted in bad faith or engaged in
fraud or willful misconduct. The indemnification, exculpation and liability limitations of this Section 2.4 shall apply to the Administrative
Record Keeper only to the extent that the Administrative Record Keeper is or was a director, officer or employee of the Company.
SECTION 3
PARTICIPATION
3.1 Continuing Participants . Any individual on the Restatement Date who was participating in the Prior Plan
immediately prior to the Restatement Date shall continue to be a Participant in the Plan on the Restatement Date.
3.2 Mandatory Participation . An Eligible Employee shall be required to commence participation in the Plan as of
the effective date of his first election to defer Base Salary under the DCP.
3.3 Voluntary Participation
. An Eligible Employee may voluntarily elect to defer from one to six percent of Excess
Compensation under the Plan. In the event that an Eligible Employee elects to participate in the Plan in accordance with this Section 3.3,
participation shall commence as of the first payroll period during a Plan Year in which such Eligible Employee’s compensation exceeds
Covered Compensation.
7

3.4 Exclusions . No Employee who is not an Eligible Employee shall be eligible to participate in the Plan. In
addition, the Committee may, if it determines it to be necessary or advisable to comply with ERISA, the Code or other applicable law,
exclude one or more Eligible Employees or one or more classes of Eligible Employees from Plan participation.
SECTION 4
ELECTIONS
4.1 Deferral Elections . All deferrals under the Plan shall be evidenced by the Eligible Employee properly executing
and submitting such Election Forms as may be required by the Administrative Record Keeper in accordance with the Administrative
Procedures and this Section 4.
4.2 Deferrals .
(a) Mandatory Deferrals
. If an Eligible Employee is required to participate in the Plan because he has elected to make
Base Salary deferrals under the DCP, he shall complete such Election Forms as may be required by the Administrative Record Keeper in
accordance with the Administrative Procedures.
(b) Voluntary Deferrals . Except for the first Plan Year in which an individual becomes an Eligible Employee, an
Eligible Employee’
s voluntary election to defer Excess Compensation under the Plan with respect to a particular Plan Year must be received
by the Administrative Record Keeper no later than December 31 of the prior Plan Year. If a Participant fails to make a new deferral election
for a subsequent Plan Year, a Participant’s deferral election for such subsequent Plan Year shall be the deferral election in effect as of
December 31 of the preceding Plan Year. With respect to the first Plan Year in which an individual becomes an Eligible Employee,
elections to voluntarily defer Excess Compensation into the Plan must be made within 30 days after the earlier of the date the Eligible
Employee becomes eligible to participate in the Plan or any other Company Account Plan.
(c) Rehired Employees . Notwithstanding the foregoing provisions of this Section 4.2, an Eligible Employee who is
rehired by the Company or otherwise again becomes an Eligible Employee after deferring amounts under the Plan or under another
Company Account Plan and prior to receiving a distribution of his entire 409A Account and his entire balance under any other Company
Account Plan shall not be entitled to make deferrals under the Plan until the Plan Year following the Plan Year that includes the date such
individual again becomes an Eligible Employee. In the event such an Eligible Employee previously Separated from Service with the
Company, payment of his 409A Account shall not be suspended or otherwise delayed. In the event an Eligible Employee received a
distribution of his entire 409A Account and his entire balance under any other Company Account Plan prior to the date he was rehired by
the Company or otherwise again became an Eligible Employee, he shall be entitled to make a deferral election within 30 days of the date he
again becomes eligible to participate in the Plan; provided , however , that if such Eligible Employee is rehired on or after January 1, 2009,
any additional deferrals shall be paid on the applicable Participant’s Payment Date following the date he next Separates from Service with
the Company, unless the Participant elects to redefer such amounts pursuant to Section 8.
8

(d) Amount of Deferral . If an Eligible Employee is required to participate in the Plan as a result of his election to
defer Base Salary under the DCP, the first six percent of Base Salary he elected to be deferred under the DCP shall instead automatically be
deferred under the Plan. Except as provided in Section 4.2(b) above, if an Eligible Employee has compensation that exceeds Covered
Compensation, such Eligible Employee may voluntarily elect to defer from one to six percent of the amount of his Excess Compensation
into the Plan.
(e) Vesting . A Participant shall be fully vested at all times in the Base Salary or Excess Compensation deferred
(adjusted to reflect Investment Earnings/Losses) into the Plan.
4.3 Contingent Distribution Election for Transition Election Eligible Employees Who Become Participants
Prior to January 1, 2009 .
(a) An Eligible Employee who is a Participant prior to January 1, 2009 or is hired prior to November 1, 2008 with an
annual base salary of $230,000 or more (the “ 2008 New Executives ”) may make, by no later then December 31, 2008, a Transition
Election with respect to this 409A Account; provided , however , that an election made in 2008 shall apply solely to the amount that would
not otherwise be payable to him in 2008 and shall not cause any amounts to be paid to him in 2008 that would not otherwise be payable to
him in 2008. The Administrative Record Keeper may, in accordance with the requirements of Section 409A and the Administrative
Procedures, permit Eligible Employees who are Participants prior to January 1, 2009 and the 2008 New Executives to make one or more
additional elections to transfer, in a Valid Notional Rollover, deferrals made under the Plan; provided , however
, that any such election shall
only apply to deferrals made for Plan Years subsequent to the date of such election. The Administrative Record Keeper may also, in
accordance with the requirements of Section 409A and the Administrative Procedures, permit Eligible Employees who become Participants
prior to January 1, 2009 and the 2008 New Executives to make one or more elections to receive distribution of their 409A Accounts on the
Payment Date in lieu of a Valid Notional Rollover; provided , however , that any such election shall only apply to deferrals made for Plan
Years subsequent to the date of such election. A Participant may not revoke his contingent election to transfer all or a portion of the vested
balance of his 409A Account in a Valid Notional Rollover. If a Participant who has elected to make a Valid Notional Rollover is not
Retirement Eligible at the time of his Separation from Service, then the election shall be void and of no further force and effect and the
Participant’s 409A Account shall be paid on the Payment Date. An Eligible Employee who first becomes a Participant on or after January 1,
2009 (other than the 2008 New Executives) shall not be entitled to make a Transition Election and shall receive the vested balance of his
409A Account on the Payment Date, unless he elects to redefer the vested balance of his 409A Account in accordance with Section 8.
(b) The Transition Elections made by a Participant shall supplement and, to the extent inconsistent therewith, shall
supersede the corresponding provisions of this Section 4.
9

4.4 Cancellation of Deferral Election Upon Unforeseeable Emergency or Hardship Distribution
(a) Unforeseeable Emergency
. The Committee shall cancel a deferral election with respect to a Plan Year in the event
that the Participant demonstrates that an Unforeseeable Emergency has occurred.
(b) Savings Plan Hardship Distribution . Notwithstanding anything to the contrary herein, in the event a Participant
receives a hardship distribution under the Savings Plan, the Participant’s deferral election shall be cancelled for the remainder of the Plan
Year and the Participant shall not be entitled to make further deferrals under the Plan until the second Plan Year subsequent to the date such
Participant receives a hardship distribution under the Savings Plan.
(c) Effect of Cancellation . If the Participant’s election is cancelled pursuant to this Section 4.4, the Participant’s
election shall be cancelled, and not postponed or otherwise delayed, such that any later deferral election will be subject to the provisions
governing deferral elections as provided in Section 4.2 and the Administrative Procedures.
SECTION 5
MATCHING CONTRIBUTIONS
5.1 Matching Contributions . Subject to the provisions regarding vesting in Section 5.2 below, the Company shall
make a notional matching contribution in an amount equal to fifty percent of the Base Salary or Excess Compensation deferred by the
Participant under the Plan (the “ Matching Contribution ”). Matching Contributions shall be credited to a Participant’s Deferral Account on
the same date as Base Salary or Excess Compensation deferrals and shall be accounted for by the Company separately from Base Salary or
Excess Compensation deferrals.
5.2 Vesting . A Participant shall be fully vested in the Company’s Matching Contributions if he has five or more
Years of Vesting Service. If a Participant has less than five Years of Vesting Service, he shall become vested in his Matching Contributions
to the 409A Account, according to the following schedule:
Regardless of the number of Years of Vesting Service, a Participant shall be fully vested in his Matching Contributions, and such Matching
Contributions shall be non-forfeitable, when he attains age 65 or upon his death, if earlier, provided that upon such event he is still an
Employee. If a Participant incurs a Separation from Service or otherwise receives a distribution from the Plan at a time when such
Participant is less than 100% vested in Matching Contributions, the unvested portion of such Matching Contributions shall be forfeited in
their entirety.
10
Years of Vesting Service
Cumulative Vesting Percentage
Prior to 2 years
0%
On or after 2 years
25%
On or after 3 years
50%
On or after 4 years
75%
5 or more years
100%

SECTION 6
DEFERRAL ACCOUNTS
6.1 Plan Accounts – In General
. An individual Deferral Account shall be established and maintained under the Plan
on behalf of each Participant by or on behalf of whom deferrals have been made. The Deferral Account of each Participant shall be divided
into a separate Grandfathered Account and a 409A Account, as applicable, which accounts shall track the Base Salary deferrals, Excess
Compensation deferrals, Matching Contributions, Investment Earnings/Losses, distributions, forfeitures or other elections applicable to such
accounts. The Grandfathered Account and the 409A Account shall have sub-accounts established and maintained as appropriate to reflect
Matching Contributions and the Investment Option(s) selected by the Participant.
6.2 Crediting/Debiting of Deferral Account . Base Salary, Excess Compensation and Matching Contribution
deferrals under the Plan shall be credited to a Participant’s Deferral Account in accordance with the Administrative Procedures. A
Participant’s Deferral Account shall be credited or debited with Investment Earnings/Losses based upon the Investment Options selected by
the Participant pursuant to Section 6.3 and in accordance with the Administrative Procedures.
6.3 Election of Investment Options . A Participant shall elect, in connection with his initial deferral election under
the Plan, one or more Investment Option(s) from a menu of Investment Options provided by the Committee to be used to determine
Investment Earnings/Losses credited or debited to his Deferral Account. A Participant may reallocate the existing balance of his Deferral
Account among the available Investment Options and change Investment Options with respect to future deferrals under the Plan in
accordance with the Administrative Procedures. In the event that a Participant fails to select one or more Investment Options for all or a
portion of his Deferral Account (including in the situation where the Investment Option is discontinued and the Participant fails to designate
an alternative in accordance with the Administrative Procedures), such amounts shall be deemed invested in the default Investment Option
specified in the Savings Plan, or if no default is specified, in such Investment Option as may be specified by the Committee from time to
time. In addition to the blackout periods and other restrictions set forth in the Company’s Securities Transactions Policy, as amended from
time to time, the Company may impose such additional restrictions on transfers by Participants in the Company Stock Fund as it deems
necessary or advisable in order to comply with federal or state securities laws (including, but not limited to Rule 16b-3 of the Securities
Exchange Act of 1934, as amended). Any Participant subject to such restrictions shall be notified by the Company.
6.4 Investment Options . The Committee shall select the Investment Options that are used as hypothetical
investment options among which Participants may allocate all or a portion of their Deferral Account. The Committee shall be permitted to
add, remove or change Investment Options as it deems appropriate, provided
that any such addition, deletion or change shall not be effective
with respect to any period prior to the effective date of the change. Each Participant, as a condition to his participation in the Plan, agrees to
indemnify and hold harmless the Committee, the Administrative Record Keeper, and the Company, and their agents and representatives,
from any losses or damages of any kind relating to the Investment Options made available hereunder.
11

6.5 Crediting or Debiting Method
. The performance of each elected Investment Option (either positive or negative)
will be determined based on the performance of the actual Investment Option. A Participant’s Deferral Account shall be credited or debited
with Investment Earnings/Losses on each Business Day, or as otherwise determined by the Administrative Record Keeper in accordance
with the Administrative Procedures.
6.6 Valuation . The Administrative Record Keeper shall establish procedures for valuing the balance of a
Participant’s Deferral Account in accordance with the Administrative Procedures.
6.7 No Actual Investment . Notwithstanding any other provision of the Plan, the Investment Options are to be used
for measurement purposes only, and a Participant’s election of any such Investment Options and the crediting or debiting of Investment
Earnings/Losses to a Participant’s Deferral Account shall not be considered or construed in any manner as an actual investment of his
Deferral Account in any such Investment Options. In the event that the Company decides to invest funds in any or all of the Investment
Options, no Participant shall have any rights in or to such investments themselves. Without limiting the foregoing, a Participant’s Deferral
Account shall at all times be a bookkeeping entry only and shall not represent any investment made on his behalf by the Company. The
Participant shall at all times remain an unsecured creditor of the Company.
SECTION 7
DISTRIBUTIONS
7.1 Distribution of Grandfathered Accounts .
(a) Payout under the SESP . Unless a Participant makes an election in accordance with Section 7.1(b), a Participant
shall receive a lump sum cash payment equal to the balance of his Grandfathered Account upon twelve months advance written notice to the
Administrative Record Keeper; provided , however , that no payment of the Grandfathered Account may be made prior to the first
anniversary of the date such Participant incurs a Separation from Service.
(b) Rollover to Prior DCP . In lieu of receiving a lump sum cash distribution in accordance with Section 7.1(a), the
Participant may elect, prior to his Separation from Service, to transfer the balance of his Grandfathered Account in a Valid Notional
Rollover; provided , however , that no payment may be made to the Participant under the Prior DCP prior to the first anniversary of the date
such Participant incurs a Separation from Service.
(c) Death . Notwithstanding the foregoing, in the event of a Participant’s death, his benefit shall be payable on the
Death Payment Date.
12

(d) Loss of Grandfathering . In the event that a Participant’s Grandfathered Account shall, for any reason, become
subject to Section 409A, such account shall be paid out to the Participant in the same manner as such Participant’s 409A Account.
7.2 Distribution of 409A Accounts .
(a) Payout under the SESP . A Participant shall receive a lump sum cash payment equal to the vested balance of his
409A Account on the Participant’s Payment Date, unless (i) he became a Participant prior to January 1, 2009 or is a 2008 New Executive
and such balance is transferred in a Valid Notional Rollover in accordance with an election made by the Participant under Section 4.3, or
(ii) he redefers his 409A Account prior to his Separation from Service in accordance with Section 8.
(b) Death . Notwithstanding the foregoing, in the event of a Participant’s death, his Beneficiary shall receive the
vested balance of his 409A Account on the Death Payment Date.
7.3 Applicability of Prior DCP or DCP to Amounts Rolled Over to the Prior DCP or DCP . A Participant who
elects to transfer his Grandfathered Account and/or 409A Account in a Valid Notional Rollover shall be subject to the applicable terms and
provisions of the Prior DCP or the DCP, as the case may be, and shall be required to make his payment elections thereunder at the time he
elects such notional rollover. Once the amount constituting the Participant’s Grandfathered Account and/or 409A Account is credited under
the Prior DCP or the DCP, as the case may be, such crediting shall constitute a full and complete settlement with respect to the Company’s
obligations to the Participant under the Plan with respect to his Grandfathered Account and/or 409A Account.
7.4 No Duplicate Benefits . Nothing in the Plan, including the ability of a Participant to make separate elections with
respect to his Grandfathered Account and his 409A Account, shall obligate the Company to pay duplicate benefits to any Participant.
SECTION 8
REDEFERRALS
8.1 409A Account .
(a) Redeferrals to the DCP . Subject to this Section 8, instead of being paid on the Payment Date, a Participant shall
be permitted to elect, prior to his Separation from Service, to have all or a portion of the vested balance of his 409A Account transferred in a
Valid Notional Rollover on the Participant’s Separation from Service.
(b) Redeferral Requirements . Subject to Section 8.2, the elections described in this Section 8.1 shall be subject to the
following requirements:
13
1. The election to transfer the vested balance of a Participant’s 409A Account in a Valid Notional Rollover
must be made and become irrevocable (other than in the case of the death of the Participant) at least one
year prior to the Payment Date.

8.2 Limitations on Redeferrals . Notwithstanding the foregoing provisions of Section 8.1, no Participant shall be
permitted to elect a notional rollover to the DCP for any portion of his 409A Account following the date of the Participant’
s Separation from
Service.
SECTION 9
CLAIMS PROCEDURE
9.1 General . If a Participant or his Beneficiary or the authorized representative of one of the foregoing (hereinafter,
the “ Claimant ”) does not receive the timely payment of the benefits which he believes are due under the Plan, the Claimant may make a
claim for benefits in the manner hereinafter provided.
9.2 Claims . All claims for benefits under the Plan shall be made in writing and shall be signed by the Claimant.
Claims shall be submitted to the Administrative Record Keeper. If the Claimant does not furnish sufficient information with the claim for
the Administrative Record Keeper (or such other person who is delegated the responsibility by the Committee to review claims) to
determine the validity of the claim, the Administrative Record Keeper shall indicate to the Claimant any additional information which is
necessary for the Administrative Record Keeper to determine the validity of the claim.
9.3 Review of Claims . Each claim hereunder shall be acted on and approved or disapproved by the Administrative
Record Keeper within 90 days following the receipt by the Administrative Record Keeper of the information necessary to process the claim.
If special circumstances require an extension of the time needed to process the claim, this 90-day period may be extended to 180 days after
the claim is received. The Claimant shall be notified before the end of the original period if an extension is necessary, the reason for the
extension and the date by
14
2.
The election shall not become effective for at least one year after the election is made.
3. Any transfer of the 409A Account in a Valid Notional Rollover must be made in accordance with the
applicable terms and provisions of the DCP as then in effect and, once the deferred amount constituting
such 409A Account is credited under the DCP, shall constitute a full and complete settlement of the
Company
’
s obligations to the Participant under the Plan with respect to the 409A Account.
4. If the 409A Account is transferred in a Valid Notional Rollover, the payment commencement date elected
by the Participant under the DCP for the 409A Account must not be earlier than the fifth anniversary of the
Payment Date.

which it is expected that a decision will be made. In the event the Administrative Record Keeper denies a claim for benefits in whole or in
part, the Administrative Record Keeper shall notify the Claimant in writing of the denial of the claim and notify the Claimant of his right to
a review of the Administrative Record Keeper’s decision by the Committee. Such notice by the Administrative Record Keeper shall also set
forth, in a manner calculated to be understood by the Claimant, the specific reason for such denial, the specific provisions of the Plan on
which the denial is based, and a description of any additional material or information necessary to perfect the claim with an explanation of
the Plan’s appeals procedure as set forth in this Section 9.
9.4 Appeals . Any Claimant whose claim for benefits is denied in whole or in part may appeal to the Committee for a
review of the decision by the Administrative Record Keeper. Such appeal must be made within 60 days after the applicant has received
actual or constructive notice of the denial as provided above. An appeal must be submitted in writing within such period and must:
9.5 Review of Appeals
. The Committee shall act upon each appeal within 60 days after receipt thereof unless special
circumstances require an extension of the time for processing, in which case a decision shall be rendered by the Committee as soon as
possible but not later than 120 days after the appeal is received by it. If such an extension of time for processing is required because of
special circumstances, written notice of the extension shall be furnished prior to the commencement of the extension describing the reasons
an extension is needed and the date when the determination will be made. The Committee may require the Claimant to submit such
additional facts, documents or other evidence as the Committee in its discretion deems necessary or advisable in making its review. The
Claimant shall be given the opportunity to review pertinent documents or materials upon submission of a written request to the Committee,
provided that the Committee finds the requested documents or materials are pertinent to the appeal.
9.6 Final Decisions . On the basis of its review, the Committee shall make an independent determination of the
Participant’s eligibility for benefits under the Plan. The decision of the Committee on any appeal of a claim for benefits shall be final and
conclusive upon all parties thereto.
9.7 Denial of Appeals
. In the event the Committee denies an appeal in whole or in part, it shall give written notice of
the decision to the Claimant, which notice shall set forth, in a manner calculated to be understood by the Claimant, the specific reasons for
such denial and which shall make specific reference to the pertinent provisions of the Plan on which the Committee’s decision is based.
15
1.
request a review by the Committee of the claim for benefits under the Plan;
2. set forth all of the grounds upon which the Claimant’s request for review is based and any facts in support
thereof; and
3.
set forth any issues or comments which the Claimant deems pertinent to the appeal.

9.8 Statute of Limitations
. A Claimant wishing to seek judicial review of an adverse benefit determination under the
Plan, whether in whole or in part, must file any suit or legal action, including, without limitation, a civil action under Section 502(a) of
ERISA, within three years of the date the final decision on the adverse benefit determination on review is issued or should have been issued
under Section 9.6 or lose any rights to bring such an action. If any such judicial proceeding is undertaken, the evidence presented shall be
strictly limited to the evidence timely presented to the Committee. Notwithstanding anything in the Plan to the contrary, a Claimant must
exhaust all administrative remedies available to such Claimant under the Plan before such Claimant may seek judicial review pursuant to
Section 502(a) of ERISA.
SECTION 10
AMENDMENT AND TERMINATION
10.1 Amendment or Termination . The Plan may be amended or terminated at any time, by the Board of Directors or
the Committee; provided, however, no amendment or termination may reduce the amount of a Participant’s Deferral Account as of the date
of the amendment or termination without the Participant’s written consent. Upon termination of the Plan, payment of a Participant’
s Deferral
Account shall be made in accordance with the terms of the Plan and the elections in effect prior to such termination unless the Board of
Directors or the Committee, in its discretion, determines to accelerate payment and such acceleration may be effected in a manner that will
not result in the imposition on any Participant of additional tax or penalties under Section 409A (“ Section 409A Compliance ”).
10.2 409A Account Amendments . Notwithstanding any provision in the Plan to the contrary, the Board of Directors,
the Committee or the Deferred Compensation Tax Compliance Committee shall have the independent right, prospectively and/or
retroactively, to amend or modify the Plan in accordance with Section 409A, in each case, without the consent of any Participant, to the
extent that the Board of Directors, the Committee or the Deferred Compensation Tax Compliance Committee deems such action to be
necessary or advisable to address regulatory or other changes or developments that affect the terms of the Plan with the intent of effecting
Section 409A Compliance. Any determinations made by the Board of Directors, the Committee or the Deferred Compensation Tax
Compliance Committee under this Section 10.2 shall be final, conclusive and binding on all persons.
16

SECTION 11
MISCELLANEOUS
11.1 No Effect on Employment Rights . Nothing contained herein shall be construed as a contract of employment
with any person. The Plan and its establishment shall not confer upon any person the right to be retained in the service of the Company or
limit the right of the Company to discharge or otherwise deal with any person without regard to the existence of the Plan.
11.2 Funding . The Plan at all times shall be entirely unfunded, and no provision shall at any time be made with
respect to segregating any assets of the Company for payment of any benefits hereunder. No Participant, Beneficiary or other person shall
have any interest in any particular assets of the Company by reason of a right to receive a benefit under the Plan, and any such Participant,
Beneficiary or other person shall have the rights of a general unsecured creditor of the Company with respect to any rights under the Plan.
Notwithstanding the foregoing, the Committee or the Board of Directors, in its discretion, may establish a grantor trust to fund benefits
payable under the Plan and administrative costs relating to the Plan. The assets of said trust shall be held separate and apart from other
Company funds and shall be used exclusively for the purposes set forth in the Plan and the applicable trust agreement, subject to the
following conditions:
11.3 Anti-assignment
. To the maximum extent permitted by law, no benefit payable under the Plan shall be subject in
any manner to anticipation, alienation, sale, transfer, assignment, pledge, encumbrance, or charge, and any attempt to do so shall be void,
nor shall any such benefit be in any manner liable for or subject to garnishment, attachment, execution or levy, or liable for or subject to the
debts, contracts, liabilities, engagements or torts of the Participant.
11.4 Taxes . The Company shall have the right to deduct any required employment, income or other withholding
Taxes from a Participant’s Deferral Account.
11.5 Construction . This Plan is intended to satisfy the requirements of Section 409A with respect to amounts subject
thereto and shall be interpreted and construed accordingly. The Plan is intended to be an unfunded deferred compensation arrangement for a
select group of management or highly compensated employees within the meaning of ERISA and therefore exempt from the requirements of
Sections 201, 301 and 401 of ERISA. Whenever the terms of the Plan require the payment of an amount by a specified date, the Company
shall use reasonable efforts to make payment by that date. The Company shall not be (i) liable to the
17
1.
the creation of said trust shall not cause the Plan to be other than
“
unfunded
”
for purposes of ERISA;
2. the Company shall be treated as the “grantor” of said trust for purposes of Sections 671 and 677 of the
Code; and
3. said trust agreement shall provide that the trust fund assets may be used to satisfy claims of the Company’
s
general creditors.

Participant or any other person if such payment is delayed for administrative or other reasons to a date that is later than the date so specified
by the Plan or (ii) required to pay interest or any other amount in respect of such delayed payment except to the extent specifically
contemplated by the terms of the Plan.
11.6 Incapacity of Participant . In the event a Participant is declared incompetent and a conservator or other person
legally charged with the care of his person or his estate is appointed, any benefits under the Plan to which such Participant is entitled shall be
paid to such conservator or other person legally charged with the care of his person or estate.
11.7 Severability . In the event that one or more provisions of the Plan shall be or become invalid, illegal or
unenforceable in any respect, the validity, legality and enforceability of the remaining provisions of the Plan shall not be affected thereby.
11.8 Governing Law . The Plan is established under and shall be governed and construed in accordance with the laws
of the State of New Jersey, to the extent that such laws are not preempted by ERISA.
18

FORM OF AMENDMENT TO THE
WYETH SUPPLEMENTAL EMPLOYEE SAVINGS PLAN
The Wyeth Supplemental Employee Savings Plan (the “Plan”)
is hereby amended as follows, effective as of the Closing
Date (as defined in the Amended and Restated Asset Purchase Agreement, dated September 17, 2009) by and among Pfizer Inc., Wyeth and
Boehringer Ingelheim Vetmedica, Inc.:
1. Section 5.2 of the Plan is hereby amended by adding the following to the end thereof:
“Regardless of the number of Years of Vesting Service, a Participant who is treated as an Affected
Employee under the terms of the Amended and Restated Asset Purchase Agreement, dated
September 17, 2009, by and among Pfizer Inc., Wyeth and Boehringer Ingelheim Vetmedica, Inc. (the
“Boehringer Agreement”) shall become 100% vested in his Matching Contributions as of the Closing
Date (as defined under the Boehringer Agreement).”

FORM OF AMENDMENT TO THE
WYETH SUPPLEMENTAL EMPLOYEE SAVINGS PLAN
The Wyeth Supplemental Employee Savings Plan (the “Plan”)
is hereby amended as follows, effective as of the Closing
Date (as defined in the Agreement and Plan of Merger, dated as of January 25, 2009, by and among Pfizer, Inc., Wagner Acquisition Corp.,
and Wyeth):
1. Section 1.2(o) of the Plan is hereby amended in its entirety to read as follows:
“(o) “ Company Stock Fund ” means the Investment Option available under the Plan that is designed
to track the performance of the common stock of Pfizer Inc., par value $0.05 per share, and any
successor thereof.”
2. Section 5.2 of the Plan is hereby amended by adding the following to the end thereof:
“Notwithstanding the foregoing, all Participants who are employed by the Company on the Closing
Date (as defined in the Agreement and Plan of Merger, dated as of January 25, 2009, by and among
Pfizer, Inc., Wagner Acquisition Corp., and Wyeth) shall become 100% vested in the Matching
Contributions credited to their 409A Account as of the Closing Date. Participants who are on an
approved leave of absence on the Closing Date shall become 100% vested in the Matching
Contributions credited to their 409A Account as of the Closing Date.
”

FORM OF AMENDMENTS TO THE
WYETH SUPPLEMENTAL EMPLOYEE SAVINGS PLAN, WYETH
SUPPLEMENTAL EXECUTIVE RETIREMENT PLAN, WYETH
DEFERRED COMPENSATION PLAN AND THE WYETH
EXECUTIVE RETIREMENT PLAN (COLLECTIVELY, THE “PLANS”)
The Plans are hereby amended, effective as of January 1, 2011, by deleting the following language “within ninety (90) days after
the date of the Participant’s death,” in each of the plan sections listed below, and replacing it with “in the January following the calendar
year in which the Participant’s death occurs”:
1.
Wyeth Supplemental Employee Savings Plan
–
Section 1.2(r);
2.
Wyeth Supplemental Executive Retirement Plan
–
Sections 5.7, 6.5, 6.6 and 6.8;
3.
Wyeth Executive Retirement Plan
–
Sections 5.7, 6.5, 6.6 and 6.8; and
4.
Wyeth Deferred Compensation Plan
–
Sections 7.4 and 7.6.

FORM OF WRITTEN ACKNOWLEDGEMENT AND CONSENT
TERMINATION OF PARTICIPATION
WYETH SUPPLEMENTAL EMPLOYEE SAVINGS PLAN (THE “PLAN”)
Effective on and after January 1, 2012, it is hereby acknowledged that the Plan is “frozen” and no further deferrals may be made
under the Plan.

Exhibit 10(12)
WYETH
SUPPLEMENTAL EXECUTIVE RETIREMENT PLAN
(amended and restated effective as of January 1, 2005)
PURPOSE
The Plan supplements the benefits of Participants whose benefits under the Retirement Plan are limited as a result of Deferrals or by operation of the Code
Limits. The Plan is intended to constitute an unfunded deferred compensation plan for a select group of management or highly compensated employees within the
meaning of ERISA and shall be construed and administered accordingly.
The Plan is an amendment and restatement of the Prior Plan, effective as of the Restatement Date.
Capitalized terms not otherwise defined in the text hereof shall have the meanings set forth in Section 1.
SECTION 1 DEFINITIONS
1.1 Rules of Construction . Except where the context indicates otherwise, any masculine terminology used herein shall also include the feminine gender,
and the definition of any term herein in the singular shall also include the plural. All references to sections and appendices are, unless otherwise indicated, to sections or
appendices of the Plan.
1.2 Terms Defined in the Plan . Whenever used herein, the following terms shall have the meanings set forth below:
(a) “ 25, 50, 75 or 100% Joint and Survivor Annuity ” has the meaning set forth in Section 5.6(a)(2).
(b) “ 409A Benefit ” has the meaning set forth in Section 4.4(b).
(c) “ Administrative Record Keeper ” means the person or persons designated by the Committee in accordance with Section 2.
(d) “ Affiliate ” means any corporation which is included in a controlled group of corporations (within the meaning of Section 414(b) of the Code) which
includes Wyeth and any trade or business (whether or not incorporated) which is under common control with Wyeth (within the meaning of Section 414(c) of the
Code); provided , however , that in applying Section 1563(a)(1), (2), and (3) of the Code for purposes of determining a controlled group of corporations under
Section 414(b) of the Code the language “at least 50 percent” shall be used instead of “at least 80 percent” in each place it appears in Section 1563(a)(1), (2) and (3) of
the Code, and in applying Section 1.414(c)-2 of the Treasury Regulations, for purposes of determining trades or businesses (whether or not incorporated) that are under
common control for purposes of Section 414(c) of the Code, “at least 50 percent” shall be used instead of “at least 80 percent” in each place it appears in Section 1.414
(c)-2 of the Treasury Regulations.
(e) “ Beneficiary ” means, with respect to death benefits payable under Sections 5.2(c), 5.3(e), 5.6(a)(3), 5.6(a)(4) and 5.7, as applicable, a Participant’s
Surviving Spouse or, if there is no Surviving Spouse, the Participant’s estate. Participants shall not be permitted or required to make Beneficiary designations under the
Plan. If the Surviving Spouse of a Participant is legally impaired or prohibited from receiving any amounts under the Plan otherwise payable to a Beneficiary, the
Participant’s Beneficiary shall be the Participant’s estate. The term Beneficiary shall not refer to any “contingent annuitant” applicable to a Participant in connection
with a Payment Form.
(f) “ Board of Directors ” means the Board of Directors of Wyeth (or any Committee of the Board of Directors to whom the Board of Directors delegates,
from time to time, its authority hereunder).
(g)
“
Business Day
”
means each day on which the New York Stock Exchange is open for business.

(h) “ Claimant ” has the meaning set forth in Section 8.1.
(i) “ Code ” means the Internal Revenue Code of 1986, as amended, and any applicable rulings and regulations promulgated thereunder.
(j) “ Code ” means the Internal Revenue Code of 1986, as amended, and any applicable rulings and regulations promulgated thereunder.
(k) “ Code Limits ” means Sections 401(a)(17) and 415 of the Code and any other provisions of the Code which limit the amount of benefits that a
Participant may accrue or receive under or from the Retirement Plan.
(l) “ Committee ”
means the committee of such officers and/or employees of the Company as shall be designated from time to time by Wyeth to administer
the Plan and any successor thereto.
(m) “ Company ” means Wyeth and its Affiliates.
(n) “ Company Non-Account Plan ” means any arrangement sponsored by the Company, other than the Plan, that is a “non-account balance plan,” as such
term is defined under Section 409A and that is required to be aggregated with the Plan under Treasury Regulation 1.409A-1(c)(2)(C).
(o) “ DCP ” means the Prior DCP and the New DCP.
(p) “ DCP Option ” has the meaning set forth in Section 5.6(a)(6).
(q) “ Default Payment Form ” means (i) with respect to a Participant’s Grandfathered Benefit, the form of payment elected by such Participant under the
Retirement Plan in connection with the Participant’s Separation from Service; and (ii) with respect to a Participant’s 409A Benefit, the Lump-Sum Option.
(r) “ Deferral Plan ” means each of the DCP, the Wyeth Supplemental Employee Savings Plan, as amended from time to time, and/or any other non-
qualified plan of the Company designated from time to time by the Committee pursuant to which Participants may elect to defer annual, base compensation or annual,
cash bonus compensation, sales bonuses or sales commissions.
(s) “ Deferrals ” means any cash compensation earned by a Participant from the Company that is not taken into account in determining a Participant’s
accrued benefit under the Retirement Plan because of the Participant’s election under a Deferral Plan to defer the receipt of such compensation.
(t) “ Deferred Compensation Tax Compliance Committee ” means a committee of such officers and/or employees of the Company as shall be designated
from time to time by the Board of Directors.
(u) “ Delayed Payment Amount ” has the meaning set forth in Section 5.7.
(v) “ Early Commencement Factors ” means the factors set forth in Appendix A .
(w) “ Elected Payment Date ” means (i) with respect to the Grandfathered Benefit, the first day of any month after a Participant’s Separation from Service
elected by the Participant in accordance with Section 5.2 and/or (ii) with respect to the 409A Benefit, the Normal Payment Date, unless the Participant elects the DCP
Option in accordance with Section 5.3, or elects to redefer his 409A Benefit into the DCP in accordance with Section 7, in which case Elected Payment Dates shall be
determined in accordance with the applicable terms of the DCP.
(x) “ Elected Payment Form ” means the Payment Form elected by a Participant (i) for the payment of his Grandfathered Benefit in accordance with
Section 5.2, and/or (ii) for the payment of his 409A Benefit in accordance with Section 5.3 or Section 7.
(y) “ Eligible Employee ” means an employee of the Company (i) whose terms and conditions of employment are not subject to a collective bargaining
agreement, (ii) whose rate of annual base compensation for a calendar year equals or exceeds $155,000.00, and (iii) who is eligible to participate in the Retirement Plan.
Notwithstanding the foregoing, an individual shall not become an
“Eligible Employee” until the first day of the month following the date on which such individual
satisfies the requirement of clause (iii) of the previous sentence. Further, the term “ Eligible Employees ” shall exclude individuals classified by the Company as leased
employees, independent contractors or consultants or any individuals who are not paid through the Company
’
s regular payroll.

(z) “ ERISA ” means the Employee Retirement Income Security Act of 1974, as amended from time to time, including any applicable rulings and
regulations promulgated thereunder.
(aa) “ Grandfathered Benefit ” means the portion of a Participant’s Plan Benefit that, for purposes of Section 409A, was both earned and vested as of
December 31, 2004.
(bb) “ Guaranteed Death Benefit Option ” has the meaning set forth in Section 5.6(a)(4).
(cc) “ Key Employee ” means (i) each “specified employee,” as defined in Section 409A(a)(2)(B)(i) of the Code, who meets the requirements of
Section 416(i)(1)(A)(i), (ii) or (iii) of the Code (applied in accordance with the regulations thereunder and disregarding Section 416(i)(5) of the Code) at any time during
the 12-month period ending on December 31 of a calendar year and (ii) to the extent not otherwise included in (i) hereof, each of the top-100 paid individuals (based
on taxable wages for purposes of Section 3401(a) of the Code as reported in Box 1 of Form W-2 for the 12-month period ending on December 31 of such calendar
year plus amounts that would be included in wages for such 12 month period but for pre-tax deferrals to a tax-qualified retirement plan or cafeteria plan or for qualified
transportation benefits) who performed services for the Company at any time during the 12-month period ending on December 31 of such calendar year. A Participant
shall be treated as a Key Employee for the 12-month period beginning on April 1 of the calendar year following the calendar year for which the determination under
clause (i) or (ii) of this definition is made.
(dd) “ Lump-Sum Option ” has the meaning set forth in Section 5.6(a)(5).
(ee) “ New DCP ” means the Wyeth 2005 (409A) Deferred Compensation Plan, as amended and restated as of the Restatement Date, and as subsequently
amended from time to time thereafter.
(ff) “ Normal Retirement Date ” means the first day of the first month following a Participant’s 65 birthday, unless such birthday falls on the first of the
month, in which case Normal Retirement Date means the Participant’s 65 birthday.
(gg) “ Normal Payment Date ” means (i) with respect to a Participant’s Grandfathered Benefit, the first day of the month on which benefits commence to
be paid to the Participant under the Retirement Plan; and (ii) with respect to a Participant’s 409A Benefit, the following: (A) for a Participant who incurs a Separation
from Service with a Vested Plan Benefit prior to attaining age 55, the first day of the month coincident with or next following the month in which he attains age 55; and
(B) for a Participant who incurs a Separation from Service with a Vested Plan Benefit on or after attaining age 55, the first day of the month following his Separation
from Service.
(hh) “ Participant ” means an Eligible Employee who has met the requirements for participation in the Plan in accordance with Section 3.
(ii) “ Payment Date ” means the Elected Payment Date or, if no such date has been elected or is permitted to be elected by the Participant, the Normal
Payment Date, in each case for the commencement of payment of a Plan Benefit.
(jj) “ Payment Delay Period ” means, solely with respect to a Lump-Sum Option payment of a Participant’s Grandfathered Benefit, the twelve-month
period beginning on the first day of the month following the month in which occurs the Participant’s Separation from Service.
(kk) “ Payment Election ” means the elections made by a Participant for his Grandfathered Benefit and/or 409A Benefit, as applicable, under Section 5 or
Section 7, as applicable.
(ll) “ Payment Form ” means the Elected Payment Form or, if no such form is elected or is permitted to be elected by the Participant, the Default Payment
Form, in each case for the payment of a Plan Benefit.
(mm) “ Plan ” means this Wyeth Supplemental Executive Retirement Plan, as amended from time to time.
(nn) “ Plan Benefit ” means, as of a given date, the benefit, expressed as a Single Life Annuity commencing at the Participant’s Normal Retirement Date,
that a Participant has accrued under the Plan in accordance with Section 4.2.
st
st
st
st
th
th

(oo) “ Prior DCP ” means the terms of the Wyeth Deferred Compensation Plan (as amended and restated as of November 20, 2003), as set forth in the
Company’s written documentation, rules, practices and procedures applicable to such plan (but without regard to any amendments thereto after October 3, 2004 that
would result in any material modification of such plan, within the meaning of Section 409A).
(pp) “ Prior Plan ” means the terms of the Plan in effect immediately prior to the Restatement Date, as set forth in the Company’s written documentation,
rules, practices and procedures applicable to the Plan (but without regard to any amendments thereto after October 3, 2004 that would result in any material modification
of the Grandfathered Benefit, within the meaning of Section 409A).
(qq) “ Puerto Rico Participant ” means a Participant employed by the Company in Puerto Rico and who resides in Puerto Rico.
(rr) “ Restatement Date ” means January 1, 2005.
(ss) “ Retirement Eligible ” means a Participant who, as of the date of his Separation from Service, is (i) at least age 55 with at least five Years of Vesting
Service or (ii) at least age 65.
(tt) “ Retirement Plan ” means the Wyeth Retirement Plan – United States, as amended from time to time.
(uu) “ Rule of 70 Participant ” means a Participant who as of the date of his Separation from Service has a Vested Plan Benefit (other than a Participant
employed at the Company’s facilities in Rouses Point, New York) and who (i) is involuntarily terminated by the Company prior to February 6, 2011 in connection with
Project Impact and (ii) as of the date of his Separation from Service, has a combined age and Years of Vesting Service equal to or in excess of 70; provided , however ,
that with respect to Participants employed by Genetics Institute prior to American Home Products purchase of Genetics Institute, and solely for purposes of determining
whether a Participant is a Rule of 70 Participant, such Participant’s service with Genetics Institute on or after January 1, 1992 shall be taken into account in determining
such Participant’s Years of Vesting Service.
(vv) “ Section 409A ” means Section 409A of the Code and the applicable notices, rulings and regulations promulgated thereunder.
(ww) “ Section 409A Compliance ” has the meaning set forth in Section 9.1.
(xx) “ Separation from Service ”
means a separation from service with the Company for purposes of Section 409A, determined using the default provisions
set forth in Treasury Regulation Section 1.409A-1(h); provided , however , that, for purposes of the Grandfathered Benefit, “ Separation from Service ” shall be
determined in accordance with the terms of the Prior Plan. Notwithstanding the foregoing, if a Participant would otherwise incur a Separation from Service in
connection with a sale of assets of the Company, the Company shall retain the discretion to determine whether a Separation from Service has occurred in accordance
with Treasury Regulation Section 1.409A-1(h)(4).
(yy) “ Single Life Annuity ” has the meaning set forth in Section 5.6(a)(1).
(zz) “ Surviving Spouse ” means the individual to whom a Participant was legally married, for federal law purposes, for a continuous period of at least one
year as of the date of the Participant’s death.
(aaa) “ Ten Year Certain and Life Option ” has the meaning set forth in Section 5.6(a)(3).
(bbb) “ Transition Elections ” means elections made by a Participant prior to January 1, 2009 in accordance with the provisions of Notices 2005-1, 2006-
79
and 2007-86 promulgated by the U.S. Treasury Department and the Internal Revenue Service and the Proposed Regulations under Section 409A, 70 Fed. Reg. 191 (Oct
4, 2005).
(ccc) “ Treasury Regulations ” means the regulations adopted by the Internal Revenue Service under the Code, as they may be amended from time to time.
(ddd) “ Valid Notional Rollover ” means a notional rollover constituting a full and complete settlement of the Company’s obligations to the Participant
with respect to the portion of the Grandfathered Benefit credited to the Prior DCP or the 409A Benefit credited to the New DCP by a Participant who is Retirement
Eligible at the time of his Separation from Service.

(eee) “ Vested Plan Benefit ” means a Plan Benefit that has vested in accordance with Section 4.3.
(fff) “ Wyeth ” means Wyeth, a Delaware corporation, and any successor thereto.
(ggg) “ Wyeth Retirement Plans ” means the Retirement Plan, the American Cyanamid and Subsidiaries Supplemental Employees Retirement Plan and the
American Cyanamid and Subsidiaries ERISA Excess Plan.
(hhh) “ Year of Vesting Service ” has the meaning ascribed to it in the Retirement Plan as of January 1, 2006 and, prior to such date, has the meaning
ascribed to “Continuous Service”, as such term was defined in the Retirement Plan prior to January 1, 2006.
SECTION 2 ADMINISTRATION
2.1 General Authority . The general supervision of the Plan shall be the responsibility of the Committee, which, in addition to such other powers as it may
have as provided herein, shall have the power, subject to the terms of the Plan: (i) to determine eligibility to participate in, and the amount of benefit to be provided to
any Participant under, the Plan; (ii) to make and enforce such rules and regulations as it shall deem necessary or proper for the efficient administration of the Plan;
(iii) to determine all questions arising in connection with the Plan, to interpret and construe the Plan, to resolve ambiguities, inconsistencies or omissions in the text of
the Plan, to correct any defects in the text of the Plan and to take such other action as may be necessary or advisable for the orderly administration of the Plan; (iv) to
make any and all legal and factual determinations in connection with the administration and implementation of the Plan; (v) to designate the Administrative Record
Keeper and to review actions taken by the Administrative Record Keeper or any other person to whom authority is delegated under the Plan; and (vi) to employ and rely
on legal counsel, actuaries, accountants and any other agents as may be deemed to be advisable to assist in the administration of the Plan. All such actions of the
Committee shall be conclusive and binding upon all persons. The Committee shall be entitled to rely conclusively upon all tables, valuations, certificates, opinions, and
reports furnished by any actuary, accountant, controller, counsel, or other person employed or engaged by the Company with respect to the Plan. If any member of the
Committee is a Participant, such member shall not resolve, or participate in the resolution of, any matter relating specifically to such Committee member’s eligibility to
participate in the Plan or the calculation or determination of such member’s Plan Benefit.
2.2 Delegation
. The Committee shall have the power to delegate to any person or persons the authority to carry out such administrative duties, powers and
authority relative to the administration of the Plan as the Committee may from time to time determine. Any action taken by any person or persons to whom the
Committee makes such a delegation shall, for all purposes of the Plan, have the same force and effect as if undertaken directly by the Committee. If any individual to
whom the Committee delegates authority is a Participant, such individual shall not resolve, or participate in the resolution of, any matter specifically relating to such
individual’s eligibility to participate in the Plan or the calculation or determination of such individual’s Plan Benefit.
2.3 Administrative Record Keeper . The Administrative Record Keeper shall be responsible for the day-to-day operation of the Plan, having the power
(except to the extent such power is reserved to the Committee) to take all action and to make all decisions necessary or proper in order to carry out his duties and
responsibilities under the provisions of the Plan. If the Administrative Record Keeper is a Participant, the Administrative Record Keeper shall not resolve, or participate
in the resolution of, any question which relates directly or indirectly to him and which, if applied to him, would significantly vary his eligibility for, or the amount of,
any benefit to him under the Plan. The Administrative Record Keeper shall report to the Committee at such times and in such manner as the Committee shall request
concerning the operation of the Plan.
2.4 Actions; Indemnification . The members of the Board of Directors, the Committee, the Administrative Record Keeper, the members of the Deferred
Compensation Tax Compliance Committee, the members of any other committee and any director, officer or employee of the Company to whom responsibilities are
delegated by the Committee shall not be liable for any actions or failure to act with respect to the administration or interpretation of the Plan, unless such person acted in
bad faith or engaged in fraud or willful misconduct. The Company shall indemnify and hold harmless, to the fullest extent permitted by law, the Board of Directors (and
each member thereof), the Committee (and each member thereof), the Deferred Compensation Tax Compliance Committee (and each member thereof), the
Administrative Record Keeper, the members of any other committee and any director, officer or employee of the Company to whom responsibilities are delegated by the
Committee from and against any liabilities, damages, costs and expenses (including attorneys
’
fees and amounts paid in settlement of

any claims approved by the Company) incurred by or asserted against it or him by reason of its or his duties performed in connection with the administration or
interpretation of the Plan, unless such person acted in bad faith or engaged in fraud or willful misconduct. The indemnification, exculpation and liability limitations of
this Section 2.4 shall apply to the Administrative Record Keeper only to the extent that the Administrative Record Keeper is or was a director, officer or employee of the
Company.
SECTION 3 PARTICIPATION
3.1 Continuing Participants . Any individual who participated in the Prior Plan immediately prior to the Restatement Date shall continue to be a
Participant in the Plan on the Restatement Date.
3.2 New Participants . An employee of the Company who does not become a Participant in the Plan in accordance with Section 3.1 shall commence
participation in the Plan as of the date on which such employee first becomes an Eligible Employee. Eligible Employees shall not accrue any Plan Benefit prior to their
commencement of participation in the Plan; provided that when participation commences a Participant’s accrued Plan Benefit shall be calculated as of the later of the
date the Participant was first employed by the Company and the date the Participant reached age 21.
3.3 Enrollment . Each Participant shall complete, execute and return to the Administrative Record Keeper such forms as are required from time to time by
the Administrative Record Keeper, and such forms shall be submitted to the Administrative Record Keeper within such time periods specified by the Administrative
Record Keeper. A Participant’s failure to submit in a complete and timely manner any such forms to the Administrative Record Keeper shall subject the Participant to
the default rules specified in the Plan. For purposes of the Plan, “forms” prescribed by the Administrative Record Keeper can be in paper, electronic or such other media
(or combination thereof) as the Administrative Record Keeper shall specify from time to time.
3.4 Exclusions . No employee of the Company who is not an Eligible Employee shall be eligible to participate in the Plan. In addition, the Committee
may, if it determines it to be necessary or advisable to comply with ERISA, the Code or other applicable law, exclude one or more Eligible Employees or one or more
classes of Eligible Employees from Plan participation.
SECTION 4 PLAN FORMULA AND VESTING
4.1 Applicability of Prior Plan
. The benefit payable to a Participant who had a Separation from Service prior to the Restatement Date shall be governed by
the terms of the Prior Plan as in effect on the date of his Separation from Service.
4.2. Plan Benefit Formula . The Plan Benefit of a Participant who has a Separation from Service on or after the Restatement Date shall equal the positive
difference, if any, that results from subtracting the amount determined under Section 4.2(b) from the amount determined under Section 4.2(a):
(a) The Participant’s annual accrued benefit under the terms of the “Final Average Annual Pension Earnings” formula of the Retirement Plan calculated as
of the date of the Participant’s Separation from Service as if:
less
(b) The Participant’s annual accrued benefit under the Wyeth Retirement Plans, as of the date of the Participant’s Separation from Service.
4.3 Vesting . Anything in the Plan to the contrary notwithstanding, no Plan Benefit or other amount shall be payable to a Participant under the Plan unless
the Participant has either (i) completed five Years of Vesting Service or (ii) is at least age 65, in each case, as of the date of the Participant’s Separation from Service.
4.4
Plan Benefit Components
.
1. for purposes of calculating such accrued benefit, the Participant’s compensation for each calendar year included the Participant’s Deferrals for each
such calendar year; and
2.
for purposes of calculating such accrued benefit, the Code Limits did not apply.

(a) Grandfathered Benefit .
(b) 409A Benefit . A Participant’s 409A Benefit shall mean any portion of the Participant’s Plan Benefit which is not a Grandfathered Benefit.
(c) Special Adjustment at Separation from Service to the 409A Benefit . Solely to the extent necessary to comply with Section 409A, a special allocation
shall be made to the Plan Benefit of a Participant who was not eligible to retire under the Plan as of December 31, 2004 with a subsidized early retirement benefit (solely
by reason of the Participant as of December 31, 2004 not having ten or more Years of Vesting Service as of such date) and who subsequently becomes eligible to retire
under the Plan with a subsidized early retirement benefit (including on account of becoming a Rule of 70 Participant) at a later date. For such a Participant, any early
retirement subsidy earned by the Participant based on Years of Vesting Service credited for periods after December 31, 2004 and attributable to the Participant’s
Grandfathered Benefit shall be treated for all purposes of the Plan as part of the Participant’
s 409A Benefit. The adjusted 409A Benefit (including the subsidized portion
of the Grandfathered Benefit that is treated by operation of this Section 4.4(c) as part of the 409A Benefit) shall be determined at the time of the Participant’s Separation
from Service by the formula [(X – Y)/Z], where “ X ” is the Plan Benefit multiplied by the applicable subsidized Early Commencement Factor set forth in Appendix A
;
where “ Y ” is the Grandfathered Benefit multiplied by the applicable unsubsidized Early Commencement Factor set forth in Appendix A ; and where “ Z ” is the
applicable subsidized Early Commencement Factor set forth in Appendix A (all such Early Commencement Factors to be determined based upon the Participant’s
(including on account of becoming a Rule of 70 Participant) age and Years of Vesting Service at Separation from Service).
(d) Other Actuarial Rules and Procedures . The Committee shall from time to time promulgate such additional rules and procedures as the Committee
deems necessary or advisable to facilitate the calculation and allocation of a Participant’s Plan Benefit between the Grandfathered Benefit and the 409A Benefit in a
manner that is intended to result in Section 409A Compliance.
4.5 Payment Prior to Normal Retirement . If the Payment Date for a Participant’s Grandfathered Benefit and/or 409A Benefit, as applicable, is prior to the
Participant’s Normal Retirement Date, then the amount of the Grandfathered Benefit and/or 409A Benefit, as applicable, shall be reduced for early commencement by
the applicable Early Commencement Factors set forth in Appendix A .
SECTION 5 PAYMENT ELECTIONS
5.1 General Rules .
(a) Separate Elections . Subject to Section 5.3 hereof, a Participant shall be permitted to make a separate Payment Election for his Grandfathered Benefit
and his 409A Benefit. The rules applicable to Payment Elections for Grandfathered Benefits are set forth in Section 5.2. The rules applicable to Payment Elections for
409A Benefits are set forth in Section 5.3.
(b) Section 409A Transition . The Transition Elections made by a Participant shall supplement and, to the extent inconsistent therewith, shall supersede
the corresponding provisions of this Section 5.
1. The portion of a Participant’s Plan Benefit which is a Grandfathered Benefit (and the procedures applicable to a Participant’s election to receive such
Grandfathered Benefit, which are set forth in Section 5.2) shall be based upon the terms of the Prior Plan and the Retirement Plan in effect immediately
prior to the Restatement Date, disregarding for this purpose any change or amendment to the terms of the Retirement Plan effective after October 3,
2004 that would result in any material modification, within the meaning of Section 409A of the Grandfathered Benefit.
2. The Grandfathered Benefit of a Puerto Rico Participant shall comprise (i) the portion of his Plan Benefit that was earned and vested as of December 31,
2004 and (ii) the portion of his Plan Benefit that was earned or vested on or after January 1, 2005, but only in the event such Puerto Rico Participant
does not become employed by the Company in the United States (other than in Puerto Rico) on or after January 1, 2005.
3. A Participant’s Grandfathered Benefit shall not be increased if the payment of the Grandfathered Benefit is made after the Participant’s Normal
Retirement Date.

(c) No Duplicate Benefits
. Nothing in the Plan, including the ability of a Participant to make separate Payment Elections with respect to his Grandfathered
Benefit and his 409A Benefit, shall obligate the Company to pay duplicate benefits to any Participant.
5.2 Payment Elections for Grandfathered Benefits .
(a) Election Form and Election Timing . A Participant may elect prior to or in connection with his Separation from Service to have his Grandfathered
Benefit paid in any of the available forms of payment described in Section 5.6. The Elected Payment Form for a Grandfathered Benefit may be different from the form
of payment elected by the Participant under the Retirement Plan. A Participant shall make his Payment Election for his Grandfathered Benefit prior to the date of, or in
connection with, the Participant’s Separation from Service, and if no Payment Election is made prior to the date of, or in connection with, the Participant’s Separation
from Service, the Participant’s Grandfathered Benefit shall be payable in the Default Payment Form on the applicable Normal Payment Date.
(b) Payment Date for Annuities . If the Payment Form for a Participant’s Grandfathered Benefit is other than the Lump-Sum Option or the DCP Option,
the payment of the Participant’s Grandfathered Benefit shall commence on the Participant’s applicable Normal Payment Date, unless the Participant has specified an
Elected Payment Date. An Elected Payment Date for an annuity shall not be earlier than the first day of the month coincident with or next following the month in which
a Participant attains age 55, and shall not be later than the Participant’s Normal Retirement Date (or, if the Participant’s Separation from Service is later, the first day of
the month following the month in which occurs the Participant’s Separation from Service).
(c) Payment Dates for Lump-Sum Option . A Participant shall not be permitted to specify an Elected Payment Date for his Grandfathered Benefit if such
Grandfathered Benefit is payable in the Lump-Sum Option. The Payment Date for such Lump-Sum Option shall be determined in accordance with the following
provisions:
If payment of a Participant’s Lump-Sum Option is delayed under this Section 5.2(c) solely by operation of the Payment Delay Period, the Participant’s Grandfathered
Benefit shall be credited with interest on a quarterly basis during the applicable portion of the Payment Delay Period based upon the interest rate being used to determine
Lump-Sum Option payments under the Retirement Plan for each such quarter. In the event a Participant dies during the Payment Delay Period, his Grandfathered
Benefit shall be paid to his Beneficiary together with any interest credited thereto in a lump-sum payment as soon as administratively practicable after such Participant’s
death.
(d) Valid Notional Rollovers to the Prior DCP . A Participant who elects prior to, or in connection with, his Separation from Service to receive his
Grandfathered Benefit in the Lump-Sum Option shall be permitted, in accordance with the deferral rules of the Prior Plan, to elect prior to, or in connection with, his
Separation from Service the DCP Option for some or all of the amount otherwise payable in the Lump-Sum Option. The effective date of the Valid Notional Rollover
made in connection with the DCP Option will be the date that the portion of the Lump-Sum Option subject to the Valid Notional Rollover would otherwise have been
paid to the Participant under Section 5.2(c) (determined, solely for this purpose, without regard to the Payment Delay Period). Any such Valid Notional Rollover shall
be subject to the applicable terms and provisions of the Prior DCP. Notwithstanding anything herein to the contrary, no amount shall be distributed under the Prior DCP
on account of a Valid Notional Rollover prior to the conclusion of the Payment Delay Period.
(e) Special Default Rule . If the portion of a Participant’s Plan Benefit that is intended to be a Grandfathered Benefit shall, for any reason, become subject
to Section 409A, such benefit shall be paid in accordance with the Payment Election (or applicable default payment rule) for such Participant
’
s 409A Benefit.
1. Participants Who Are Not Retirement Eligible . If a Participant who is not Retirement Eligible at the time of his Separation from Service has elected prior
to, or in connection with, his Separation from Service the Lump-Sum Option for the payment of his Grandfathered Benefit, such Lump-Sum Option shall
be paid on the later of (i) the first day of the first month following the expiration of the Payment Delay Period and (ii) the first day of the month coincident
with or next following the month in which the Participant attains age 55.
2. Participants Who Are Retirement Eligible . If a Participant who is Retirement Eligible at the time of his Separation from Service has elected prior to, or in
connection with, his Separation from Service the Lump-Sum Option for the payment of his Grandfathered Benefit, such Lump-Sum Option shall be paid
on the first day of the first month following the end of the Payment Delay Period.

5.3 Payment Elections for 409A Benefits .
(a) Election Timing; Participants Who Accrue a Plan Benefit Prior to January 1, 2009 . An employee who first becomes a Participant and accrues a 409A
Benefit prior to January 1, 2009, and an employee who is hired prior to November 1, 2008 with an annual base salary of $230,000.00 or more (the “ 2008 New
Executives ”) shall make, by no later than December 31, 2008, a Transition Election with respect to his 409A Benefit; provided , however , that an election made in
2008 shall apply solely to the amount that would not otherwise be payable to him in 2008 and shall not cause any amounts to be paid to him in 2008 that would not
otherwise be payable to him in 2008. For purposes of clarification, a Participant accrues a benefit under the Plan only to the extent that a Participant’s benefits under the
Retirement Plan are limited as a result of Deferrals or by operation of Code Limits.
(b) Payment Date for Participants Who Accrue a Plan Benefit Prior to January 1, 2009 . An employee who first becomes a Participant and accrues a Plan
Benefit prior to January 1, 2009 shall receive or commence receiving payment of his 409A Benefit on the Participant’s applicable Normal Payment Date, unless (i) the
Participant (A) elects in accordance with his Transition Election the DCP Option for all or a portion of his 409A Benefit and (B) specifies an Elected Payment Date in
accordance with this Section 5.3 or (ii) the Participant makes a redeferral election in accordance with Section 7.
(c) Payment Forms for Participants Who Accrue a Plan Benefit Prior to January 1, 2009
. An employee who first becomes a Participant and accrues a Plan
Benefit prior to January 1, 2009 may elect to receive his 409A Benefit in any of the available forms of payment described in Section 5.6. The Elected Payment Form for
a 409A Benefit may be different than the form of payment elected by the Participant under the Retirement Plan. If a Participant does not specify an Elected Payment
Form for his 409A Benefit, such Participant’s 409A Benefit shall be paid in the Default Payment Form. A Participant may only elect one payment form for his
409A Benefit, unless he elects the DCP Option. In the event a Participant elects to receive a portion of his 409A Benefit in the form of the DCP Option, the remainder of
the Participant’s Plan Benefit shall be paid in the Default Payment Form.
(d) Special Rule for Certain Executives Hired in 2008 . A 2008 New Executive shall be entitled to make a contingent Payment Election prior to
December 31, 2008 with respect to any 409A Benefit to which he may be entitled in the future. A 2008 New Executive shall be permitted to make the same Payment
Elections with respect to his 409A Benefit, as an employee who first becomes a Participant and accrues a Plan Benefit prior to January 1, 2009.
(e) Separation from Service in 2009 . If a Participant described in Section 5.3(a) makes a Payment Election during 2008, incurs a Separation from Service
between January 1, 2009 and December 31, 2009 and has elected to receive his 409A Benefit in a Lump-Sum Option, such payment of the Lump-Sum Option shall not
be made until January 1, 2010. If the payment of a Lump-Sum Option is delayed beyond the Normal Payment Date in accordance with the previous sentence, a
Participant’s 409A Benefit shall be credited with interest on a quarterly basis based upon the interest rate being used to determine Lump-Sum Option payments under
the Retirement Plan for each quarter of such delay. In the event a Participant dies during the period of any such delay, his 409A Benefit shall be paid to his Beneficiary
together with any interest credited thereto in a lump-sum payment on the tenth day of the month following the date of such Participant’s death.
(f) Payment Date and Payment Form for Participants Who Accrue a Plan Benefit On or After January 1, 2009 . A Participant who first accrues a Plan
Benefit on or after January 1, 2009 (other than a 2008 New Executive), shall receive his 409A Benefit on the Normal Payment Date and in the Default Payment Form.
Such Participant shall not be permitted to select an Elected Payment Date or an Elected Payment Form; provided , however , that such Participant shall be permitted to
make a redeferral election in accordance with Section 7.
(g) Payment Date and Payment Form for Participants Who Transfer from Puerto Rico to the United States
. Notwithstanding anything in Section 5.3 to the
contrary, a Puerto Rico Participant shall receive his 409A Benefit on the Normal Payment Date and in the Default Payment Form. Such Puerto Rico Participant shall not
be permitted to select an Elected Payment Date or an Elected Payment Form; provided , however , that such Puerto Rico Participant shall be permitted to make a
redeferral election in accordance with Section 7.
(h) Rehire
. Notwithstanding the foregoing provisions of Section 5.3, an Eligible Employee who is rehired by the Company or otherwise again becomes an
Eligible Employee, after accruing a 409A Benefit under the Plan or a benefit under any other Company Non-Account Plan shall not be entitled to make a Payment
Election. In

the event such an Eligible Employee previously Separated from Service with the Company, payment of his 409A Benefit accrued prior to such Separation from Service
shall not be suspended or otherwise delayed and any additional 409A Benefit accrued by such an Eligible Employee shall be paid on the Normal Payment Date and in
the Default Payment Form. In the event such an Eligible Employee did not incur a Separation from Service, the additional benefit accrued by the Participant shall be
distributed on the Payment Date and in the Payment Form applicable to the 409A Benefit previously accrued by the Participant.
(i) Modifying a Payment Form
. A employee who first becomes a Participant and accrues a Plan Benefit prior to January 1, 2009 and who elects to receive
his 409A Benefit in an annuity Payment Form described in Section 5.6(a)(1) or (2) may, at any time prior to the Payment Date for such 409A Benefit, elect to have his
409A Benefit paid in another annuity Payment Form described in Section 5.6(a)(1) or (2) that is the actuarial equivalent of the original annuity elected by the
Participant. For this purpose, actuarial equivalence shall be determined in accordance with Section 5.6(b). Except as permitted by Section 7, a Participant who elects to
have his 409A Benefit paid in the form of a Ten-Year Certain and Life Option, Guaranteed Death Benefit Option, Lump-Sum Option or DCP Option shall not be
permitted to change the Payment Form so elected.
(j) Valid Notional Rollovers to the New DCP . An employee who first becomes a Participant and accrues a Plan Benefit prior to January 1, 2009 shall be
permitted to elect the DCP Option for some or all of the amount otherwise payable under the Plan, provided that in the event that such Participant elects the DCP
Option for only a portion of his 409A Benefit, he shall receive the remaining portion of his 409A Benefit in the Lump Sum Option. The effective date of the Valid
Notional Rollover made in connection with the DCP Option will be the first day of the month following the Participant
’s Separation from Service, even if the portion of
the Participant’s 409A Benefit subject to the Valid Notional Rollover would otherwise have been paid to the Participant at a later date. Any such Valid Notional
Rollover shall be subject to the terms of the New DCP. If a Participant who has elected the DCP Option is not Retirement Eligible at the time of his Separation from
Service, then (i) the election of the DCP Option shall be void and of no force and effect and (ii) the Participant’s 409A Benefit shall be paid on the Default Payment
Date and in the Default Payment Form.
5.4 Payment of De Minimis Grandfathered Amounts . Notwithstanding a Participant’s Payment Date, the Company shall make a distribution of de
minimis Grandfathered Benefits according to the following rules:
(a) Grandfathered Benefit . Each Participant who (i) incurs a Separation from Service and (ii) as of the date of such Separation from Service has a
Grandfathered Benefit with an actuarial equivalent Lump-Sum Option value that does not exceed $5,000 shall receive a distribution of his entire Grandfathered Benefit
in a cash lump-sum as soon as administratively practicable after his Separation from Service.
(b) 409A Benefit . Each Participant who (i) incurs a Separation from Service and (ii) as of the date of such Separation from Service has a 409A Benefit
with an actuarial equivalent Lump-Sum Option value which, when aggregated with such Participant’s benefit subject to Section 409A under each other Company Non-
Account Plan in which the Participant participates, does not exceed $5,000 shall receive a distribution of his entire 409A Benefit in a cash lump-
sum on the last Business
Day of the month following the month in which the Separation from Service occurs.
(c) Lump-Sum Option Values . Lump-sum values under this Section 5.4 shall be determined using the same actuarial assumptions as would be applied
under the Retirement Plan for the purpose of determining the actuarial equivalent Lump-Sum Option value of Retirement Plan benefits of the Participant as of the date
of his Separation from Service.
5.5 Certain Accelerated Payments of 409A Amounts . Notwithstanding a Participant’s Payment Date, the Company in its sole discretion may accelerate
payment of all or a portion of a Participant’s 409A Benefit as permitted by Treasury Regulation Section 1.409A-j(4).
5.6 Available Forms of Payment .
(a) Forms of Payment . A Participant’
s Grandfathered Benefit and/or 409A Benefit, as applicable, may be paid in the forms of payment available under the
Retirement Plan as follows; provided , however , that a Participant who first accrues a Plan Benefit on or after January 1, 2009 (other than the 2008 New Executives)
may only receive payment of his 409A Benefit in the Lump-Sum Option:
1. “ Single Life Annuity ” means a Participant’s Grandfathered Benefit and/or 409A Benefit, as applicable, payable as an annuity in equal monthly
installments over the life of the Participant, commencing as of the Payment Date and terminating in the month in which the Participant dies, with
no further payments thereafter.

(b) Actuarial Equivalence . The actuarial equivalence of forms of payment in Sections 5.6(a) (1) through (4) above of a Grandfathered Benefit and/or
409A Benefit, as applicable, shall be determined in accordance with the factors and assumptions specified in the Retirement Plan (or such other factors or assumptions
specified from time to time by the Committee), in a manner which is intended to result in Section 409A Compliance.
5.7 Six-Month Delay in Commencement of 409A Benefits . Notwithstanding a Participant’
s Payment Election and the default rules hereunder effective for
Separations from Service (other than by reason of death) occurring on or after the Restatement Date, if, at the time of a Participant’s Separation from Service, the
Participant
2. “ 25, 50, 75 or 100% Joint and Survivor Annuity ” means a Participant’s actuarially reduced Grandfathered Benefit and/or 409A Benefit, as
applicable, payable as an annuity in equal monthly installments over the life of the Participant, commencing as of the Payment Date and
terminating in the month in which the Participant dies, with a survivor contingent annuity for the life of the Participant’s surviving contingent
annuitant, commencing in the month following the month in which the Participant died and terminating in the month in which the Participant’s
surviving contingent annuitant dies, which is either 25%, 50%, 75% or 100% of the monthly payment to the Participant, as elected by the
Participant. Following such contingent annuitant
’
s death, no further payments shall be made.
3. “ Ten Year Certain and Life Option ” means a Participant’s actuarially reduced Grandfathered Benefit and/or 409A Benefit, as applicable,
payable in monthly installments over the life of the Participant, commencing as of the Payment Date, with a guarantee that if the Participant dies
within 120 months ( i.e. , ten years) of the applicable Payment Date, such reduced Grandfathered Benefit and/or 409A Benefit, as applicable,
shall be paid to the Participant’s Beneficiary for the balance of the 120 month ( i.e. , ten year) guaranteed period in the month following the
month in which the date of the Participant’s death occurs, or, upon the Participant’s death, if the Participant’s Beneficiary so elects with respect
to the Grandfathered Benefit, the commuted value of the remaining payments shall be paid to such Beneficiary in a lump-sum amount. If the
Participant survives the 120 month ( i.e. , ten year) guaranteed period, he shall continue to receive the actuarially reduced Grandfathered Benefit
and/or 409A Benefit, as applicable, through the month in which the Participant dies.
4. “ Guaranteed Death Benefit Option ” means a Participant’s actuarially reduced lifetime monthly Grandfathered Benefit and/or 409A Benefit, as
applicable, commencing as of the Payment Date, in return for a death benefit guarantee. If the Participant dies on or after the Payment Date, the
Participant’s Beneficiary shall receive the excess, if any, of the initial death benefit (defined in a manner consistent with the terms of the
comparable payment option set forth in the Retirement Plan) over the aggregate Grandfathered Benefit or 409A Benefit, as applicable, payments
made to the Participant after the Payment Date and prior to the date of the Participant’s death. With respect to a Participant’s Grandfathered
Benefit only, a Participant shall be permitted, in the manner designated by the Committee, to make any of the alternative payment elections
related to this distribution option in the Retirement Plan.
5. “ Lump-Sum Option ” means the actuarial equivalent of a Participant’s Grandfathered Benefit and/or 409A Benefit, as applicable, payable in a
cash lump sum on the Payment Date.
6. “ DCP Option ” means the actuarial equivalent of a Participant’s Grandfathered Benefit and/or 409A Benefit, as applicable (or the applicable
portion thereof) that the Participant elects, in accordance with the terms of the Plan, to convert into a cash lump-sum amount to be credited in a
Valid Notional Rollover to the DCP. A Participant who elects the DCP Option with respect to some or all of his Grandfathered Benefit shall be
subject to the applicable terms and provisions of the Prior DCP and shall have the amount of the Valid Notional Rollover credited to the Prior
DCP. A Participant who elects or contingently elects the DCP Option with respect to some or all of his 409A Benefit shall be subject to the
applicable terms and provisions of the New DCP, shall be required to make his payment elections under the New DCP at the time the DCP
Option is elected and shall have the amount of the Valid Notional Rollover credited to the New DCP.

is a Key Employee, then, any amounts payable to the Participant under the Plan with respect to his 409A Benefit during the period beginning on the date of the
Participant’s Separation from Service and ending on the six-month anniversary of such date (the “ Delayed Payment Amount ”) shall be delayed and not paid to the
Participant until the first Business Day of the month following such six-month anniversary date, at which time such delayed amounts shall be paid to the Participant in a
lump-sum. If payment of an amount is delayed as a result of this Section 5.7, such amount shall be increased with interest from the date on which such amount would
otherwise have been paid to the Participant but for this Section 5.7 to the day immediately prior to the date the Delayed Payment Amount is paid. Interest on the Delayed
Payment Amount shall be credited on a quarterly basis based upon the interest rate being used to determine lump-
sum payments under the Retirement Plan for each such
quarter. If a Participant dies on or after the date of the Participant’s Separation from Service and prior to payment of the Delayed Payment Amount, any amount delayed
pursuant to this Section 5.7 shall be paid to the Participant’s joint annuitant (if the benefit form elected by the Participant is a joint annuity) or, if there is no joint
annuitant, the Participant’s Beneficiary, as applicable, together with any interest credited thereon, within 90 days of the date of the Participant’s death.
SECTION 6 DEATH BENEFITS
6.1 No Vesting Solely as a Result of Death . No survivor or death benefit shall be payable to any person under this Section 6 in respect of a Participant
unless the Participant had a Vested Plan Benefit on the date of the Participant’s death (or, if earlier, the date of the Participant’s Separation from Service). If a death
benefit is payable under this Section 6, no other amounts shall be payable in respect of a Participant under the Plan, and the default payment rules and any prior Payment
Elections made by the Participant shall be disregarded.
6.2 Death on or After Payment Date . If a Participant dies on or after his Payment Date, (i) no survivor or death benefit shall be payable under this
Section 6, (ii) any survivor or death benefits payable under the Plan shall be based solely upon the Payment Form applicable to the Participant, and (iii) no survivor or
death benefits shall be payable under the Plan if the applicable Payment Form ( e.g. , a Single Life Annuity) does not contemplate the payment of any survivor or death
benefits. The terms and provisions of the DCP (and not the Plan) shall govern the payment of any death benefit in respect of the portion of a Participant’s Plan Benefit
that has been credited under the DCP in connection with a Valid Notional Rollover. Solely for purposes of this Section 6, the Payment Date for the portion of a
Participant’s Plan Benefit that is transferred to the DCP in a Valid Notional Rollover shall be the date as of which the amount subject to the Valid Notional Rollover is
first credited to the DCP.
6.3 Death on or After Attaining Age 55 and Prior to Payment Date; Participants Who Accrue a Plan Benefit Prior to January 1, 2009 . If a Participant with
a Vested Plan Benefit, who first becomes a Participant and accrues a Plan Benefit prior to January 1, 2009 (or who is a 2008 New Executive), dies on or after attaining
age 55 and prior to the Participant’s Payment Date, the Participant’s Surviving Spouse, if any, shall be eligible, subject to a Participant’
s election under Section 6.8, for a
survivor annuity under the Plan calculated under Section 4.2 (and reduced for early commencement in accordance with the applicable Early Commencement Factor
from Appendix A) as if (i) the Participant had elected a 50% Joint and Survivor Annuity commencing immediately prior to the date of the Participant’
s death and (ii) the
Participant died immediately following the commencement of such annuity. The survivor annuity contemplated by this Section 6.3 shall commence in the month
following the month in which the Participant died and shall terminate in the month in which the Surviving Spouse dies.
6.4 Death Prior to Attaining Age 55 and Prior to Payment Date; Participants Who Accrue a Plan Benefit Prior to January 1, 2009 . If a Participant with a
Vested Plan Benefit, who first becomes a Participant and accrues a Plan Benefit prior to January 1, 2009 (or who is a 2008 New Executive), dies prior to attaining age
55 and prior to the Participant’s Payment Date, the Participant’s Surviving Spouse, if any, shall be eligible, subject to a Participant’s election under Section 6.8, for a
survivor annuity under the Plan calculated under Section 4.2 (and reduced for early commencement in accordance with the applicable Early Commencement Factor
from Appendix A) as if (i) the Participant incurred a Separation from Service on the date of death or, if earlier, on the date of Separation from Service, (ii) the
Participant survived until age 55, (iii) the Participant incurred a Separation from Service having elected a 50% Joint and Survivor Annuity commencing in the month
following the month in which the Participant attained age 55, and (iv) the Participant died on the day after attaining age 55. The survivor annuity contemplated by this
Section 6.4 shall commence in the month following the month in which the Participant would have attained age 55 and shall terminate in the month in which the
Surviving Spouse dies.

6.5 Death Benefits for Participants Who First Accrues a Plan Benefit On or After January 1, 2009 . If a Participant with a Vested Plan Benefit, who first
accrues a Plan Benefit on or after January 1, 2009 (other than a 2008 New Executive), dies prior to his Payment Date, the Participant’s Surviving Spouse, if any, shall
receive a cash lump-sum payment under the Plan equal to the actuarial equivalent (determined in accordance with Section 5.6(b)) of the death benefit described in
Section 6.3 or Section 6.4, as applicable, within 90 days of the date of the Participant’s death.
6.6 Death Benefits to Participants Who Die Without a Surviving Spouse . The provisions of this Section 6.6 shall apply effective July 24, 2006 to a
Participant described in Section 6.3 or 6.4 and a Participant described in Section 6.5 who, at the time of death while employed by the Company, is not survived by a
Surviving Spouse:
6.7 Rules of Application . The provisions of this Section 6 shall be applied separately with respect to a Participant’s Grandfathered Benefit and 409A
Benefit. Except as provided in Section 6.6(3), the payment of the survivor annuity under Section 6.3 or 6.4, as applicable, attributable to a Participant’s Grandfathered
Benefit may not be accelerated or deferred or paid in any alternative Payment Form.
6.8 Special Lump-Sum Election . An employee who first becomes a Participant and accrues a Plan Benefit prior to January 1, 2009 (or who is a 2008
New Executive) may irrevocably elect at the time that the Participant makes his Payment Election to have the actuarial equivalent (determined in accordance with
Section 5.6(b)) of the death benefit attributable to his 409A Benefit payable under Section 6.3 or 6.4, as applicable, paid to the Participant’s Surviving Spouse
(determined without regard to Section 6.6) within 90 days of the date of the Participant’s death. The consent of the Surviving Spouse shall not be required for any such
election by the Participant.
SECTION 7 REDEFERRAL OF 409A BENEFITS
7.1 Redeferrals to the DCP . Subject to this Section 7, a Participant who will be Retirement Eligible at his Separation from Service, shall be permitted to
elect, prior to his Separation from Service and in the manner contemplated by Section 7.2, to transfer in a Valid Notional Rollover all of the amount of his 409A Benefit
to the New DCP instead of having such amount paid to the Participant on the applicable Payment Date. The amount transferred to the New DCP in a Valid Notional
Rollover shall be credited to the New DCP as of the first day of the month following the Participant’s Separation from Service, even if the Payment Date for the 409A
Benefit is a later date. Subject to this Section 7, a Participant who will be Retirement Eligible at his Separation from Service and who has previously elected to receive
all or a portion of his 409A Benefit in the DCP Option shall be permitted to redefer payment, in the manner contemplated by Section 7.2, of the amount subject to the
DCP Option, subject to the applicable payment terms of the New DCP.
7.2 Redeferral Requirements . Subject to Section 7.3, the elections described in Sections 7.1 shall be subject to the following requirements:
1. For purposes of calculating the amount of the death benefit under Section 6.3 or 6.4, as applicable, the Participant shall be deemed to have been
survived by a Surviving Spouse of the opposite gender with a date of birth that is the same as the date of birth of the Participant.
2.
The actuarial equivalent (determined in accordance with Section 5.6(b)) of the benefit described in Section 6.3 or Section 6.4, as applicable, shall
be paid to the estate of the Participant within 90 days of the date of the Participant
’
s death.
3. Any survivor benefit provided by this Section 6.6 shall be treated as a 409A Benefit for purposes of the Plan (even if it is calculated with respect
to the Participant
’
s Grandfathered Benefit) and shall be payable only in a lump
-
sum and not in any other form of payment.
(a) The election to transfer the 409A Benefit in a Valid Notional Rollover to the New DCP must be made and become irrevocable (other than in the
case of the death of the Participant) at least one year prior to the then effective Payment Date.
(b)
The election shall not become effective for at least one year after the election is made.
(c) Any transfer to the New DCP of the 409A Benefit in connection with a Valid Notional Rollover must be made in accordance with the applicable
terms and provisions of the New DCP as then in

7.3 Limitations on Redeferrals . Notwithstanding the foregoing provisions of this Section 7, no Participant shall be permitted to elect a Valid Notional
Rollover for any portion of his Plan Benefit following the date of the Participant’s Separation from Service. A Valid Notional Rollover shall be void and of no effect if
the Participant is not Retirement Eligible at the time of his Separation from Service.
SECTION 8 CLAIMS PROCEDURE
8.1 General . If a Participant or his Surviving Spouse, Beneficiary or contingent annuitant or the authorized representative of one of the foregoing
(hereinafter, the “Claimant”) does not receive the timely payment of the benefits which he believes are due under the Plan, the Claimant may make a claim for benefits
in the manner hereinafter provided.
8.2 Claims . All claims for benefits under the Plan shall be made in writing and shall be signed by the Claimant. Claims shall be submitted to the
Administrative Record Keeper. If the Claimant does not furnish sufficient information with the claim for the Administrative Record Keeper to determine the validity of
the claim, the Administrative Record Keeper shall indicate to the Claimant any additional information which is necessary for the Administrative Record Keeper to
determine the validity of the claim.
8.3 Review of Claims . Each claim hereunder shall be acted on and approved or disapproved by the Administrative Record Keeper within 90 days
following the receipt by the Administrative Record Keeper of the information necessary to process the claim. If special circumstances require an extension of the time
needed to process the claim, this 90-day period may be extended to 180 days after the claim is received. The Claimant shall be notified before the end of the original
period if an extension is necessary, the reason for the extension and the date by which it is expected that a decision will be made. In the event the Administrative Record
Keeper denies a claim for benefits, in whole or in part, the Administrative Record Keeper shall notify the Claimant in writing of the denial of the claim and notify the
Claimant of his right to a review of the Administrative Record Keeper’s decision by the Committee. Such notice by the Administrative Record Keeper shall also set
forth, in a manner calculated to be understood by the Claimant, the specific reason for such denial, the specific provisions of the Plan on which the denial is based, and a
description of any additional material or information necessary to perfect the claim with an explanation of the Plan’s appeals procedure as set forth in this Section 8.
8.4 Appeals . Any Claimant whose claim for benefits is denied in whole or in part may appeal to the Committee for a review of the decision by the
Administrative Record Keeper. Such appeal must be made within 60 days after the applicant has received actual or constructive notice of the denial as provided above.
An appeal must be submitted in writing within such period and must:
8.5 Review of Appeals . The Committee shall act upon each appeal within 60 days after receipt thereof unless special circumstances require an extension
of the time for processing, in which case a decision shall be rendered by the Committee as soon as possible but not later than 120 days after the appeal is received by it.
If such an extension of time for processing is required because of special circumstances, written notice of the extension shall be furnished prior to the commencement of
the extension describing the reasons an extension is needed and the date when the determination will be made. The Committee may require the Claimant to submit such
additional facts, documents or other evidence as the Committee in its discretion deems necessary or advisable in making its review. The Claimant shall be given the
opportunity to review pertinent documents or materials upon submission of a written request to the Committee, provided that the Committee finds the requested
documents or materials are pertinent to the appeal.
effect and, once the deferred amount constituting the 409A Benefit is credited under the New DCP, shall constitute a full and complete
settlement of the Company
’
s obligations to the Participant under the Plan.
(d) If the 409A Benefit is transferred to the New DCP in a Valid Notional Rollover, the payment commencement date elected by the Participant
under the New DCP for the 409A Benefit for the amount so transferred must not be earlier than the fifth anniversary of the original Payment
Date.
1.
request a review by the Committee of the claim for benefits under the Plan;
2.
set forth all of the grounds upon which the Claimant
’
s request for review is based and any facts in support thereof; and
3.
set forth any issues or comments which the Claimant deems pertinent to the appeal.

8.6 Final Decisions . On the basis of its review, the Committee shall make an independent determination of the Participant’s eligibility for benefits under
the Plan. The decision of the Committee on any appeal of a claim for benefits shall be final and conclusive upon all parties thereto.
8.7 Denial of Appeals . In the event the Committee denies an appeal in whole or in part, it shall give written notice of the decision to the Claimant, which
notice shall set forth, in a manner calculated to be understood by the Claimant, the specific reasons for such denial and which shall make specific reference to the
pertinent provisions of the Plan on which the Committee’s decision is based.
8.8 Statute of Limitations . A Claimant wishing to seek judicial review of an adverse benefit determination under the Plan, whether in whole or in part,
must file any suit or legal action, including, without limitation, a civil action under Section 502(a) of ERISA, within three years of the date the final decision on the
adverse benefit determination on review is issued or should have been issued under Section 8.6 or lose any rights to bring such an action. If any such judicial proceeding
is undertaken, the evidence presented shall be strictly limited to the evidence timely presented to the Committee. Notwithstanding anything in the Plan to the contrary, a
Claimant must exhaust all administrative remedies available to such Claimant under the Plan before such Claimant may seek judicial review pursuant to Section 502(a)
of ERISA.
SECTION 9 AMENDMENT AND TERMINATION
9.1 Amendment or Termination . The Plan may be amended or terminated at any time, by the Board of Directors or the Committee; provided , however ,
no amendment or termination may reduce the amount of a Participant’s Plan Benefit as of the date of the amendment or termination without the Participant’s written
consent; and provided further that it shall not be a reduction of a Participant’s Plan Benefit if the amount of the Plan Benefit is reduced pursuant to Section 4.2 solely
as a result in an increase in the value of Participant’s accrued benefit under the Retirement Plan. Upon termination of the Plan, payment of a Participant’s 409A Benefit
shall be made on the Payment Date and in the Payment Form applicable to the Participant unless the Board of Directors or the Committee, in its discretion, determines to
accelerate payment and such acceleration may be effected in a manner that will not result in the imposition on any Participant of additional taxes or penalties under
Section 409A (“ Section 409A Compliance ”).
9.2 409A Benefit Amendments . Notwithstanding any provision in the Plan to the contrary, with respect to a Participant’s 409A Benefit, the Board of
Directors, the Committee or the Deferred Compensation Tax Compliance Committee shall have the independent right, prospectively and/or retroactively, to amend or
modify the Plan in accordance with Section 409A, in each case, without the consent of any Participant, to the extent that the Board of Directors, the Committee or the
Deferred Compensation Tax Compliance Committee deems such action to be necessary or advisable to address regulatory or other changes or developments that affect
the terms of the Plan with the intent of effecting Section 409A Compliance. Any determinations made by the Board of Directors, the Committee or the Deferred
Compensation Tax Compliance Committee under this Section 9.2 shall be final, conclusive and binding on all persons.
SECTION 10 MISCELLANEOUS
10.1 No Effect on Employment Rights . Nothing contained herein shall be construed as a contract of employment with any person. The Plan and its
establishment shall not confer upon any person the right to be retained in the service of the Company or limit the right of the Company to discharge or otherwise deal
with any person without regard to the existence of the Plan.
10.2 Funding . The Plan at all times shall be entirely unfunded, and no provision shall at any time be made with respect to segregating any assets of the
Company for payment of any benefits hereunder. No Participant, Surviving Spouse, Beneficiary or other person shall have any interest in any particular assets of the
Company by reason of a right to receive a benefit under the Plan, and any such Participant, Surviving Spouse, Beneficiary or other person shall have the rights of a
general unsecured creditor of the Company with respect to any rights under the Plan. Notwithstanding the foregoing, the Committee or the Board of Directors, in its
discretion, may establish a grantor trust to fund benefits payable under the Plan and administrative costs relating to the Plan. The assets of said trust shall be held
separate and apart from other Company funds and shall be used exclusively for the purposes set forth in the Plan and the applicable trust agreement, subject to the
following conditions:
1.
the creation of said trust shall not cause the Plan to be other than
“
unfunded
”
for purposes of ERISA;

10.3 Anti-assignment . To the maximum extent permitted by law, no benefit payable under the Plan shall be subject in any manner to anticipation,
alienation, sale, transfer, assignment, pledge, encumbrance, or charge, and any attempt to do so shall be void, nor shall any such benefit be in any manner liable for or
subject to garnishment, attachment, execution or levy, or liable for or subject to the debts, contracts, liabilities, engagements or torts of the Participant.
10.4 Taxes . The Company shall have the right to pay any required employment, income or other withholding taxes from a Participant’s Plan Benefit.
10.5 Construction . The Plan is intended to satisfy the requirements of Section 409A with respect to amounts subject thereto and shall be interpreted and
construed accordingly. The Plan is intended to be an unfunded deferred compensation arrangement for a select group of management or highly compensated employees
within the meaning of ERISA and, therefore, exempt from the requirements of Sections 201, 301 and 401 of ERISA. Whenever the terms of the Plan or of a Payment
Election require the payment of an amount by a specified date, the Company shall use reasonable efforts to make or commence the payment by that date. The Company
shall not be (i) liable to the Participant or any other person if such payment or payment commencement is delayed for administrative or other reasons to a date that is
later than the date so specified by the Plan or the Payment Election or (ii) required to pay interest or any other amount in respect of such delayed payment except to the
extent specifically contemplated by the terms of the Plan.
10.6 Incapacity of Participant . In the event a Participant or Surviving Spouse is declared incompetent and a conservator or other person legally charged
with the care of his person or his estate is appointed, any benefits under the Plan to which such Participant or Surviving Spouse is entitled shall be paid to such
conservator or other person legally charged with the care of his person or estate.
10.7 Severability . In the event that one or more provisions of the Plan shall be or become invalid, illegal or unenforceable in any respect, the validity,
legality and enforceability of the remaining provisions of the Plan shall not be affected thereby.
10.8 Governing Law . The Plan is established under and shall be governed and construed in accordance with the laws of the State of New Jersey, to the
extent that such laws are not preempted by ERISA.
2.
the Company shall be treated as the
“
grantor
”
of said trust for purposes of Sections 671 and 677 of the Code; and
3.
said trust agreement shall provide that the trust fund assets may be used to satisfy claims of the Company
’
s general creditors.

APPENDIX A
EARLY COMMENCEMENT FACTORS
Subsidized Early Commencement Factor (used for (A) the 409A Benefit for a Participant whose Separation from Service occurs on or after attaining age 55 and
completing ten or more Years of Vesting Service; (B) for the Grandfathered Benefit of a Participant whose Separation from Service occurs on or after attaining age 55
and completing ten or more Years of Vesting Service and who, as of December 31, 2004, had at least ten Years of Vesting Service); and (C) for the 409A Benefit of a
Rule of 70 Participant.
1.00 less /
4
% for each month by which the Payment Date precedes the Normal Retirement Date.
Unsubsidized Early Commencement Factor (used for all other purposes):
The actuarially equivalent factor applicable to the accrued benefit of a terminated vested participant under the Retirement Plan.
1

Form of First Amendment to Wyeth Supplemental Executive Retirement Plan
This Amendment (the “ Amendment ”) to the Wyeth Supplemental Executive Retirement Plan (amended and restated effective as of January 1, 2005) (the “ Plan ”) is
hereby entered into effective as of September 1, 2009:
Capitalized terms used, but not otherwise defined, shall have the meanings set forth in the Plan.
W I T N E S S E T H
WHEREAS, the Plan supplements the benefits of Participants whose benefits under the Retirement Plan are limited as a result of Deferrals or by operation of
the Code Limits;
WHEREAS, the Deferred Compensation Tax Compliance Committee has the right to amend or modify the Plan in accordance with Section 409A to the extent
necessary or advisable to address developments that affect the terms of the Plan;
WHEREAS, the Retirement Plan has been amended to permit Participants to elect to receive their benefit in a lump sum after their Separation from Service;
WHEREAS, Wyeth desires to ensure that such amendment to the Retirement Plan shall not result in a material modification of the Plan within the meaning of
Section 409A;
NOW THEREFORE, in consideration of the premises and other good and valuable consideration, the receipt and sufficiency of which is hereby
acknowledged, the parties hereto hereby agree as follows, effective as of September 1, 2009.
1. The definition of “Default Payment Form” is amended as follows:
“ Default Payment Form ”
means (i) with respect to a Participant's Grandfathered Benefit, the form of payment elected by the Participant under the Retirement
Plan; provided , however , that if the Participant elects, following his Separation from Service, to receive his benefit under the Retirement Plan in the Lump-
Sum Option, the form of annuity elected by the Participant under the Plan; and (ii) with respect to a Participant’s 409A Benefit, the Lump-Sum Option.
2. The Definition of “Normal Payment Date” is amended as follows:
“
Normal Payment Date
”
means (i) with respect to a Participant
’
s Grandfathered Benefit, the following: (A) if the Payment Form is other

than the Lump-Sum Option or the DCP Option, the first day of the first period for which an amount is payable to the Participant under the Retirement Plan;
and (B) if the payment form is the Lump-Sum Option, the Payment Date specified in Section 5.2(c); and (ii) with respect to a Participant's 409A Benefit, the
following: (A) for a Participant who incurs a Separation from Service with a Vested Plan Benefit prior to attaining age 55, the first day of the month
coincident with or next following the month in which he attains age 55; and (B) for a Participant who incurs a Separation from Service with a Vested Plan
Benefit on or after attaining age 55, the first day of the month following his Separation from Service.
2

FORM OF AMENDMENT TO THE
WYETH SUPPLEMENTAL EXECUTIVE RETIREMENT PLAN
The Wyeth Supplemental Executive Retirement Plan (the “Plan”)
is hereby amended as follows, effective as of the Closing Date as defined in the
Agreement and Plan of Merger, dated as of January 25, 2009, by and among Pfizer, Inc., Wagner Acquisition Corp., and Wyeth:
1. Section 1.2 of the Plan is hereby amended by adding the following as a new subsection (vv) and renumbering existing subsections
(vv) through (hhh) as subsections (ww) through (iii):
An otherwise Eligible Employee who is employed at the Rouses Point location (other than an Eligible Employee who is covered
by a transition plan related to the Pfizer Agreement) or incurs a Separation from Service in connection with Project Impact, shall
not be treated as a Pfizer Rule of 70 Participant.
“
(
vv)
“
Pfizer Rule of 70 Participant
”
means an Eligible Employee who as of the date of his Separation from Service:
(i)
Is fully vested in his Plan Benefit on the date that he incurs a Separation from Service;
(ii)
Is involuntarily terminated by the Company prior to the end of the two period that commences on the Closing
Date as defined in the Agreement and Plan of Merger, dated as of January 25, 2009, by and among Pfizer, Inc.,
Wagner Acquisition Corp., and Wyeth (the
“
Pfizer Agreement
”
),
(iii) Is notified by the Company that he is eligible for the Rule of 70 in connection with Pfizer Inc.’
s acquisition of
the Company (the
“
Pfizer Rule of 70 Benefit
”
),
and
(iv)
As of the date of his Separation from Service, has a combined age and Years of Vesting Service equal to or in
excess of 70.

2. Section 4 of the Plan is hereby amended by adding the following as new Section 4.6:
3. Appendix A to the Plan is hereby amended by deleting the period after the first paragraph and adding the following:
“, and (D) for the Pfizer Rule of 70 Benefits.”
2
“ 4.6 Pfizer Rule of 70 Benefit
. The Pfizer Rule of 70 Benefit shall be equal to the benefit that the Pfizer Rule of 70 Participant
would receive under the Retirement Plan if he was eligible for the Rule of 70 benefit under Section 4.3(d) of the Retirement
Plan. The Pfizer Rule of 70 Benefit shall be paid on the Normal Payment Date and in the Default Payment Form.
”

FORM OF AMENDMENT TO THE
WYETH SUPPLEMENTAL EXECUTIVE RETIREMENT PLAN
The Wyeth Supplemental Executive Retirement Plan (the “Plan”)
is hereby amended as follows, effective as of the Closing Date (as defined in the
Amended and Restated Asset Purchase Agreement, dated September 17, 2009, by and among Pfizer Inc., Wyeth and Boehringer Ingelheim Vetmedica, Inc.):
1. Section 1.2 of the Plan is hereby amended by adding the following as new subsection (g) and renumbering existing subsections (g) through
(iii) as subsections (h) through (jjj):
An otherwise Eligible Employee who is employed at the Rouses Point location (other than an Eligible Employee who is
covered by a transition benefit plan related to the Pfizer Agreement) or incurs a Separation from Service in connection
with Project Impact, shall not be treated as a Boehringer Rule of 70 Participant.
”
“
(
g)
“
Boehringer Rule of 70 Participant
”
means an Eligible Employee who as of the date of his Separation from Service:
(i)
Is fully vested in his Plan Benefit on the date that he incurs a Separation from Service;
(ii)
Is notified by the Company that he is eligible for the Boehringer Rule of 70 Benefit;
(iii)
Does not incur a Separation from Service in connection with Project Impact and is not eligible for the Pfizer
Rule of 70 Benefit; and
(iv) As of the date of the Participant’
s Separation from Service his combined age and Years of Vesting Service
equals or exceeds 70.

2. Section 4.3 of the Plan is hereby amended by adding the following to the end thereof:
“Notwithstanding the foregoing, all Affected Employees (as defined in the Amended and Restated Asset Purchase Agreement, dated
September 17, 2009, by and among Pfizer Inc., Wyeth and Boehringer Ingelheim Vetmedica, Inc. (the “Boehringer Agreement”)) shall
become 100% vested in their Plan Benefits as of the Closing Date (as defined in the Boehringer Agreement).”
3. Section 4 of the Plan is hereby amended by adding the following as new Section 4.6:
4. Appendix A to the Plan is hereby amended by deleting the period after the first paragraph and adding the following:
“ and (E) for the Boehringer Rule of 70 Benefits.”
2
“
4.6
Boehringer Rule of 70 Benefit
. The Boehringer Rule of 70 Benefit shall be equal to the benefit that the Boehringer Rule of
70 Participant would receive under the Retirement Plan if he was eligible for the Rule of 70 benefit under Section 4.3(d) of the
Retirement Plan. The Boehringer Rule of 70 Benefit shall be paid on the Normal Payment Date and in the Default Payment
Form.
”

FORM OF AMENDMENTS TO THE
WYETH SUPPLEMENTAL EXECUTIVE RETIREMENT PLAN (“SERP”)
Notwithstanding the foregoing, any payment made within 90 days of the Normal Payment Date shall be considered to be made
on the Normal Payment Date.
(tt) “Retirement Plan” means the Wyeth Retirement Plan – United States, as amended from time to time, and with respect to a
Participant who effective December 30, 2010, became a participant in the Pfizer Consolidated Pension Plan for Employees
Resident in Puerto Rico (the “PCPP PR”), Retirement Plan shall mean the PCPP PR.
Rate of Annual Earnings. The Rate of Earnings to be included in the determination of “Final Average Annual Pension
Earnings” under Section 2.19 of the Retirement Plan means the sum of (a) base salary rate (including 401(k) salary deferral
contributions, elective contributions to a plan subject to Section 125 of the Code and elective amounts that are not includible
in the gross income of the Employee by reason of Section 132(f)(4) of the Code) as of April 1 of each Plan Year starting on
or after January 1, 2011, and January 1
of each Plan Year
1.
Section 1.2(gg) of the SERP is hereby amended by adding the following language to the end thereof to read as follows:
2.
Section 1.2(tt) of the SERP is hereby amended to read as follows:
3.
Effective December 30, 2010, Section 4.2(a)2. of the SERP is hereby amended to read as follows:
2.
for purposes of calculating such accrued benefit, except with respect to a Participant who effective December 30, 2010,
became a participant in the Pfizer Consolidated Pension Plan for Employees Resident in Puerto Rico (the “PCPP PR”)
and consented to the transfer of his benefit hereunder to the PCPP PR, the Code limits did not apply.
4. For purposes of calculating the “Final Average Annual Pension Earnings” formula of the Retirement Plan referenced in
Section 4.2 of the SERP, Section 4.2 is amended to add to the end thereof as follows:
st
st

starting prior to January 1, 2011 (except that for any Participant with a Severance From Service Date between January 1, 2011
and March 31, 2011, it shall be the base salary rate in effect on January 1, 2011), (b) cash bonuses paid by the Company or a
Participating Company in such Plan Year, including any payments under the Wyeth Performance Incentive Award Program
(“PIA”) or its successor plan, and (c) overtime earnings, shift differentials and premium pay, sales commissions, and sales
bonuses paid in the prior Plan Year. Notwithstanding the foregoing, the Rate of Annual Earnings shall exclude any amounts
deferred under any nonqualified deferred compensation plan. For a Participant whose base salary on the date of his Severance
From Service is equal to or more than $155,000, and who has attained his Early Retirement Date prior to Severance From
Service (b) in the first sentence shall be substituted with the following if such substitution shall result in a larger Accrued
Benefit for the Participant: “(b) cash bonuses paid by the Company or a Participating Company in the year earned, including
any payments under the Wyeth Performance Incentive Award Program (“PIA”) or its successor plan, and for the year of
retirement the annualized PIA bonus received in the year of retirement shall be used provided that the annualized PIA bonus in
the year of retirement cannot be greater than the largest PIA bonus percentage received in either of the previous two years
multiplied by the final year’s annual base salary rate.”
4.3 Vesting . Anything in the Plan to the contrary notwithstanding, no Plan Benefit or other amounts shall be
payable to a Participant under the Plan unless the Participant has either (i) completed three Years of Vesting Service or (ii) is
at least age 60, in each case as of the date of the Participant’s Separation From Service.
4.7 Benchmark Rule of 70 Benefit . The Benchmark Rule of 70 Benefit hereunder shall be equal to the benefit that a
Participant would have been eligible for provided in Section 4.3(f) of the Retirement Plan but for the fact that such Participant
does not make less than $155,000. The Benchmark Rule of 70 Benefit shall be paid on the Normal Payment Date in the
Default Payment Form.
4.8 Wyeth Change in Control Plan Benefit . Any benefit payable pursuant to Section 4(iv)(D), shall be payable
hereunder.
5.
Effective January 1, 2011, Section 4.3 of the SERP is amended to read as follows:
6.
New Section 4.7 is added to the end of Section 4 to read as follows:
7.
New Section 4.8 is added to the end of Section 4 to read as follows:

9.3 Errors in Calculating Lump-Sum Option Payments. Whenever due to (1) a bona fide mathematical or
actuarial error, or (2) additional compensation for purposes of the Plan for the taxable year of the Participant in which the
Participant has a Separation from Service which has been administratively impracticable to take into account at the time of
such Separation from Service, the amount of such 409A Benefit is determined after such payment to have been less than if
such error had not been made or such compensation taken into account, then a supplemental corrective lump sum payment
correcting such error or taking into such additional compensation may be made by the Company prior to December 31st of the
year in which the lump sum payment was made, After such December 31
, no further corrective payment shall be made.
8.
Effective January 1, 2011, new Section 9.3 is added to the end of Section 9 of the SERP to read as follows:
st

FORM OF AMENDMENTS TO THE
WYETH SUPPLEMENTAL EMPLOYEE SAVINGS PLAN, WYETH SUPPLEMENTAL
EXECUTIVE RETIREMENT PLAN, WYETH DEFERRED COMPENSATION PLAN
AND THE WYETH EXECUTIVE RETIREMENT PLAN (COLLECTIVELY, THE “PLANS”)
The Plans are hereby amended, effective as of January 1, 2011, by deleting the following language “within ninety (90) days after
the date of the Participant’s death,” in each of the plan sections listed below, and replacing it with “in the January following the calendar
year in which the Participant’s death occurs”:
1.
Wyeth Supplemental Employee Savings Plan
–
Section 1.2(r);
2.
Wyeth Supplemental Executive Retirement Plan
–
Sections 5.7, 6.5, 6.6 and 6.8;
3.
Wyeth Executive Retirement Plan
–
Sections 5.7, 6.5, 6.6 and 6.8; and
4.
Wyeth Deferred Compensation Plan
–
Sections 7.4 and 7.6.
Exhibit 12
PFIZER INC. AND SUBSIDIARY COMPANIES
COMPUTATION OF RATIO OF EARNINGS TO FIXED CHARGES
Year Ended December 31,
(millions except ratios)
2011
2010
2009
2008
2007
Determination of Earnings:
Income from continuing operations before provision for taxes on income, noncontrolling interests and
cumulative effect of a change in accounting principles
$
12,762
$
9,282
$
10,674
$
9,520
$
9,127
Less:
Noncontrolling interests
42
31
8
22
40
Income attributable to Pfizer Inc.
12,720
9,251
10,666
9,498
9,087
Add:
Fixed charges
1,813
1,932
1,358
647
541
Total earnings as defined
$
14,533
$
11,183
$
12,024
$
10,145
$
9,628
Fixed charges:
Interest expense (a)
$
1,681
$
1,797
$
1,232
$
516
$
397
Preferred stock dividends (b)
5
6
7
8
11
Rents (c)
127
129
119
123
133
Fixed charges
1,813
1,932
1,358
647
541
Capitalized interest
50
36
34
46
43
Total fixed charges
$
1,863
$
1,968
$
1,392
$
693
$
584
Ratio of earnings to fixed charges
7.8
5.7
8.6
14.6
16.5
(a) Interest expense includes amortization of debt premium, discount and expenses. Interest expense does not include interest related to uncertain tax positions of
$346 million for 2011; $384 million for 2010; $337 million for 2009; $333 million for 2008 and $331 million for 2007.
(b)
Preferred stock dividends are from our Series A convertible perpetual preferred stock held by an Employee Stock Ownership Plan assumed in connection with our
acquisition of Pharmacia in 2003.
(c) Rents included in the computation consist of one-third of rental expense which we believe to be a conservative estimate of an interest factor in our leases, which
are not material.

EXHIBIT 13
Pfizer Inc.
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
INTRODUCTION
Our Financial Review is provided to assist readers in understanding the results of operations, financial condition and cash flows of Pfizer Inc. (the Company). It should be read in conjunction with the
Consolidated Financial Statements and Notes to Consolidated Financial Statements. The discussion in this Financial Review contains forward-looking statements that involve substantial risks and
uncertainties. Our actual results could differ materially from those anticipated in these forward-looking statements as a result of various factors such as those discussed in Part 1, Item 1A, “Risk Factors”
of our 2011 Annual Report on Form 10-K and in the “Forward-Looking Information and Factors That May Affect Future Results”, “Our Operating Environment” and “Our Strategy” sections of this Financial
Review.
The Financial Review is organized as follows:
• Overview of Our Performance, Operating Environment, Strategy and Outlook . This section, beginning on page 2, provides information about the following: our business; our 2011 performance; our
operating environment; our strategy; our business development initiatives, such as acquisitions, dispositions, licensing and collaborations; and our financial guidance for 2012.
• Significant Accounting Policies and Application of Critical Accounting Estimates . This section, beginning on page 11, discusses those accounting policies and estimates that we consider important in
understanding Pfizer’s consolidated financial statements. For additional discussion of our accounting policies, see Notes to Consolidated Financial Statements—
Note 1. Significant Accounting Policies
.
•
Analysis of the Consolidated Statements of Income.
This section begins on page 16, and consists of the following sections:
o
Revenues. This section, beginning on page 16, provides an analysis of our revenues and products for the three years ended December 31, 2011, including an overview of important product
developments.
o
Costs and Expenses
. This section, beginning on page 30, provides a discussion about our costs and expenses.
o
Provision for Taxes on Income.
This section, beginning on page 35, provides a discussion of items impacting our tax provisions.
o
Discontinued Operations.
This section, beginning on page 36, provides an analysis of the financial statement impact of our discontinued operations.
o
Adjusted Income
. This section, beginning on page 36, provides a discussion of an alternative view of performance used by management.
•
Analysis of the Consolidated Balance Sheets.
This section begins on page 40 and provides a discussion of changes in certain balance sheet accounts.
•
Analysis of the Consolidated Statements of Cash Flows.
This section begins on page 41 and provides an analysis of our consolidated cash flows for the three years ended December 31, 2011.
•
Analysis of Financial Condition, Liquidity and Capital Resources
. This section, beginning on page 42, provides an analysis of our financial assets and liabilities as of December 31, 2011 and
December 31, 2010, as well as a discussion of our outstanding debt and other commitments that existed as of December 31, 2011. Included in the discussion of outstanding debt is a discussion of the
amount of financial capacity available to help fund Pfizer
’
s future activities.
•
New Accounting Standards
. This section, on page 45, discusses accounting standards that we recently have adopted, as well as those that recently have been issued, but not yet adopted by us.
• Forward-Looking Information and Factors That May Affect Future Results . This section, beginning on page 45, provides a description of the risks and uncertainties that could cause actual results to
differ materially from those discussed in forward-looking statements presented in this Financial Review relating to our financial and operating performance, business plans and prospects, in-line
products and product candidates, strategic review, capital allocation, and share-repurchase and dividend-rate plans. Such forward-looking statements are based on management’s current expectations
about future events, which are inherently susceptible to uncertainty and changes in circumstances. Also included in this section are discussions of Financial Risk Management and Legal Proceedings
and Contingencies.
2011 Financial Report
1
Financial Review
Pfizer Inc. and Subsidiary Companies
OVERVIEW OF OUR PERFORMANCE, OPERATING ENVIRONMENT, STRATEGY AND OUTLOOK
Our Business
Our mission is to apply science and our global resources to improve health and well-being at every stage of life. We strive to set the standard for quality, safety and value in the discovery, development
and manufacturing of medicines for people and animals. Our diversified global healthcare portfolio includes human and animal biologic and small molecule medicines and vaccines, as well as nutritional
products and many of the world’s best-known consumer products. Every day, we work across developed and emerging markets to advance wellness, prevention, treatments and cures that challenge the
most feared diseases of our time. We also collaborate with other biopharmaceutical companies, healthcare providers, governments and local communities to support and expand access to reliable,
affordable healthcare around the world. Our revenues are derived from the sale of our products, as well as through alliance agreements, under which we co-promote products discovered by other
companies.
The majority of our revenues come from the manufacture and sale of biopharmaceutical products. The biopharmaceutical industry is highly competitive and we face a number of industry-specific
challenges, which can significantly impact our results. These factors include, among others: the loss or expiration of intellectual property rights, the regulatory environment and pipeline productivity, pricing
and access pressures, and increasing competition among branded products. (For more information about these challenges, see the “Our Operating Environment” section of this Financial Review.)
The financial information included in our consolidated financial statements for our subsidiaries operating outside the United States (U.S.) is as of and for the year ended November 30 for each year
presented.
The assets, liabilities, operating results and cash flows of acquired businesses, such as King Pharmaceuticals, Inc. (King) (acquired on January 31, 2011) and Wyeth (acquired on October 15, 2009) are
included in our results on a prospective basis only commencing from the acquisition date. As such, our consolidated financial statements for the year ended December 31, 2011 reflect approximately 11
months of King’s U.S. operations and approximately 10 months of King’s international operations, and our consolidated financial statements for the year ended December 31, 2009 reflect approximately
two-and-a-half months of Wyeth’s U.S. operations and approximately one-and-a-half months of Wyeth’s international operations. (For more information about these acquisitions, see the “Our Business
Development Initiatives” section of this Financial Review.)
On August 1, 2011, we completed the sale of our Capsugel business. In connection with our decision to sell, the operating results associated with the Capsugel business are classified as Discontinued
operations––net of tax in our consolidated statements of income for all periods presented, and the assets and liabilities associated with this business are classified as Assets of discontinued operations
and other assets held for sale and Liabilities of discontinued operations , as appropriate, in our consolidated balance sheets as of December 31, 2010. (See “Our Business Development Initiatives” and
“Discontinued Operations” sections of this Financial Review for more information.)
On July 7, 2011, we announced our decision to explore strategic alternatives for our Animal Health and Nutrition businesses, which may include, among other things, a full or partial separation of each of
these businesses from Pfizer through a spin-off, sale or other transaction. We expect to announce our strategic decision for each business in 2012. (For further information, see the “Our Business
Development Initiatives” section of this Financial Review.)
2
2011 Financial Report
Financial Review
Pfizer Inc. and Subsidiary Companies
Our 2011 Performance
Revenues increased 1% in 2011 to $67.4 billion, compared to $67.1 billion in 2010, due to the favorable impact of foreign exchange, which increased revenues by approximately $1.9 billion, or 3%, and
the inclusion of revenues of $1.3 billion or 2% from our acquisition of King, partially offset by a net operational decline of $2.9 billion, or 4%, primarily due to the loss of exclusivity of certain products.
The significant impacts on revenues for 2011, compared to 2010, are as follows:
Income from continuing operations was $8.7 billion in 2011 compared to $8.2 billion in 2010, primarily reflecting:
partially offset by:
2011 vs. 2010
(MILLIONS OF DOLLARS)
INCREASE/
(DECREASE)
%
CHANGE
Prevnar 13/Prevenar13
$1,241
51
Lyrica
630
21
Enbrel (Outside the U.S. and Canada)
392
12
Skelaxin
203
*
Celebrex
149
6
Sutent
121
11
Pristiq
111
24
Zyvox
107
9
ReFacto AF/Xyntha
102
25
Medrol
55
12
Norvasc
(61
)
(4
)
Vfend
(78
)
(9
)
Aromasin
(122
)
(25
)
Detrol/Detrol LA
(130
)
(13
)
Zosyn/Tazocin
(316
)
(33
)
Protonix
(482
)
(70
)
Xalatan/Xalacom
(499
)
(29
)
Prevnar/Prevenar (7-valent)
(765
)
(61
)
Effexor
(1,040
)
(61
)
Lipitor
(1,156
)
(11
)
Alliance revenues
(454
)
(11
)
All other biopharmaceutical products
1,056
19
Animal Health products
609
17
Consumer Healthcare products
285
10
Nutrition products
271
15
2011 reflects the inclusion of revenues from legacy King products.
Lipitor lost exclusivity in the U.S. in November 2011, Canada in May 2010, Spain in July 2010, Brazil in August 2010 and Mexico in December 2010. Aromasin lost exclusivity in the U.S. in April 2011.
Xalatan lost exclusivity in the U.S. in March 2011. Vfend tablets lost exclusivity in the U.S. in February 2011. Effexor XR lost exclusivity in the U.S. in July 2010. The basic U.S. patent (including the six-
month exclusivity period) for Protonix expired in January 2011. Zosyn lost exclusivity in the U.S. in September 2009. We lost exclusivity for Aricept 5mg and 10mg tablets, which are included in Alliance
revenues, in November 2010.
Includes the “All other” category included in the Revenues — Major Biopharmaceutical Products table presented in this Financial Review.
*
Calculation not meaningful.
• higher impairment charges of $1.3 billion (pre-tax) in 2010 compared to 2011, (see further discussion in the “Costs and Expenses––Other (Income)/Deductions––Net” section of this Financial Review
and Notes to Consolidated Financial Statements—
Note 4. Other Deductions
–
–
net
);
•
lower purchase accounting impacts of $1.5 billion (pre
-
tax) in 2011 compared to 2010, primarily related to inventory sold that had been recorded at fair value;
•
lower merger restructuring and transaction costs of $2.0 billion (pre
-
tax) in 2011 compared to 2010; and
• the non-recurrence of a charge of $1.3 billion (pre-tax) in 2010 for asbestos litigation related to our wholly owned subsidiary Quigley Company, Inc. (see Notes to Consolidated Financial Statements––
Note 17. Commitments and Contingencies
),
• higher charges of $2.5 billion (pre-tax) in 2011 compared to 2010 related to our non-acquisition related cost-reduction and productivity initiatives; and
•
the non
-
recurrence of a favorable settlement with the U.S. Internal Revenue Service in 2010.
2011 Financial Report
3
(a)
(b)
(b)
(b)
(b)
(b)
(b)
(b)
(b)
(a), (c)
(a)
(a)
(b)
(c)

Financial Review
Pfizer Inc. and Subsidiary Companies
Our Operating Environment
U.S. Healthcare Legislation
Principal Provisions Affecting Us
In March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act (together, the U.S. Healthcare Legislation), was enacted in the U.S. This
legislation has resulted in both current and longer-term impacts on us, as discussed below.
Certain provisions of the U.S. Healthcare Legislation became effective in 2010 or on January 1, 2011, while other provisions will become effective on various dates. The principal provisions affecting us
provide for the following:
In addition, the U.S. Healthcare Legislation includes provisions that affect the cost of certain of our postretirement benefit plans. Companies currently permitted to take a deduction for federal income tax
purposes in an amount equal to the subsidy received from the federal government related to their provision of prescription drug coverage to Medicare-eligible retirees will no longer be eligible to do so
effective for tax years beginning after December 31, 2012. While the loss of this deduction will not take effect until 2013, under U.S. generally accepted accounting principles, we were required to account
for the impact in the first quarter of 2010, the period when the provision was enacted into law, through a write-off of the deferred tax asset associated with those previously expected future income tax
deductions. Other provisions of the U.S. Healthcare Legislation relating to our postretirement benefit plans will affect the measurement of our obligations under those plans, but those impacts are not
expected to be significant.
Impacts to our 2011 Results
We recorded the following amounts in 2011 as a result of the U.S. Healthcare Legislation:
Impacts to our 2010 Results
We recorded the following amounts in 2010 as a result of the U.S. Healthcare Legislation:
Anticipated Future Financial Impacts
We expect to record the following amounts in 2012 as a result of the U.S. Healthcare Legislation:
These estimated impacts on our 2012 results are reflected in our 2012 financial guidance (see the “Our Financial Guidance for 2012” section of this Financial Review for additional information).
•
an increase, from 15.1% to 23.1%, in the minimum rebate on branded prescription drugs sold to Medicaid beneficiaries (effective January 1, 2010);
• extension of Medicaid prescription drug rebates to drugs dispensed to enrollees in certain Medicaid managed care organizations (effective March 23, 2010);
• expansion of the types of institutions eligible for the “Section 340B discounts” for outpatient drugs provided to hospitals meeting the qualification criteria under Section 340B of the Public Health Service
Act of 1944 (effective January 1, 2010);
•
discounts on branded prescription drug sales to Medicare Part D participants who are in the Medicare
“
coverage gap,
”
also known as the
“
doughnut hole
”
(
effective January 1, 2011); and
• a fee payable to the federal government (which is not deductible for U.S. income tax purposes) based on our prior-calendar-year share relative to other companies of branded prescription drug sales to
specified government programs (effective January 1, 2011, with the total fee to be paid each year by the pharmaceutical industry increasing annually through 2018).
•
$
648 million recorded as a reduction to
Revenues,
related to the higher, extended and expanded rebate provisions and the Medicare
“
coverage gap
”
discount provision; and
• $248 million recorded in
Selling, informational and administrative expenses
, related to the fee payable to the federal government referred to above.
•
$
289 million recorded as a reduction to
Revenues,
related to the higher, extended and expanded rebate provisions; and
• approximately $270 million recorded in Provision for taxes on income , related to the write-off of the deferred tax asset associated with the loss of the deduction, for tax years beginning after
December 31, 2012, of an amount equal to the subsidy from the federal government related to our prescription drug coverage offered to Medicare-eligible retirees. For additional information on the
impact of this write
-
off on our effective tax rate for 2010, see the
“
Provision for Taxes on Income
”
section of this Financial Review.
•
approximately $500 million recorded as a reduction to
Revenues,
related to the higher, extended and expanded rebate provisions and the Medicare
“
coverage gap
”
discount provision; and
• approximately $300 million recorded in
Selling, informational and administrative expenses
, related to the fee payable to the federal government referred to above.
4
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
In addition:
The Loss or Expiration of Intellectual Property Rights
As is inherent in the biopharmaceutical industry, the loss or expiration of intellectual property rights can have a significant adverse effect on our revenues. Many of our products have multiple patents that
expire at varying dates, thereby strengthening our overall patent protection. However, once patent protection has expired or has been lost prior to the expiration date as a result of a legal challenge, we
lose exclusivity on these products, and generic pharmaceutical manufacturers generally produce similar products and sell them for a lower price. This price competition can substantially decrease our
revenues for products that lose exclusivity, often in a very short period of time. While small molecule products are impacted in such a manner, biologics currently have additional barriers to entry related to
the manufacture of such products and, unlike small molecule generics, biosimilars are not necessarily identical to the reference products. Therefore, generic competition with respect to biologics may not
be as significant. A number of our current products are expected to face significantly increased generic competition over the next few years.
Our financial guidance for 2012 reflects the anticipated impact of the loss of exclusivity of various products and the expiration of certain alliance product contract rights discussed below (see the “Our
Financial Guidance for 2012” section of this Financial Review). Specifically:
• Individual Mandate —The financial impact of U.S. healthcare reform may be affected by certain additional developments over the next few years, including pending implementation guidance relating to
the U.S. Healthcare Legislation and certain healthcare reform proposals. In addition, the U.S. Healthcare Legislation requires that, except in certain circumstances, individuals obtain health insurance
beginning in 2014, and it also provides for an expansion of Medicaid coverage in 2014. It is expected that, as a result of these provisions, there will be a substantial increase in the number of Americans
with health insurance beginning in 2014, a significant portion of whom will be eligible for Medicaid. We anticipate that this will increase demand for pharmaceutical products overall. However, because of
the substantial mandatory rebates we pay under the Medicaid program, we do not anticipate that implementation of the coverage expansion will generate significant additional revenues for Pfizer. The
individual mandate is currently the subject of a legal challenge before the U.S. Supreme Court. If the Supreme Court strikes down the mandate, but allows the other provisions of the U.S. Healthcare
Legislation to remain in force, the benefits of the U.S. Healthcare Legislation to Pfizer will diminish. However, we do not expect the impact on us of any such decision to be material because we
anticipate that many Americans will choose coverage even in the absence of a mandate as a result of the government subsidies that will make purchasing coverage more affordable.
• Biotechnology Products— The U.S. Healthcare Legislation provides an abbreviated legal pathway to approve biosimilars (also referred to as “follow-on biologics”). Innovator biologics were granted 12
years of exclusivity, with a potential six-month pediatric extension. After the exclusivity period expires, the U.S. Food and Drug Administration (FDA) could approve biosimilar versions of innovator
biologics. The regulatory implementation of these provisions is ongoing and expected to take several years. However, the FDA has begun to clarify its expectations for approval via the biosimilar
pathway with the recent issuance of three draft guidance documents. Among other things, these draft guidance documents confirm that the FDA will allow biosimilar applicants to use a non-U.S.
licensed comparator in certain studies to support a demonstration of biosimilarity to a U.S.-
licensed reference product. If competitors are able to obtain marketing approval for biosimilars referencing our
biotechnology products, our biotechnology products may become subject to competition from biosimilars, with the attendant competitive pressures. Concomitantly, a better-defined biosimilars approval
pathway will assist us in pursuing approval of our own biosimilar products in the U.S.
The budget proposal submitted to Congress by President Obama in February 2012 includes a provision that would reduce the base exclusivity period for a biologics product from 12 years to seven
years. There is no corresponding pending bill designed to amend the U.S. Healthcare Legislation to alter the biologics provisions.
• Lipitor overview – In 2011, worldwide revenues from Lipitor were approximately $9.6 billion, or approximately 14% of total Pfizer revenues. Of this amount, approximately $5.0 billion was generated in
the U.S. and approximately $4.6 billion was generated in international markets, including approximately $859 million in emerging markets. We expect that the losses of exclusivity for Lipitor in the U.S.
and various international markets discussed below will have a significant adverse impact on our revenues in 2012 and subsequent years.
•
Lipitor in the U.S.
–
In November 2011, we lost exclusivity in the U.S. for Lipitor.
Pfizer announced in June 2008 that we entered into an agreement providing a license to Ranbaxy to sell generic versions of Lipitor and Caduet in the U.S effective November 30, 2011. In addition, the
agreement provides a license for Ranbaxy to sell a generic version of Lipitor beginning on varying dates in several additional countries. (See Notes to Consolidated Financial Statements – Note 17.
Commitments and Contingencies for a discussion of certain litigation relating to this agreement.) We also granted Watson Pharmaceuticals, Inc. (Watson) the exclusive right to sell the authorized
generic version of Lipitor in the U.S. for a period of five years, which commenced on November 30, 2011. As Watson’s exclusive supplier, we manufacture and sell generic atorvastatin tablets to
Watson. We expect the entry of multi-source generic competition in the U.S., with attendant increased competitive pressures, following the end of Ranbaxy’s 180-day generic exclusivity period in late
May 2012.
Through the end of 2011, sales of Lipitor in the U.S. were reported in our Primary Care business unit. Beginning in 2012, sales of Lipitor in the U.S. will be reported in our Established Products business
unit.
2011 Financial Report
5

Financial Review
Pfizer Inc. and Subsidiary Companies
In addition, we expect to lose exclusivity for various other products in various markets over the next few years, including the following in 2012:
For additional information, including with regard to the expiration of the patents for various products in the U.S., EU and Japan, see the “Patents and Intellectual Property Rights” section of our 2011
Annual Report on Form 10-K.
In Alliance revenues, we expect to be negatively impacted by the following over the next few years.
Pipeline Productivity and Regulatory Environment
The discovery and development of safe, effective new products, as well as the development of additional uses for existing products, are necessary for the continued strength of our businesses. We are
confronted by increasing regulatory scrutiny of drug safety and efficacy, even as we continue to gather safety and other data on our products, before and after the products have been launched. Our
product lines must be replenished over time in order to offset revenue losses when products lose their exclusivity, as well as to provide for revenue and earnings growth. We devote considerable
resources to research and development (R&D) activities. These activities involve a high degree of risk and may take many years, and with respect to any specific research and development project, there
can be no assurance that the development of any particular product candidate or new indication for an in-line product will achieve desired clinical endpoints and safety profile, will be approved by
regulators or will be successful commercially. On February 1, 2011, we announced a new research and productivity initiative to accelerate our strategies to improve innovation and overall productivity in
R&D by prioritizing areas with the greatest scientific and commercial promise, utilizing appropriate risk/return profiles and focusing on areas with the highest potential to deliver value in the near term and
over time.
During the development of a product, we conduct clinical trials to provide data on the drug’s safety and efficacy to support the evaluation of its overall benefit-risk profile for a particular patient
population. In addition, after a product has been approved and launched, we continue to monitor its safety as long as it is available to patients, and post-marketing trials may be conducted, including trials
requested by regulators and trials that we do voluntarily to gain additional medical knowledge. For the entire life of the product, we collect safety data and report potential problems to the FDA. The FDA
may evaluate potential safety concerns and take regulatory actions in response, such as updating a product’s labeling, restricting the use of the product, communicating new safety information to the
public, or, in rare cases, removing a product from the market.
• Lipitor in international markets—Lipitor lost exclusivity in Australia in February 2012; in Japan in 2011; and in Brazil, Canada, Spain and Mexico in 2010; and it has lost exclusivity in nearly all emerging
market countries. We do not expect that Lipitor revenues in emerging markets will be materially impacted over the next several years by the loss of exclusivity. Lipitor will have lost exclusivity in the
majority of European markets by May 2012.
Prior to loss of exclusivity, sales of Lipitor in international markets, except for those in emerging markets, are reported in our Primary Care business unit. Typically, as of the beginning of the fiscal year
following loss of exclusivity, sales of Lipitor in international markets, except for those in emerging markets, are reported in our Established Products business unit.
• Other loss of exclusivity impacts—In the U.S., we lost exclusivity for Effexor XR in July 2010, for Aricept 5mg and 10mg tablets (included in Alliance revenue) in November 2010, for Vfend tablets in
February 2011, for Xalatan in March 2011 and for Caduet in November 2011. The basic U.S. patent (including the six-month pediatric exclusivity period) for Protonix expired in January 2011. The basic
patent for Vfend tablets in Brazil expired in January 2011. We lost exclusivity for Aromasin in the U.S. in April 2011 and in the European Union (EU) in July 2011. We lost exclusivity for Xalatan and
Xalacom in 15 major European markets in January 2012. We lost exclusivity for Aricept in many of the major European markets in February 2012.
• Geodon in the U.S. in March 2012;
•
Revatio tablet in the U.S. in September 2012, which reflects the extension of the exclusivity period from March to September 2012 as the result of a pediatric extension; and
•
Detrol IR in the U.S. in September 2012.
•
Aricept
—
Our rights to Aricept in Japan will return to Eisai Co., Ltd. in December 2012. We expect to lose exclusivity for the Aricept 23mg tablet in the U.S. in July 2013.
•
Spiriva—Our collaboration with Boehringer Ingelheim (BI) for Spiriva will expire on a country-by-country basis between 2012 and 2016. As a result, we expect to experience a graduated decline in
revenues from Spiriva during that period. Our collaboration with BI for Spiriva will expire in the EU from 2012 and 2016, in 2014 in the U.S. and Japan, and by 2016 in all other countries where the
collaboration exists.
•
Enbrel—Our U.S. and Canada collaboration agreement with Amgen Inc. for Enbrel will expire in October 2013. While we are entitled to royalties for 36 months thereafter, we expect that those
royalties will be significantly less than our current share of Enbrel profits from U.S. and Canada sales. Outside of the U.S. and Canada, our exclusive rights to Enbrel continue in perpetuity.
•
Rebif—Our collaboration agreement with EMD Serono Inc. (Serono) to co-promote Rebif in the U.S. will expire either at the end of 2013 or the end of 2015, depending on the outcome of pending
litigation between Pfizer and Serono concerning the interpretation of the agreement. We believe that we are entitled to a 24-month extension of the agreement to the end of 2015. Serono believes
that we are not entitled to the extension and that the agreement will expire at the end of 2013. The lower court ruled in our favor and dismissed Serono’s complaint, and Serono has appealed the
decision. For additional information, see Notes to Consolidated Financial Statements
–
–
Note 17. Commitments and Contingencies
.
6
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
Pricing and Access Pressures
Governments, managed care organizations and other payer groups continue to seek increasing discounts on our products through a variety of means such as leveraging their purchasing power,
implementing price controls, and demanding price cuts (directly or by rebate actions). In particular, as a result of the economic environment, the industry has experienced significant pricing pressures in
certain European and emerging market countries. There were government-mandated price reductions for certain biopharmaceutical products in certain European and emerging market countries in 2011,
and we anticipate continuing pricing pressures in Europe and emerging markets in 2012. Also, health insurers and benefit plans continue to limit access to certain of our medicines by imposing formulary
restrictions in favor of the increased use of generics. In prior years, Presidential advisory groups tasked with reducing healthcare spending have recommended and legislative changes have been
proposed that would allow the U.S. government to directly negotiate prices with pharmaceutical manufacturers on behalf of Medicare beneficiaries, which we expect would restrict access to and
reimbursement for our products. There have also been a number of legislative proposals seeking to allow importation of medicines into the U.S. from countries whose governments control the price of
medicines, despite the increased risk of counterfeit products entering the supply chain. If importation of medicines is allowed, an increase in cross-border trade in medicines subject to foreign price
controls in other countries could occur and negatively impact our revenues.
In August 2011, the federal Budget Control Act of 2011 (the Act) was enacted in the U.S. The Act includes provisions to raise the U.S. Treasury Department’s borrowing limit, known as the debt ceiling,
and provisions to reduce the federal deficit by $2.4 trillion between 2012 and 2021. Deficit-reduction targets include $900 billion of discretionary spending reductions associated with the Department of
Health and Human Services and various agencies charged with national security, but those discretionary spending reductions do not include programs such as Medicare and Medicaid or direct changes
to pharmaceutical pricing, rebates or discounts. A Joint Select Committee of Congress (the Committee) was appointed to identify the remaining $1.5 trillion of deficit reductions by November 23, 2011, but
no recommendations were made by the Committee prior to the deadline. As a result, the Office of Management and Budget (OMB) is now responsible for identifying the remaining $1.5 trillion of deficit
reductions, which will be divided evenly between defense and non-defense spending. Under this OMB fallback review process, Social Security, Medicaid, Veteran Benefits and certain other spending
categories are excluded from consideration, but reductions in payments to Medicare providers may be made, although any such reductions are prohibited by law from exceeding 2%. Additionally, certain
payments to Medicare Part D plans, such as low-income subsidy payments, are exempt from reduction. While we do not know the specific nature of the spending reductions under the Act that will affect
Medicare, we do not expect that those reductions will have a material adverse impact on our results of operations. However, any significant spending reductions affecting Medicare, Medicaid or other
publicly funded or subsidized health programs that may be implemented, and/or any significant additional taxes or fees that may be imposed on us, as part of any broader deficit-reduction effort could
have an adverse impact on our results of operations.
Competition Among Branded Products
Many of our products face competition in the form of branded products, which treat similar diseases or indications. These competitive pressures can have an adverse impact on our future revenues.
The Global Economic Environment
In addition to the industry-specific factors discussed above, we, like other businesses, continue to face the effects of the challenging economic environment, which have impacted our biopharmaceutical
operations in the U.S. and Europe, affecting the performance of products such as Lipitor, Celebrex and Lyrica. We believe that patients, experiencing the effects of the challenging economic environment,
including high unemployment levels, and increases in co-pays, sometimes are switching to generics, delaying treatments, skipping doses or using less effective treatments to reduce their costs.
Challenging economic conditions in the U.S. also have increased the number of patients in the Medicaid program, under which sales of pharmaceuticals are subject to substantial rebates and, in many
states, to formulary restrictions limiting access to brand-name drugs, including ours. In addition, during 2011, we continued to experience pricing pressure as a result of the economic environment in
Europe and in a number of emerging markets, with government-mandated reductions in prices for certain biopharmaceutical products in certain European and emerging market countries.
Significant portions of our revenues and earnings are exposed to changes in foreign exchange rates. We seek to manage our foreign exchange risk in part through operational means, including managing
same-currency revenues in relation to same-currency costs and same-currency assets in relation to same-currency liabilities. Depending on market conditions, foreign exchange risk also is managed
through the use of derivative financial instruments and foreign currency debt. As we operate in multiple foreign currencies, including the euro, the U.K. pound, the Japanese yen, the Canadian dollar and
approximately 100 other currencies, changes in those currencies relative to the U.S. dollar will impact our revenues and expenses. If the U.S. dollar weakens against a specific foreign currency, our
revenues will increase, having a positive impact, and our overall expenses will increase, having a negative impact, on net income. Likewise, if the U.S. dollar strengthens against a specific foreign
currency, our revenues will decrease, having a negative impact, and our overall expenses will decrease, having a positive impact on net income. Therefore, significant changes in foreign exchange rates
can impact our results and our financial guidance.
Despite the challenging financial markets, Pfizer maintains a strong financial position. Due to our significant operating cash flows, financial assets, access to capital markets and available lines of credit
and revolving credit agreements, we continue to believe that we have the ability to meet our liquidity needs for the foreseeable future. Our long-term debt is rated high quality by both Standard & Poor’s
and Moody’s Investors Service. As market conditions change, we continue to monitor our liquidity position. We have taken and will continue to take a conservative approach to our financial investments.
Both short-term and long-term investments consist primarily of high-quality, highly liquid, well-diversified, available-for-sale debt securities. For further discussion of our financial condition, see the
“Analysis of Financial Condition, Liquidity and Capital Resources” section of this Financial Review.
2011 Financial Report
7
Financial Review
Pfizer Inc. and Subsidiary Companies
Our Strategy
We believe that our medicines provide significant value for both healthcare providers and patients, not only from the improved treatment of diseases but also from a reduction in other healthcare costs,
such as emergency room or hospitalization costs, as well as improvements in health, wellness and productivity. We continue to actively engage in dialogues about the value of our products and how we
can best work with patients, physicians and payers to prevent and treat disease and improve outcomes. We will work within the current legal and pricing structures, as well as continue to review our
pricing arrangements and contracting methods with payers, to maximize access to patients and minimize any adverse impact on our revenues.
If a decision is made to separate Animal Health and Nutrition from the Company, then, following those separations, Pfizer will be a global biopharmaceutical company with a portfolio of innovative in-line
products and a productive R&D organization; a portfolio of unpatented products that help meet the global need for less expensive, quality medicines; and a complementary Consumer Healthcare business
with several well
-known brands.
In response to the challenging operating environment, we have taken and continue to take many steps to strengthen our Company and better position ourselves for the future. We believe in a
comprehensive approach to our challenges—organizing our business to maximize research, development and commercial opportunities, improving the performance of our innovative core, making the
right capital allocation decisions, and protecting our intellectual property.
We continue to closely evaluate our global research and development function and pursue strategies to improve innovation and overall productivity by prioritizing areas with the greatest scientific and
commercial promise, utilizing appropriate risk/return profiles and focusing on areas with the highest potential to deliver value in the near term and over time. To that end, our research primarily focuses on
five high-priority areas that have a mix of small and large molecules—immunology and inflammation; oncology; cardiovascular, metabolic and endocrine diseases; neuroscience and pain; and vaccines.
In addition to reducing the number of disease areas of focus, we are realigning and reducing our research and development footprint, and outsourcing certain functions that do not drive competitive
advantage for Pfizer. As a result of these actions, we expect significant reductions in our annual research and development expenses, which are reflected in our 2012 financial guidance, and we expect to
incur significant costs, which are also reflected in our 2012 financial guidance. For additional information, see the “Our Financial Guidance for 2012” and “Costs and Expenses—Restructuring Charges
and Other Costs Associated with Acquisitions and Cost-Reduction/Productivity Initiatives” sections of this Financial Review.
While a significant portion of R&D is done internally, we continue to seek to expand our pipeline by entering into agreements with other companies to develop, license or acquire promising compounds,
technologies or capabilities. Collaboration, alliance and license agreements and acquisitions allow us to capitalize on these compounds to expand our pipeline of potential future products. In addition,
collaborations and alliances allow us to share risk and to access external scientific and technological expertise.
For information about our pending new drug applications (NDA) and supplemental filings, see the “Revenues—Product Developments-Biopharmaceutical” section of this Financial Review.
Our acquisition strategy included the acquisition of Wyeth in 2009. We continue to build on our broad portfolio of businesses through various business development transactions. See the “Our Business
Development Initiatives” section of this Financial Review for information on our recent transactions and strategic investments that we believe complement our businesses.
We continue to aggressively defend our patent rights against increasingly aggressive infringement whenever appropriate (see Notes to Consolidated Financial Statements— Note 17. Commitments and
Contingencies ), and we will continue to support efforts that strengthen worldwide recognition of patent rights while taking necessary steps to ensure appropriate patient access. In addition, we will
continue to employ innovative approaches to prevent counterfeit pharmaceuticals from entering the supply chain and to achieve greater control over the distribution of our products, and we will continue to
participate in the generics market for our products, whenever appropriate, once they lose exclusivity.
We remain focused on achieving an appropriate cost structure for the Company. For information regarding our cost-reduction and productivity initiatives, see the “Costs and Expenses—Restructuring
Charges and Other Costs Associated with Acquisitions and Cost-Reduction/Productivity Initiatives” section of this Financial Review.
Our strategy also includes directly enhancing shareholder value through dividends and share repurchases. On December 12 2011, our Board of Directors declared a first-quarter 2012 dividend of $0.22
per share, an increase from the $0.20 per-share quarterly dividend paid during 2011. Also on December 12, 2011, our Board of Directors authorized a new $10 billion share-repurchase plan. We expect
to repurchase approximately $5 billion of our common stock during 2012, with the remaining authorized amount available in 2013 and beyond.
8
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
Our Business Development Initiatives
We are committed to capitalizing on growth opportunities by advancing our own pipeline and maximizing the value of our in-
line products, as well as through various forms of business development, which
can include alliances, licenses, joint ventures, dispositions and acquisitions. We view our business-
development activity as an enabler of our strategies, and we seek to generate profitable revenue growth
and enhance shareholder value by pursuing a disciplined, strategic and financial approach to evaluating business development opportunities. We are especially interested in opportunities in our five high-
priority therapeutic areas—immunology and inflammation; oncology; cardiovascular, metabolic and endocrine diseases; neuroscience and pain; and vaccines. The most significant recent transactions are
described below.
• In early 2011, we announced that we were conducting a strategic review of all of our businesses and assets. On July 7, 2011, we announced our decisions to explore strategic alternatives for our
Animal Health and Nutrition businesses that may include, among other things, a full or partial separation of each of these businesses through a spin-off, sale, or other transaction. We believe these
potential actions may create greater shareholder value, enable us to become a more focused organization and optimize capital allocation. Given the separate and distinct nature of Animal Health and
Nutrition, we may pursue a different strategic alternative for each of these businesses. Although the timeline for each evaluation may differ, we expect to announce our strategic decision for each of
these businesses in 2012 and to complete any separation of these businesses between July 2012 and July 2013.
We will continue to assess our businesses and assets as part of our regular, ongoing portfolio review process and also continue to consider business development activities for our businesses.
• On February 26, 2012, we completed our acquisition of Alacer Corp., a privately owned company that manufactures, markets and distributes vitamin supplements, including Emergen-C, primarily in the
U.S.
• On December 1, 2011, we completed our acquisition of the consumer healthcare business of Ferrosan Holding A/S, a Danish company engaged in the sale of science-based consumer healthcare
products, including dietary supplements and lifestyle products, primarily in the Nordic region and the emerging markets of Russia and Central and Eastern Europe. Our acquisition of Ferrosan’s
consumer healthcare business strengthens our presence in dietary supplements with a new set of brands and pipeline products. Also, we believe that the acquisition allows us to expand the marketing
of Ferrosan
’
s brands through Pfizer
’
s global footprint and provide greater distribution and scale for certain Pfizer brands, such as Centrum and Caltrate, in Ferrosan
’
s key markets.
•
On November 30, 2011, we completed our acquisition of Excaliard Pharmaceuticals, Inc. (Excaliard), a privately owned biopharmaceutical company focused on developing novel drugs for the treatment
of skin fibrosis, more commonly referred to as skin scarring. Excaliard‘s lead compound, EXC-
001, is an antisense oligonucleotide designed to interrupt the process of fibrosis by inhibiting expression of
connective tissue growth factor (CTGF) and has produced positive clinical results in reducing scar severity in certain Phase 2 trials. For additional information, see Notes to Consolidated Financial
Statements
—
Note 2C. Acquisitions, Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Other Acquisitions
.
• In October 2011, we entered into an agreement with GlycoMimetics, Inc. for their investigational compound GMI-1070. GMI-1070 is a pan-selectin antagonist currently in Phase 2 development for the
treatment of vaso-occlusive crisis associated with sickle cell disease. GMI-1070 has received Orphan Drug and Fast Track status from the FDA. Under the terms of the agreement, Pfizer will receive an
exclusive worldwide license to GMI-1070 for vaso-occlusive crisis associated with sickle cell disease and for other diseases for which the drug candidate may be developed. GlycoMimetics will remain
responsible for completion of the ongoing Phase 2 trial under Pfizer’s oversight, and Pfizer will then assume all further development and commercialization responsibilities. GlycoMimetics would be
entitled to payments up to approximately $340 million, including an upfront payment as well as development, regulatory and commercial milestones. GlycoMimetics is also eligible for royalties on any
sales.
• On September 20, 2011, we completed our cash tender offer for the outstanding shares of Icagen, Inc. (Icagen), resulting in an approximately 70% ownership of the outstanding shares of Icagen, a
biopharmaceutical company focused on discovery, development and commercialization of novel orally-administered small molecule drugs that modulate ion channel targets. On October 27, 2011, we
acquired all of the remaining shares of Icagen. For additional information, see Notes to Consolidated Financial Statements— Note 2C. Acquisitions, Divestitures, Collaborative Arrangements and
Equity
-
Method Investments: Other Acquisitions
.
• On August 1, 2011, we sold our Capsugel business for approximately $2.4 billion in cash. For additional information, see Notes to Consolidated Financial Statements— Note 2D. Acquisitions,
Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Divestitures
.
•
On January 31, 2011 (the acquisition date), we completed a tender offer for the outstanding shares of common stock of King at a purchase price of $14.25 per share in cash and acquired approximately
92.5% of the outstanding shares. On February 28, 2011, we acquired all of the remaining shares of King for $14.25 per share in cash. As a result, the total fair value of consideration transferred for King
was approximately $3.6 billion in cash ($3.2 billion, net of cash acquired). Our acquisition of King complements our current portfolio of pain treatments in our Primary Care business unit and provides
potential growth opportunities in our Established Products and Animal Health business units. For additional information, see Notes to Consolidated Financial Statements— Note 2B. Acquisitions,
Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Acquisition of King Pharmaceuticals, Inc.
King’s principal businesses consist of a prescription pharmaceutical business focused on delivering new formulations of pain treatments designed to discourage common methods of misuse and abuse;
the Meridian auto-injector business for emergency drug delivery, which develops and manufactures the EpiPen; an established products portfolio; and an animal health business that offers a variety of
feed-additive products for a wide range of species.
2011 Financial Report
9

Financial Review
Pfizer Inc. and Subsidiary Companies
As a result of our acquisition of King, we recorded Inventories of $340 million, Property, plant and equipment (PP&E) of $412 million, Identifiable intangible assets of $2.1 billion and Goodwill of $765
million. For additional information, see Notes to Consolidated Financial Statements— Note 2B. Acquisitions, Divestitures, Collaborative Arrangements and Equity-Method Investments: Acquisition of King
Pharmaceuticals, Inc.
As of the acquisition date, Identifiable intangible assets included the following:
Our Financial Guidance for 2012
We forecast 2012 revenues of $60.5 billion to $62.5 billion, Reported diluted earnings per common share (EPS) of $1.37 to $1.52 and Adjusted diluted EPS of $2.20 to $2.30. The current exchange rates
assumed in connection with the 2012 financial guidance are the mid-January 2012 exchange rates. For an understanding of Adjusted income, see the “Adjusted Income” section of this Financial Review.
o
Developed technology rights of approximately $1.8 billion, which includes EpiPen, Thrombin, Bicillin, Levoxyl, Skelaxin and Flector Patch, among others.
o
In
-
Process Research and Development (IPR&D) of approximately $300 million, which includes Vanquix, Embeda and Remoxy, among others.
• On November 8, 2010 we consummated our partnership to develop and commercialize generic medicines with Laboratório Teuto Brasileiro S.A. (Teuto) a leading generics company in Brazil. As part of
the transaction, we acquired a 40 percent equity stake in Teuto, and entered into a series of commercial agreements. The partnership is enhancing our position in Brazil, a key emerging market, by
providing access to Teuto’s portfolio of products. Through this partnership, we have access to significant distribution networks in rural and suburban areas in Brazil and the opportunity to register and
commercialize Teuto’s products in various markets outside of Brazil. For additional information, see also Notes to Consolidated Financial Statements—
Note 2F. Acquisitions, Divestitures, Collaborative
Arrangements and Equity
-
Method Investments: Equity
-
Method Investments
.
• On October 18, 2010, we entered into a strategic global agreement with Biocon, a biotechnology company based in India, for the worldwide commercialization of Biocon’s biosimilar versions of insulin
and insulin analog products: Recombinant Human Insulin, Glargine, Aspart and Lispro. We will have exclusive rights to commercialize these products globally, with certain exceptions, including co-
exclusive rights for all of the products with Biocon in Germany, India and Malaysia. We will also have co-
exclusive rights with existing Biocon licensees with respect to certain of these products, primarily
in a number of developing markets. Biocon will remain responsible for the clinical development, manufacture and supply of these biosimilar insulin products, as well as for regulatory activities to secure
approval for these products in various markets.
• On October 6, 2010, we completed our acquisition of FoldRx Pharmaceuticals, Inc. (FoldRx), a privately held drug discovery and clinical development company, whose portfolio includes clinical and
preclinical programs for investigational compounds to treat diseases caused by protein misfolding. FoldRx’s lead product candidate, Vyndaqel (tafamidis meglumine), was approved in the EU in
November 2011 and our new drug application was accepted for review in the U.S. in February 2012. This product is a first-in-class oral therapy for the treatment of transthyretin familial amyloid
polyneuropathy (TTR-FAP), a progressively fatal genetic neurodegenerative disease, for which liver transplant is the only treatment option currently available. Our acquisition of FoldRx is expected to
strengthen our presence in the growing rare medical disease market, which complements our Specialty Care unit.
For additional information regarding Vyndaqel (tafamidis meglumine), see the “Product Developments – Biopharmaceutical” section of this Financial Review. For additional information about the
acquisition, see Notes to Consolidated Financial Statements—
Note 2C. Acquisitions, Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Other Acquisitions
.
• On October 30, 2009, we and GlaxoSmithKline plc (GSK) created a new company, ViiV Healthcare Limited (ViiV), which is focused solely on research, development and commercialization of human
immunodeficiency virus (HIV) medicines. We and GSK have contributed certain HIV-
related product and pipeline assets to the new company. ViiV has a broad product portfolio of 11 marketed products,
including innovative leading therapies such as Combivir and Kivexa products and Selzentry/Celsentri (maraviroc), and has a pipeline of three medicines. ViiV has contracted R&D and manufacturing
services directly from GSK and us and also has entered into a research alliance agreement with GSK and us. Under this alliance, ViiV is investing in our and GSK’
s programs for discovery research and
development into HIV medicines. ViiV has exclusive rights of first negotiation in relation to any new HIV-related medicines developed by either GSK or us. For additional information, see Notes to
Consolidated Financial Statements––
Note 2F. Acquisitions, Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Equity
-
Method Investments
.
• On October 15, 2009 (the acquisition date), we acquired all of the outstanding equity of Wyeth in a cash-and-stock transaction, valued at $50.40 per share of Wyeth common stock, or a total of
approximately $68.2 billion, based on the closing market price of Pfizer common stock on the acquisition date. In connection with our acquisition of Wyeth, we are required to divest certain animal health
assets. Certain of these assets were sold in 2009. In addition, in 2010, we completed the divestiture of certain animal health products and related assets in Australia, China, the EU, Switzerland and
Mexico, and in 2011, we divested certain animal health products and related assets in South Korea. It is possible that additional divestitures of animal health assets may be required based on ongoing
regulatory reviews in other jurisdictions worldwide, but they are not expected to be significant to our business. For additional information, see the “Acquisition of Wyeth” section of this Financial Review
and see Notes to Consolidated Financial Statements—
Note 2A. Acquisitions, Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Acquisition of Wyeth.
10
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
A reconciliation of 2012 Adjusted income and Adjusted diluted EPS guidance to 2012 Reported Net income attributable to Pfizer Inc. and Reported diluted EPS attributable to Pfizer Inc. common
shareholders guidance follows:
Our 2012 financial guidance is subject to a number of factors and uncertainties—as described in the “Forward-Looking Information and Factors That May Affect Future Results”, “Our Operating
Environment” and “Our Strategy” sections of this Financial Review and in Part I, Item 1A, “Risk Factors”, of our 2011 Annual Report on Form 10-K.
SIGNIFICANT ACCOUNTING POLICIES AND APPLICATION OF CRITICAL ACCOUNTING ESTIMATES
For a description of our significant accounting policies, see Notes to Consolidated Financial Statements–– Note 1. Significant Accounting Policies .
Of these policies, the following are considered critical to an understanding of Pfizer’s Consolidated Financial Statements as they require the application of the most difficult, subjective and complex
judgments: (i) Acquisitions (Note 1D); (ii) Fair Value (Note 1E); (iii) Revenues (Note 1G); (iv) Asset Impairment Reviews (Note 1K); (v) Tax Contingencies (Note 1O); (vi) Benefit Plans (Note 1P);
(vii) Legal and Environmental Contingencies (Note 1Q).
Below are some of our more critical accounting estimates. See also Estimates and Assumptions (Note 1C) for a discussion about the risks associated with estimates and assumptions.
Acquisitions and Fair Value
For a discussion about the application of Fair Value to our recent acquisitions, see Notes to Consolidated Financial Statements— Note 2. Acquisitions, Divestitures, Collaborative Arrangements and
Equity-Method Investments .
For a discussion about the application of Fair Value to our investments, see Notes to Consolidated Financial Statements— Note 7. Financial Instruments .
For a discussion about the application of Fair Value to our benefit plan assets, see Notes to Consolidated Financial Statements–– Note 11. Pension and Postretirement Benefit Plans and Defined
Contribution Plans .
For a discussion about the application of Fair Value to our asset impairment reviews, see “Asset Impairment Reviews––Long-Lived Assets” below.
Revenues
As is typical in the biopharmaceutical industry, our gross product sales are subject to a variety of deductions that are generally estimated and recorded in the same period that the revenues are
recognized and primarily represent rebates and discounts to government agencies, wholesalers, distributors and managed care organizations with respect to our biopharmaceutical products. See also
Notes to Consolidated Financial Statements–– Note 1G. Significant Accounting Policies: Revenues for a detailed description of the nature of our sales deductions and our procedures for estimating our
obligations. For example,
If any of our ratios, factors, assessments, experiences or judgments, are not indicative or accurate predictors of our future experience, our results could be materially affected. Although the amounts
recorded for these sales deductions are heavily dependent on estimates and assumptions, historically, our adjustments to actual have not been material; on a quarterly basis, they generally have been
less than 1.0% of biopharmaceutical net sales and can result in a net increase to income or a net decrease to income. The sensitivity of our estimates can vary by program, type of customer and
geographic location. However, estimates associated with U.S. Medicaid and contract rebates are most at-risk for material adjustment because of the extensive time delay between the recording of the
accrual and its ultimate settlement, an interval that can range up to one year. Because of this time lag, in any given quarter, our adjustments to actual can incorporate revisions of several prior quarters.
Amounts recorded for sales deductions can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For further information
about the risks associated with estimates and assumptions, see Notes to Consolidated Financial Statements— Note1C. Significant Accounting Policies: Estimates and Assumptions.
FULL
-
YEAR 2012 GUIDANCE
(BILLIONS OF DOLLARS, EXCEPT PER SHARE AMOUNTS)
NET INCOME
DILUTED EPS
Adjusted income/diluted EPS
guidance
~$16.5
-
$17.3
~$2.20
-
$2.30
Purchase accounting impacts of transactions completed as of 12/31/11
(4.1)
(0.54)
Acquisition-related costs
(1.0 - 1.2)
(0.13 - 0.15)
Non
-
acquisition
-
related restructuring costs
(0.9
-
1.1)
(0.11
-
0.14)
Reported net income attributable to Pfizer Inc./diluted EPS guidance
~$10.1
-
$
11.3
~$1.37
-
$
1.52
Does not assume the completion of any business-development transactions not completed as of December 31, 2011, including any one-
time upfront payments associated with such transactions. Also
excludes the potential effects of the resolution of litigation
-
related matters not substantially resolved as of December 31, 2011.
For an understanding of Adjusted income, see the “Adjusted Income” section of this Financial Review.
Includes amounts related to our initiatives to reduce R&D spending, including our realigned R&D footprint, and amounts related to other cost-
reduction and productivity initiatives. In our reconciliation
between Net income attributable to Pfizer Inc.
, as reported under principles generally accepted in the United States of America (U.S. GAAP), and Adjusted income, and in our reconciliation between
diluted EPS, as reported under U.S. GAAP, and Adjusted diluted EPS, these amounts will be categorized as Certain Significant Items.
• For Medicaid, Medicare and contract rebates, we use experience ratios, which may be adjusted to better match our current experience or our expected future experience.
• For contractual or legislatively mandated deductions outside of the U.S., we use estimated allocation factors, based on historical payments and some third-party reports, to project the expected level of
reimbursement.
• For sales returns, we perform calculations in each market that incorporate the following, as appropriate: local returns policies and practices; returns as a percentage of sales; an understanding of the
reasons for past returns; estimated shelf life by product; and an estimate of the amount of time between shipment and return or lag time; and any other factors that could impact the estimate of future
returns, such as loss of exclusivity, product recalls or a changing competitive environment.
•
For sales incentives, we use our historical experience with similar incentives programs to predict customer behavior.
2011 Financial Report
11
(a)
(a)
(b)
(c)
(a)
(
b)
(c)

Financial Review
Pfizer Inc. and Subsidiary Companies
Asset Impairment Reviews––Long-Lived Assets
We review all of our long-lived assets, including goodwill and other intangible assets, for impairment indicators throughout the year and we perform detailed impairment testing for goodwill and indefinite-
lived assets annually and for all other long-lived assets whenever impairment indicators are present. When necessary, we record charges for impairments of long-lived assets for the amount by which the
fair value is less than the carrying value of these assets.
Our impairment review processes are described in the Notes to Consolidated Financial Statements––
Note 1K. Significant Accounting Policies: Amortization of Intangible Assets, Depreciation and Certain
Long-Lived Assets and, for deferred tax assets, in Note 1O. Significant Accounting Policies: Deferred Tax Assets and Liabilities and Income Tax Contingencies.
Examples of events or circumstances that may be indicative of impairment include:
Our impairment reviews of most of our long-lived assets depend heavily on the determination of fair value, as defined by U.S. GAAP, and these judgments can materially impact our results of operations.
A single estimate of fair value can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For information about the risks
associated with estimates and assumptions, see Notes to Consolidated Financial Statements–– Note 1C. Significant Accounting Policies: Estimates and Assumptions.
Intangible Assets Other than Goodwill
As a result of our intangible asset impairment review work, described in detail below, we recognized a number of impairments of intangible assets other than goodwill.
We recorded the following intangible asset impairment charges in Other deductions––net:
• A significant adverse change in legal factors or in the business climate that could affect the value of the asset. For example, a successful challenge of our patent rights likely would result in generic
competition earlier than expected.
•
A significant adverse change in the extent or manner in which an asset is used. For example, restrictions imposed by the FDA or other regulatory authorities could affect our ability to manufacture or sell
a product.
• A projection or forecast that demonstrates losses or reduced profits associated with an asset. This could result, for example, from a change in a government reimbursement program that results in an
inability to sustain projected product revenues and profitability. This also could result from the introduction of a competitor’s product that results in a significant loss of market share or the inability to
achieve the previously projected revenue growth, as well as the lack of acceptance of a product by patients, physicians and payers. For IPR&D projects, this could result from, among other things, a
change in outlook based on clinical trial data, a delay in the projected launch date or additional expenditures to commercialize the product.
• In 2011, $863 million, which includes (i) approximately $475 million of IPR&D assets, primarily related to two compounds for the treatment of certain autoimmune and inflammatory diseases;
(ii) approximately $195 million related to our indefinite-lived biopharmaceutical brand, Xanax; and (iii) approximately $185 million of Developed Technology Rights comprising the impairments of five
other assets. These impairment charges reflect, among other things, the impact of new scientific findings and the increased competitive environment. The impairment charges are associated with the
following: Worldwide Research and Development ($394 million); Established Products ($193 million); Specialty Care ($135 million); Primary Care ($56 million); Oncology ($56 million); Animal Health
($17 million); and other ($12 million).
• In 2010, $2.2 billion, which included (i) approximately $950 million of IPR&D assets, primarily Prevnar 13/Prevenar 13 Adult, a compound for the prevention of pneumococcal disease in adults age 50
and older, and Neratinib, a compound for the treatment of breast cancer; (ii) approximately $700 million of indefinite-lived Brands, related to Third Age, infant formulas for the first 12-36 months of age,
and Robitussin, a cough suppressant; and (iii) approximately $550 million of Developed Technology Rights, primarily Thelin, a product that treated pulmonary hypertension and Protonix, a product that
treats erosive gastroesophageal reflux disease. These impairment charges, most of which occurred in the third quarter of 2010, reflect, among other things, the following: for IPR&D assets, the impact
of changes to the development programs, the projected development and regulatory timeframes and the risk associated with these assets; for Brand assets, the current competitive environment and
planned investment support; and, for Developed Technology Rights, in the case of Thelin, we voluntarily withdrew the product in regions where it was approved and discontinued all clinical studies
worldwide and, for the others, an increased competitive environment.
12
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
Accounting Policy and Specific Procedures
For a description of our accounting policy, see Notes to Consolidated Financial Statements–– Note 1K. Significant Accounting Policies: Amortization of Intangible Assets, Depreciation and Certain Long-
Lived Assets .
When we are required to determine the fair value of intangible assets other than goodwill, we use an income approach, specifically the multi-
period excess earnings method, also known as the discounted
cash flow method. We start with a forecast of all the expected net cash flows associated with the asset, which includes the application of a terminal value for indefinite-lived assets, and then we apply an
asset-specific discount rate to arrive at a net present value amount. Some of the more significant estimates and assumptions inherent in this approach include: the amount and timing of the projected net
cash flows, which includes the expected impact of competitive, legal and/or regulatory forces on the projections and the impact of technological risk associated with in-process research and development
assets, as well as the selection of a long-term growth rate; the discount rate, which seeks to reflect the various risks inherent in the projected cash flows; and the tax rate, which seeks to incorporate the
geographic diversity of the projected cash flows.
Future Impairment Risks
While all intangible assets other than goodwill can confront events and circumstances that can lead to impairment, in general, intangible assets other than goodwill that are most at risk of impairment
include in-process research and development assets ($1.2 billion as of December 31, 2011) and newly acquired or recently impaired indefinite-lived brand assets ($1.2 billion as of December 31, 2011).
In-process research and development assets are high-risk assets, as research and development is an inherently risky activity. Newly acquired and recently impaired indefinite-lived assets are more
vulnerable to impairment as the assets are recorded at fair value and are then subsequently measured at the lower of fair value or carrying value at the end of each reporting period. As such, immediately
after acquisition or impairment, even small declines in the outlook for these assets can negatively impact our ability to recover the carrying value and can result in an impairment charge.
Goodwill
As a result of our goodwill impairment review work, described in detail below, we concluded that none of our goodwill is impaired as of December 31, 2011, and we do not believe the risk of impairment is
significant at this time.
Accounting Policy and Specific Procedures
For a description of our accounting policy, see Notes to Consolidated Financial Statements— Note 1K. Significant Accounting Policies: Amortization of Intangible Assets, Depreciation and Certain Long-
Lived Assets .
In determining the fair value of a reporting unit, as appropriate for the individual reporting unit, we may use the market approach, the income approach or a weighted-average combination of both
approaches.
The impairment charges are associated with the following: Specialty Care ($708 million); Oncology ($396 million); Nutrition ($385 million); Consumer Healthcare ($292 million); Established Products
($182 million); Primary Care ($145 million); Worldwide Research and Development ($54 million); and other ($13 million).
• In 2009, the impairment charge of $417 million primarily relates to certain materials used in our research and development activities that were no longer considered recoverable.
• One of our indefinite-lived biopharmaceutical brands, Xanax, was written down to its fair value of $1.2 billion at the end of 2011. This asset continues to be at risk for future impairment. Any negative
change in the undiscounted cash flows, discount rate and/or tax rate could result in an impairment charge. Xanax, which was launched in the mid 1980’s and acquired in 2003, must continue to remain
competitive against its generic challengers or the associated asset may become impaired again. We re-considered and confirmed the classification of this asset as indefinite-lived. We will continue to
closely monitor this asset.
• One of our indefinite-
lived Consumer Healthcare brands, Robitussin, has a fair value that approximates its carrying value of about $500 million, which reflects an impairment charge that was taken in the
third quarter of 2010. This asset continues to be at risk for future impairment. Any negative change in the undiscounted cash flows, discount rate and/or tax rate could result in an impairment charge.
Robitussin, launched in the mid 1950’s, enjoys strong brand recognition, and is one of the leading over-the-counter cold and cough remedies in the world. Robitussin must continue to remain
competitive against its market challengers or the associated asset may become impaired again. We re-considered and confirmed the classification of this asset as indefinite-lived. We will continue to
closely monitor this asset.
•
The market approach is a historical approach to estimating fair value and relies primarily on external information. Within the market approach are two methods that we may use:
o
Guideline public company method—this method employs market multiples derived from market prices of stocks of companies that are engaged in the same or similar lines of business and that are
actively traded on a free and open market and the application of the identified multiples to the corresponding measure of our reporting unit
’
s financial performance.
o
Guideline transaction method—this method relies on pricing multiples derived from transactions of significant interests in companies engaged in the same or similar lines of business and the
application of the identified multiples to the corresponding measure of our reporting unit’s financial performance.
2011 Financial Report
13

Financial Review
Pfizer Inc. and Subsidiary Companies
Specifically, our 2011 goodwill impairment assessment involved the following:
Future Impairment Risks
While all reporting units can confront events and circumstances that can lead to impairment, we do not believe that the risk of goodwill impairment for any of our reporting units is significant at this time.
At the end of 2011, our Consumer Healthcare reporting unit has the smallest difference between fair value and book value. However, we estimate that it would take a significant negative change in the
undiscounted cash flows, the discount rate and/or the market multiples in the consumer industry for the Consumer Healthcare reporting unit goodwill to be impaired. Our Consumer Healthcare reporting
unit performance and consumer healthcare industry market multiples are highly correlated with the overall economy and our specific performance is also dependent on our and our competitors’
innovation
and marketing effectiveness, and on regulatory developments affecting claims, formulations and ingredients of our products.
For all of our reporting units, there are a number of future events and factors that may impact future results and that could potentially have an impact on the outcome of subsequent goodwill impairment
testing. For a list of these factors, see the “Forward-Looking Information and Factors That May Affect Future Results” section of this Financial Review.
Pension and Postretirement Benefit Plans
The majority of our employees worldwide are covered by defined benefit pension plans, defined contribution plans or both. In the U.S., we have both qualified and supplemental (non-qualified) defined
benefit plans, as well as other postretirement benefit plans, consisting primarily of healthcare and life insurance for retirees (see Notes to Consolidated Financial Statements— Note 1P. Pension and
Postretirement Benefit Plans and Note 11. Pension and Postretirement Benefit Plans and Defined Contribution Plans ). Beginning on January 1, 2011, for employees hired in the U.S. and Puerto Rico
after December 31, 2010, we no longer offer a defined benefit plan and, instead, offer an enhanced benefit under our defined contribution plan. In addition to the standard matching contribution by the
Company, the enhanced benefit provides an automatic Company contribution for such eligible employees based on age and years of service.
The accounting for benefit plans is highly dependent on actuarial estimates, assumptions and calculations, which result from a complex series of judgments about future events and uncertainties. For
information about the risks associated with estimates and assumptions, see Notes to Consolidated Financial Statements–– Note 1C. Significant Accounting Policies: Estimates and Assumptions. The
assumptions and actuarial estimates required to estimate the employee benefit obligations for the defined benefit and postretirement plans may include the discount rate; expected salary increases;
certain employee-related factors, such as turnover, retirement age and mortality (life expectancy); expected return on assets; and healthcare cost trend rates.
The market approach is only appropriate when the available external information is robust and deemed to be a reliable proxy for the specific reporting unit being valued; however, these assessments
may prove to be incomplete or inaccurate. Some of the more significant estimates and assumptions inherent in this approach include: the selection of appropriate guideline companies and transactions
and the determination of applicable premiums and discounts based on any differences in ownership percentages, ownership rights, business ownership forms or marketability between the reporting unit
and the guideline companies and transactions.
• The income approach is a forward-looking approach to estimating fair value and relies primarily on internal forecasts. Within the income approach, the method that we use is the discounted cash flow
method. We start with a forecast of all the expected net cash flows associated with the reporting unit, which includes the application of a terminal value, and then we apply a reporting unit-specific
discount rate to arrive at a net present value amount. Some of the more significant estimates and assumptions inherent in this approach include: the amount and timing of the projected net cash flows,
which includes the expected impact of technological risk and competitive, legal and/or regulatory forces on the projections, as well as the selection of a long
-term growth rate; the discount rate, which
seeks to reflect the various risks inherent in the projected cash flows; and the tax rate, which seeks to incorporate the geographic diversity of the projected cash flows.
• To estimate the fair value of our five biopharmaceutical reporting units, we relied solely on the income approach. We used the income approach exclusively as many of our products are sold in multiple
reporting units and as one reporting unit is geographic-based while the others are product and/or customer-based. Further, the projected cash flows from a single product may reside in up to three
reporting units at different points in future years and the discounted cash flow method would reflect the movement of products among reporting units. As such, the use of the comparable guideline
company method was not practical or reliable. However, on a limited basis and as deemed reasonable, we did attempt to corroborate our outcomes with the market approach. For the income approach,
we used the discounted cash flow method.
• To estimate the fair value of our Consumer Healthcare reporting unit, we used a combination of approaches and methods. We used the income approach and the market approach, which were
weighted equally in our analysis. We weighted them equally as we have equal confidence in the appropriateness of the approaches for this reporting unit. For the income approach, we used the
discounted cash flow method and for the market approach, we used both the guideline public company method and the guideline transaction method, which were weighted equally to arrive at our
market approach value.
• To estimate the fair value of our Nutrition and Animal Health reporting units, we used the income approach, relying exclusively on the discounted cash flow method. We relied exclusively on the income
approach as the discounted cash flow method provides a more reliable outlook of the business. However, on a limited basis and as deemed reasonable, we did attempt to corroborate our outcomes
with the market approach. (On July 7, 2011, we announced our decision to explore strategic alternatives for our Nutrition and Animal Health businesses. See the
“Our Business Development initiatives”
section of this Financial Review.)
14
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
Our assumptions reflect our historical experiences and our best judgment regarding future expectations that have been deemed reasonable by management. The judgments made in determining the
costs of our benefit plans can materially impact our results of operations.
The following table shows the expected versus actual rate of return on plan assets and the discount rate used to determine the benefit obligations for the U.S. qualified pension plans:
As a result of the global financial market downturn during 2008, the fair value of the assets held in our pension plans decreased by approximately 21% in 2008 and we estimate those losses will be
amortized over a 10-year period. In early 2009, we shifted from an explicit target asset allocation to asset allocation ranges in order to maintain flexibility in meeting minimum funding requirements and
achieving our expected return on assets. However, we did not significantly change the asset allocation during 2009 and the allocation was largely consistent with that of 2008. No further changes to the
strategic asset allocation ranges have been made, and actual allocations have remained stable throughout 2010 and 2011. Therefore, we maintained the 8.5% expected long-
term rate of return on assets
in 2011 and 2010. Any changes in the expected long-term rate of return on assets would impact net periodic benefit cost.
The assumption for the expected rate of return on assets for our U.S. and international plans reflects our actual historical return experience and our long-term assessment of forward-looking return
expectations by asset classes, which is used to develop a weighted-average expected return based on the implementation of our targeted asset allocation in our respective plans. The expected return for
our U.S. plans and the majority of our international plans is applied to the fair market value of plan assets at each year end. Holding all other assumptions constant, the effect of a 0.5 percentage-point
decline in the return-on-assets assumption would increase our 2012 U.S. qualified pension plans’ pre-tax expense by approximately $59 million.
The discount rate used in calculating our U.S. defined benefit plan obligations as of December 31, 2011, is 5.1%, which represents a 0.8 percentage-point decrease from our December 31, 2010 rate of
5.9%. The discount rate for our U.S. defined benefit plans is determined annually and evaluated and modified to reflect at year-end the prevailing market rate of a portfolio of high-quality corporate bond
investments rated AA or better that would provide the future cash flows needed to settle benefit obligations as they come due. For our international plans, the discount rates are set by benchmarking
against investment grade corporate bonds rated AA or better, including where there is sufficient data, a yield curve approach. These rate determinations are made consistent with local requirements.
Holding all other assumptions constant, the effect of a 0.1 percentage-point decrease in the discount rate assumption would increase our 2012 U.S. qualified pension plans’ pre-tax expense by
approximately $29 million and increase the U.S. qualified pension plans’ projected benefit obligations as of December 31, 2011 by approximately $233 million.
Contingencies
For a discussion about income tax contingencies, see Notes to Consolidated Financial Statements— Note 5D. Taxes on Income: Tax Contingencies.
For a discussion about legal and environmental contingencies, guarantees and indemnifications, see Notes to Consolidated Financial Statements— Note 17. Commitments and Contingencies .
2011
2010
2009
Expected annual rate of return
8.5
%
8.5
%
8.5%
Actual annual rate of return
3.8
10.8
14.2
Discount rate
5.1
5.9
6.3
2011 Financial Report
15

Financial Review
Pfizer Inc. and Subsidiary Companies
ANALYSIS OF THE CONSOLIDATED STATEMENTS OF INCOME
Revenues-Overview
Total revenues were $67.4 billion in 2011, an increase of 1% compared to 2010, due to:
partially offset by:
Total revenues of $67.1 billion in 2010 increased by approximately $17.8 billion compared to 2009, primarily due to:
partially offset by:
In 2011, Lipitor (which lost exclusivity in the U.S in November 2011), Lyrica, Prevnar 13/Prevenar 13, Enbrel and Celebrex each delivered at least $2 billion in revenues, while Viagra, Norvasc, Zyvox,
Xalatan/Xalacom (Xalatan lost exclusivity in the U.S. in March 2011), Sutent, Geodon/Zeldox, and the Premarin family each surpassed $1 billion in revenues.
In 2010, Lipitor, Enbrel, Lyrica, Prevnar 13/Prevenar 13 and Celebrex each delivered at least $2 billion in revenues, while Viagra, Xalatan/Xalacom, Effexor (Effexor XR lost exclusivity in the U.S. in July
2010), Norvasc, Prevnar/Prevenar (7-valent), Zyvox, Sutent, the Premarin family, Geodon/Zeldox and Detrol/Detrol LA each surpassed $1 billion in revenues.
In 2009, Lipitor, Lyrica and Celebrex each delivered at least $2 billion in revenues, while Norvasc, Viagra, Xalatan/Xalacom, Detrol/Detrol LA, Zyvox and Geodon/Zeldox each surpassed $1 billion in
revenues. In 2009, we did not record more than $1 billion in revenues for any individual legacy Wyeth product since the Wyeth acquisition date of October 15, 2009.
Revenues exceeded $500 million in each of 18 countries outside the U.S. in 2011 and 2010, and in each of 13 countries outside the U.S. in 2009. The increase in the number of countries outside the U.S.
in which revenues exceeded $500 million in 2010 and 2011 compared to 2009 was due to the inclusion of revenues from legacy Wyeth products for the full year in 2010. The U.S. was the only country to
contribute more than 10% of total revenues in each year.
Our policy relating to the supply of pharmaceutical inventory at domestic wholesalers, and in major international markets, is to generally maintain stocking levels under one month on average and to keep
monthly levels consistent from year to year based on patterns of utilization. We historically have been able to closely monitor these customer stocking levels by purchasing information from our customers
directly or by obtaining other third-party information. We believe our data sources to be directionally reliable but cannot verify their accuracy. Further, as we do not control this third-party data, we cannot
be assured of continuing access. Unusual buying patterns and utilization are promptly investigated.
YEAR ENDED DECEMBER 31,
% CHANGE
(MILLIONS OF DOLLARS)
2011
2010
2009
11/10
10/09
Revenues
$
67,425
$
67,057
$
49,269
1
36
Cost of sales
15,085
15,838
8,459
(5
)
87
% of revenues
22.4
%
23.6
%
17.2
%
Selling, informational and administrative expenses
19,468
19,480
14,752
—
32
% of revenues
28.9
%
29.0
%
29.9
%
Research and development expenses
9,112
9,392
7,824
(3
)
20
% of revenues
13.5
%
14.0
%
15.9
%
Amortization of intangible assets
5,585
5,403
2,877
3
88
% of revenues
8.3
%
8.1
%
5.8
%
Acquisition
-
related in
-
process research and development charges
—
125
68
(100
)
84
% of revenues
—
0.2
%
0.1
%
Restructuring charges and certain acquisition
-
related costs
2,934
3,201
4,330
(8
)
(26
)
% of revenues
4.4
%
4.8
%
8.8
%
Other deductions
—
net
2,479
4,336
285
(43
)
*
Income from continuing operations before provision for taxes on income
12,762
9,282
10,674
37
(13
)
% of revenues
18.9
%
13.8
%
21.7
%
Provision for taxes on income
4,023
1,071
2,145
276
(50
)
Effective tax rate
31.5
%
11.5
%
20.1
%
Plus: Gain from discontinued operations
—
net of tax
1,312
77
114
*
(32
)
Less: Net income attributable to noncontrolling interests
42
31
8
35
288
Net income attributable to Pfizer Inc.
$
10,009
$
8,257
$
8,635
21
(4
)
% of revenues
14.8
%
12.3
%
17.5
%
Percentages
may reflect rounding adjustments.
*
Calculation not meaningful.
•
the favorable impact of foreign exchange, which increased revenues by approximately $1.9 billion, or 3%; and
•
the inclusion of revenues of $1.3 billion, or 2% from our acquisition of King,
•
an operational decline of $2.9 billion or 4%, primarily due to the loss of exclusivity of certain products.
• the inclusion of revenues from legacy Wyeth products of $18.1 billion; and
•
the favorable impact of foreign exchange, which increased revenues by approximately $1.1 billion,
• the net revenue decrease from legacy Pfizer products of $1.4 billion resulting primarily from continuing generic competition and the loss of exclusivity on certain products.
16
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
As is typical in the biopharmaceutical industry, our gross product sales are subject to a variety of deductions, that generally are estimated and recorded in the same period that the revenues are
recognized and primarily represent rebates and discounts to government agencies, wholesalers, distributors and managed care organizations with respect to our pharmaceutical products. These
deductions represent estimates of the related obligations and, as such, judgment and knowledge of market conditions and practice are required when estimating the impact of these sales deductions on
gross sales for a reporting period. Historically, our adjustments to actual results have not been material to our overall business. On a quarterly basis, our adjustments to actual results generally have been
less than 1% of biopharmaceutical net sales and can result in either a net increase or a net decrease in income. Product-specific rebate charges, however, can have a significant impact on year-over-
year
individual product growth trends.
Certain deductions from revenues follow:
The rebates and chargebacks for 2011 were higher than 2010, primarily as a result of:
partially offset by, among other factors:
Our accruals for Medicaid rebates, Medicare rebates, performance-based contract rebates and chargebacks were $3.3 billion as of December 31, 2011 and $3.0 billion as of December 31, 2010, and
primarily are all included in Other current liabilities in our Consolidated Balance Sheets.
YEAR ENDED DECEMBER 31,
(BILLIONS OF DOLLARS)
2011
2010
2009
Medicaid and related state program rebates
$1.2
$1.2
$0.6
Medicare rebates
1.4
1.3
0.9
Performance
-
based contract rebates
3.0
2.6
2.3
Chargebacks
3.2
3.0
2.3
Total
$8.8
$8.1
$6.1
Rebates are product-specific and, therefore, for any given year are impacted by the mix of products sold.
Performance-based contracts are with managed care customers, including health maintenance organizations and pharmacy benefit managers, who receive rebates based on the achievement of
contracted performance terms for products.
Chargebacks primarily represent reimbursements to wholesalers for honoring contracted prices to third parties.
•
the impact of increased rebates under the U.S. Healthcare Legislation, which includes increased Medicaid rates and discounts to Medicare Part D participants who are in the Medicare
“
coverage gap
”
;
• an increase in chargebacks for our branded products as a result of increasing competitive pressures and increasing sales for certain branded products and certain generic products sold by our
Greenstone unit that are subject to chargebacks,
• the impact of decreased Medicare, Medicaid and performance-based contract rebates contracted for certain products that have lost exclusivity;
•
changes in product mix; and
• the impact on chargebacks of decreased sales for products that have lost exclusivity.
2011 Financial Report
17
(a)
(a)
(a), (b )
(c)
(a)
(b)
(c)

Financial Review
Pfizer Inc. and Subsidiary Companies
Revenues by Segment and Geographic Area
Biopharmaceutical Revenues
Revenues from biopharmaceutical products contributed approximately 86% of our total revenues in 2011, 87% of our total revenues in 2010 and 92% of our total revenues in 2009.
We recorded direct product sales of more than $1 billion for each of 12 biopharmaceutical products in 2011, each of 15 biopharmaceutical products in 2010 and each of nine legacy Pfizer
biopharmaceutical products in 2009. These products represented 56% of our revenues from biopharmaceutical products in 2011, 60% of our revenues from biopharmaceutical products in 2010 and 56%
of our revenues from biopharmaceutical products in 2009. We did not record more than $1 billion in revenues for any individual legacy Wyeth product in 2009 as the Wyeth acquisition date was
October 15, 2009.
2011 vs. 2010
Worldwide revenues from biopharmaceutical products in 2011 were $57.7 billion, a decrease of 1% compared to 2010, primarily due to:
partially offset by:
Geographically,
Worldwide revenues by operating segment, business unit and geographic area follow:
YEAR ENDED DECEMBER 31,
% CHANGE
WORLDWIDE
U.S.
INTERNATIONAL
WORLDWIDE
U.S.
INTERNATIONAL
(MILLIONS OF
DOLLARS)
2011
2010
2009
2011
2010
2009
2011
2010
2009
11/10
10/09
11/10
10/09
11/10
10/09
Biopharmaceutical revenues:
Primary Care Operating Segment
$22,670
$
23,328
$
22,576
$12,819
$
13,536
$
13,045
$ 9,851
$ 9,792
$ 9,531
(3
)
3
(5
)
4
1
3
Specialty Care
15,245
15,021
7,414
6,870
7,419
3,853
8,375
7,602
3,561
1
103
(7
)
93
10
113
Oncology
1,323
1,414
1,511
391
506
456
932
908
1,055
(6
)
(6
)
(23
)
11
3
(14
)
SC&O Operating Segment
16,568
16,435
8,925
7,261
7,925
4,309
9,307
8,510
4,616
1
84
(8
)
84
9
84
Emerging Markets
9,295
8,662
6,157
—
—
—
9,295
8,662
6,157
7
41
—
—
7
41
Established Products
9,214
10,098
7,790
3,627
4,501
2,656
5,587
5,597
5,134
(9
)
30
(19
)
69
—
9
EP&EM Operating Segment
18,509
18,760
13,947
3,627
4,501
2,656
14,882
14,259
11,291
(1
)
35
(19
)
69
4
26
57,747
58,523
45,448
23,707
25,962
20,010
34,040
32,561
25,438
(1
)
29
(9
)
30
5
28
Other product revenues:
Animal Health
4,184
3,575
2,764
1,648
1,382
1,106
2,536
2,193
1,658
17
29
19
25
16
32
Consumer Healthcare
3,057
2,772
494
1,490
1,408
331
1,567
1,364
163
10
*
6
*
15
*
AH&CH Operating Segment
7,241
6,347
3,258
3,138
2,790
1,437
4,103
3,557
1,821
14
95
12
94
15
95
Nutrition Operating Segment
2,138
1,867
191
—
—
—
2,138
1,867
191
15
*
—
—
15
*
Pfizer CentreSource
299
320
372
88
103
93
211
217
279
(7
)
(14
)
(15
)
11
(3
)
(22
)
Total Revenues
$67,425
$
67,057
$
49,269
$26,933
$
28,855
$
21,540
$40,492
$38,202
$27,729
1
36
(7
)
34
6
38
2011 includes revenues from legacy King U.S. operations for 11 months and from legacy King international operations for ten months, commencing on the King acquisition date, January 31, 2011.
Legacy Wyeth revenues are included for a full year in each of 2011 and 2010. 2009 includes revenues from legacy Wyeth products commencing on the Wyeth acquisition date, October 15, 2009.
Our contract manufacturing and bulk pharmaceutical chemical sales organization.
*
Calculation not meaningful.
• the decrease of $4.7 billion in operational revenues from Lipitor, Effexor, Protonix, Xalatan, Caduet, Vfend, Aromasin and Zosyn, and lower Alliance revenues for Aricept, all due to loss of exclusivity in
certain markets; and
•
a reduction in revenues of $359 million due to the U.S. Healthcare Legislation,
•
the solid performance of Lyrica, the Prevnar/Prevenar franchise and Enbrel;
•
the inclusion of operational revenues from legacy King products of approximately $950 million, which favorably impacted biopharmaceutical revenues by 2%; and
• the favorable impact of foreign exchange of $1.7 billion, or 3%.
18
2011 Financial Report
(a), (b) (b) (b) (a) , (b) (b) (b) (a) , (b) (b) (b)
(c)
(a)
(b)
(c)

Financial Review
Pfizer Inc. and Subsidiary Companies
During 2011, international revenues from biopharmaceutical products represented 59% of total revenues from biopharmaceutical products, compared to 56% in 2010.
Primary Care Operating Segment
Specialty Care and Oncology Operating Segment
Established Products and Emerging Markets Operating Segment
Total revenues from established products in both the Established Products and Emerging Markets units were $13.0 billion, with $3.8 billion generated in emerging markets in 2011.
2010 vs. 2009
Worldwide revenues from biopharmaceutical products in 2010 were $58.5 billion, an increase of 29% compared to 2009, primarily due to:
partially offset by:
Geographically,
•
in the U.S., revenues from biopharmaceutical products decreased 9% in 2011, compared to 2010, reflecting lower revenues from Lipitor, Protonix, Effexor, Zosyn, Xalatan, Vfend, Caduet and Aromasin,
all due to loss of exclusivity, lower Alliance revenues due to loss of exclusivity of Aricept 5mg and 10mg tablets in November 2010 and lower revenues from Detrol/Detrol LA, as well as the reduction in
revenues of $359 million in 2011 due to the U.S. Healthcare Legislation. The impact of these adverse factors was partially offset by the strong performance of certain other biopharmaceutical products
and the addition of U.S. revenues from legacy King products of approximately $904 million in 2011.
• in our international markets, revenues from biopharmaceutical products increased 5% in 2011, compared to 2010, reflecting the favorable impact of foreign exchange of 6% in 2011, partially offset by a
net operational decrease. Operationally, revenues were favorably impacted by increases in the Prevenar franchise, Lyrica, Enbrel, Celebrex and Alliance revenues and unfavorably impacted by
declines in Lipitor, Effexor, Norvasc and Xalatan/Xalacom. International revenues from legacy King products were not significant to our international revenues in 2011.
• Primary Care unit revenues decreased 3% in 2011 compared to 2010, due to lower operational revenues of 6%, partially offset by the favorable impact of foreign exchange of 3%. Primary Care unit
revenues were favorably impacted by higher revenues from certain patent-
protected products, including Lyrica, Celebrex, Pristiq and Spiriva (in Alliance revenues), among others, as well as the addition
of revenues from legacy King products of $404 million, or 2% in 2011. Operational revenues in 2011 were negatively impacted by the loss of exclusivity of Lipitor and Caduet in the U.S. in November
2011, Lipitor in various other developed markets during 2010, as well as Aricept 5mg and 10 mg tablets in the U.S. in November 2010. Taken together, these losses of exclusivity reduced Primary Care
unit revenues by approximately $2.1 billion, or 9%, in comparison 2010.
• Specialty Care unit revenues increased 1% compared to 2010 due to the favorable impact of foreign exchange of 3%, partially offset by lower operational revenues of 2%. Operational revenues were
favorably impacted by strong growth in the Prevnar/Prevenar franchise and Enbrel, and unfavorably impacted by the loss of exclusivity of Vfend and Xalatan in the U.S. in February and March 2011,
respectively. Collectively, these losses of exclusivity reduced Specialty Care unit revenues by $624 million, or 4%, in comparison with 2010.
• Oncology unit revenues decreased 6%, compared to 2010, due to lower operational revenues of 10%, partially offset by the favorable impact of foreign exchange of 4%. The decrease in the Oncology
unit operational revenues in 2011 was primarily due to the transfer of Aromasin’s U.S. business to the Established Products unit effective January 1, 2011 as a result of its loss of exclusivity in April
2011. This loss of exclusivity reduced Oncology unit revenues by $160 million, or 11%, in comparison with 2010.
• Established Products unit revenues decreased 9% in 2011 compared to 2010 due to lower operational revenues of 13%, partially offset by a 4% favorable impact of foreign exchange. The decrease in
Established Products unit operational revenues in 2011 was mainly due to the loss of exclusivity of Effexor XR, Protonix and Zosyn in the U.S. Taken together, these losses of exclusivity decreased
Established Products unit revenues by $1.7 billion, or 17%, in comparison with 2010. These declines were partially offset by the addition of revenues from legacy King products of $546 million, or 5% in
2011.
• Emerging Markets unit revenues increased 7%, compared to 2010, due to higher operational revenues of 5%, as well as a 2% favorable impact of foreign exchange. The increase in Emerging Markets
unit operational revenues in 2011 was due to growth in certain key innovative brands, primarily the Prevenar franchise, Lyrica, Enbrel, Celebrex, Vfend and Zyvox. These increases were partially offset
by lower revenues from Lipitor, which lost exclusivity in Brazil in August 2010 and Mexico in December 2010, as well as the impact of price reductions for certain products in certain emerging market
countries. These losses of exclusivity reduced Emerging Market unit revenues by $118 million, or 1%, in comparison with 2010.
•
the inclusion of operational revenues from legacy Wyeth products of approximately $13.7 billion, which favorably impacted biopharmaceutical revenues by 30%; and
• the weakening of the U.S. dollar relative to other currencies, primarily the Canadian dollar, Australian dollar, Japanese yen and Brazilian real, which favorably impacted biopharmaceutical revenues by
approximately $900 million, or 2%,
•
the decrease in operational revenues of approximately $1.5 billion, or 3%, from legacy Pfizer products overall, including Norvasc, Camptosar, Lipitor and Detrol/Detrol LA.
•
in the U.S., biopharmaceutical revenues increased 30% in 2010, compared to 2009, reflecting the inclusion of revenues from legacy Wyeth products of $6.6 billion, which had a favorable impact of 33%,
partially offset by lower overall revenues from legacy Pfizer products, including Lipitor, Detrol/Detrol LA, Celebrex, Lyrica, Chantix and Caduet and the impact of increased rebates in 2010 as a result of
the U.S. Healthcare Legislation, all of which had an unfavorable impact of $664 million, or 3%; and
2011 Financial Report
19

Financial Review
Pfizer Inc. and Subsidiary Companies
Primary Care Operating Segment
Specialty Care and Oncology Operating Segment
Established Products and Emerging Markets Operating Segment
Total revenues from established products in both the Established Products and Emerging Markets units were $13.8 billion, with $3.7 billion generated in emerging markets in 2010.
Effective July 1, 2011, January 1, 2011, July 1, 2010, January 1, 2010, August 14, 2009, and January 3, 2009, we increased the published prices for certain U.S. biopharmaceutical products. These price
increases had no material effect on wholesaler inventory levels in comparison to the prior year.
Other Product Revenues
2011 vs. 2010
Animal Health and Consumer Healthcare Operating Segment
• in our international markets, biopharmaceutical revenues increased 28% in 2010, compared to 2009, reflecting the inclusion of operational revenues from legacy Wyeth products of $7.1 billion, which
had a favorable impact of 28%, and the favorable impact of foreign exchange on international biopharmaceutical revenues of approximately $900 million, or 3%, partially offset by lower operational
revenues from legacy Pfizer products of $819 million, or 3%. The decrease in operational revenues of legacy Pfizer products was due to lower operational revenues from, among other products, Lipitor,
Norvasc and Camptosar, all of which were impacted by the loss of exclusivity in certain international markets.
• Primary Care unit revenues increased 3% in 2010 compared to 2009, due to higher operational revenues of 2% and the favorable impact of foreign exchange of 1%. Primary Care unit revenues were
favorably impacted by the addition of legacy Wyeth products, primarily Premarin and Pristiq. Operational revenues in 2010 were negatively impacted by the loss of exclusivity of Lipitor in Canada in May
2010 and Spain in July 2010, which reduced Primary Care unit revenues by approximately $534 million, or 2%, in comparison 2009. Additionally, legacy Pfizer Primary Care revenues were negatively
impacted by developed Europe pricing pressures and the U.S. Healthcare Legislation and positively impacted by growth from select brands, including Lyrica, Champix and Celebrex, among others, in
key international markets, most notably Japan.
• Specialty Care unit revenues increased 103% in 2010 compared to 2009, due to higher operational revenues of 103%. Foreign exchange was flat. Specialty Care unit revenues in 2010 were favorably
impacted by the addition of legacy Wyeth products, primarily Enbrel and the Prevnar/Prevenar franchise and were negatively impacted by developed Europe pricing pressures and the U.S. Healthcare
Legislation, as well as an overall decline in certain therapeutic markets.
• Oncology unit revenues decreased 6% in 2010, compared to 2009, due to lower operational revenues of 6%. Foreign exchange was flat. Legacy Pfizer Oncology unit revenues in 2010 do not include
Camptosar’s European revenues due to Camptosar’s loss of exclusivity in Europe in July 2009. The reclassification of those revenues to the Established Products unit effective January 1, 2010
negatively impacted the Oncology unit performance by 17% in 2010 compared to 2009.
• Established Products unit revenues increased 30% in 2010 compared to 2009 due to higher operational revenues of 28% and the favorable impact of foreign exchange of 2%. The increase in
Established Products unit operational revenues in 2010 was mainly due to the addition of legacy Wyeth products, primarily Protonix, and was negatively impacted by 4% due to the loss of exclusivity of
Norvasc in Canada in July 2009.
• Emerging Markets unit revenues increased 41%, compared to 2009, due to higher operational revenues of 35%, as well as a 6% favorable impact of foreign exchange. The increases in Emerging
Markets unit operational revenues in 2010 was due to the addition of legacy Wyeth products, most notably Enbrel and the Prevenar franchise, as well as growth in key markets, including China and
Brazil. These increases were partially offset by the impact of price reductions for certain products in certain emerging market countries.
• Animal Health unit revenues increased 17% in 2011, compared to 2010, reflecting higher operational revenues of 14% and the favorable impact of foreign exchange of 3%. Operational revenues from
Animal Health products were favorably impacted by approximately $329 million, or 9%, due to the addition of revenues from legacy King animal health products. Legacy Pfizer products grew 7%
primarily driven by improving market conditions and resulting increased demand for products across the livestock business, as well as deeper market penetration in emerging markets. This was partially
offset by the adverse impact of required product divestitures in 2010 related to the acquisition of Wyeth.
• Consumer Healthcare unit revenues increased 10% in 2011, compared to 2010, reflecting higher operational revenues of 8% and the favorable impact of foreign exchange of 2%. The operational
revenue increase in 2011 was primarily driven by increased sales of core brands including Advil, Caltrate and Robitussin, as well as the temporary voluntary withdrawal of Centrum in Europe in the third
quarter of 2010.
20
2011 Financial Report
Financial Review
Pfizer Inc. and Subsidiary Companies
Nutrition Operating Segment
2010 vs. 2009
Animal Health and Consumer Healthcare Operating Segment
Revenues—Major Biopharmaceutical Products
• Nutrition unit revenues increased 15% in 2011, compared to 2010, reflecting higher operational revenues of 11% and the favorable impact of foreign exchange of 4%. The operational revenue increase
was primarily due to increased demand for premium products, launches of new products and strength in China and the Middle East.
• Revenues from Animal Health increased 29% in 2010, compared to 2009, reflecting the inclusion of operational revenues from legacy Wyeth Animal Health products of 22%, higher operational
revenues from legacy Pfizer Animal Health products of 4% due primarily to growth in the companion animal and livestock businesses, as well as the favorable impact of foreign exchange of 3%.
Revenue information for several of our major biopharmaceutical products follows:
(MILLIONS OF DOLLARS)
PRODUCT
PRIMARY INDICATIONS
YEAR ENDED DECEMBER 31,
% CHANGE
2011
2010
2009
11/10
10/09
Lipitor
Reduction of LDL cholesterol
$
9,577
$
10,733
$
11,434
(11
)
(6
)
Lyrica
Epilepsy, post-herpetic neuralgia and diabetic peripheral neuropathy,
fibromyalgia
3,693
3,063
2,840
21
8
Prevnar 13/Prevenar 13
Vaccine for prevention of pneumococcal disease
3,657
2,416
—
51
*
Enbrel (Outside the U.S. and Canada)
Rheumatoid, juvenile rheumatoid and psoriatic arthritis, plaque psoriasis and
ankylosing spondylitis
3,666
3,274
378
12
*
Celebrex
Arthritis pain and inflammation, acute pain
2,523
2,374
2,383
6
—
Viagra
Erectile dysfunction
1,981
1,928
1,892
3
2
Norvasc
Hypertension
1,445
1,506
1,973
(4
)
(24
)
Zyvox
Bacterial infections
1,283
1,176
1,141
9
3
Xalatan/Xalacom
Glaucoma and ocular hypertension
1,250
1,749
1,737
(29
)
1
Sutent
Advanced and/or metastatic renal cell carcinoma (mRCC) and refractory
gastrointestinal stromal tumors (GIST) and advanced pancreatic
neuroendocrine tumor
1,187
1,066
964
11
11
Geodon/Zeldox
Schizophrenia; acute manic or mixed episodes associated with bipolar
disorder; maintenance treatment of bipolar mania
1,022
1,027
1,002
—
2
Premarin family
Menopause
1,013
1,040
213
(3
)
*
Genotropin
Replacement of human growth hormone
889
885
887
—
—
Detrol/Detrol LA
Overactive bladder
883
1,013
1,154
(13
)
(12
)
Vfend
Fungal infections
747
825
798
(9
)
3
Chantix/Champix
An aid to smoking cessation treatment
720
755
700
(5
)
8
BeneFIX
Hemophilia
693
643
98
8
*
Effexor
Depression and certain anxiety disorders
678
1,718
520
(61
)
*
Zosyn/Tazocin
Antibiotic
636
952
184
(33
)
*
Pristiq
Depression
577
466
82
24
*
Zoloft
Depression and certain anxiety disorders
573
532
516
8
3
Caduet
Reduction of LDL cholesterol and hypertension
538
527
548
2
(4
)
Revatio
Pulmonary arterial hypertension (PAH)
535
481
450
11
7
Medrol
Inflammation
510
455
457
12
—
ReFacto AF/Xyntha
Hemophilia
506
404
47
25
*
Prevnar/Prevenar (7
-
valent)
Vaccine for prevention of pneumococcal disease
488
1,253
287
(61
)
*
Zithromax/Zmax
Bacterial infections
453
415
430
9
(3
)
Aricept
Alzheimer
’
s disease
450
454
435
(1
)
(4
)
Fragmin
Anticoagulant
382
341
359
12
(5
)
Cardura
Hypertension/Benign prostatic hyperplasia
380
413
457
(8
)
(10
)
Rapamune
Immunosuppressant
372
388
57
(4
)
*
Aromasin
Breast cancer
361
483
483
(25
)
—
BMP2
Development of bone and cartilage
340
400
81
(15
)
*
Relpax
Treat the symptoms of migraine headache
341
323
326
6
(1
)
Xanax XR
Anxiety disorders
306
307
318
—
(
3
)
Tygacil
Antibiotic
298
324
54
(8
)
*
Neurontin
Seizures
289
322
327
(10
)
(2
)
Diflucan
Fungal infections
265
278
281
(5
)
(1
)
Arthrotec
Osteoarthritis and rheumatoid arthritis
242
250
270
(3
)
(7
)
Unasyn
Injectable antibacterial
231
244
245
(5
)
—
Sulperazon
Antibiotic
218
213
204
2
4
Skelaxin
Muscle relaxant
203
—
—
*
*
Inspra
High blood pressure
195
157
130
24
21
Dalacin/Cleocin
Antibiotic for bacterial infections
192
214
241
(10
)
(11
)
Methotrexate
Severe psoriasis
191
164
21
16
*
Toviaz
Overactive bladder
187
137
59
36
132
Somavert
Acromegaly
183
157
147
17
7
Alliance revenues
Various
3,630
4,084
2,925
(11
)
40
All other
Various
6,768
6,194
4,913
9
26
2011 Financial Report
21
(a)
(a)
(a)
(a)
(a)
(a)
(a)
(a)
(a)
(b)
(a)
(a)
(a)
(c)
(d)
(e)
Financial Review
Pfizer Inc. and Subsidiary Companies
Certain amounts and percentages may reflect rounding adjustments.
Biopharmaceutical—Selected Product Descriptions
Legacy Wyeth product. Legacy Wyeth operations are included for a full year in each of 2010 and 2011. In 2009, includes approximately two-and-a-half months of Wyeth’s U.S. operations and
approximately one
-
and
-
a
-
half months of Wyeth
’
s international operations.
Represents direct sales under license agreement with Eisai Co., Ltd.
Legacy King product. King’s operations are included in our financial statements commencing from the acquisition date of January 31, 2011. Therefore, our results for 2010 and 2009 do not include
King
’
s results of operations.
Enbrel (in the U.S. and Canada) , Aricept, Exforge, Rebif and Spiriva.
Includes legacy Pfizer products in 2011, 2010 and 2009. Also includes legacy Wyeth and King products, as described in notes (a) and (c) above.
*
Calculation not meaningful.
•
Lipitor
, for the treatment of elevated LDL-cholesterol levels in the blood, is the most widely used branded prescription treatment for lowering cholesterol. Lipitor recorded worldwide revenues of $9.6
billion, or a decrease of 11%, in 2011, compared to 2010 due to:
o
the impact of loss of exclusivity in Canada in May 2010, Spain in July 2010, Brazil in August 2010, Mexico in December 2010 and the U.S. in November 2011;
o
the continuing impact of an intensely competitive lipid
-
lowering market with competition from generics and branded products worldwide; and
o
increased payer pressure worldwide, including the need for flexible rebate policies,
partially offset by:
o
the favorable impact of foreign exchange, which increased revenues by $257 million, or 2%.
Geographically,
o
in the U.S., Lipitor revenues were $5.0 billion, a decrease of 6% in 2011, compared to 2010; and
o
in our international markets, Lipitor revenues were $4.6 billion, a decrease of 15%, in 2011, compared to 2010. Foreign exchange had a favorable impact on international revenues of 5% in 2011,
compared to 2010.
See the “Our Operating Environment” section of this Financial Review for a discussion concerning losses and expected losses of exclusivity for Lipitor in various markets.
• Lyrica , indicated for the management of post-herpetic neuralgia, neuropathic pain associated with diabetic peripheral neuropathy, the management of fibromyalgia, and as adjunctive therapy for adult
patients with partial onset seizures in the U.S., and for neuropathic pain (peripheral and central), adjunctive treatment of epilepsy and general anxiety disorder in certain countries outside the U.S.,
recorded an increase in worldwide revenues of 21% in 2011, compared to 2010. Lyrica had a strong operational performance in international markets in 2011, including Japan, where Lyrica was
launched in 2010 as the first product approved for the peripheral neuropathic pain indication. In the U.S., revenues increased 6% in 2011, compared to 2010. Notwithstanding this increase, U.S.
revenues continue to be affected by increased competition from generic versions of competitive medicines, as well as managed care pricing and formulary pressures.
•
Prevnar 13/Prevenar 13
is our 13-
valent pneumococcal conjugate vaccine for the prevention of various syndromes of pneumococcal disease in infants and young children and in adults 50 years of age
and older. Prevnar 13/Prevenar 13 for use in infants and young children has been launched in the U.S. for the prevention of invasive pneumococcal disease caused by the 13 serotypes in Prevnar 13
and otitis media caused by the seven serotypes in Prevnar, and in the EU and many other international markets for the prevention of invasive pneumococcal disease, otitis media and pneumococcal
pneumonia caused by the vaccine serotypes. Worldwide revenues for Prevnar 13/Prevenar 13 increased 51% in 2011, compared to 2010. The launch of the Prevnar 13/Prevenar 13 pediatric indication
has reduced our Prevnar/Prevenar (7-valent) revenues (see discussion below), and we expect this trend to continue. In addition, in 2011, we received approval of Prevnar 13/Prevenar 13 for use in
adults 50 years of age and older in the U.S. for the prevention of pneumococcal pneumonia and invasive pneumococcal disease caused by the 13 serotypes in Prevnar 13, and in the EU for the
prevention of invasive pneumococcal disease caused by the vaccine serotypes. Prevenar 13 for use in adults 50 years of age and older also has been approved in many other international markets. We
expect to commence commercial launches for the adult indication in 2012.
We currently are conducting the Community-Acquired Pneumonia Immunization Trial in Adults (CAPiTA) to fill requirements in connection with the FDA’s approval of the Prevnar 13 adult indication
under its accelerated approval program. CAPiTA is an efficacy trial involving subjects 65 years of age and older that is designed to evaluate whether Prevnar 13 is effective in preventing the first
episode of community-acquired pneumonia caused by the serotypes contained in the vaccine. We estimate that this event-driven trial will be completed in 2013. At its regular meeting held on
February 22, 2012, the U.S. Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) indicated that it will defer voting on a recommendation for the routine
use of Prevnar 13 in adults 50 years of age and older until the results of CAPiTA, as well as data on the impact of pediatric use of Prevnar 13 on the disease burden and serotype distribution among
adults, are available. We expect that the rate of uptake for the use of Prevnar 13 in adults 50 years of age and older will be impacted by ACIP’s decision to defer voting on a recommendation for the
routine use of Prevnar 13 by that population.
•
Enbrel
, for the treatment of moderate-to-severe rheumatoid arthritis, polyarticular juvenile rheumatoid arthritis, psoriatic arthritis, plaque psoriasis and ankylosing spondylitis, a type of arthritis affecting
the spine, recorded increases in worldwide revenues, excluding the U.S. and Canada, of 12% in 2011, compared to 2010, primarily due to increased penetration of Enbrel in developed Europe,
developed Asia and emerging markets. Enbrel revenues from the U.S. and Canada are included in Alliance revenues.
22
2011 Financial Report
(a)
(b)
(c)
(d)
(a)
(e)

Financial Review
Pfizer Inc. and Subsidiary Companies
Under our co-promotion agreement with Amgen Inc. (Amgen), we co-promote Enbrel in the U.S. and Canada and share in the profits from Enbrel sales in those countries, which we include in Alliance
revenues. Our co-
promotion agreement with Amgen will expire in October 2013, and, subject to the terms of the agreement, we are entitled to a royalty stream for 36 months thereafter, which we expect
will be significantly less than our current share of Enbrel profits from U.S. and Canadian sales. Following the end of the royalty period, we will not be entitled to any further revenues from Enbrel sales in
the U.S. and Canada. Our exclusive rights to Enbrel outside the U.S. and Canada will not be affected by the expiration of the co-promotion agreement with Amgen.
• Celebrex , indicated for the treatment of the signs and symptoms of osteoarthritis and rheumatoid arthritis worldwide and for the management of acute pain in adults in the U.S. and certain markets in
the EU, recorded increases in worldwide revenues of 6% in 2011, compared to 2010. In the U.S., revenues have been adversely affected by increased competition from generic versions of competitive
medicines and managed care formulary pressures. Celebrex is supported by continued educational and promotional efforts highlighting its efficacy and safety profile for appropriate patients.
•
Viagra
remains the leading treatment for erectile dysfunction. Viagra worldwide revenues increased 3% in 2011, compared to 2010, primarily due to the favorable impact of foreign exchange.
•
Norvasc
, for treating hypertension, lost exclusivity in the U.S. and other major markets in 2007 and in Canada in 2009. Norvasc worldwide revenues decreased 4% in 2011, compared to 2010.
•
Zyvox
is the world’s best-selling agent among those used to treat serious Gram-positive pathogens, including methicillin-resistant staphylococcus-aureus. Zyvox worldwide revenues increased 9% in
2011, compared to 2010, primarily due to growth in emerging markets, as well as growth in certain other markets driven by secondary bacterial infections arising from the stronger flu season in 2011.
• Xalabrands consists of Xalatan , a prostaglandin, which is a branded agent used to reduce elevated eye pressure in patients with open-angle glaucoma or ocular hypertension, and Xalacom, a fixed
combination prostaglandin (Xalatan) and beta blocker (timolol) available outside the U.S. Xalatan/Xalacom worldwide revenues decreased 29% in 2011, compared to 2010. Lower revenues in the U.S.
were due to the loss of exclusivity in March 2011. Lower operational revenues internationally were due to the launch of generic latanoprost (generic Xalatan) in Japan in May 2010 and in Italy in July
2010. Xalatan and
Xalacom lost exclusivity in 15 major European markets in January 2012.
•
Sutent
is for the treatment of advanced renal cell carcinoma, including metastatic renal cell carcinoma (mRCC) and gastrointestinal stromal tumors after disease progression on, or intolerance to,
imatinib mesylate and advanced pancreatic neuroendocrine tumor. Sutent worldwide revenues increased 11% in 2011, compared to 2010, due to strong operational performance and the favorable
impact of foreign exchange. We continue to drive total revenue and prescription growth, supported by cost-effectiveness data and efficacy data in first-line mRCC––including two-year survival data,
which represent the first time that overall survival of two years has been seen in the treatment of advanced kidney cancer, as well as through increasing access and healthcare coverage. As of
December 31, 2011, Sutent was the most prescribed oral mRCC therapy in the U.S.
• Geodon/Zeldox , an atypical antipsychotic, is indicated for the treatment of schizophrenia, as monotherapy for the acute treatment of bipolar manic or mixed episodes, and as an adjunct to lithium or
valproate for the maintenance treatment of bipolar disorder. Geodon worldwide revenues were relatively flat in 2011, compared to 2010, which reflects higher rebates in 2011 due to the impact of the
U.S. Healthcare Legislation and moderate growth in the U.S. antipsychotic market. Geodon will lose exclusivity in the U.S. in March 2012.
• Our
Premarin
family of products remains the leading therapy to help women address moderate-to-severe menopausal symptoms. It recorded a decrease in worldwide revenues of 3% in 2011,
compared to 2010.
• Genotropin , one of the world’s leading human growth hormones, is used in children for the treatment of short stature with growth hormone deficiency, Prader-Willi Syndrome, Turner Syndrome, Small
for Gestational Age Syndrome, Idiopathic Short Stature (in the U.S. only) and Chronic Renal Insufficiency (outside the U.S. only), as well as in adults with growth hormone deficiency. Genotropin is
supported by a broad platform of innovative injection
-
delivery devices and patient
-
support programs. Genotropin worldwide revenues were relatively flat in 2011, compared to 2010.
•
Detrol/Detrol LA,
a muscarinic receptor antagonist, is one of the most prescribed branded medicines worldwide for overactive bladder. Detrol LA is an extended-release formulation taken once a day.
Detrol/Detrol LA worldwide revenues declined 13% in 2011, compared to 2010, primarily due to increased competition from other branded medicines and a shift in promotional focus to our Toviaz
product in most major markets. Detrol immediate release (Detrol IR) will lose exclusivity in the U.S. in September 2012.
•
Vfend
is a broad-spectrum agent for treating yeast and molds. Vfend worldwide revenues decreased 9% in 2011, compared to 2010. While international revenues of Vfend continued to be driven in
2011 by its acceptance as an excellent broad-spectrum agent for treating serious yeast and molds, revenues in the U.S. declined primarily due to a loss of exclusivity of Vfend tablets and the launch of
generic voriconazole (generic Vfend) in February 2011.
•
Chantix/Champix
is an aid to smoking-cessation treatment in adults 18 years of age and older. Chantix/Champix worldwide revenues decreased 5% in 2011, compared to 2010. Revenues in 2011
were favorably impacted by foreign exchange, which was more than offset by the impact of changes to the product
’s label and other factors. We are continuing our educational and promotional efforts,
which are focused on addressing the significant health consequences of smoking highlighting the Chantix benefit
-risk proposition and emphasizing the importance of the physician-patient dialogue in
helping patients quit smoking.
In July 2011, the U.S. prescribing information was revised to include clinical data showing that Chantix is an effective aid to smoking-cessation treatment for smokers with stable cardiovascular disease
(CVD) and mild-to-moderate chronic obstructive pulmonary disease (COPD). The revised label also includes a warning/precaution advising smokers with CVD to inform their physician of any new or
worsening symptoms of cardiovascular disease, and to seek emergency medical help if they experience any symptoms of a heart attack.
2011 Financial Report
23

Financial Review
Pfizer Inc. and Subsidiary Companies
See Notes to Consolidated Financial Statements— Note 17. Commitments and Contingencies for a discussion of recent developments concerning patent and product litigation relating to certain of the
products discussed above.
Embeda —On February 23, 2011, we stopped distribution of our Embeda product due to failed specification tolerance related to naltrexone degradation identified in post-manufacturing testing. On
March 10, 2011, we initiated a voluntary recall to wholesale and retail customers of all Embeda products. We are committed to returning this important product to the market as quickly as possible, once
the stability issue is resolved.
This safety information was added at the FDA’s request following an observation of a small numeric increase in certain cardiovascular events in patients treated with Chantix versus those taking a
placebo in a study of 700 smokers with stable cardiovascular disease. Approval of the EU labeling, revised at the European Medicine’s Agency’s (EMA’s) request to include a similar cardiovascular-
related warning/precaution, was received in late December 2011, with regulators reaffirming the positive benefit/risk profile of the medication. Approval of the Japan labeling, which includes a similar
precaution, occurred in late October 2011. In December 2011, Pfizer received a positive opinion from the EMA’s Committee for Medical Products for Human Use for changes to the EU label regarding
schizophrenia data.
•
BeneFIX and ReFacto AF/Xyntha
are hemophilia products using state-of-the-
art manufacturing that assist patients with a lifelong bleeding disorder. BeneFIX is the only available recombinant factor IX
product for the treatment of hemophilia B, while ReFacto AF/Xyntha are recombinant factor VIII products for the treatment of hemophilia A. Both products are indicated for the control and prevention of
bleeding in patients with these disorders and in some countries also are indicated for prophylaxis in certain situations, such as surgery. BeneFIX recorded an increase in worldwide revenues of 8% in
2011, compared to 2010. ReFacto AF/Xyntha recorded an increase in worldwide revenues of 25% in 2011, compared to 2010. The increases for all of these products were due to strong operational
performance and the favorable impact of foreign exchange.
• Effexor , an antidepressant for treating adult patients with major depressive disorder, generalized anxiety disorder, social anxiety disorder and panic disorder, recorded a decrease in worldwide
revenues of 61% in 2011, compared to 2010. Effexor and Effexor XR, an extended-
release formulation, face generic competition in most markets, including in the U.S., where Effexor XR lost exclusivity
on July 1, 2010. This generic competition had a negative impact in 2011, and will continue to have a significant adverse impact on our revenues for Effexor and Effexor XR.
• Zosyn / Tazocin , our broad-spectrum intravenous antibiotic, faces generic global competition. U.S. exclusivity was lost in September 2009. Zosyn/Tazocin recorded a decrease in worldwide revenues
of 33% in 2011, compared to 2010.
• Pristiq is approved for the treatment of major depressive disorder in the U.S. and in various other countries. Pristiq has also been approved for treatment of moderate-to-severe vasomotor symptoms
(VMS) associated with menopause in Thailand, Mexico, the Philippines and Ecuador. Pristiq recorded an increase in worldwide revenues of 24% in 2011, compared to 2010, primarily driven by
promotional activities in the U.S., and targeted international markets where Pristiq was recently launched. The activities are designed to educate physicians and pharmacists about the benefit-
risk profile
of Pristiq.
•
Caduet
is a single-pill therapy combining Lipitor and Norvasc for the prevention of cardiovascular events. Caduet worldwide revenues increased 2% in 2011, compared to 2010, due to strong
operational performance in international markets and the favorable impact of foreign exchange, partially offset by the impact of increased generic competition, as well as an overall decline in U.S.
hypertension market volume. Caduet lost U.S. exclusivity in November 2011.
•
Revatio
, for the treatment of pulmonary arterial hypertension (PAH), had an increase in worldwide revenues of 11% in 2011, compared to 2010, due in part to increased PAH awareness driving earlier
diagnosis in the U.S. and EU and the favorable impact of foreign exchange. In the U.S., Revatio tablet will lose exclusivity in September 2012, and Revatio IV injection will lose exclusivity in May 2013.
•
Prevnar/Prevenar (7
-
valent),
our 7-valent pneumococcal conjugate vaccine for preventing invasive, and, in certain international markets, non-invasive pneumococcal disease in infants and young
children, recorded a decrease in worldwide revenues of 61% in 2011, compared to 2010. Many markets have transitioned from the use of Prevnar/Prevenar (7-valent) to Prevnar 13/Prevenar 13 (see
discussion above), resulting in lower revenues for Prevnar/Prevenar (7
-
valent). We expect this trend to continue.
•
Xalkori,
the first-ever therapy targeting anaplastic lymphoma kinase (ALK), for the treatment of patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) that is ALK-positive as
detected by an FDA-approved test, was approved by the FDA in August 2011. In December 2011, Xalkori was approved in Korea for the treatment of ALK-positive locally advanced or metastatic
NSCLC.
•
Inlyta
was approved by the FDA in January 2012 for the treatment of patients with advance renal cell carcinoma after failure of one prior systemic therapy.
• Alliance revenues worldwide decreased 11% in 2011, compared to 2010, mainly due to the loss of exclusivity for Aricept 5mg and 10mg tablets in the U.S. in November 2010, partially offset by the
strong performance of Spiriva and Enbrel in the U.S. and Canada. We expect that the Aricept 23mg tablet will have exclusivity in the U.S. until July 2013. See the “The Loss or Expiration of Intellectual
Property Rights” section of this Financial Review for a discussion regarding the expiration of various contract rights relating to Aricept, Spiriva, Enbrel and Rebif. ELIQUIS (apixaban) is being jointly
developed and commercialized by Pfizer and Bristol-Myers Squibb (BMS). The two companies share with respect to the approved indication in the EU and, if and when indications for ELIQUIS are
approved in various markets, will share on a global basis commercialization expenses and profit/losses equally.
24
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
Research and Development
Research and Development Operations
Innovation is critical to the success of our company and drug discovery and development is time-consuming, expensive and unpredictable, particularly for human health products. As a result, and also
because we are predominately a human health company, the vast majority of our R&D spending is associated with human health products, compounds and activities.
We incurred the following expenses in connection with our Research and Development (R&D) operations (see also Notes to Consolidated Financial Statements–– Note 18. Segment, Geographic and
Revenue Information ):
Our human health R&D spending is conducted through a number of matrix organizations––Research Units, within our Worldwide Research and Development organization, that are generally responsible
for research assets (assets that have not yet achieved proof-of-concept); Business Units that are generally responsible for development assets (assets that have achieved proof-of-concept); and science-
based and other platform-services organizations.
We take a holistic approach to our human health R&D operations and manage the operations on a total-
company basis through our matrix organizations described above. Specifically, a single committee,
co-chaired by members of our R&D and commercial organizations, is accountable for aligning resources among all of our human health R&D projects and for ensuring that our company is focusing its
R&D resources in the areas where we believe that we can be most successful and maximize our return on investment. We believe that this approach also serves to maximize accountability and flexibility.
Our Research Units are organized in a variety of ways (by therapeutic area or combinations of therapeutic areas, by discipline, by location, etc.) to enhance flexibility, cohesiveness and focus. Because of
our structure, we can rapidly redeploy resources, within a Research Unit, between various projects as necessary because the workforce shares similar skills, expertise and/or focus.
Our platform-services organizations, where a significant portion of our R&D spending occurs, provide technical expertise and other services to the various R&D projects, and are organized into science-
based functions such as Pharmaceutical Sciences, Chemistry, Drug Safety, and Development Operations, and non-science-based functions, such as Facilities, Business Technology and Finance. As a
result, within each of these functions, we are able to migrate resources among projects, candidates and/or targets in any therapeutic area and in most phases of development, allowing us to react quickly
in response to evolving needs.
Generally, we do not disaggregate total R&D expense by development phase or by therapeutic area since, as described above, we do not manage a significant portion of our R&D operations by
development phase or by therapeutic area. Further, as we are able to adjust a significant portion of our spending quickly, as conditions change, also as described above, we believe that any prior-period
information about R&D expense by development phase or by therapeutic area would not necessarily be representative of future spending.
Product Developments—Biopharmaceutical
We continue to invest in R&D to provide potential future sources of revenues through the development of new products, as well as through additional uses for in-line and alliance products. We have
achieved our previously announced goal of 15 to 20 regulatory submissions in the 2010-to-2012 period. Notwithstanding our efforts, there are no assurances as to when, or if, we will receive regulatory
approval for additional indications for existing products or any of our other products in development.
We continue to closely evaluate our global research and development function and to pursue strategies to improve innovation and overall productivity by prioritizing areas with the greatest scientific and
commercial promise, utilizing appropriate risk/return profiles and focusing on areas with the highest potential to deliver value in the near term and over time.
RESEARCH AND DEVELOPMENT EXPENSES
YEAR ENDED DECEMBER 31,
% INCR./(DECR.)
(
MILLIONS
OF
DOLLARS
)
2011
2010
2009
11/10
10/09
Primary Care Operating Segment
$1,307
$1,473
$1,407
(11
)
5
Specialty Care and Oncology Operating Segment
1,561
1,624
1,060
(4
)
53
Established Products and Emerging Markets Operating Segment
441
452
392
(2
)
15
Animal Health and Consumer Healthcare Operating Segment
425
428
297
(1
)
44
Nutrition and Pfizer CentreSource
41
34
8
17
*
Worldwide Research and Development/Pfizer Medical
3,337
3,709
2,698
(10
)
37
Corporate and other
2,000
1,672
1,962
20
(15)
$9,112
$9,392
$7,824
(3
)
20
Our operating segments, in addition to their sales and marketing responsibilities, are responsible for certain development activities. Generally, these responsibilities relate to additional indications for in-
line products and IPR&D projects that have achieved proof
-
of
-
concept. R&D spending may include upfront and milestone payments for intellectual property rights.
Worldwide Research and Development is generally responsible for human health research projects until proof-of-concept is achieved, and then for transitioning those projects to the appropriate
business unit for possible clinical and commercial development. R&D spending may include upfront and milestone payments for intellectual property rights. This organization also has responsibility for
certain science-based and other platform-services organizations, which provide technical expertise and other services to the various R&D projects. Pfizer Medical is responsible for all human-health-
related regulatory submissions and interactions with regulatory agencies, including all safety event activities, for conducting clinical trial audits and readiness reviews and for providing Pfizer-related
medical information to healthcare providers.
Corporate and other includes unallocated costs, primarily facility costs, information technology, share-based compensation, and restructuring related costs.
2011 Financial Report
25
(a)
(a)
(a)
(a)
(a)
(b)
(c)
(a)
(b)
(c)

Financial Review
Pfizer Inc. and Subsidiary Companies
To that end, our research primarily focuses on five high-priority areas that have a mix of small and large molecules –– immunology and inflammation; oncology; cardiovascular, metabolic and endocrine
diseases; neuroscience and pain; and vaccines.
Our development pipeline, which is updated quarterly, can be found at www.pfizer.com/pipeline. It includes an overview of our research and a list of compounds in development with targeted indication,
phase of development and, for late-stage programs, mechanism of action. The information currently in our development pipeline is accurate as of February 28, 2012.
Below are significant regulatory actions by, and filings pending with, the FDA and regulatory authorities in the EU and Japan, as well as new drug candidates and additional indications in late-stage
development:
Recent FDA approvals:
PRODUCT
INDICATION
DATE APPROVED
INLYTA (Axitinib)
Treatment of advanced renal cell carcinoma after failure of one prior systemic therapy
January 2012
Prevnar 13 Adult
Prevention of pneumococcal pneumonia and invasive disease in adults 50 years of age and older
December 2011
Xalkori (Crizotinib)
Treatment of ALK-positive advanced non-small cell lung cancer
August 2011
Oxecta––Immediate
release
oxycodone with
Aversion
technology
(formerly
Acurox) (without
niacin)
Management of moderate-to-severe pain where the use of an opioid analgesic is appropriate
June 2011
Sutent
Treatment of unresectable pancreatic neuroendocrine tumor
May 2011
In early 2011, we acquired King, which has an exclusive license from Acura Pharmaceuticals, Inc. (Acura) to sell Oxecta in the U.S., Canada and Mexico.
Pending U.S. new drug applications (NDA) and supplemental filings:
PRODUCT
INDICATION
DATE FILED*
Tafamidis meglumine
Treatment of transthyretin familial amyloid polyneuropathy (TTR
-
FAP)
February 2012
Lyrica
Treatment of central neuropathic pain due to spinal cord injury
February 2012
Revatio
Pediatric PAH
January 2012
Bosutinib
Treatment of previously treated chronic myelogenous leukemia
January 2012
Tofacitinib
Treatment of moderate
-
to
-
severe active rheumatoid arthritis
December 2011
Apixaban
Prevention of stroke and systemic embolism in patients with atrial fibrillation
November 2011
Taliglucerase alfa
Treatment of Gaucher disease
April 2010
Genotropin
Replacement of human growth hormone deficiency (Mark VII multidose disposable device)
December 2009
Celebrex
Chronic pain
October 2009
Geodon
Treatment of bipolar disorder
–
–
pediatric filing
December 2008
Remoxy
Management of moderate-to-severe pain when a continuous, around-the-clock opioid analgesic is needed for an extended period of time
August 2008
Spiriva
Respimat device for chronic obstructive pulmonary disease
January 2008
Zmax
Treatment of bacterial infections––sustained release––acute otitis media (AOM) and sinusitis––pediatric filing
January 2007
Viviant
Osteoporosis treatment and prevention
August 2006
Vfend
Treatment of fungal infections––pediatric filing
August 2005
*
The dates set forth in this column are the dates on which the FDA accepted our submissions.
This indication for apixaban is being developed in collaboration with our alliance partner, BMS.
In November 2009, we entered into a license and supply agreement with Protalix BioTherapeutics (Protalix), which provides us exclusive worldwide rights, except in Israel, to develop and
commercialize taliglucerase alfa for the treatment of Gaucher disease. In April 2010, Protalix completed a rolling NDA with the FDA for taliglucerase alfa. Taliglucerase alfa was granted orphan drug
designation in the U.S. in September 2009. In February 2011, Protalix received a “complete response” letter from the FDA for the taliglucerase alfa NDA that set forth additional requirements for
approval. On August 1, 2011, Protalix announced that it had submitted its response to the FDA letter.
In April 2010, we received a “complete response” letter from the FDA for the Genotropin Mark VII multidose disposable device submission. In August 2010, we submitted our response to address the
requests and recommendations included in the FDA letter. In April 2011, we received a second “complete response” letter from the FDA, requesting additional information. We are assessing the
requests and recommendations included in the FDA’s letter.
In June 2010, we received a “complete response” letter from the FDA for the Celebrex chronic pain supplemental NDA. The supplemental NDA remains pending while we await the completion of
ongoing studies to determine next steps.
In October 2009, we received a “complete response” letter from the FDA with respect to the supplemental NDA for Geodon for the treatment of acute bipolar mania in children and adolescents aged 10
to 17 years. In October 2010, we submitted our response. In April 2010, we received a “warning letter”
from the FDA with respect to the clinical trial in support of this supplemental NDA. We are working
to address the issues raised in the letter. In April 2011, we received a second “complete response” letter from the FDA in which the FDA indicated that, in its view, the reliability of the data supporting
the filing had not yet been demonstrated. We are working to better understand the issues raised in the letter.
26
2011 Financial Report
(a)
(a)
(a)
(b)
(c)
(d)
(e)
(f)
(g)
(h)
(i)
(j)
(a)
(b)
(c)
(d)
(e)

Financial Review
Pfizer Inc. and Subsidiary Companies
In July 2007, Wyeth received an “approvable” letter from the FDA with respect to its supplemental NDA for the use of Pristiq in the treatment of moderate-to-severe vasomotor symptoms (VMS)
associated with menopause. The FDA requested an additional one-year study of the safety of Pristiq for this indication. This study was completed, and the results were provided to the FDA in December
2010. In September 2011, we received a “complete response” letter from the FDA regarding our supplemental NDA. In February 2012, we decided to withdraw our supplemental NDA for Pristiq for the
treatment of moderate-to-severe VMS associated with menopause. Pristiq continues to be available in the U.S. for the treatment of major depressive disorder (MDD) in appropriate adult patients, and
around the world, for the respective indications approved in each market.
In 2005, King entered into an agreement with Pain Therapeutics, Inc. (PT) to develop and commercialize Remoxy. In August 2008, the FDA accepted the NDA for Remoxy that had been submitted by
King and PT. In December 2008, the FDA issued a “complete response” letter. In March 2009, King exercised its right under the agreement with PT to assume sole control and responsibility for the
development of Remoxy. In December 2010, King resubmitted the NDA for Remoxy with the FDA. In June 2011, we and PT announced that a “complete response” letter was received from the FDA
with regard to the resubmission of the NDA. We are working to address the issues raised in the letter, which primarily relate to manufacturing. There are several key decision points over the next
several months that will determine the timing and the nature of our response to the FDA
’
s
“
complete response
”
letter.
Boehringer Ingelheim (BI), our alliance partner, holds the NDAs for Spiriva Handihaler and Spiriva Respimat. In September 2008, BI received a “complete response” letter from the FDA for the Spiriva
Respimat submission. The FDA is seeking additional data, and we are coordinating with BI, which is working with the FDA to provide the additional information. A full response will be submitted to the
FDA upon the completion of planned and ongoing studies.
In September 2007, we received an “approvable” letter from the FDA for Zmax that set forth requirements to obtain approval for the pediatric acute otitis media (AOM) indication based on
pharmacokinetic data. In January 2010, we filed a supplemental NDA, which proposed the inclusion of the new indications for AOM and acute bacterial sinusitis in pediatric patients. In May 2011, we
received a
“
complete response
”
letter from the FDA with respect to the supplemental NDA. We are working to determine the next steps.
Two “approvable” letters were received by Wyeth in April and December 2007 from the FDA for Viviant (bazedoxifene), for the prevention of post-menopausal osteoporosis, that set forth the additional
requirements for approval. In May 2008, Wyeth received an “approvable” letter from the FDA for the treatment of post-menopausal osteoporosis. The FDA is seeking additional data, and we have been
systematically working through these requirements and seeking to address the FDA’s concerns. A full response will be provided to the FDA. In February 2008, the FDA advised Wyeth that it expects to
convene an advisory committee to review the pending NDAs for both the treatment and prevention indications after we submit our response to the “approvable” letters. In April 2009, Wyeth received
approval in the EU for CONBRIZA (the EU trade name for Viviant) for the treatment of post-menopausal osteoporosis in women at increased risk of fracture. Viviant was also approved in Japan in July
2010 for the treatment of post
-
menopausal osteoporosis and in Korea in November 2011 for the treatment and prevention of post
-
menopausal osteoporosis.
In December 2005, we received an “approvable” letter from the FDA for our Vfend pediatric filing that set forth the additional requirements for approval to extend Vfend exclusivity in the U.S. for an
additional six months. In April 2010, based on data from a new pharmacokinetics study, we and the FDA agreed on a pediatric dosing regimen, which was subsequently incorporated into the three
ongoing pediatric trials. Depending on the results of those trials, we may pursue a pediatric indication for Vfend; however, this would not extend Vfend exclusivity for an additional six months because
we lost exclusivity for Vfend tablets in the U.S. in February 2011.
2011 Financial Report
27
(f)
(g)
(h)
(i)
(j)

Financial Review
Pfizer Inc. and Subsidiary Companies
In March 2011, we decided to withdraw our application in Japan for Toviaz for the treatment of overactive bladder due to required stability testing. We intend to resubmit the application in the first half of
2012.
In March 2010, we withdrew our application in Japan for Prevenar 13 for the prevention of invasive pneumococcal disease in infants and young children due to a request by the Pharmaceutical and
Medical Devices Agency (PMDA) for an additional study of this indication in Japanese subjects. We are conducting the requested additional study and, if the results are positive, we plan to resubmit the
application.
Regulatory approvals and filings in the EU and Japan:
PRODUCT
DESCRIPTION OF EVENT
DATE
APPROVED
DATE FILED*
Tofacitinib
Application filed in Japan for treatment of moderate-to-severe active rheumatoid arthritis
—
December 2011
Celebrex
Approval in Japan for treatment of acute pain
December 2011
—
Apixaban
Application filed in Japan for prevention of stroke and systemic embolism in patients with non-valvular atrial
fibrillation
—
December 2011
Vyndaqel (Tafamidis meglumine)
Approval in the EU for treatment of TTR-FAP in adult patients with stage 1 symptomatic polyneuropathy
November 2011
—
Tofacitinib
Application filed in the EU for treatment of moderate-to-severe active rheumatoid arthritis
—
November 2011
Prevenar 13 Adult
Approval in the EU for prevention of invasive pneumococcal disease in adults 50 years of age and older
October 2011
—
Sutent
Application filed in Japan for treatment of pancreatic neuroendocrine tumor
—
October 2011
Lyrica
Application filed in Japan for treatment of fibromyalgia
—
October 2011
ELIQUIS (Apixaban)
Application filed in the EU for prevention of stroke in patients with atrial fibrillation
—
October 2011
Bosutinib
Application filed in the EU for treatment of newly diagnosed chronic myelogenous leukemia
—
August 2011
Crizotinib
Application filed in the EU for treatment of previously treated ALK
-
positive advanced non
-
small cell lung cancer
—
August 2011
Axitinib
Application filed in Japan for treatment of advanced renal cell carcinoma after failure of prior systemic treatment
—
July 2011
ELIQUIS
(Apixaban)
Approval in the EU for prevention of venous thromboembolism following elective hip or knee-
replacement surgery
May 2011
—
Axitinib
Application filed in the EU for treatment of advanced renal cell carcinoma after failure of prior systemic treatment
—
May 2011
Revatio
Approval in the EU for pediatric PAH
May 2011
—
Crizotinib
Application filed in Japan for treatment of ALK
-
positive advanced non
-
small cell lung cancer
—
March 2011
Xiapex
Approval in the EU for treatment of Dupuytren
’
s contracture
February 2011
—
Sutent
Approval in the EU for treatment of unresectable pancreatic neuroendocrine tumor
December 2010
—
Taliglucerase alfa
Application filed in the EU for treatment of Gaucher disease
—
November 2010
*
For applications in the EU, the dates set forth in this column are the dates on which the European Medicines Agency (EMA) validated our submissions.
This indication for ELIQUIS (apixaban) is being developed in collaboration with BMS.
This indication for ELIQUIS (apixaban) was developed and is being commercialized in collaboration with BMS.
28
2011 Financial Report
(a)
(a)
(b)
(a)
(b)

Financial Review
Pfizer Inc. and Subsidiary Companies
In October 2011, an independent Data Monitoring Committee (DMC) for a Phase 3 efficacy and safety study of Lyrica as monotherapy for epilepsy patients with partial onset seizures recommended that
the study be stopped based on positive findings for the primary efficacy endpoint. We have accepted the DMC’s recommendation and stopped the study. We intend to submit the results of the study for
publication in a medical journal. We do not intend to seek an indication for Lyrica as monotherapy for epilepsy patients with partial onset seizures.
In March 2010, we and Medivation, Inc. announced that a Phase 3 trial of dimebon (latrepiridine) did not meet its co-primary or secondary endpoints. Subsequently, we and Medivation, Inc. agreed to
discontinue the CONSTELLATION and CONTACT Phase 3 trials in patients with moderate-to-severe Alzheimer’s disease. In April 2011, we and Medivation, Inc. announced that the Phase 3 HORIZON
trial in patients with Huntington’s disease did not meet its co-primary endpoints and that, as a result, development of dimebon in Huntington’s disease has been discontinued. In January 2012, we and
Medivation, Inc. announced that the CONCERT trial in patients with mild-to-moderate Alzheimer’s disease did not meet the primary efficacy endpoints and that the two companies will discontinue
development of dimebon for all indications, terminate the ongoing open label extension study in Alzheimer’s disease and terminate their collaboration to co-develop and market dimebon.
Additional product-related programs are in various stages of discovery and development. Also, see the discussion in the “Our Business Development Initiatives” section of this Financial Review.
Late
-
stage clinical trials for additional uses and dosage forms for in
-
line and in
-
registration products:
PRODUCT
INDICATION
ELIQUIS (Apixaban)
For the prevention and treatment of venous thromboembolism, which is being developed in collaboration with BMS
Eraxis/Vfend Combination
Aspergillosis fungal infections
INLYTA (Axitinib)
Oral and selective inhibitor of vascular endothelial growth factor (VEGF) receptor 1, 2 & 3 for the treatment of renal cell carcinoma in treatment
-
naïve patients
Lyrica
Peripheral neuropathic pain; CR (once
-
a
-
day) dosing
Sutent
Adjuvant renal cell carcinoma
Tofacitinib
A JAK kinase inhibitor for the treatment of psoriasis
Torisel
Renal cell carcinoma 2nd line
Xalkori (Crizotinib)
An oral ALK and c
-
Met inhibitor for the treatment of ALK
-
positive 1st and 2nd line non
-
small cell lung cancer
Xiapex
Peyronie
’
s disease
Zithromax/chloroquine
Malaria
New drug candidates in late
-
stage development:
CANDIDATE
INDICATION
ALO-02
A Mu-type opioid receptor agonist for the management of moderate-to-severe pain when a continuous, around-the-clock opioid analgesic is needed for an extended
period of time
Bapineuzumab
A beta amyloid inhibitor for the treatment of mild-to-moderate Alzheimer’s disease being developed in collaboration with Janssen Alzheimer Immunotherapy
Research & Development, LLC (Janssen AI), a subsidiary of Johnson & Johnson
Bazedoxifene-conjugated
estrogens
A tissue
-
selective estrogen complex for the treatment of menopausal vasomotor symptoms
Dacomitinib
A pan-HER tyrosine kinase inhibitor for the treatment of advanced non-small cell lung cancer
Inotuzumab ozogamicin
An antibody drug conjugate, consisting of an anti-CD22 monotherapy antibody linked to a cytotoxic agent, calicheamycin, for the treatment of aggressive Non-
Hodgkin
’
s Lymphoma
Tanezumab
An anti
-
nerve growth factor monoclonal antibody for the treatment of pain (on clinical hold)
Our collaboration with Janssen AI on bapineuzumab, a potential treatment for mild-to-moderate Alzheimer’s disease, continues with four Phase 3 studies. In December 2010, Janssen AI confirmed that
enrollment was complete for its two Phase 3 primarily North American studies (301 and 302), including the biomarker sub-
studies. The other two Phase 3 primarily international studies (3000 and 3001)
continue to enroll. Johnson & Johnson expects that the two Janssen AI primarily North American studies will be completed (last patient out) in mid-2012. We expect that the last patient will have
completed our two primarily international 18
-
month trials, including associated biomarker studies, in 2014.
Following requests by the FDA in 2010, we suspended and subsequently terminated worldwide the osteoarthritis, chronic low back pain and painful diabetic peripheral neuropathy studies of
tanezumab. The FDA’s requests followed a small number of reports of osteoarthritis patients treated with tanezumab who experienced the worsening of osteoarthritis leading to joint replacement and
also reflected the FDA’s concerns regarding the potential for such events in other patient populations. In December 2010, the FDA placed a clinical hold on all other anti-nerve growth factor therapies
under clinical investigation in the U.S. Studies of tanezumab in cancer pain were allowed to continue. We continue to work with the FDA to reach an understanding about the appropriate scope of
continued clinical investigation of tanezumab. In July 2011, we submitted our response to the “clinical hold” letter from the FDA, and we anticipate that an FDA Arthritis Advisory Committee meeting will
be held to discuss the anti
-
nerve growth factor class of investigational drugs.
2011 Financial Report
29
(a)
(b)
(a)
(b)

Financial Review
Pfizer Inc. and Subsidiary Companies
COSTS AND EXPENSES
Cost of Sales
2011 vs. 2010
Cost of sales decreased 5% in 2011, compared to 2010, primarily as a result of:
partially offset by:
2010 vs. 2009
Cost of sales increased 87% in 2010, compared to 2009, primarily as a result of:
partially offset by:
Foreign exchange had a minimal impact on cost of sales during 2010.
Selling, Informational and Administrative (SI&A) Expenses
2011 vs. 2010
SI&A expenses were largely unchanged in 2011, compared to 2010, primarily as a result of:
• the fee provided for under the U.S. Healthcare Legislation beginning in 2011;
• the addition of legacy King operating costs; and
• the unfavorable impact of foreign exchange of 2%,
offset by:
2010 vs. 2009
YEAR ENDED DECEMBER 31,
INCR./(DECR.)
(MILLIONS OF DOLLARS)
2011
2010
2009
11/10
10/09
Cost of sales
$15,085
$15,838
$8,459
(5)
%
87%
•
lower purchase accounting charges of $1.7 billion, primarily reflecting the fair value adjustments to acquired inventory from Wyeth that was subsequently sold; and
•
savings associated with our cost
-
reduction and productivity initiatives,
•
the addition of costs from legacy King
’
s operations;
• the Puerto Rico excise tax (for additional information, see the “Provision for Taxes on Income” section of this Financial Review);
•
a shift in geographic and business mix; and
• the unfavorable impact of foreign exchange of 2% in 2011
• purchase accounting charges of approximately $2.9 billion in 2010, compared to approximately $970 million in 2009, primarily reflecting the fair value adjustments to inventory acquired from Wyeth that
was subsequently sold;
• a write-off of inventory of $212 million (which includes a purchase accounting fair value adjustment of $104 million), primarily related to biopharmaceutical inventory acquired from Wyeth that became
unusable after the acquisition date;
• the inclusion of Wyeth’s manufacturing operations for a full year in 2010, compared to part of the year in 2009; and
•
the change in the mix of products and businesses as a result of the Wyeth acquisition,
•
lower costs as a result of our cost
-
reduction and productivity initiatives.
YEAR ENDED DECEMBER 31,
INCR./(DECR.)
(MILLIONS OF DOLLARS)
2011
2010
2009
11/10
10/09
Selling, informational and administrative expenses
$
19,468
$
19,480
$
14,752
—
32%
•
savings associated with our cost
-
reduction and productivity initiatives.
SI&A
expenses increased 32% in 2010, compared to 2009, primarily as a result of:
• the inclusion of Wyeth operating costs for a full year in 2010, compared to part of the year in 2009; and
•
the unfavorable impact of foreign exchange of $236 million.
30
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
Research and Development (R&D) Expenses
2011 vs. 2010
R&D expenses decreased 3% in 2011, compared to 2010, primarily as a result of:
partially offset by:
2010 vs. 2009
R&D expenses increased 20% in 2010, compared to 2009, primarily as a result of:
Foreign exchange had a minimal impact on R&D expenses during 2010.
R&D expenses also include payments for intellectual property rights of $306 million in 2011, $393 million in 2010 and $489 million in 2009 (for further discussion, see the “Our Business Development
Initiatives” section of this Financial Review).
Acquisition-Related In-Process Research and Development Charges
In 2010 and 2009, we resolved certain contingencies and met certain milestones associated with the CovX acquisition and recorded $125 million in 2010 and $68 million in 2009 of Acquisition-related in-
process research and development charges . As of December 31, 2011, we have no unresolved contingencies that could result in charges to Acquisition-related in-process research and development
charges.
Restructuring Charges and Other Costs Associated with Acquisitions and Cost-Reduction/Productivity Initiatives
We incur significant costs in connection with acquiring businesses and restructuring and integrating acquired businesses and in connection with our global cost-reduction and productivity initiatives. For
example:
All of our businesses and functions can be impacted by these actions, including sales and marketing, manufacturing and research and development, as well as functions such as information technology,
shared services and corporate operations.
Since the acquisition of Wyeth, our cost-reduction initiatives announced on January 26, 2009, but not completed as of December 31, 2009, were incorporated into a comprehensive plan to integrate
Wyeth’s operations, acquired on October 15, 2009, to generate cost savings and to capture synergies across the combined company. And, on February 1, 2011, we announced a new research and
productivity initiative to accelerate our strategies to improve innovation and overall productivity in R&D by prioritizing areas with the greatest scientific and commercial promise, utilizing appropriate
risk/return profiles and focusing on areas with the highest potential to deliver value in the near term and over time.
YEAR ENDED DECEMBER 31,
INCR./(DECR.)
(MILLIONS OF DOLLARS)
2011
2010
2009
11/10
10/09
Research and development expenses
$9,112
$9,392
$7,824
(3
)%
20%
•
savings associated with our cost
-
reduction and productivity initiatives,
•
higher charges related to implementing our cost
-
reduction and productivity initiatives;
• the addition of legacy King expenses; and
•
the unfavorable impact of foreign exchange of 1%.
• the inclusion of Wyeth operating costs for a full year in 2010, compared to part of the year in 2009; and
•
continued investment in the late
-
stage development portfolio.
YEAR ENDED DECEMBER 31,
INCR./(DECR.)
(MILLIONS OF DOLLARS)
2011
2010
2009
11/10
10/09
Cost-reduction/productivity initiatives and acquisition activity expenses
$4,520
$3,989
$4,821
13
%
(17)%
• for our cost-reduction and productivity initiatives, we typically incur costs and charges associated with site closings and other facility rationalization actions, workforce reductions and the expansion of
shared services, including the development of global systems; and
• for our acquisition activity, we typically incur costs that can include transaction costs, integration costs (such as expenditures for consulting and the integration of systems and processes) and
restructuring charges, related to employees, assets and activities that will not continue in the combined company.
2011 Financial Report
31

Financial Review
Pfizer Inc. and Subsidiary Companies
Cost-Reduction Goals
With respect to the January 26, 2009 announcements, and our acquisition of Wyeth on October 15, 2009, in the aggregate, we set a goal to generate cost reductions, net of investments in the business,
of approximately $4 billion to $5 billion, by the end of 2012, at 2008 average foreign exchange rates, in comparison with the 2008 pro forma combined adjusted total costs of the legacy Pfizer and legacy
Wyeth operations. (For an understanding of adjusted total costs, see the “Adjusted Income” section of this Financial Review.) We achieved this goal by the end of 2011, a year earlier than expected.
With respect to the new R&D productivity initiative announced on February 1, 2011, we set a goal to achieve significant reductions in our annual research and development expenses by the end of 2012.
Adjusted R&D expenses were $8.4 billion in 2011, and we expect adjusted R&D expenses to be approximately $6.5 billion to $7.0 billion in 2012. (For an understanding of adjusted research and
development expenses, see the “Adjusted Income” section of this Financial Review.) We are on track to meet this 2012 goal.
In addition to these major initiatives, we continuously monitor our organizations for cost reduction and/or productivity opportunities.
Expected Total Costs
We have incurred and will continue to incur costs in connection with these announced actions. We estimate that the total costs of both of the aforementioned initiatives could range up to $16.4 billion
through 2012, of which we have incurred approximately $12.7 billion in cost-reduction and acquisition-related costs (excluding transaction costs) through December 31, 2011.
Key Activities
The targeted cost reductions have been and are being achieved through the following actions:
• The closing of duplicative facilities and other site rationalization actions Company-wide, including research and development facilities, manufacturing plants, sales offices and other corporate facilities.
Among the more significant actions are the following:
•
Manufacturing: After the acquisition of Wyeth, our operational manufacturing sites totaled 81 and in mid-2010, we announced our plant network strategy for our Global Supply division,
excluding Capsugel. Excluding the 14 plants acquired as part of our acquisition activity in 2011, as of December 31, 2011, we operated plants in 74 locations around the world that
manufacture products for our businesses. Locations with major manufacturing facilities include Belgium, China, Germany, Ireland, Italy, Japan, Philippines, Puerto Rico, Singapore and the
United States. Our Global Supply division
’
s plant network strategy has targeted the exiting of ten additional sites over the next several years.
•
Research and Development: After the acquisition of Wyeth, we operated in 20 R&D sites and announced that we would close a number of sites. We have completed a number of site
closures. In addition, in 2011, we closed our Sandwich, U.K. research and development facility, except for a small presence, and rationalized several other sites to reduce and optimize the
overall R&D footprint. We disposed of our toxicology site in Catania, Italy; exited our R&D sites in Aberdeen and Gosport, U.K.; and disposed of a vacant site in St. Louis, MO. We are
presently marketing for sale, lease or sale/lease-back, either a portion of or all of certain of our R&D campuses. Locations with R&D operations are in the U.S., Europe, Canada and China,
with five major research sites in addition to a number of specialized units. We also re
-prioritized our commitments to disease areas and have reduced efforts in areas where we do not
currently have or expect to have a competitive advantage.
• Workforce reductions across all areas of our business and other organizational changes. We identified areas for a reduction in workforce across all of our businesses. After the closing of the Wyeth
acquisition, the combined workforce was approximately 120,700. As of December 31, 2011, the workforce totaled approximately 103,700, a decrease of 17,000, primarily in the U.S. field force,
manufacturing, R&D and corporate operations. We have exceeded our original target for reducing the combined Pfizer/Wyeth workforce.
• The increased use of shared services.
•
Procurement savings.
32
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
Details of Actual Costs Incurred
The components of costs incurred in connection with our acquisitions and our cost
-
reduction/productivity initiatives follow:
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Transaction costs
$ 30
$ 22
$ 768
Integration costs
730
1,004
569
Restructuring charges
Employee termination costs
1,791
1,114
2,564
Asset impairments
256
870
159
Other
127
191
270
Restructuring charges and certain acquisition
-
related costs
$2,934
$3,201
$4,330
Additional depreciation––asset restructuring, recorded in our Consolidated
Statements of Income as follows :
Cost of Sales
$ 557
$ 527
$ 133
Selling, informational and administrative expenses
75
227
53
Research and development expenses
607
34
55
Total additional depreciation––asset restructuring
1,239
788
241
Implementation costs
:
Cost of sales
250
—
46
Selling, informational and administrative expenses
25
—
159
Research and development expenses
72
—
36
Other deductions
—
net
—
—
9
Total implementation costs
347
—
250
Total costs associated with cost-reduction/productivity initiatives and acquisition activity
$4,520
$3,989
$4,821
Transaction costs represent external costs directly related to our business combinations and primarily include expenditures for banking, legal, accounting and other similar services. Substantially all of
the costs incurred in 2009 were fees related to a $22.5 billion bridge term loan credit agreement entered into with certain financial institutions on March 12, 2009 to partially fund our acquisition of
Wyeth. The bridge term loan credit agreement was terminated in June 2009 as a result of our issuance of approximately $24.0 billion of senior unsecured notes in the first half of 2009.
Integration costs represent external, incremental costs directly related to integrating acquired businesses and primarily include expenditures for consulting and the integration of systems and processes.
From the beginning of our cost-reduction and transformation initiatives in 2005 through December 31, 2011, Employee termination costs represent the expected reduction of the workforce by
approximately 57,400 employees, mainly in manufacturing, sales and research, of which approximately 42,800 employees have been terminated as of December 31, 2011. Employee termination costs
are generally recorded when the actions are probable and estimable and include accrued severance benefits, pension and postretirement benefits, many of which may be paid out during periods after
termination.
Asset impairments
primarily include charges to write down property, plant and equipment to fair value.
Other
primarily includes costs to exit certain assets and activities.
The restructuring charges in 2011 are associated with the following:
• Primary Care operating segment ($593 million), Specialty Care and Oncology operating segment ($220 million), Established Products and Emerging Markets operating segment ($110 million),
Animal Health and Consumer Healthcare operating segment ($51 million), Nutrition operating segment ($4 million), research and development operations ($489 million), manufacturing operations
($280 million) and Corporate ($427 million).
The restructuring charges in 2010 are associated with the following:
• Primary Care operating segment ($71 million), Specialty Care and Oncology operating segment ($197 million), Established Products and Emerging Markets operating segment ($43 million), Animal
Health and Consumer Healthcare operating segment ($46 million), Nutrition operating segment ($4 million), research and development operations ($292 million), manufacturing operations ($1.1
billion) and Corporate ($455 million).
The restructuring charges in 2009 are associated with the following:
•
Our three biopharmaceutical operating segments ($1.3 billion), Animal Health and Consumer Healthcare operating segment ($250 million), Nutrition operating segment ($4 million income), research
and development operations ($339 million), manufacturing operations ($292 million) and Corporate ($781 million).
Additional depreciation––asset restructuring represents the impact of changes in the estimated useful lives of assets involved in restructuring actions.
Implementation costs generally represent external, incremental costs directly related to implementing our non-acquisition-related cost-reduction and productivity initiatives.
2011 Financial Report
33
(a)
(b)
(c)
(d)
(e)
(a)
(b)
(c)
(d)
(e)

Financial Review
Pfizer Inc. and Subsidiary Companies
Other Deductions—Net
2011 vs. 2010
Other deductions—net changed favorably by $1.9 billion in 2011, compared to 2010, which primarily reflects:
2010 vs. 2009
Other deductions––net increased by $4.1 billion in 2010, compared to 2009, which primarily reflects:
partially offset primarily by:
Asset Impairment Charges
For information about the asset impairment charges in each year, see the “Significant Accounting Policies and Application of Critical Accounting Estimates—Asset Impairment Reviews—Long-Lived
Assets” section of this Financial Review as well as Notes to Consolidated Financial Statements Note 4. Other Deductions—Net and Note 10B. Goodwill and Other Intangible Assets: Other Intangible
Assets.
The components of restructuring charges associated with all of our cost-reduction and productivity initiatives and acquisition activity follow:
COSTS
INCURRED
ACTIVITY
THROUGH
DECEMBER 31,
ACCRUAL
AS OF
DECEMBER 31,
(MILLIONS OF DOLLARS)
2005
-
2011
2011
2011
Employee termination costs
$10,602
$ 8,167
$2,434
Asset impairments
2,564
2,564
—
Other
1,022
931
92
Total
$14,188
$11,662
$2,526
Includes adjustments for foreign currency translation.
Included in O ther current liabilities ($1.6 billion) and Other noncurrent liabilities ($928 million).
YEAR ENDED DECEMBER 31,
INCR./(DECR.)
(MILLIONS OF DOLLARS)
2011
2010
2009
11/10
10/09
Other Deductions
—
Net
$2,479
$4,336
$285
(43
)%
*
*
Calculation not meaningful
•
asset impairment charges that were approximately $1.3 billion higher in 2010 than in 2011, (see below); and
• charges for litigation-related matters that were $947 million higher in 2010 than in 2011, which reflects charges recorded in 2010 for asbestos litigation related to our wholly owned subsidiary, Quigley
Company, Inc. (see below).
•
higher asset impairment charges of $1.8 billion in 2010, (see below);
• higher charges for litigation-related matters of $1.5 billion in 2010, primarily associated with the additional $1.3 billion (pre-tax) charge for asbestos litigation related to our wholly owned subsidiary,
Quigley Company, Inc. (for additional information, see Notes to Consolidated Financial Statements
—
Note 17. Commitments and Contingencies
);
• higher interest expense of $565 million in 2010, primarily associated with the $13.5 billion of senior unsecured notes that we issued in March 2009 and the approximately $10.5 billion of senior
unsecured notes that we issued in June 2009 to partially finance the acquisition of Wyeth, as well as the addition of legacy Wyeth debt;
• lower interest income of $345 million in 2010, primarily due to lower interest rates coupled with lower average investment balances; and
•
the non
-
recurrence of a $482 million gain recorded in 2009 related to ViiV (see further discussion in the
“
Our Business Development Initiatives
”
section of this Financial Review),
•
higher royalty
-
related income of $336 million in 2010, primarily due to the addition of legacy Wyeth royalties.
34
2011 Financial Report
(a) (b)
(a)
(b)

Financial Review
Pfizer Inc. and Subsidiary Companies
PROVISION FOR TAXES ON INCOME
During the fourth quarter of 2010, we reached a settlement with the U.S. Internal Revenue Service (IRS) related to issues we had appealed with respect to the audits of the Pfizer Inc. tax returns for the
years 2002 through 2005, as well as the Pharmacia audit for the year 2003 through the date of merger with Pfizer (April 16, 2003). The IRS concluded its examination of the aforementioned tax years and
issued a final Revenue Agent’s Report (RAR). We agreed with all of the adjustments and computations contained in the RAR. As a result of settling these audit years, in the fourth quarter of 2010, we
reduced our unrecognized tax benefits by approximately $1.4 billion and reversed the related interest accruals by approximately $600 million, both of which had been classified in Other taxes payable ,
and recorded a corresponding tax benefit in Provision for taxes on income (see Notes to Consolidated Financial Statements–– Note 5. Taxes on Income ).
2011 vs. 2010
The higher effective tax rate in 2011 compared to 2010 is primarily the result of:
partially offset by:
2010 vs. 2009
The lower tax rate for 2010, compared to 2009, is primarily due to:
partially offset by:
Tax Law Changes
On August 10, 2010, the President of the United States signed into law the Education Jobs and Medicaid Assistance Act of 2010 (the Act), which includes education and Medicaid funding provisions, the
cost of which is offset with revenues that result from changes to certain aspects of the tax treatment of the foreign-source income of U.S.-based companies. Given the effective dates of the various
provisions of the Act, it had no impact on our 2010 results. The Act did not have a significant negative impact on our results in 2011 and is not expected to have a significant negative impact on results in
2012. The impact of the Act is recorded in Provision for taxes on income. The impact this year is reflected in our financial guidance for 2012.
YEAR ENDED DECEMBER 31,
INCR./(DECR.)
(MILLIONS OF DOLLARS)
2011
2010
2009
11/10
10/09
Provision for taxes on income
$4,023
$1,071
$2,145
276
%
(50)%
Effective tax rate on continuing operations
31.5
%
11.5
%
20.1
%
•
the non-
recurrence of the aforementioned $1.4 billion reduction in unrecognized tax benefits and $600 million in interest on those unrecognized tax benefits in 2010, which were recorded as a result
of the favorable tax audit settlement pertaining to prior years; and
•
the non-recurrence of a $320 million reduction in unrecognized tax benefits and $140 million in interest on those unrecognized tax benefits in 2010 resulting from the resolution of certain tax
positions pertaining to prior years with various foreign tax authorities as well as from the expiration of the statute of limitations;
•
the decrease and jurisdictional mix of certain impairment charges related to assets acquired in connection with the Wyeth acquisition; and
•
the change in the jurisdictional mix of earnings.
•
the aforementioned $1.4 billion reduction in unrecognized tax benefits and $600 million in interest on those unrecognized tax benefits in 2010, which were recorded as a result of the favorable tax
audit settlement pertaining to prior years;
•
the aforementioned $320 million reduction in unrecognized tax benefits and $140 million in interest on those unrecognized tax benefits in 2010 resulting from the resolution of certain tax positions
pertaining to prior years with various foreign tax authorities, as well as from the expiration of the statute of limitations; and
•
the tax impact of the charge incurred in 2010 for asbestos litigation;
•
the tax impact of higher expenses, incurred as a result of our acquisition of Wyeth, and the mix of jurisdictions in which those expenses were incurred;
•
the write-off in 2010 of the deferred tax asset of approximately $270 million related to the Medicare Part D subsidy for retiree prescription drug coverage, resulting from the provisions of the U.S.
Healthcare Legislation concerning the tax treatment of that subsidy effective for tax years beginning after December 31, 2012; and
•
the non-recurrence of a tax benefit of $174 million that was recorded in the third quarter of 2009 related to the final resolution of certain investigations concerning Bextra and various other products
that resulted in the receipt of information that raised our assessment of the likelihood of prevailing on the technical merits of our tax position, and the non-recurrence of the $556 million tax benefit
recorded in the fourth quarter of 2009 related to the sale of one of our biopharmaceutical companies, Vicuron Pharmaceuticals, Inc.
2011 Financial Report
35

Financial Review
Pfizer Inc. and Subsidiary Companies
On October 25, 2010, the Governor of Puerto Rico signed into law Act 154 to modify the Puerto Rico source-of-income rules and implement an excise tax on the purchase of products by multinational
corporations and their subsidiaries from their Puerto Rico affiliates that will be in effect from 2011 through 2016. Act 154 had no impact on our results in 2010, since it did not become effective until 2011.
Act 154 had a negative impact on our results in 2011 and will continue to negatively impact results through 2016. The impact of Act 154 is recorded in Cost of sales and Provision for taxes on income.
The impact this year is reflected in our financial guidance for 2012.
DISCONTINUED OPERATIONS
For additional information about our discontinued operations, see Notes to Consolidated Financial Statements— Note 2D. Acquisitions, Divestitures, Collaborative Arrangements and Equity-Method
Investments: Divestitures .
ADJUSTED INCOME
General Description of Adjusted Income Measure
Adjusted income is an alternative view of performance used by management, and we believe that investors’ understanding of our performance is enhanced by disclosing this performance measure. We
report Adjusted income in order to portray the results of our major operations––
the discovery, development, manufacture, marketing and sale of prescription medicines for humans and animals, consumer
healthcare (over-the-counter) products, vaccines and nutrition products––prior to considering certain income statement elements. We have defined Adjusted income as Net income attributable to Pfizer
Inc. before the impact of purchase accounting for acquisitions, acquisition-related costs, discontinued operations and certain significant items. The Adjusted income measure is not, and should not be
viewed as, a substitute for U.S. GAAP net income. Adjusted total costs represent the total of Adjusted cost of sales, Adjusted SI&A expenses and Adjusted R&D expenses, which are income statement
line items prepared on the same basis as, and are components of, the overall Adjusted income measure.
The Adjusted income measure is an important internal measurement for Pfizer. We measure the performance of the overall Company on this basis in conjunction with other performance metrics. The
following are examples of how the Adjusted income measure is utilized:
Despite the importance of this measure to management in goal setting and performance measurement, we stress that Adjusted income is a non-GAAP financial measure that has no standardized
meaning prescribed by U.S. GAAP and, therefore, has limits in its usefulness to investors. Because of its non-standardized definition, Adjusted income (unlike U.S. GAAP net income) may not be
comparable to the calculation of similar measures of other companies. Adjusted income is presented solely to permit investors to more fully understand how management assesses performance.
The components of
Discontinued operations
—
net of tax,
substantially all of which relate to our Capsugel business, follow:
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Revenues
$ 507
$752
$740
Pre
-
tax income from discontinued operations
31
140
148
Provision for taxes on income
23
52
51
Income from discontinued operations—net of tax
8
88
97
Pre
-
tax gain/(loss) on sale of discontinued operations
1,688
(11
)
15
Provision for taxes on income
384
—
(
2)
Gain/(loss) on sale of discontinued operations
—
net of tax
1,304
(11
)
17
Discontinued operations
—
net of tax
$1,312
$ 77
$114
Deferred tax amounts are not significant for 2011.
Includes a deferred tax expense of $190 million for 2011.
Includes deferred tax expense of $16 million and $8 million, respectively for 2010 and 2009.
Deferred tax amounts are not significant for 2010 and 2009.
• senior management receives a monthly analysis of our operating results that is prepared on an Adjusted income basis;
•
our annual budgets are prepared on an Adjusted income basis; and
• senior management’s annual compensation is derived, in part, using this Adjusted income measure. Adjusted income is one of the performance metrics utilized in the determination of bonuses under
the Pfizer Inc. Executive Annual Incentive Plan that is designed to limit the bonuses payable to the Executive Leadership Team (ELT) for purposes of Internal Revenue Code Section 162(m). Subject to
the Section 162(m) limitation, the bonuses are funded from a pool based on the achievement of three financial metrics, including adjusted diluted earnings per share, which is derived from Adjusted
income. Beginning in 2011, this metric accounts for 40% of the bonus pool made available to ELT members and other members of senior management and will constitute a factor in determining each of
these individual
’
s bonus.
36
2011 Financial Report
(a), (c)
(b), (d)
(a)
(b)
(c)
(d)

Financial Review
Pfizer Inc. and Subsidiary Companies
We also recognize that, as an internal measure of performance, the Adjusted income measure has limitations, and we do not restrict our performance-management process solely to this metric. A
limitation of the Adjusted income measure is that it provides a view of our operations without including all events during a period, such as the effects of an acquisition or amortization of purchased
intangibles, and does not provide a comparable view of our performance to other companies in the biopharmaceutical industry. We also use other specifically tailored tools designed to achieve the highest
levels of performance. For example, our R&D organization has productivity targets, upon which its effectiveness is measured. In addition, the earn-out of Performance Share Award grants is determined
based on a formula that measures our performance using relative total shareholder return.
Purchase Accounting Adjustments
Adjusted income is calculated prior to considering certain significant purchase accounting impacts resulting from business combinations and net asset acquisitions. These impacts can include the
incremental charge to cost of sales from the sale of acquired inventory that was written up to fair value, amortization related to the increase in fair value of the acquired finite-lived intangible assets
acquired from Pharmacia, Wyeth and King, depreciation related to the increase/decrease in fair value of the acquired fixed assets, amortization related to the increase in fair value of acquired debt,
charges for purchased IPR&D and the fair value changes associated with contingent consideration. Therefore, the Adjusted income measure includes the revenues earned upon the sale of the acquired
products without considering the aforementioned significant charges.
Certain of the purchase accounting adjustments associated with a business combination, such as the amortization of intangibles acquired as part of our acquisition of King in 2011, Wyeth in 2009 and
Pharmacia in 2003, can occur through 20 or more years, but this presentation provides an alternative view of our performance that is used by management to internally assess business performance. We
believe the elimination of amortization attributable to acquired intangible assets provides management and investors an alternative view of our business results by trying to provide a degree of parity to
internally developed intangible assets for which research and development costs previously have been expensed.
However, a completely accurate comparison of internally developed intangible assets and acquired intangible assets cannot be achieved through Adjusted income. This component of Adjusted income is
derived solely from the impacts of the items listed in the first paragraph of this section. We have not factored in the impacts of any other differences in experience that might have occurred if we had
discovered and developed those intangible assets on our own, and this approach does not intend to be representative of the results that would have occurred in those circumstances. For example, our
research and development costs in total, and in the periods presented, may have been different; our speed to commercialization and resulting sales, if any, may have been different; or our costs to
manufacture may have been different. In addition, our marketing efforts may have been received differently by our customers. As such, in total, there can be no assurance that our Adjusted income
amounts would have been the same as presented had we discovered and developed the acquired intangible assets.
Acquisition-Related Costs
Adjusted income is calculated prior to considering transaction, integration, restructuring and additional depreciation costs associated with business combinations because these costs are unique to each
transaction and represent costs that were incurred to restructure and integrate two businesses as a result of the acquisition decision. For additional clarity, only transaction costs, additional depreciation
and restructuring and integration activities that are associated with a business combination or a net-asset acquisition are included in acquisition-related costs. We have made no adjustments for the
resulting synergies.
We believe that viewing income prior to considering these charges provides investors with a useful additional perspective because the significant costs incurred in a business combination result primarily
from the need to eliminate duplicate assets, activities or employees––a natural result of acquiring a fully integrated set of activities. For this reason, we believe that the costs incurred to convert disparate
systems, to close duplicative facilities or to eliminate duplicate positions (for example, in the context of a business combination) can be viewed differently from those costs incurred in other, more normal,
business contexts.
The integration and restructuring costs associated with a business combination may occur over several years, with the more significant impacts ending within three years of the transaction. Because of
the need for certain external approvals for some actions, the span of time needed to achieve certain restructuring and integration activities can be lengthy. For example, due to the highly regulated nature
of the pharmaceutical business, the closure of excess facilities can take several years, as all manufacturing changes are subject to extensive validation and testing and must be approved by the FDA
and/or other global regulatory authorities.
Discontinued Operations
Adjusted income is calculated prior to considering the results of operations included in discontinued operations, as well as any related gains or losses on the sale of such operations such as the sale of
our Capsugel business, which we sold in August 2011. We believe that this presentation is meaningful to investors because, while we review our businesses and product lines for strategic fit with our
operations, we do not build or run our businesses with the intent to sell them. (Restatements due to discontinued operations do not impact compensation or change the adjusted income measure for the
compensation of the restated periods but are presented here on a restated basis for consistency across all periods.)
2011 Financial Report
37
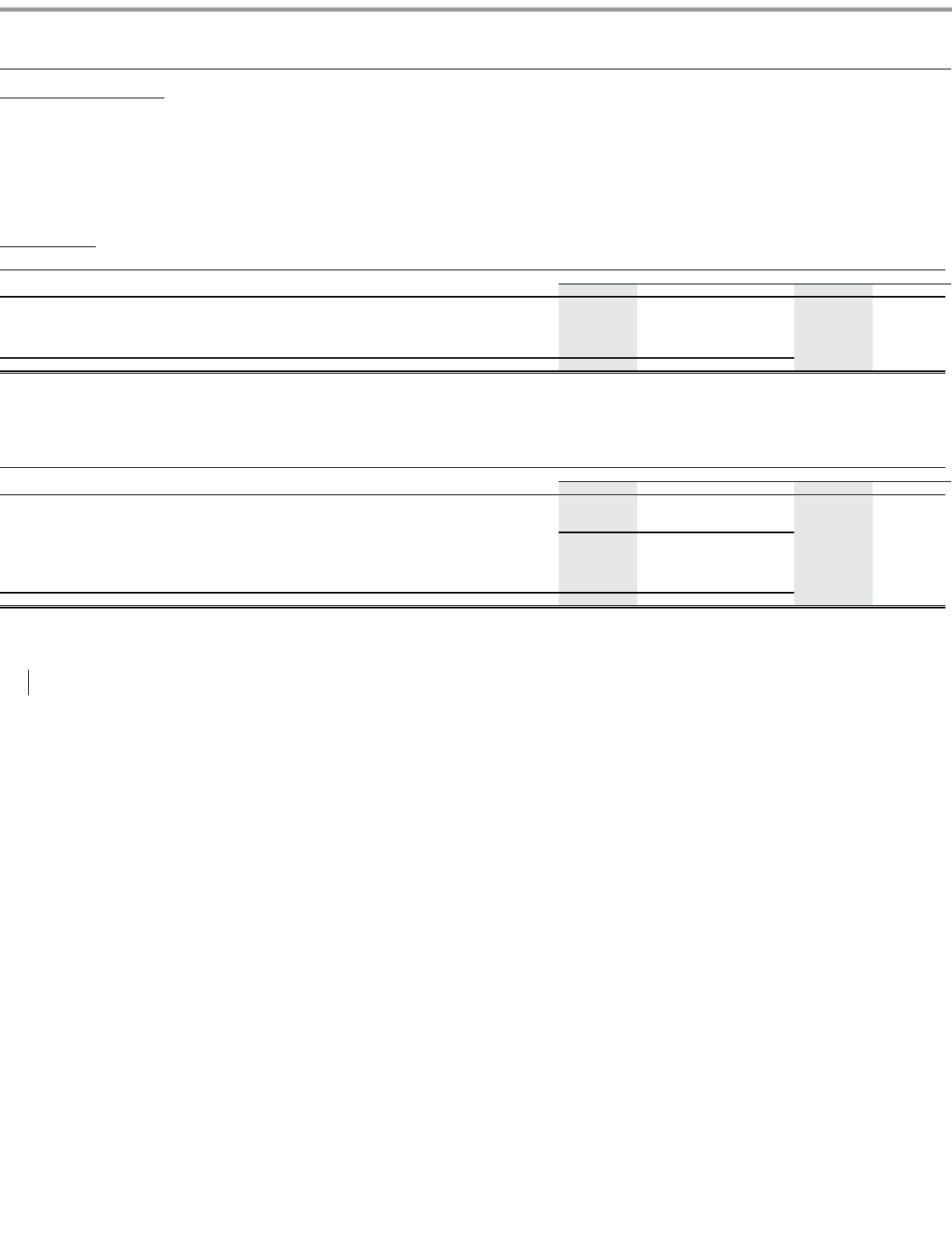
Financial Review
Pfizer Inc. and Subsidiary Companies
Certain Significant Items
Adjusted income is calculated prior to considering certain significant items. Certain significant items represent substantive, unusual items that are evaluated on an individual basis. Such evaluation
considers both the quantitative and the qualitative aspect of their unusual nature. Unusual, in this context, may represent items that are not part of our ongoing business; items that, either as a result of
their nature or size, we would not expect to occur as part of our normal business on a regular basis; items that would be non-recurring; or items that relate to products we no longer sell. While not all-
inclusive, examples of items that could be included as certain significant items would be a major non-acquisition-related restructuring charge and associated implementation costs for a program that is
specific in nature with a defined term, such as those related to our non-acquisition-related cost-reduction and productivity initiatives; charges related to certain sales or disposals of products or facilities
that do not qualify as discontinued operations as defined by U.S. GAAP; amounts associated with transition service agreements in support of discontinued operations after sale; certain intangible asset
impairments; adjustments related to the resolution of certain tax positions; the impact of adopting certain significant, event-driven tax legislation; net interest expense incurred through the consummation
date of the acquisition of Wyeth on acquisition-related borrowings made prior to that date; or possible charges related to legal matters, such as certain of those discussed in Notes to Consolidated
Financial Statements— Note 17. Commitments and Contingencies and in Part II—Other Information; Item 1. Legal Proceedings in our Quarterly Reports on Form 10-Q filings. Normal, ongoing defense
costs of the Company or settlements of and accruals on legal matters made in the normal course of our business would not be considered certain significant items.
Reconciliation
A reconciliation of Net income attributable to Pfizer Inc. , as reported under U.S. GAAP to Adjusted income follows:
Certain amounts and percentages may reflect rounding adjustments.
A reconciliation of Reported diluted EPS, as reported under U.S. GAAP, to Adjusted diluted EPS follows:
Certain amounts and percentages may reflect rounding adjustments.
YEAR ENDED DECEMBER 31,
% CHANGE
(MILLIONS OF DOLLARS)
2011
2010
2009
11/10
10/09
Reported net income attributable to Pfizer Inc.
$10,009
$ 8,257
$ 8,635
21
(4)
Purchase accounting adjustments
—
net of tax
5,032
6,109
2,633
(18)
132
Acquisition
-
related costs
—
net of tax
1,458
2,897
2,858
(50)
1
Discontinued operations—net of tax
(1,312)
(77)
(114)
*
32
Certain significant items
—
net of tax
3,030
699
83
*
*
Adjusted income
$18,217
$17,885
$14,095
2
27
The effective tax rate on Adjusted income was 29.5% in 2011, 29.7% in 2010 and 29.5% in 2009. The lower effective tax rate on Adjusted income in 2011 is primarily due to the change in the
jurisdictional mix of earnings and the write
-
off in 2010 of the deferred tax asset of approximately $270 million related to the Medicare Part D subsidy for retiree prescription drug coverage resulting from
the provisions of the U.S. Healthcare Legislation concerning the tax treatment of that subsidy effective for tax years beginning after December 31, 2012, partially offset by $460 million in tax benefits in
2010 for the resolution of certain tax positions pertaining to prior years with various foreign tax authorities.
*
Calculation not meaningful.
YEAR ENDED DECEMBER 31,
% CHANGE
2011
2010
2009
11/10
10/09
Earnings per common share
—
diluted:
Reported income from continuing operations attributable to Pfizer Inc. common shareholders
$1.11
$1.01
$1.21
10
(17)
Income from discontinued operations—net of tax
0.17
0.01
0.02
*
(50)
Reported net income attributable to Pfizer Inc. common shareholders
1.27
1.02
1.23
25
(17)
Purchase accounting adjustments
—
net of tax
0.64
0.76
0.37
(16)
105
Acquisition
-
related costs
—
net of tax
0.19
0.36
0.41
(47)
(12)
Discontinued operations
—
net of tax
(0.17)
(0.01)
(0.02)
*
50
Certain significant items—net of tax
0.39
0.09
0.01
*
*
Adjusted Net income attributable to Pfizer Inc. common shareholders
$2.31
$2.22
$2.00
4
11
Reported and Adjusted diluted earnings per share in 2011 and 2010 were impacted by the decrease in the number of shares outstanding in comparison with 2009, primarily due to the Company’s
ongoing share repurchase program, offset by the impact of shares issued to partially fund the Wyeth acquisition in 2009.
*
Calculation not meaningful.
38
2011 Financial Report
(a)
(a)
(a)
(a)

Financial Review
Pfizer Inc. and Subsidiary Companies
Adjusted income, as shown above, excludes the following items:
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Purchase accounting adjustments:
Amortization, depreciation and other
$ 5,563
$ 5,228
$ 2,743
Cost of sales, primarily related to fair value adjustments of acquired inventory
1,238
2,904
976
In
-
process research and development charges
—
125
68
Total purchase accounting adjustments, pre
-
tax
6,801
8,257
3,787
Income taxes
(1,769)
(2,148)
(1,154)
Total purchase accounting adjustments—net of tax
5,032
6,109
2,633
Acquisition
-
related costs:
Transaction costs
30
22
768
Integration costs
730
1,004
569
Restructuring charges
598
2,175
2,607
Additional depreciation
—
asset restructuring
625
788
81
Total acquisition
-
related costs, pre
-
tax
1,983
3,989
4,025
Income taxes
(525)
(1,092)
(1,167)
Total acquisition
-
related costs
—
net of tax
1,458
2,897
2,858
Discontinued operations:
Loss/(income) from operations—net of tax
(8)
(88)
(97)
(Gain)/loss on sale of discontinued operations
(1,304)
11
(17)
Total discontinued operations
—
net of tax
(1,312)
(77)
(114)
Certain significant items:
Restructuring charges
1,576
—
386
Implementation costs and additional depreciation
—
asset restructuring
961
—
410
Certain legal matters
828
1,703
294
Net interest expense
—
—
589
Certain asset impairment charges
848
2,151
294
Inventory write
-
off
8
212
—
Gain related to ViiV
—
—
(
482)
Other
133
(102)
20
Total certain significant items, pre
-
tax
4,354
3,964
1,511
Income taxes
(1,324)
(3,265)
(1,428)
Total certain significant items
—
net of tax
3,030
699
83
Total purchase accounting adjustments, acquisition
-
related costs, discontinued operations and certain significant items
—
net of tax
$8,208
$9,628
$5,460
Included primarily in
Amortization of intangible assets
(see Notes to Consolidated Financial Statements
—
Note 10. Goodwill and Other Intangible Assets
).
Included in Acquisition-related in-process research and development charges (see Notes to Consolidated Financial Statements— Note 2. Acquisitions, Divestitures, Collaborative Arrangements and
Equity
-
Method Investments)
.
Included in Restructuring charges and certain acquisition-related costs (see Notes to Consolidated Financial Statements— Note 3. Restructuring Charges and Other Costs Associated with
Acquisitions and Cost
-
Reduction/Productivity Initiatives
).
Represents the impact of changes in the estimated useful lives of assets involved in restructuring actions related to acquisitions. For 2011, included in Cost of sales ($557 million), Selling,
informational and administrative expenses ($45 million) and Research and development expenses ($23 million). For 2010, included in Cost of sales ($527 million), Selling, informational and
administrative expenses ($227 million) and Research and development expenses ($34 million). For 2009, included in Cost of sales ($31 million), Selling, informational and administrative expenses
($37 million) and
Research and development expenses
($13 million).
Represents restructuring charges incurred for our cost-reduction and productivity initiatives. Included in Restructuring charges and certain acquisition-related costs (see Notes to Consolidated
Financial Statements—
Note 3. Restructuring Charges and Other Costs Associated with Acquisitions and Cost
-
Reduction/Productivity Initiatives
).
Amounts primarily relate to our cost-reduction and productivity initiatives (see Notes to Consolidated Financial Statements— Note 3. Restructuring Charges and Other Costs Associated with
Acquisitions and Cost
-Reduction/Productivity Initiatives ). For 2011, included in Cost of sales ($250 million), Selling, informational and administrative expenses ($55 million), Research and
development expenses ($656 million). For 2009, included in Cost of sales ($148 million), Selling, informational and administrative expenses ($175 million), Research and development expenses ($78
million) and
Other deductions
—
net
($9 million).
Included in Other deductions—net . For 2011, includes approximately $700 million related to hormone-replacement therapy litigation. For 2010, includes an additional $1.3 billion charge for asbestos
litigation related to our wholly owned subsidiary Quigley Company, Inc. (for additional information, see Notes to Consolidated Financial Statements
Note 17. Commitments and Contingencies
).
Included in Other deductions—net. Includes interest expense on the senior unsecured notes issued in connection with our acquisition of Wyeth, less interest income earned on the proceeds of the
notes.
Included in Other deductions—net. In 2011 and 2010, the majority relates to certain Wyeth intangible assets, including IPR&D intangible assets. In 2011, also includes a charge related to our
indefinite-lived brand asset, Xanax. In 2010, also includes a charge related to an intangible asset associated with our product, Thelin . In 2009, primarily relates to certain materials used in our
research and development activities that were no longer considered recoverable. (See also the “Other (Income)/Deductions—Net” section of this Financial Review and Notes to Consolidated
Financial Statements—
Note 4. Other Deductions
—
Net
.)
Included in
Cost of sales
(see also the
“
Costs and Expenses
–
–
Cost of Sales
”
section of this Financial Review
).
Included in Other deductions––net and represents a gain related to ViiV, an equity method investment (see Notes to Consolidated Financial Statements— Note 2F. Acquisitions, Divestitures,
Collaborative Arrangements and Equity
-
Method Investments: Equity
-
Method Investments).
Included in Provision for taxes on income. In 2011, primarily includes the tax impacts and jurisdictional mix of restructuring, implementation and impairment charges. Amounts in 2010 include a $2.0
billion tax benefit recorded in the fourth quarter as a result of a settlement of certain audits covering the years 2002-2005 (see Notes to Consolidated Financial Statements–– Note 5D. Taxes on
Income: Tax Contingencies). Amounts in 2009 include tax benefits of approximately $556 million related to the sale of one of our biopharmaceutical companies, Vicuron, which were recorded in the
fourth quarter of 2009, and tax benefits of approximately $174 million related to the final resolution of investigations concerning Bextra and various other products, which were recorded in the third
quarter of 2009. This resolution resulted in the receipt of information that raised our assessment of the likelihood of prevailing on the technical merits of our tax position.
2011 Financial Report
39
(a)
(b)
(c)
(c)
(c)
(d)
(e)
(f)
(g)
(h)
( i )
(j)
(k)
(l)
(a)
(b)
(c)
(d)
(e)
(f)
(g)
(h)
( i )
(j)
(k)
(l)
Financial Review
Pfizer Inc. and Subsidiary Companies
ANALYSIS OF THE CONSOLIDATED BALANCE SHEETS
Discussion of Changes
Virtually all changes in our asset and liability accounts as of December 31, 2011, compared to December 31, 2010, reflect, among other things, increases associated with our acquisition of King (see
Notes to Consolidated Financial Statements— Note 2B. Acquisitions, Divestitures, Collaborative Arrangements and Equity-Method Investments: Acquisition of King Pharmaceuticals, Inc. ) and increases
due to the impact of foreign exchange.
For information about certain of our financial assets and liabilities, including cash and cash equivalents, short-term investments, short-term loans, long-term investments and loans, short-term borrowings,
including current portion of long-term debt , and long-term debt , see “Analysis of Financial Condition, Liquidity and Capital Resources” below.
For Accounts Receivable, net , see “Selected Measures of Liquidity and Capital Resources: Accounts Receivable” below.
For Inventories , the change also reflects inventory sold during 2011 that was acquired from Wyeth and that had been recorded at fair value.
For Assets of discontinued operations and other assets held for sale , the decrease reflects the sale of Capsugel (see Notes to Consolidated Financial Statements— Note 2D. Acquisitions, Divestitures,
Collaborative Arrangements and Equity-Method Investments: Divestitures ).
For Identifiable intangible assets, less accumulated amortization, the change also includes the impact of impairments of certain assets (see Notes to Consolidated Financial Statements— Note 4. Other
Deductions—Net ).
For Other current liabilities, the change also includes the charges recorded for hormone-replacement therapy litigation (see Notes to Consolidated Financial Statements— Note 4. Other Deductions––Net
and Note 17. Commitments and Contingencies ).
For Pension benefit obligations, the change also reflects the impact of $2.9 billion of company contributions in 2011 (see Notes to Consolidated Financial Statements— Note 11. Pension and
Postretirement Benefit Plans and Defined Contribution Plans ).
For Other noncurrent liabilities, the change also reflects an increase in the fair value of derivative financial instruments in a liability position (see Notes to Consolidated Financial Statements – Note 7A.
Financial Instruments: Selected Financial Assets and Liabilities).
Goodwill
Goodwill –
Our company was previously managed through two operating segments (Biopharmaceutical and Diversified), and is now managed through five operating segments (see Notes to Consolidated
Financial Statements – Note 18. Segment, Geographic and Other Revenue Information for further information). As a result of this change, the goodwill previously associated with our Biopharmaceutical
operating segment has been allocated among the Primary Care, Specialty Care and Oncology, and Established Products and Emerging Markets operating segments.
While all reporting units can confront events and circumstances that can lead to impairments (such as, among other things, unanticipated competition, an adverse action or assessment by a regulator, a
significant adverse change in legal matters or in the business climate and/or a failure to replace the contributions of products that lose exclusivity), in general, the increased number of biopharmaceutical
reporting units significantly increases our risk of goodwill impairment charges as smaller reporting units are inherently less able to absorb negative developments that might affect certain operating assets
but not others. However, as a result of our goodwill impairment review work, we concluded that none of our goodwill is impaired as of December 31, 2011, and we do not believe the risk of impairment is
significant at this time (see also the “Significant Accounting Policies and Application of Critical Accounting Estimates” section of this Financial Review).
The allocation of biopharmaceutical goodwill and goodwill impairment testing depend heavily on the determination of fair value, as defined by U.S. GAAP, and these judgments can materially impact our
results of operations. A single estimate of fair value can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimate and assumptions. For information
about the risks associated with estimates and assumptions, see Notes to Consolidated Financial Statements— Note 1C. Significant Accounting Policies: Estimates and Assumptions.
40
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
ANALYSIS OF THE CONSOLIDATED STATEMENTS OF CASH FLOWS
Operating Activities
2011 vs. 2010
Our net cash provided by operating activities was $20.2 billion in 2011, compared to $11.5 billion in 2010. The increase in operating cash flows was primarily attributable to:
In 2010, the cash flow line item called Other tax accounts, net, reflects the $11.8 billion tax payment described above.
2010 vs. 2009
Our net cash provided by continuing operating activities was $11.5 billion in 2010, compared to $16.6 billion in 2009. The decrease in net cash provided by operating activities was primarily attributable to:
partially offset by:
• the timing of receipts and payments in the ordinary course of business.
Investing Activities
2011 vs. 2010
Our net cash provided by investing activities was $2.2 billion in 2011, compared to $492 million net cash used in 2010. The increase in cash provided by investing activities was primarily attributable to:
partially offset by:
2010 vs. 2009
Our net cash used in investing activities was $492 million in 2010, compared to $31.3 billion in 2009. The decrease in net cash used in investing activities was primarily attributable to:
partially offset by:
YEAR ENDED DECEMBER 31,
% INCR./(DECR.)
(MILLIONS OF DOLLARS)
2011
2010
2009
11/10
10/09
Cash provided by/(used in):
Operating activities
$
20,240
$
11,454
$
16,587
77
(31)
Investing activities
2,200
(492
)
(31,272
)
*
(98)
Financing activities
(20,607
)
(11,174
)
14,481
(84
)
*
Effect of exchange
-
rate changes on cash and cash equivalents
(29
)
(31
)
60
6
*
Net increase/decrease in cash and cash equivalents
1,804
(243
)
(144
)
*
(69)
* Calculation
not meaningful
•
income tax payments made in 2010 of approximately $11.8 billion, primarily associated with certain business decisions executed to finance the Wyeth acquisition, including the decision to repatriate
certain funds earned outside the U.S., compared with $2.9 billion in 2011; and
•
the timing of receipts and payments in the ordinary course of business.
•
income tax payments in 2010 of approximately $11.8 billion, primarily associated with certain business decisions executed to finance the Wyeth acquisition, including the decision to repatriate certain
funds earned outside the U.S., compared with $2.3 billion in 2009;
•
the inclusion of operating cash flows from legacy Wyeth operations for a full year in 2010;
•
the non-recurrence of payments in 2009 in connection with the resolution of certain legal matters related to Bextra and certain other products and our NSAID pain medicines of approximately $3.2
billion; and
•
net proceeds from redemptions, purchases and sales of investments of $4.1 billion in 2011, which were primarily used to finance our acquisitions, compared to net proceeds from redemptions,
purchases and sales of investments of $23 million in 2010; and
•
net proceeds of $2.4 billion received from the sale of Capsugel in 2011 (see Notes to Consolidated Financial Statements— Note 2D. Acquisitions, Divestitures, Collaborative Arrangements and Equity-
Method Investments: Divestitures)
;
•
net cash of $3.3 billion paid for the acquisitions of King, Excaliard and Icagen in 2011, compared to $273 million paid for the acquisitions of FoldRx, Vetnex and Synbiotics in 2010.
•
net cash paid for acquisitions of $273 million in 2010 compared to $43.1 billion in 2009 for the acquisition of Wyeth;
•
net proceeds from redemptions and sales of investments of $23 million in 2010, compared to net proceeds from redemptions and sales of investments of $12.4 billion in 2009.
2011 Financial Report
41

Financial Review
Pfizer Inc. and Subsidiary Companies
Financing Activities
2011 vs. 2010
Our net cash used in financing activities was $20.6 billion in 2011, compared to $11.2 billion in 2010. The increase in net cash used in financing activities was primarily attributable to:
2010 vs. 2009
Our net cash used in financing activities was $11.2 billion in 2010 compared to net cash provided by financing activities of $14.5 billion in 2009. The change in financing cash flows was primarily
attributable to:
ANALYSIS OF FINANCIAL CONDITION, LIQUIDITY AND CAPITAL RESOURCES
Net Financial Liabilities, as shown below:
We rely largely on operating cash flows, short-term investments, short-term commercial paper borrowings and long-term debt to provide for our liquidity requirements. We believe that we have the ability
to obtain both short-term and long-term debt to meet our financing needs for the foreseeable future. Due to our significant operating cash flows, including the impact on cash flows of the anticipated cost
savings from our cost-reduction and productivity initiatives, as well as our financial assets, access to capital markets and available lines of credit and revolving credit agreements, we further believe that
we have the ability to meet our liquidity needs for the foreseeable future, which include:
Our long-term debt is rated high quality by both Standard & Poor’s and Moody’s Investors Service. As market conditions change, we continue to monitor our liquidity position. We have taken and will
continue to take a conservative approach to our financial investments. Both short-term and long-term investments consist primarily of high-quality, highly liquid, well-diversified, available-for-sale debt
securities. Our short-term and long-term loans are due from companies with highly rated securities (Standard & Poor’s ratings of mostly AA or better).
Net financial liabilities decreased during 2011 primarily due to a reduction in short-term borrowings and long-term debt. For additional information, see the “Analysis of the Consolidated Statements of
Cash Flows” section of this Financial Review.
•
net repayments of borrowings of $5.5 billion in 2011, compared to net repayments of borrowings of $4.2 billion in 2010; and
•
purchases of our common stock of $9.0 billion in 2011, compared to purchases of $1.0 billion in 2010.
•
net repayments of borrowings of $4.2 billion in 2010, compared to net proceeds from borrowings of $20.1 billion in 2009, primarily associated with our acquisition of Wyeth; and
•
purchases of our common stock of $1.0 billion in 2010, compared to no purchases in 2009.
AS OF DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
Financial assets:
Cash and cash equivalents
$ 3,539
$ 1,735
Short
-
term investments
23,219
26,277
Short-term loans
51
467
Long-term investments and loans
9,457
9,747
Total financial assets
36,266
38,226
Debt:
Short
-
term borrowings, including current portion of long
-
term debt
4,018
5,603
Long
-
term debt
34,931
38,410
Total debt
38,949
44,013
Net financial liabilities
$ (2,683
)
$ (5,787
)
•
the working capital requirements of our operations, including our research and development activities;
•
investments in our business;
•
dividend payments and potential increases in the dividend rate;
•
share repurchases, including our plan to repurchase approximately $5 billion of our common stock in 2012;
•
the cash requirements associated with our cost
-
reduction/productivity initiatives;
•
paying down outstanding debt;
•
contributions to our pension and postretirement plans; and
•
business
-
development activities.
42
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
Credit Ratings
Two major corporate debt-rating organizations, Moody’s Investors Service (Moody’s) and Standard & Poor’s (S&P), assign ratings to our short-term and long-term debt.
The current ratings assigned by these rating agencies to our commercial paper and senior unsecured non-credit-enhanced long-term debt follow:
Debt Capacity
We have available lines of credit and revolving credit agreements with a group of banks and other financial intermediaries. We maintain cash and cash equivalent balances and short-term investments in
excess of our commercial paper and other short-term borrowings. As of December 31, 2011, we had access to $9.4 billion of lines of credit, of which $2.3 billion expire within one year. Of these lines of
credit, $8.6 billion are unused, of which our lenders have committed to loan us $7.5 billion at our request. Also, $7.0 billion of our unused lines of credit, all of which expire in 2016, may be used to support
our commercial paper borrowings.
Global Economic Conditions
The challenging economic environment has not had, nor do we anticipate it will have, a significant impact on our liquidity. Due to our significant operating cash flows, financial assets, access to capital
markets and available lines of credit and revolving credit agreements, we continue to believe that we have the ability to meet our liquidity needs for the foreseeable future. As markets change, we continue
to monitor our liquidity position. There can be no assurance that the challenging economic environment or a further economic downturn would not impact our ability to obtain financing in the future.
Selected Measures of Liquidity and Capital Resources
Certain relevant measures of our liquidity and capital resources follow:
In fiscal 2012, we funded our acquisition of Ferrosan’s consumer healthcare business, which closed in December 2011 (which falls in the first fiscal quarter of 2012 for our international operations), with
available cash and the proceeds from short-term investments. For additional information on this transaction, see the “Our Business Development Initiatives” section of this Financial Review.
For additional information about the sources and uses of our funds, see the “Analysis of Consolidated Balance Sheets” and “Analysis of Consolidated Statements of Cash Flows” sections of this Financial
Review.
Domestic and International Short-Term Funds
Many of our operations are conducted outside the U.S., and significant portions of our cash, cash equivalents and short-term investments are held internationally. We generally hold approximately 10%-
30% of these short-term funds in U.S. tax jurisdictions. The amount of funds held in U.S. tax jurisdictions can fluctuate due to the timing of receipts and payments in the ordinary course of business and
due to other reasons, such as business-development activities. As part of our ongoing liquidity assessments, we regularly monitor the mix of domestic and international cash flows (both inflows and
outflows). Repatriation of overseas funds can result in additional U.S. federal, state and local income tax payments. We record U.S. deferred tax liabilities for certain unremitted earnings, but when
amounts earned overseas are expected to be permanently reinvested outside of the U.S., no accrual for U.S. taxes is provided.
Accounts Receivable
We continue to monitor developments regarding government and government agency receivables in several European markets, where economic conditions remain uncertain. Historically, payments from
a number of European governments and government agencies extend beyond the contractual terms of sale and the trend is worsening. In Greece, certain of our accounts receivable have been
restructured into bonds with maturities that further lengthened the repayment timeline.
NAME OF RATING AGENCY
COMMERCIAL
PAPER
LONG
-
TERM DEBT
DATE OF LAST
ACTION
RATING
OUTLOOK
Moody
’
s
P
-
1
A1
Stable
October 2009
S&P
A1+
AA
Stable
October 2009
AS OF DECEMBER 31,
(MILLIONS OF DOLLARS, EXCEPT RATIOS AND PER COMMON SHARE DATA)
2011
2010
Cash and cash equivalents and short-term investments and loans
$26,809
$28,479
Working capital
$29,659
$32,377
Ratio of current assets to current liabilities
2.06:1
2.13:1
Shareholders
’
equity per common share
$10.85
$10.96
See Notes to Consolidated Financial Statements – Note 7. Financial Instruments for a description of investment assets held and for a description of credit risk related to our financial instruments held.
Working capital includes assets held for sale of $101 million as of December 31, 2011, and $1.4 billion as of December 31, 2010. Working capital also includes liabilities of discontinued operations of
$151 million as of December 31, 2010.
Represents total Pfizer Inc. shareholders’ equity divided by the actual number of common shares outstanding (which excludes treasury shares and share held by our employee benefit trust).
2011 Financial Report
43
(a)
(b)
(c)
(a)
(b)
(c)

Financial Review
Pfizer Inc. and Subsidiary Companies
We believe that our allowance for doubtful accounts is appropriate. Our assessment is based on an analysis of the following: (i) payments received to date; (ii) the consistency of payments from
customers; (iii) direct and observed interactions with the governments (including court petitions) and with market participants (for example, the factoring industry); and (iv) various third-party assessments
of repayment risk (for example, rating agency publications and the movement of rates for credit default swap instruments).
As of December 31, 2011, we had about $1.5 billion in aggregate gross accounts receivable from governments and/or government agencies in Spain, Italy, Greece, Portugal and Ireland, where economic
conditions remain uncertain. Such receivables in excess of one year from the invoice date were as follows: $290 million in Spain; $139 million in Italy; $81 million in Greece; and $10 million in Portugal.
Although certain European governments and government agencies sometimes delay payments beyond the contractual terms of sale, we seek to appropriately balance repayment risk with the desire to
maintain good relationships with our customers and to ensure a humanitarian approach to local patient needs.
We will continue to closely monitor repayment risk and, when necessary, we will continue to adjust our allowance for doubtful accounts and/or write-down our holdings in Greek bonds.
Our assessments about the recoverability of accounts receivables can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions.
For information about the risks associated with estimates and assumptions, see Notes to Consolidated Financial Statements— Note 1C. Significant Accounting Policies: Estimates and Assumptions.
Share Purchase Plans
From June 2005 through year-end 2011, we purchased approximately 1.2 billion shares of our common stock for approximately $28 billion. On February 1, 2011, we announced that the Board of
Directors authorized a new $5 billion share-purchase plan. On December 12, 2011, we announced that the Board of Directors authorized an additional $10 billion share-purchase plan. In 2011, we
purchased approximately 459 million shares of our common stock for approximately $9.0 billion. In 2010, we purchased approximately 61 million shares of our common stock for approximately $1.0
billion. We did not purchase any shares of our common stock in 2009.
After giving effect to share purchases through year-end 2011, our remaining share-purchase authorization is approximately $10 billion at December 31, 2011. During 2012, we anticipate purchasing
approximately $5 billion of our common stock, with the remaining authorized amount available in 2013 and beyond.
Contractual Obligations
Payments due under contractual obligations as of December 31, 2011, mature as follows:
The above table excludes amounts for potential milestone payments under collaboration, licensing or other arrangements unless the payments are deemed reasonably likely to occur. Payments under
these agreements generally become due and payable only upon the achievement of certain development, regulatory and/or commercialization milestones, which may span several years and which may
never occur.
YEARS
(MILLIONS OF DOLLARS)
TOTAL
2012
2013-
2014
2015-
2016
Thereafter
Long-term debt, including interest obligations
$54,870
$1,658
$10,936
$10,066
$32,210
Other long
-
term liabilities reflected on our consolidated balance sheet under U.S. GAAP
5,553
506
1,132
1,099
2,816
Lease commitments
1,430
190
314
191
735
Purchase obligations and other
3,835
1,291
1,561
616
367
Uncertain tax positions
491
491
—
—
—
Our long-term debt obligations include both our expected principal and interest obligations. Our calculations of expected interest payments incorporate only current period assumptions for interest
rates, foreign currency translation rates and hedging strategies (see Notes to Consolidated Financial Statements— Note 9. Financial Instruments ). Long-term debt consists of senior unsecured notes
including fixed and floating rate, foreign currency denominated, and other notes.
Includes expected payments relating to our unfunded U.S. supplemental (non-qualified) pension plans, postretirement plans and deferred compensation plans.
Includes operating and capital lease obligations.
Includes agreements to purchase goods and services that are enforceable and legally binding and includes amounts relating to advertising, information technology services, employee benefit
administration services, and potential milestone payments deemed reasonably likely to occur.
Except for amounts reflected in Income taxes payable , we are unable to predict the timing of tax settlements, as tax audits can involve complex issues and the resolution of those issues may span
multiple years, particularly if subject to negotiation or litigation.
44
2011 Financial Report
(a)
(b)
(c)
(d)
(e)
( a )
( b )
(c)
(d)
(e)

Financial Review
Pfizer Inc. and Subsidiary Companies
In 2012, we expect to spend approximately $1.5 billion on property, plant and equipment. Planned capital spending mostly represents investment to maintain existing facilities and capacity. We rely
largely on operating cash flows to fund our capital investment needs. Due to our significant operating cash flows, we believe we have the ability to meet our capital investment needs and anticipate no
delays to planned capital expenditures.
Off-Balance Sheet Arrangements
In the ordinary course of business and in connection with the sale of assets and businesses, we often indemnify our counterparties against certain liabilities that may arise in connection with a transaction
or that are related to activities prior to a transaction. These indemnifications typically pertain to environmental, tax, employee and/or product-related matters, and patent-infringement claims. If the
indemnified party were to make a successful claim pursuant to the terms of the indemnification, we would be required to reimburse the loss. These indemnifications generally are subject to threshold
amounts, specified claim periods and other restrictions and limitations. Historically, we have not paid significant amounts under these provisions and, as of December 31, 2011, recorded amounts for the
estimated fair value of these indemnifications are not significant.
Certain of our co-promotion or license agreements give our licensors or partners the rights to negotiate for, or in some cases to obtain under certain financial conditions, co-promotion or other rights in
specified countries with respect to certain of our products.
Dividends on Common Stock
We paid dividends of $6.2 billion in 2011 and $6.1 billion in 2010 on our common stock. In December 2011, our Board of Directors declared a first-quarter 2012 dividend of $0.22 per share, payable on
March 6, 2012, to shareholders of record at the close of business on February 3, 2012. The first-quarter 2012 cash dividend will be our 293 consecutive quarterly dividend.
Our current and projected dividends provide a return to shareholders while maintaining sufficient capital to invest in growing our businesses and increasing shareholder value. Our dividends are not
restricted by debt covenants. While the dividend level remains a decision of Pfizer’
s Board of Directors and will continue to be evaluated in the context of future business performance, we currently believe
that we can support future annual dividend increases, barring significant unforeseen events.
NEW ACCOUNTING STANDARDS
Recently Adopted Accounting Standards
See Notes to Consolidated Financial Statements— Note 1B. Significant Accounting Policies: New Accounting Standards.
Recently Issued Accounting Standard, Not Adopted as of December 31, 2011
In June 2011, the Financial Accounting Standards Board (FASB) issued an accounting standards update regarding the presentation of comprehensive income in financial statements. The provisions of
this standard provide an option to present the components of net income and other comprehensive income either as one continuous statement of comprehensive income or as two separate but
consecutive statements. This standard was amended December 2011. The provisions of this new disclosure standard are effective January 1, 2012 and, beginning in 2012, we will provide a separate
Statement of Other Comprehensive Income.
In September 2011, the FASB issued an accounting standards update to the guidelines that address the accounting for goodwill to permit a qualitative approach to determining the likelihood of a goodwill
impairment charge. The provisions of this new standard are permitted to be adopted early.
FORWARD-LOOKING INFORMATION AND FACTORS THAT MAY AFFECT FUTURE RESULTS
The SEC encourages companies to disclose forward-looking information so that investors can better understand a company’s future prospects and make informed investment decisions. This report and
other written or oral statements that we make from time to time contain such forward-looking statements that set forth anticipated results based on management’s plans and assumptions. Such forward-
looking statements involve substantial risks and uncertainties. We have tried, wherever possible, to identify such statements by using words such as “will,” “anticipate,” “estimate,” “expect,” “project,”
“intend,” “plan,” “believe,” “target,” “forecast,” “goal”, “objective” and other words and terms of similar meaning or by using future dates in connection with any discussion of future operating or financial
performance, business plans and prospects, in-line products and product candidates, strategic review, capital allocation, and share-repurchase and dividend-rate plans. In particular, these include
statements relating to future actions, business plans and prospects, prospective products or product approvals, future performance or results of current and anticipated products, sales efforts, expenses,
interest rates, foreign exchange rates, the outcome of contingencies, such as legal proceedings, share-repurchase and dividend-rate plans, and financial results, including, in particular, the financial
guidance and anticipated cost savings set forth in the “Restructuring Charges and Other Costs Associated with Acquisitions and Cost-Reduction/Productivity Initiatives” and “Our Financial Guidance for
2012” sections of this Financial Review. Among the factors that could cause actual results to differ materially from past and projected future results are the following:
•
Success of research and development activities including, without limitation, the ability to meet anticipated clinical trial completion dates, regulatory submission and approval dates, and launch dates
for product candidates;
2011 Financial Report
45
rd

Financial Review
Pfizer Inc. and Subsidiary Companies
•
Decisions by regulatory authorities regarding whether and when to approve our drug applications, as well as their decisions regarding labeling, ingredients and other matters that could affect the
availability or commercial potential of our products;
•
Speed with which regulatory authorizations, pricing approvals and product launches may be achieved;
•
Success of external business
-
development activities;
•
Competitive developments, including the impact on our competitive position of new product entrants, in-line branded products, generic products, private label products and product candidates that treat
diseases and conditions similar to those treated by our in
-
line drugs and drug candidates;
•
Implementation by the FDA of an abbreviated legal pathway to approve biosimilar products, which could subject our biologic products to competition from biosimilar products in the U.S., with attendant
competitive pressures, after the expiration of any applicable exclusivity period and patent rights;
•
Ability to meet generic and branded competition after the loss of patent protection for our products or competitor products;
•
Ability to successfully market both new and existing products domestically and internationally;
•
Difficulties or delays in manufacturing;
•
Trade buying patterns;
•
Impact of existing and future legislation and regulatory provisions on product exclusivity;
•
Trends toward managed care and healthcare cost containment;
•
Impact of the U.S. Budget Control Act of 2011 (the Budget Control Act) and the deficit-reduction actions to be taken pursuant to the Budget Control Act in order to achieve the deficit-reduction targets
provided for therein, and the impact of any broader deficit
-
reduction efforts;
•
Impact of U.S. healthcare legislation enacted in 2010—the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act––and of any modification,
repeal or invalidation of any of the provisions thereof;
•
U.S. legislation or regulatory action affecting, among other things, pharmaceutical product pricing, reimbursement or access, including under Medicaid, Medicare and other publicly funded or subsidized
health programs; the importation of prescription drugs from outside the U.S. at prices that are regulated by governments of various foreign countries; direct-to-consumer advertising and interactions
with healthcare professionals; and the use of comparative effectiveness methodologies that could be implemented in a manner that focuses primarily on the cost differences and minimizes the
therapeutic differences among pharmaceutical products and restricts access to innovative medicines;
•
Legislation or regulatory action in markets outside the U.S. affecting pharmaceutical product pricing, reimbursement or access, including, in particular, continued government-
mandated price reductions
for certain biopharmaceutical products in certain European and emerging market countries;
•
Contingencies related to actual or alleged environmental contamination;
•
Claims and concerns that may arise regarding the safety or efficacy of in-line products and product candidates;
•
Significant breakdown, infiltration or interruption of our information technology systems and infrastructure;
•
Legal defense costs, insurance expenses, settlement costs, the risk of an adverse decision or settlement and the adequacy of reserves related to product liability, patent protection, government
investigations, consumer, commercial, securities, antitrust, environmental and tax issues, ongoing efforts to explore various means for resolving asbestos litigation, and other legal proceedings;
•
Ability to protect our patents and other intellectual property both domestically and internationally;
•
Interest rate and foreign currency exchange rate fluctuations;
•
Governmental laws and regulations affecting domestic and foreign operations including, without limitation, tax obligations and changes affecting the tax treatment by the U.S. of income earned outside
the U.S. that may result from pending and possible future proposals;
•
Changes in U.S. generally accepted accounting principles;
•
Uncertainties related to general economic, political, business, industry, regulatory and market conditions, including, without limitation, uncertainties related to the impact on us, our lenders, our
customers, our suppliers and counterparties to our foreign-exchange and interest-rate agreements of challenging global economic conditions and recent and possible future changes in global financial
markets; and the related risk that our allowance for doubtful accounts may not be adequate;
•
Any changes in business, political and economic conditions due to actual or threatened terrorist activity in the U.S. and other parts of the world and related U.S. military action overseas;
•
Growth in costs and expenses;
•
Changes in our product, segment and geographic mix; and
46
2011 Financial Report

Financial Review
Pfizer Inc. and Subsidiary Companies
We cannot guarantee that any forward-looking statement will be realized, although we believe we have been prudent in our plans and assumptions. Achievement of anticipated results is subject to
substantial risks, uncertainties and inaccurate assumptions. Should known or unknown risks or uncertainties materialize or should underlying assumptions prove inaccurate, actual results could vary
materially from past results and those anticipated, estimated or projected. Investors should bear this in mind as they consider forward-looking statements.
We undertake no obligation to publicly update forward-looking statements, whether as a result of new information, future events or otherwise. You are advised, however, to consult any further disclosures
we make on related subjects in our Form 10
-Q, 8-K and 10-K reports and our other filings with the SEC.
Certain risks, uncertainties and assumptions are discussed here and under the heading entitled “Risk Factors” in Part I, Item 1A. of our Annual Report on Form 10-K for the year ended December 31,
2011, which will be filed in February 2012. We note these factors for investors as permitted by the Private Securities Litigation Reform Act of 1995. You should understand that it is not possible to predict
or identify all such factors. Consequently, you should not consider any such list to be a complete set of all potential risks or uncertainties.
This report includes discussion of certain clinical studies relating to various in-line products and/or product candidates. These studies typically are part of a larger body of clinical data relating to such
products or product candidates, and the discussion herein should be considered in the context of the larger body of data. In addition, clinical trial data are subject to differing interpretations, and, even
when we view data as sufficient to support the safety and/or effectiveness of a product candidate or a new indication for an in
-line product, regulatory authorities may not share our views and may require
additional data or may deny approval altogether.
Financial Risk Management
The overall objective of our financial risk management program is to seek to minimize the impact of foreign exchange rate movements and interest rate movements on our earnings. We manage these
financial exposures through operational means and by using various financial instruments. These practices may change as economic conditions change.
Foreign Exchange Risk
A significant portion of our revenues and earnings is exposed to changes in foreign exchange rates. We seek to manage our foreign exchange risk in part through operational means, including managing
same-currency revenues in relation to same-currency costs and same-currency assets in relation to same-currency liabilities.
Foreign exchange risk is also managed through the use of foreign currency forward-exchange contracts. These contracts are used to offset the potential earnings effects from mostly intercompany short-
term foreign currency assets and liabilities that arise from operations. Foreign currency swaps are used to offset the potential earnings effects from foreign currency debt. We also use foreign currency
forward-exchange contracts and foreign currency swaps to hedge the potential earnings effects from short-term and long-term foreign currency investments, third-party loans and intercompany loans.
In addition, under certain market conditions, we protect against possible declines in the reported net investments of our Japanese yen subsidiaries. In these cases, we use currency swaps or foreign
currency debt.
Our financial instrument holdings at year-end were analyzed to determine their sensitivity to foreign exchange rate changes. The fair values of these instruments were determined using various
methodologies. For additional details, see Notes to Consolidated Financial Statements— Note 7A. Financial Instruments: Selected Financial Assets and Liabilities. In this sensitivity analysis, we assumed
that the change in one currency’s rate relative to the U.S. dollar would not have an effect on other currencies’ rates relative to the U.S. dollar; all other factors were held constant. If the dollar were to
appreciate in 2011 against all other currencies by 10%, the expected adverse impact on net income related to our financial instruments would be immaterial. For additional details, see Notes to
Consolidated Financial Statements— Note 7E. Financial Instruments: Derivative Financial Instruments and Hedging Activities .
Interest Rate Risk
Our U.S. dollar interest-bearing investments, loans and borrowings are subject to interest rate risk. We also are subject to interest rate risk on euro debt, investments and currency swaps, U.K. debt and
currency swaps, Japanese yen short and long-term borrowings and currency swaps. We seek to invest, loan and borrow primarily on a short-term or variable-rate basis. From time to time, depending on
market conditions, we will fix interest rates either through entering into fixed-rate investments and borrowings or through the use of derivative financial instruments such as interest rate swaps. In light of
current market conditions, our current borrowings are primarily on a long-term, fixed-rate basis. We may change this practice as market conditions change.
•
Impact of acquisitions, divestitures, restructurings, product recalls and withdrawals and other unusual items, including (i) our ability to realize the projected benefits of our acquisition of King
Pharmaceuticals, Inc.; (ii) our ability to realize the projected benefits of our cost-reduction and productivity initiatives, including those related to our research and development organization; and (iii) the
impact of the strategic alternatives that we decide to pursue for our Animal Health and Nutrition businesses.
2011 Financial Report
47

Financial Review
Pfizer Inc. and Subsidiary Companies
Our financial instrument holdings at year-end were analyzed to determine their sensitivity to interest rate changes. The fair values of these instruments were determined using various methodologies. For
additional details, see Notes to Consolidated Financial Statements— Note 7A. Financial Instruments: Selected Financial Assets and Liabilities . In this sensitivity analysis, we used a one hundred basis
point parallel shift in the interest rate curve for all maturities and for all instruments; all other factors were held constant. If there were a one hundred basis point decrease in interest rates, the expected
adverse impact on net income related to our financial instruments would be immaterial.
Contingencies
Legal Matters
We and certain of our subsidiaries are subject to numerous contingencies arising in the ordinary course of business, such as patent litigation, product liability and other product-related litigation,
commercial litigation, environmental claims and proceedings, government investigations and guarantees and indemnifications (see Notes to Consolidated Financial Statements— Note 17. Commitments
and Contingencies).
Certain of these contingencies could result in losses, including damages, fines and/or civil penalties, and/or criminal charges, which could be substantial.
We believe that our claims and defenses in these matters are substantial, but litigation is inherently unpredictable and excessive verdicts do occur. We do not believe that any of these matters will have a
material adverse effect on our financial position. However, we could incur judgments, enter into settlements or revise our expectations regarding the outcome of certain matters, and such developments
could have a material adverse effect on our results of operations or cash flows in the period in which the amounts are paid and/or accrued.
We have accrued for losses that are both probable and reasonably estimable. Substantially all of these contingencies are subject to significant uncertainties and, therefore, determining the likelihood of a
loss and/or the measurement of any loss can be complex. Consequently, we are unable to estimate the range of reasonably possible loss in excess of amounts accrued. Our assessments are based on
estimates and assumptions that have been deemed reasonable by management, but the assessment process relies heavily on estimates and assumptions that may prove to be incomplete or inaccurate,
and unanticipated events and circumstances may occur that might cause us to change those estimates and assumptions.
Tax Matters
We account for income tax contingencies using a benefit recognition model. If our initial assessment fails to result in the recognition of a tax benefit, we regularly monitor our position and subsequently
recognize the tax benefit: (i) if there are changes in tax law, analogous case law or there is new information that sufficiently raise the likelihood of prevailing on the technical merits of the position to more
likely than not; (ii) if the statute of limitations expires; or (iii) if there is a completion of an audit resulting in a favorable settlement of that tax year with the appropriate agency. We regularly re-evaluate our
tax positions based on the results of audits of federal, state and foreign income tax filings, statute of limitations expirations, changes in tax law or receipt of new information that would either increase or
decrease the technical merits of a position relative to the “more-likely-than-not” standard.
Our assessments are based on estimates and assumptions that have been deemed reasonable by management, but our estimates of unrecognized tax benefits and potential tax benefits may not be
representative of actual outcomes, and variation from such estimates could materially affect our financial statements in the period of settlement or when the statutes of limitations expire, as we treat these
events as discrete items in the period of resolution. Finalizing audits with the relevant taxing authorities can include formal administrative and legal proceedings, and, as a result, it is difficult to estimate
the timing and range of possible changes related to our uncertain tax positions, and such changes could be significant.
48
2011 Financial Report

Management’s Report on Internal Control Over Financial Reporting
Management’s Report
We prepared and are responsible for the financial statements that appear in our 2011 Financial Report. These financial statements are in conformity with accounting principles generally accepted in the
United States of America and, therefore, include amounts based on informed judgments and estimates. We also accept responsibility for the preparation of other financial information that is included in
this document.
Report on Internal Control Over Financial Reporting
The management of the Company is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rules 13a-15(f) and 15d-15(f) under the Securities
Exchange Act of 1934. The Company’s internal control over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial
statements for external purposes in accordance with generally accepted accounting principles in the United States of America. The Company’s internal control over financial reporting includes those
policies and procedures that: (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the Company; (ii) provide
reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles and that receipts and
expenditures of the Company are being made only in accordance with authorizations of management and directors of the Company; and (iii) provide reasonable assurance regarding prevention or timely
detection of unauthorized acquisition, use or disposition of the Company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the
risk that controls may become inadequate because of changes in conditions or that the degree of compliance with the policies or procedures may deteriorate. Management assessed the effectiveness of
the Company’s internal control over financial reporting as of December 31, 2011. In making this assessment, management used the criteria set forth by the Committee of Sponsoring Organizations of the
Treadway Commission in Internal Control—Integrated Framework. Based on our assessment and those criteria, management believes that the Company maintained effective internal control over
financial reporting as of December 31, 2011.
The Company’s independent auditors have issued their auditors’ report on the Company’s internal control over financial reporting. That report appears in our 2011 Financial Report under the heading,
Report of Independent Registered Public Accounting Firm on Internal Control Over Financial Reporting.
February 28, 2012
Ian Read
Chairman and Chief Executive Officer
Frank A. D
’
Amelio
Loretta V. Cangialosi
Principal Financial Officer
Principal Accounting Officer
2011 Financial Report
49

Audit Committee Report
The Audit Committee reviews the Company’s financial reporting process on behalf of the Board of Directors. Management has the primary responsibility for the financial statements and the reporting
process, including the system of internal controls.
In this context, the Committee has met and held discussions with management and the independent registered public accounting firm regarding the fair and complete presentation of the Company’s
results and the assessment of the Company’s internal control over financial reporting. The Committee has discussed significant accounting policies applied by the Company in its financial statements, as
well as alternative treatments. Management has represented to the Committee that the Company
’s consolidated financial statements were prepared in accordance with accounting principles generally
accepted in the United States of America, and the Committee has reviewed and discussed the consolidated financial statements with management and the independent registered public accounting firm.
The Committee has discussed with the independent registered public accounting firm matters required to be discussed by Statement on Auditing Standards No. 114.
In addition, the Committee has reviewed and discussed with the independent registered public accounting firm the auditor’s independence from the Company and its management. As part of that review,
the Committee has received the written disclosures and the letter required by applicable requirements of the Public Company Accounting Oversight Board regarding the independent accountant’s
communications with the Audit Committee concerning independence, and the Committee has discussed the independent registered public accounting firm’s independence from the Company.
The Committee also has considered whether the independent registered public accounting firm’s provision of non-audit services to the Company is compatible with the auditor’s independence. The
Committee has concluded that the independent registered public accounting firm is independent from the Company and its management.
As part of its responsibilities for oversight of the Company’s Enterprise Risk Management process, the Committee has reviewed and discussed Company policies with respect to risk assessment and risk
management, including discussions of individual risk areas, as well as an annual summary of the overall process.
The Committee has discussed with the Company’s Internal Audit Department and independent registered public accounting firm the overall scope of and plans for their respective audits. The Committee
meets with the Chief Internal Auditor, Chief Compliance Officer and representatives of the independent registered public accounting firm, in regular and executive sessions to discuss the results of their
examinations, the evaluations of the Company’s internal controls, and the overall quality of the Company’s financial reporting and compliance programs.
In reliance on the reviews and discussions referred to above, the Committee has recommended to the Board of Directors, and the Board has approved, that the audited financial statements be included in
the Company’s Annual Report on Form 10-K for the year ended December 31, 2011, for filing with the SEC. The Committee has selected, and the Board of Directors has ratified, the selection of the
Company’s independent registered public accounting firm for 2012.
February 28, 2012
The Audit Committee Report does not constitute soliciting material, and shall not be deemed to be filed or incorporated by reference into any Company filing under the Securities Act of 1933, as
amended, or the Securities Exchange Act of 1934, as amended, except to the extent that the Company specifically incorporates the Audit Committee Report by reference therein.
W. Don Cornwell
Chair, Audit Committee
50
2011 Financial Report

Report of Independent Registered Public Accounting Firm on
The Consolidated Financial Statements
The Board of Directors and Shareholders of Pfizer Inc.:
We have audited the accompanying consolidated balance sheets of Pfizer Inc. and Subsidiary Companies as of December 31, 2011 and 2010, and the related consolidated statements of income,
shareholders’ equity, and cash flows for each of the years in the three-year period ended December 31, 2011. These consolidated financial statements are the responsibility of the Company’s
management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the
financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation.
We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of Pfizer Inc. and Subsidiary Companies as of December 31, 2011 and
2010, and the results of their operations and their cash flows for each of the years in the three-year period ended December 31, 2011, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the effectiveness of Pfizer Inc. and Subsidiary Companies’ internal control
over financial reporting as of December 31, 2011, based on criteria established in Internal Control — Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway
Commission (COSO), and our report dated February 28, 2012 expressed an unqualified opinion on the effective operation of the Company’s internal control over financial reporting.
February 28, 2012
KPMG LLP
New York, New York
2011 Financial Report
51

Report of Independent Registered Public Accounting Firm on
Internal Control Over Financial Reporting
The Board of Directors and Shareholders of Pfizer Inc.:
We have audited the internal control over financial reporting of Pfizer Inc. and Subsidiary Companies as of December 31, 2011, based on criteria established in Internal Control — Integrated Framework
issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). Pfizer Inc. and Subsidiary Companies’ management is responsible for maintaining effective internal control
over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Management’s Report on Internal Control Over Financial
Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over
financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audit also
included performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for
external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (i) pertain to the
maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (ii) provide reasonable assurance that transactions are
recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only
in accordance with authorizations of management and directors of the company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or
disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the
risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
In our opinion, Pfizer Inc. and Subsidiary Companies maintained, in all material respects, effective internal control over financial reporting as of December 31, 2011, based on criteria established in
Internal Control — Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO).
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the consolidated balance sheets of Pfizer Inc. and Subsidiary Companies as
of December 31, 2011 and 2010, and the related consolidated statements of income, shareholders’ equity, and cash flows for each of the years in the three-year period ended December 31, 2011, and
our report dated February 28, 2012 expressed an unqualified opinion on those consolidated financial statements.
February 28, 2012
KPMG LLP
New York, New York
52
2011 Financial Report

Consolidated Statements of Income
Pfizer Inc. and Subsidiary Companies
See Notes to Consolidated Financial Statements, which are an integral part of these statements.
YEAR ENDED DECEMBER 31,
(MILLIONS, EXCEPT PER COMMON SHARE DATA)
2011
2010
2009
Revenues
$
67,425
$
67,057
$
49,269
Costs and expenses:
Cost of sales
15,085
15,838
8,459
Selling, informational and administrative expenses
19,468
19,480
14,752
Research and development expenses
9,112
9,392
7,824
Amortization of intangible assets
5,585
5,403
2,877
Acquisition-related in-process research and development charges
—
125
68
Restructuring charges and certain acquisition
-
related costs
2,934
3,201
4,330
Other deductions
—
net
2,479
4,336
285
Income from continuing operations before provision for taxes on income
12,762
9,282
10,674
Provision for taxes on income
4,023
1,071
2,145
Income from continuing operations
8,739
8,211
8,529
Discontinued operations:
Income from discontinued operations
—
net of tax
8
88
97
Gain/(loss) on sale of discontinued operations—net of tax
1,304
(11
)
17
Discontinued operations—net of tax
1,312
77
114
Net income before allocation to noncontrolling interests
10,051
8,288
8,643
Less: Net income attributable to noncontrolling interests
42
31
8
Net income attributable to Pfizer Inc.
$
10,009
$
8,257
$
8,635
Earnings per common share
—
basic:
Income from continuing operations attributable to Pfizer Inc. common shareholders
$
1.11
$
1.02
$
1.22
Discontinued operations
—
net of tax
0.17
0.01
0.02
Net income attributable to Pfizer Inc. common shareholders
$
1.28
$
1.03
$
1.23
Earnings per common share
—
diluted:
Income from continuing operations attributable to Pfizer Inc. common shareholders
$
1.11
$
1.01
$
1.21
Discontinued operations—net of tax
0.17
0.01
0.02
Net income attributable to Pfizer Inc. common shareholders
$
1.27
$
1.02
$
1.23
Weighted-average shares—basic
7,817
8,036
7,007
Weighted
-
average shares
—
diluted
7,870
8,074
7,045
Exclusive of amortization of intangible assets, except as disclosed in Note 1K. Significant Accounting Policies: Amortization of Intangible Assets, Depreciation and Certain Long-Lived Assets .
EPS amounts may not add due to rounding.
2011 Financial Report
53
(a)
(a)
(a)
(b)
(b)
(a)
(b)

Consolidated Balance Sheets
Pfizer Inc. and Subsidiary Companies
See Notes to Consolidated Financial Statements, which are an integral part of these statements.
AS OF DECEMBER 31,
(MILLIONS, EXCEPT PREFERRED STOCK ISSUED AND PER COMMON SHARE DATA)
2011
2010
Assets
Cash and cash equivalents
$ 3,539
$ 1,735
Short-term investments
23,219
26,277
Accounts receivable, less allowance for doubtful accounts: 2011—$227; 2010—$208
13,608
13,380
Short
-
term loans
51
467
Inventories
7,769
8,275
Taxes and other current assets
9,441
9,440
Assets of discontinued operations and other assets held for sale
101
1,439
Total current assets
57,728
61,013
Long
-
term investments and loans
9,457
9,747
Property, plant and equipment, less accumulated depreciation
16,938
18,645
Goodwill
45,067
43,928
Identifiable intangible assets, less accumulated amortization
53,833
57,555
Taxes and other noncurrent assets
4,979
4,126
Total assets
$188,002
$195,014
Liabilities and Shareholders
’
Equity
Short
-
term borrowings, including current portion of long
-
term debt: 2011
—
$
6; 2010
—
$
3,502
$ 4,018
$ 5,603
Accounts payable
3,836
3,994
Dividends payable
1,796
1,601
Income taxes payable
1,013
951
Accrued compensation and related items
2,169
2,080
Other current liabilities
15,237
14,256
Liabilities of discontinued operations
—
151
Total current liabilities
28,069
28,636
Long
-
term debt
34,931
38,410
Pension benefit obligations
6,355
6,194
Postretirement benefit obligations
3,344
3,035
Noncurrent deferred tax liabilities
19,597
18,628
Other taxes payable
6,886
6,245
Other noncurrent liabilities
6,199
5,601
Total liabilities
105,381
106,749
Commitments and Contingencies
Preferred stock, without par value, at stated value; 27 shares authorized; issued:
2011
—
1,112; 2010
—
1,279
45
52
Common stock, $0.05 par value; 12,000 shares authorized; issued: 2011—8,902;
2010
—
8,876
445
444
Additional paid
-
in capital
71,423
70,760
Employee benefit trusts
(3
)
(7
)
Treasury stock, shares at cost; 2011—1,327; 2010—864
(31,801
)
(22,712
)
Retained earnings
46,210
42,716
Accumulated other comprehensive loss
(4,129
)
(3,440
)
Total Pfizer Inc. shareholders
’
equity
82,190
87,813
Equity attributable to noncontrolling interests
431
452
Total shareholders
’
equity
82,621
88,265
Total liabilities and shareholders
’
equity
$188,002
$195,014
54
2011 Financial Report
Consolidated Statements of Shareholders’ Equity
Pfizer Inc. and Subsidiary Companies
See Notes to Consolidated Financial Statements, which are an integral part of these statements.
PFIZER INC. SHAREHOLDERS
PREFERRED
STOCK
COMMON
STOCK
EMPLOYEE
BENEFIT TRUSTS
TREASURY
STOCK
(MILLIONS, EXCEPT
PREFERRED
SHARES)
SHARES
STATED
VALUE
SHARES
PAR
VALUE
ADD’
L
PAID-
IN
CAPITAL
SHARES
FAIR
VALUE
SHARES
COST
RETAINED
EARNINGS
ACCUM.
OTHER
COMP.
INC./
(LOSS)
SHARE-
HOLDERS’
EQUITY
NON-
CONTROLLING
INTERESTS
TOTAL
SHARE-
HOLDERS’
EQUITY
Balance, January 1, 2009
1,804
$ 73
8,863
$443
$70,283
(24
)
$
(425
)
(2,117
)
$(57,391
)
$49,142
$(4,569
)
$57,556
$184
$57,740
Comprehensive income:
Net income
8,635
8,635
8
8,643
Other comprehensive
income, net of tax
5,121
5,121
6
5,127
Total comprehensive
income
13,756
14
13,770
Acquisition of Wyeth
1,319
35,733
(12,430
)
23,303
330
23,633
Cash dividends
declared—
Common stock
(4,916
)
(4,916
)
(4,916
)
Preferred stock
(5
)
(5
)
(5
)
Noncontrolling
interests
(5
)
(5
)
Stock option transactions
130
—
9
139
139
Employee benefit trust
transactions
—
net
(61
)
7
111
50
50
Preferred stock
conversions
and redemptions
(293
)
(12
)
(1
)
—
3
(10
)
(10
)
Purchase of subsidiary
shares
from noncontrolling
interests
(66
)
(66
)
(102
)
(168
)
Other
6
—
212
(2
)
(28
)
(1
)
23
207
11
218
Balance, December 31,
2009
1,511
61
8,869
443
70,497
(19
)
(333
)
(799
)
(21,632
)
40,426
552
90,014
432
90,446
Comprehensive income:
Net income
8,257
8,257
31
8,288
Other comprehensive
income/(loss), net
of tax
(3,992
)
(3,992
)
5
(3,987
)
Total comprehensive
income
4,265
36
4,301
Cash dividends
declared
—
Common stock
(5,964
)
(5,964
)
(5,964
)
Preferred stock
(3
)
(3
)
(3
)
Noncontrolling
interests
(17
)
(17
)
Stock option transactions
161
1
14
175
175
Purchases of common
stock
(61
)
(1,000
)
(1,000
)
(1,000
)
Employee benefit trust
transactions—net
(19
)
16
292
273
273
Preferred stock
conversions
and redemptions
(232
)
(9
)
(1
)
—
2
(8
)
(8
)
Other
7
1
122
2
20
(4
)
(82
)
61
1
62
Balance, December 31,
2010
1,279
52
8,876
444
70,760
—
(
7
)
(864
)
(22,712
)
42,716
(3,440
)
87,813
452
88,265
Comprehensive income:
Net income
10,009
10,009
42
10,051
Other comprehensive
loss, net of tax
(689
)
(689
)
(45
)
(734
)
Total comprehensive
income
9,320
(3
)
9,317
Cash dividends
declared
—
Common stock
(6,512
)
(6,512
)
(6,512
)
Preferred stock
(3
)
(3
)
(3
)
Noncontrolling
interests
(19
)
(19
)
Stock option
transactions
312
312
312
Purchases of common
stock
(459
)
(9,000
)
(9,000
)
(9,000
)
Preferred stock
conversions
and redemptions
(167
)
(7
)
(2
)
—
1
(8
)
(8
)
Other
26
1
353
—
4
(4
)
(90
)
—
268
1
269
Balance, December 31,
2011
1,112
$ 45
8,902
$445
$71,423
—
$ (
3
)
(1,327
)
$(31,801
)
$46,210
$(4,129
)
$82,190
$431
$82,621
2011 Financial Report
55

Consolidated Statements of Cash Flows
Pfizer Inc. and Subsidiary Companies
See Notes to Consolidated Financial Statements, which are an integral part of these statements.
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Operating Activities
Net income before allocation to noncontrolling interests
$
10,051
$
8,288
$
8,643
Adjustments to reconcile net income before allocation to noncontrolling interests to
net cash provided by operating activities:
Depreciation and amortization
9,026
8,487
4,757
Share
-
based compensation expense
419
405
349
Asset write
-
offs and impairment charges
1,198
3,486
305
Acquisition-related in-process research and development charges
—
125
68
(Gain)/loss on disposals
15
(155
)
(670)
(Gain)/loss on sale of discontinued operations
(1,688
)
11
(15)
Deferred taxes from continuing operations
264
1,937
(9,590)
Deferred taxes on discontinued operations
190
16
8
Benefit plan contributions (in excess of)/less than expense
(1,775
)
(688
)
546
Other non
-
cash adjustments
(189
)
(19
)
199
Other changes in assets and liabilities, net of acquisitions and divestitures:
Accounts receivable
(66
)
(608
)
252
Inventories
1,084
2,917
1,631
Other assets
582
(906
)
(851)
Accounts payable and other liabilities
1,147
824
1,501
Other tax accounts, net
(18
)
(12,666
)
9,454
Net cash provided by operating activities
20,240
11,454
16,587
Investing Activities
Purchases of property, plant and equipment
(1,660
)
(1,513
)
(1,205)
Purchases of short
-
term investments
(18,428
)
(10,931
)
(35,331)
Proceeds from redemptions and sales of short-term investments
13,615
4,543
42,364
Net proceeds from redemptions and sales of short-term investments with original
maturities of 90 days or less
10,874
5,950
5,775
Purchases of long
-
term investments
(4,063
)
(3,920
)
(6,888)
Proceeds from redemptions and sales of long-term investments
2,147
4,381
6,504
Proceeds from redemptions of short
-
term loans
561
1,156
1,158
Issuances of short
-
term loans
(19
)
(151
)
(565)
Proceeds from redemptions of long
-
term loans
—
356
—
Issuances of long-term loans
(200
)
(208
)
(61)
Acquisitions, net of cash acquired
(3,282
)
(273
)
(43,123)
Proceeds from sale of business
2,376
—
—
Other investing activities
279
118
100
Net cash provided by/(used in) investing activities
2,200
(492
)
(31,272)
Financing Activities
Proceeds from short-term borrowings
12,810
6,400
31,159
Principal payments on short
-
term borrowings
(3,826
)
(9,249
)
(34,969)
Net proceeds/(payments) on short-term borrowings with original maturities of 90 days
or less
(7,540
)
(1,297
)
874
Proceeds from issuances of long-term debt
1
—
24,023
Principal payments on long
-
term debt
(6,986
)
(6
)
(967)
Purchases of common stock
(9,000
)
(1,000
)
—
Cash dividends paid
(6,234
)
(6,088
)
(5,548)
Other financing activities
168
66
(91)
Net cash provided by/(used in) financing activities
(20,607
)
(11,174
)
14,481
Effect of exchange-rate changes on cash and cash equivalents
(29
)
(31
)
60
Net increase/(decrease) in cash and cash equivalents
1,804
(243
)
(144)
Cash and cash equivalents at beginning of year
1,735
1,978
2,122
Cash and cash equivalents at end of year
$
3,539
$1,735
$
1,978
Supplemental Cash Flow Information
Non
-
cash transactions:
Acquisition of Wyeth, treasury stock issued
$
—
$
—
$
23,303
Cash paid during the period for:
Income taxes
$
2,938
$
11,775
$
2,300
Interest
2,085
2,155
935
56
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
A. Consolidation and Basis of Presentation
The consolidated financial statements include our parent company and all subsidiaries, and are prepared in accordance with accounting principles generally accepted in the United States of America
(U.S. GAAP). The decision whether or not to consolidate an entity requires consideration of majority voting interests, as well as effective economic or other control over the entity. Typically, we do not
seek control by means other than voting interests. For subsidiaries operating outside the United States (U.S.), the financial information is included as of and for the year ended November 30 for each year
presented. Substantially all unremitted earnings of international subsidiaries are free of legal and contractual restrictions. All significant transactions among our businesses have been eliminated.
As a result of our decision to sell our Capsugel business, we show the operating results of Capsugel as Discontinued operations—net of tax for all periods presented (see Note 2D. Acquisitions,
Divestitures, Collaborative Arrangements and Equity-Method Investments: Divestitures ). Also, due to a change in management approach, we now have different operating segments and have restated
our prior period segment information to conform to the current period presentation (see Note 18A. Segment, Geographic and Other Revenue Information: Segment Information ). In addition, we made
certain reclassification adjustments to conform prior-period amounts to the current period presentation, primarily related to the classification of certain receivables.
On January 31, 2011 (the acquisition date), we completed the tender offer for the outstanding shares of common stock of King Pharmaceuticals, Inc. (King) and acquired approximately 92.5% of the
outstanding shares for approximately $3.3 billion in cash. On February 28, 2011, we acquired the remaining outstanding shares of King for approximately $300 million in cash (for additional information,
see Note 2B. Acquisitions, Divestitures, Collaborative Arrangements and Equity-Method Investments: Acquisition of King Pharmaceuticals, Inc. ). Commencing from the acquisition date, our financial
statements include the assets, liabilities, operating results and cash flows of King. As a result, our consolidated financial statements for the year ended December 31, 2011 reflect approximately 11
months of King’s U.S. operations and approximately 10 months of King’s international operations.
On October 15, 2009, we completed our acquisition of Wyeth in a cash-and-stock transaction valued on that date at approximately $68.2 billion (for additional information, see Note 2A. Acquisitions,
Divestitures, Collaborative Arrangements and Equity-Method Investments: Acquisition of Wyeth ). Commencing from the acquisition date, our financial statements reflect the assets, liabilities, operating
results and cash flows of Wyeth. As a result, our consolidated financial statements for the year ended December 31, 2009 reflect approximately two-and-a-half months of Wyeth’s U.S. operations and
approximately one-and-a-half months of Wyeth’s international operations.
B. New Accounting Standards
The provisions of the following new accounting standards were adopted in 2011 and did not have a significant impact on our consolidated financial statements:
C. Estimates and Assumptions
In preparing the consolidated financial statements, we use certain estimates and assumptions that affect reported amounts and disclosures, including amounts recorded and disclosed in connection with
acquisitions. These estimates and underlying assumptions can impact all elements of our financial statements. For example, in the consolidated statements of income, estimates are used when
accounting for deductions from revenues (such as rebates, chargebacks, sales returns and sales allowances), determining cost of sales, allocating cost in the form of depreciation and amortization, and
estimating restructuring charges and the impact of contingencies. On the consolidated balance sheets, estimates are used in determining the valuation and recoverability of assets, such as accounts
receivables, investments, inventories, fixed assets and intangible assets (including acquired in-process research & development (IPR&D) assets and goodwill), and estimates are used in determining the
reported amounts of liabilities, such as taxes payable, benefit obligations, the impact of contingencies, rebates, chargebacks, sales returns and sales allowances, and restructuring reserves, all of which
also impact the consolidated statements of income.
Our estimates are often based on complex judgments, probabilities and assumptions that we believe to be reasonable but that can be inherently uncertain and unpredictable. If our estimates and
assumptions are not representative of actual outcomes, our results could be materially impacted.
As future events and their effects cannot be determined with precision, our estimates and assumptions may prove to be incomplete or inaccurate, or unanticipated events and circumstances may occur
that might cause us to change those estimates and assumptions. We are subject to risks and uncertainties that may cause actual results to differ from estimated amounts, such as changes in the
healthcare environment, competition, litigation, legislation and regulations. We regularly evaluate our estimates and assumptions using historical experience and expectations about the future. We adjust
our estimates and assumptions when facts and circumstances indicate the need for change. Those changes generally will be reflected in our financial statements on a prospective basis unless they are
required to be treated retrospectively under relevant accounting standards. It is possible that other professionals, applying reasonable judgment to the same facts and circumstances, could develop and
support a range of alternative estimated amounts.
1.
Significant Accounting Policies
• Guidelines that address the recognition and presentation of the fee paid by pharmaceutical companies beginning on January 1, 2011 to the U.S. Treasury as a result of U.S. Healthcare Legislation.
As a result of adopting this new standard, we are recording the fee ratably throughout the year in
Selling, informational and administrative expenses
.
• An amendment to the guidelines that address the accounting for multiple-deliverable arrangements to enable companies to account for certain products or services separately rather than as a
combined unit.
2011 Financial Report
57

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
D. Acquisitions
Our consolidated financial statements include the operations of an acquired business after the completion of the acquisition. We account for acquired businesses using the acquisition method of
accounting, which requires, among other things, that most assets acquired and liabilities assumed be recognized at their estimated fair values as of the acquisition date and that the fair value of acquired
IPR&D be recorded on the balance sheet. Transaction costs are expensed as incurred. Any excess of the consideration transferred over the assigned values of the net assets acquired is recorded as
goodwill. When we acquire net assets that do not constitute a business as defined in U.S. GAAP, no goodwill is recognized.
Contingent consideration, if any, is included as part of the acquisition cost and is recognized at fair value as of the acquisition date. Any liability resulting from contingent consideration is remeasured to
fair value at each reporting date until the contingency is resolved. These changes in fair value are recognized in earnings.
Amounts recorded for acquisitions can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For information about the risks
associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions .
E. Fair Value
We are often required to measure certain assets and liabilities at fair value, either upon initial measurement or for subsequent accounting or reporting. For example, we use fair value extensively in the
initial measurement of net assets acquired in a business combination and when accounting for and reporting on certain financial instruments. We estimate fair value using an exit price approach, which
requires, among other things, that we determine the price that would be received to sell an asset or paid to transfer a liability in an orderly market. The determination of an exit price is considered from the
perspective of market participants, considering the highest and best use of assets and, for liabilities, assuming that the risk of non-performance will be the same before and after the transfer.
When estimating fair value, depending on the nature and complexity of the asset or liability, we may use one or all of the following approaches:
These fair value methodologies depend on the following types of inputs:
A single estimate of fair value can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For information about the risks
associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions.
F. Foreign Currency Translation
For most of our international operations, local currencies have been determined to be the functional currencies. We translate functional currency assets and liabilities to their U.S. dollar equivalents at
rates in effect at the balance sheet date and record these translation adjustments in Other comprehensive income/(loss) . We translate functional currency statement of income amounts to their U.S.
dollar equivalents at average rates for the period. The effects of converting non-functional currency assets and liabilities into the functional currency are recorded in Other deductions—net .
For operations in highly inflationary economies, we translate monetary items at rates in effect at the balance sheet date, with translation adjustments recorded in Other deductions—net , and non-
monetary items at historical rates.
•
Income approach, which is based on the present value of a future stream of net cash flows.
•
Market approach, which is based on market prices and other information from market transactions involving identical or comparable assets or liabilities.
•
Cost approach, which is based on the cost to acquire or construct comparable assets less an allowance for functional and/or economic obsolescence.
•
Quoted prices for identical assets or liabilities in active markets (Level 1 inputs).
•
Quoted prices for similar assets or liabilities in active markets or quoted prices for identical or similar assets or liabilities in markets that are not active or are directly or indirectly observable (Level 2
inputs).
•
Unobservable inputs that reflect estimates and assumptions (Level 3 inputs).
58
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
G. Revenues
Revenue Recognition —We record revenues from product sales when the goods are shipped and title passes to the customer. At the time of sale, we also record estimates for a variety of sales
deductions, such as sales rebates, discounts and incentives, and product returns. When we cannot reasonably estimate the amount of future product returns and/or other sales deductions, we record
revenues when the risk of product return and/or additional sales deductions have been substantially eliminated. We record sales of certain of our vaccines to the U.S. government as part of the Pediatric
Vaccine Stockpile program; these rules require that for fixed commitments made by the U.S. government, we record revenues when risk of ownership for the completed product has been passed to the
U.S. government. There are no specific performance obligations associated with products sold under this program.
Deductions from Revenues–– As is typical in the biopharmaceutical industry, our gross product sales are subject to a variety of deductions that generally are estimated and recorded in the same period
that the revenues are recognized and primarily represent rebates and discounts to government agencies, wholesalers, distributors and managed care organizations with respect to our biopharmaceutical
products. These deductions represent estimates of the related obligations.
Specifically:
Our accruals for Medicaid rebates, Medicare rebates, performance-based contract rebates and chargebacks were $3.3 billion as of December 31, 2011, and $3.0 billion as of December 31, 2010, and
substantially all are included in Other current liabilities .
Amounts recorded for sales deductions can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For information about the
risks associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions.
Taxes collected from customers relating to product sales and remitted to governmental authorities are presented on a net basis; that is, they are excluded from Revenues .
Collaborative Arrangements— Payments to and from our collaboration partners are presented in our consolidated statements of income based on the nature of the arrangement (including its contractual
terms), the nature of the payments and applicable accounting guidance. Under co-promotion agreements, we record the amounts received from our partners as alliance revenues, a component of
Revenues, when our co-promotion partners are the principal in the transaction and we receive a share of their net sales or profits. Alliance revenues are recorded when our co-
promotion partners ship the
product and title passes to their customers. The related expenses for selling and marketing these products are included in Selling, informational and administrative expenses. In collaborative
arrangements where we manufacture a product for our partner, we record revenues when our partner sells the product and title passes to its customer. All royalty payments to collaboration partners are
recorded as part of Cost of sales .
•
In the U.S., we record provisions for pharmaceutical Medicaid, Medicare and contract rebates based upon our experience ratio of rebates paid and actual prescriptions written during prior quarters. We
apply the experience ratio to the respective period’s sales to determine the rebate accrual and related expense. This experience ratio is evaluated regularly to ensure that the historical trends are as
current as practicable. In addition, to account for the impacts of the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act (together, U.S.
Healthcare Legislation), we also consider the increase in minimum rebate and extension of Medicaid prescription drug rebates for drugs dispensed to enrollees. We estimate discounts on branded
prescription drug sales to Medicare Part D participants in the Medicare “coverage gap,” also known as the “doughnut hole,”
based on historical experience of beneficiary prescriptions and consideration
of the utilization that is expected to result from the new discount in the coverage gap. As appropriate, we will adjust these ratios to better match our current experience or our expected future
experience. For contract rebates, we also consider current contract terms, such as changes in formulary status and discount rates.
•
Outside the U.S., the majority of our pharmaceutical rebates, discounts and price reductions are contractual or legislatively mandated, and our estimates are based on actual invoiced sales within each
period; both of these elements help to reduce the risk of variations in the estimation process. Some European countries base their rebates on the government’s unbudgeted pharmaceutical spending,
and we use an estimated allocation factor (based on historical payments) and total revenues by country against our actual invoiced sales to project the expected level of reimbursement. We obtain
third
-
party information that helps us to monitor the adequacy of these accruals.
•
Provisions for pharmaceutical chargebacks (primarily reimbursements to wholesalers for honoring contracted prices to third parties) closely approximate actual as we settle these deductions generally
within two to five weeks of incurring the liability.
•
Provisions for pharmaceutical returns are based on a calculation for each market that incorporates the following, as appropriate: local returns policies and practices; returns as a percentage of sales; an
understanding of the reasons for past returns; estimated shelf life by product; an estimate of the amount of time between shipment and return or lag time; and any other factors that could impact the
estimate of future returns, such as loss of exclusivity, product recalls or a changing competitive environment. Generally, returned products are destroyed, and customers are refunded the sales price in
the form of a credit.
•
We record sales incentives as a reduction of revenues at the time the related revenues are recorded or when the incentive is offered, whichever is later. We estimate the cost of our sales incentives
based on our historical experience with similar incentives programs.
2011 Financial Report
59

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
H. Cost of Sales and Inventories
We carry inventories at the lower of cost or market. The cost of finished goods, work in process and raw materials is determined using average actual cost. We regularly review our inventories for
impairment and reserves are established when necessary.
I. Selling, Informational and Administrative Expenses
Selling, informational and administrative costs are expensed as incurred. Among other things, these expenses include the internal and external costs of marketing, advertising, shipping and handling,
information technology and legal defense.
Advertising expenses relating to production costs are expensed as incurred, and the costs of radio time, television time and space in publications are expensed when the related advertising occurs.
Advertising expenses totaled approximately $3.9 billion in 2011, $4.0 billion in 2010 and $2.9 billion in 2009.
J. Research and Development Expenses
Research and development (R&D) costs are expensed as incurred. These expenses include the costs of our proprietary R&D efforts, as well as costs incurred in connection with certain licensing
arrangements. Before a compound receives regulatory approval in a major market, we record upfront and milestone payments made by us to third parties under licensing arrangements as expense.
Upfront payments are recorded when incurred, and milestone payments are recorded when the specific milestone has been achieved. Once a compound receives regulatory approval in a major market,
we record any milestone payments in
Identifiable intangible assets, less accumulated amortization and, unless the assets are determined to have an indefinite life, we amortize them on a straight-line
basis over the remaining agreement term or the expected product life cycle, whichever is shorter.
K. Amortization of Intangible Assets, Depreciation and Certain Long-Lived Assets
Long-lived assets include:
Amortization expense related to finite-lived acquired intangible assets that contribute to our ability to sell, manufacture, research, market and distribute products, compounds and intellectual property are
included in Amortization of intangible assets as they benefit multiple business functions. Amortization expense related to intangible assets that are associated with a single function and depreciation of
property, plant and equipment are included in Cost of sales, Selling, informational and administrative expenses and Research and development expenses, as appropriate.
We review all of our long-lived assets for impairment indicators throughout the year and we perform detailed testing whenever impairment indicators are present. In addition, we perform detailed
impairment testing for goodwill and indefinite-lived assets at least annually. When necessary, we record charges for impairments. Specifically:
•
Goodwill
—Goodwill represents the excess of the consideration transferred for an acquired business over the assigned values of its net assets. Goodwill is not amortized.
• Identifiable intangible assets, less accumulated amortization —These acquired assets are recorded at our cost. Intangible assets with finite lives are amortized on a straight-line basis over their
estimated useful lives. Intangible assets with indefinite lives that are associated with marketed products are not amortized until a useful life can be determined. Intangible assets associated with IPR&D
projects are not amortized until approval is obtained in a major market, typically either the U.S. or the European Union (EU), or in a series of other countries, subject to certain specified conditions and
management judgment. The useful life of an amortizing asset generally is determined by identifying the period in which substantially all of the cash flows are expected to be generated.
• Property, plant and equipment, less accumulated depreciation —These assets are recorded at our cost and are increased by the cost of any significant improvements after purchase. Property, plant
and equipment assets, other than land and construction in progress, are depreciated on a straight-line basis over the estimated useful life of the individual assets. Depreciation begins when the asset is
ready for its intended use. For tax purposes, accelerated depreciation methods are used as allowed by tax laws.
•
For finite-lived intangible assets, such as Developed Technology Rights, and for other long-lived assets, such as property, plant and equipment, whenever impairment indicators are present, we
calculate the undiscounted value of the projected cash flows associated with the asset, or asset group, and compare this estimated amount to the carrying amount. If the carrying amount is found to be
greater, we record an impairment loss for the excess of book value over fair value. In addition, in all cases of an impairment review, we re-evaluate the remaining useful lives of the assets and modify
them, as appropriate.
•
For indefinite-lived intangible assets, such as Brands and IPR&D assets, annually and whenever impairment indicators are present, we determine the fair value of the asset and record an impairment
loss, if any, for the excess of book value over fair value. In addition, in all cases of an impairment review other than for IPR&D assets, we re-evaluate whether continuing to characterize the asset as
indefinite-lived is appropriate.
•
For goodwill, annually and whenever impairment indicators are present, we determine the fair value of each reporting unit and compare the fair value to its book value. If the carrying amount is found to
be greater, we then determine the implied fair value of goodwill by subtracting the fair value of all the identifiable net assets other than goodwill from the fair value of the reporting unit and record an
impairment loss for the excess, if any, of the book value of goodwill over the implied fair value.
60
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Impairment reviews can involve a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For information about the risks associated with
estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions.
L. Restructuring Charges and Certain Acquisition-Related Costs
We may incur restructuring charges in connection with acquisitions when we implement plans to restructure and integrate the acquired operations or in connection with our cost-reduction and productivity
initiatives. Included in Restructuring charges and certain acquisition-related costs are all restructuring charges and certain costs associated with acquiring and integrating an acquired business. (If the
restructuring action results in a change in the estimated useful life of an asset, that incremental impact is classified in Cost of sales, Selling, informational and administrative expenses and Research and
development expenses
, as appropriate). Termination costs are a significant component of our restructuring charges and are generally recorded when the actions are probable and estimable. Transaction
costs, such as banking, legal, accounting and other costs incurred in connection with an acquisition are expensed as incurred .
Amounts recorded for restructuring charges and other associated costs can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and
assumptions. For information about the risks associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions.
M. Cash Equivalents and Statement of Cash Flows
Cash equivalents include items almost as liquid as cash, such as certificates of deposit and time deposits with maturity periods of three months or less when purchased. If items meeting this definition are
part of a larger investment pool, we classify them as Short-term investments .
Cash flows associated with financial instruments designated as fair value or cash flow hedges may be included in operating, investing or financing activities, depending on the classification of the items
being hedged. Cash flows associated with financial instruments designated as net investment hedges are classified according to the nature of the hedge instrument. Cash flows associated with financial
instruments that do not qualify for hedge accounting treatment are classified according to their purpose and accounting nature.
N. Investments, Loans and Derivative Financial Instruments
Many, but not all, of our financial instruments are carried at fair value. For example, substantially all of our cash equivalents, short-term investments and long-term investments are classified as available-
for-sale securities and are carried at fair value, with changes in unrealized gains and losses, net of tax, reported in Other comprehensive/(loss) (see Note 6. Other Comprehensive Income/(Loss)) .
Derivative financial instruments are carried at fair value in various balance sheet categories (see Note 7A. Financial Instruments: Selected Financial Assets and Liabilities ), with changes in fair value
reported in current earnings or deferred for qualifying hedging relationships. Virtually all of our valuation measurements for investments, loans and derivative financial instruments are based on the use of
quoted prices for similar instruments in active markets or quoted prices for identical or similar instruments in markets that are not active or are directly or indirectly observable.
Realized gains or losses on sales of investments are determined by using the specific identification cost method.
Investments where we have significant influence over the financial and operating policies of the investee are accounted for under the equity method. Under the equity method, we record our share of the
investee’
s income and expenses in our income statements. The excess of the cost of the investment over our share in the equity of the investee on acquisition date is allocated to the identifiable assets of
the investee, with any remainder allocated to goodwill. Such investments are initially recorded at cost, which typically does not include amounts of contingent consideration.
We regularly evaluate all of our financial assets for impairment. For investments in debt and equity securities, when a decline in fair value, if any, is determined to be other-than-temporary, an impairment
charge is recorded, and a new cost basis in the investment is established. For loans, an impairment charge is recorded if it is probable that we will not be able to collect all amounts due according to the
loan agreement.
Impairment reviews can involve a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For information about the risks associated with
estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions.
O. Deferred Tax Assets and Liabilities and Income Tax Contingencies
Deferred tax assets and liabilities are recognized for the expected future tax consequences of differences between the financial reporting and tax bases of assets and liabilities using enacted tax rates and
laws. We provide a valuation allowance when we believe that our deferred tax assets are not recoverable based on an assessment of estimated future taxable income that incorporates ongoing, prudent
and feasible tax-planning strategies.
We account for income tax contingencies using a benefit recognition model. If we consider that a tax position is more likely than not to be sustained upon audit, based solely on the technical merits of the
position, we recognize the benefit. We measure the benefit by determining the amount that is greater than 50% likely of being realized upon settlement, presuming that the tax position is examined by the
appropriate taxing authority that has full knowledge of all relevant information.
2011 Financial Report
61

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Under the benefit recognition model, if our initial assessment fails to result in the recognition of a tax benefit, we regularly monitor our position and subsequently recognize the tax benefit: (i) if there are
changes in tax law, analogous case law or there is new information that sufficiently raise the likelihood of prevailing on the technical merits of the position to more likely than not; (ii) if the statute of
limitations expires; or (iii) if there is a completion of an audit resulting in a favorable settlement of that tax year with the appropriate agency. We regularly re-evaluate our tax positions based on the results
of audits of federal, state and foreign income tax filings, statute of limitations expirations, changes in tax law or receipt of new information that would either increase or decrease the technical merits of a
position relative to the “more-likely-than-not” standard. Liabilities associated with uncertain tax positions are classified as current only when we expect to pay cash within the next 12 months. Interest and
penalties, if any, are recorded in Provision for taxes on income and are classified on our consolidated balance sheet with the related tax liability.
Amounts recorded for valuation allowances and income tax contingencies can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and
assumptions. For information about the risks associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions.
P. Pension and Postretirement Benefit Plans
The majority of our employees worldwide are covered by defined benefit pension plans, defined contribution plans or both. In the U.S., we have both qualified and supplemental (non-qualified) defined
benefit plans, as well as other postretirement benefit plans, consisting primarily of healthcare and life insurance for retirees. Beginning on January 1, 2011, for employees hired in the U.S. and Puerto Rico
after December 31, 2010, we no longer offer a defined benefit plan and, instead, offer an enhanced benefit under our defined contribution plan. We recognize the overfunded or underfunded status of
each of our defined benefit plans as an asset or liability on our consolidated balance sheet. The obligations generally are measured at the actuarial present value of all benefits attributable to employee
service rendered, as provided by the applicable benefit formula. Our pension and other postretirement obligations may include assumptions such as long-term rate of return on plan assets, expected
employee turnover and participant mortality. For our pension plans, the obligation may also include assumptions as to future compensation levels. For our other postretirement benefit plans, the obligation
may include assumptions as to the expected cost of providing the healthcare and life insurance benefits, as well as the extent to which those costs are shared with the employee or others (such as
governmental programs). Plan assets are measured at fair value. Net periodic benefit costs are recognized, as required, into Cost of sales, Selling, informational and administrative expenses and
Research and development expenses , as appropriate.
Amounts recorded for pension and postretirement benefit plans can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions.
For information about the risks associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions.
Q. Legal and Environmental Contingencies
We and certain of our subsidiaries are subject to numerous contingencies arising in the ordinary course of business, such as patent litigation, product liability and other product-related litigation,
commercial litigation, environmental claims and proceedings, government investigations and guarantees and indemnifications. We record accruals for these contingencies to the extent that we conclude
that a loss is both probable and reasonably estimable. If some amount within a range of loss appears to be a better estimate than any other amount within the range, we accrue that amount. Alternatively,
when no amount within a range of loss appears to be a better estimate than any other amount, we accrue the lowest amount in the range. We record anticipated recoveries under existing insurance
contracts when recovery is assured.
Amounts recorded for contingencies can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For information about the
risks associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions.
R. Share-Based Payments
Our compensation programs can include share-based payments. All grants under share-based payment programs are accounted for at fair value and these fair values generally are amortized on a
straight-line basis over the vesting terms into Cost of sales, Selling, informational and administrative expenses, and Research and development expenses , as appropriate.
Amounts recorded for share-
based compensation can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For information
about the risks associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions .
A. Acquisition of Wyeth
Description of the Transaction
On October 15, 2009 (the acquisition date), we acquired all of the outstanding equity of Wyeth in a cash-and-stock transaction, valued at the acquisition date at approximately $68.2 billion, in which each
share of Wyeth common stock outstanding, with certain limited exceptions, was canceled and converted into the right to receive $33.00 in cash without interest and 0.985 of a share of Pfizer common
stock. The stock component was valued at $17.40 per share of Wyeth common stock based on the closing market price of Pfizer’s common stock on the acquisition date, resulting in a total merger
consideration value of $50.40 per share of Wyeth common stock.
2.
Acquisitions, Divestitures, Collaborative Arrangements and Equity-Method Investments
62
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Wyeth’s core business was the discovery, development, manufacture and sale of prescription pharmaceutical products, including vaccines, for humans. Other operations of Wyeth included the discovery,
development, manufacture and sale of consumer healthcare products (over-the-counter products), nutritionals and animal health products. Wyeth was a diversified healthcare company, with product
offerings in human, animal, and consumer health, including vaccines, biologics, small molecules and nutrition, across developed and emerging markets. The acquisition of Wyeth added to our pipeline of
biopharmaceutical development projects endeavoring to develop medicines to help patients in critical areas, including oncology, pain, inflammation, Alzheimer’s disease, psychoses and diabetes.
In connection with the regulatory approval process, we were required to divest certain animal health assets. Certain of these assets were sold in each of the periods presented. It is possible that additional
divestitures of animal health assets may be required based on ongoing regulatory reviews in other jurisdictions worldwide, but they are not expected to be significant to our business.
Fair Value of Consideration Transferred
Certain amounts may reflect rounding adjustments.
Recording of Assets Acquired and Liabilities Assumed
The transaction has been accounted for using the acquisition method of accounting which requires, among other things, that most assets acquired and liabilities assumed be recognized at their fair values
as of the acquisition date and that the fair value of acquired IPR&D be recorded on the balance sheet.
While most assets and liabilities were measured at fair value, a single estimate of fair value results from a complex series of judgments about future events and uncertainties and relies heavily on
estimates and assumptions. Our judgments used to determine the estimated fair value assigned to each class of assets acquired and liabilities assumed, as well as asset lives, can materially impact our
results of operations.
The consideration transferred to acquire Wyeth follows:
(IN MILLIONS, EXCEPT PER SHARE AMOUNTS)
CONVERSION
CALCULATION
FAIR VALUE
FORM OF
CONSIDERATION
Wyeth common stock outstanding as of the acquisition date
1,339.6
Multiplied by Pfizer’s stock price as of the acquisition date multiplied by the
exchange ratio of 0.985 ($17.66
x 0.985)
$17.40
$23,303
Pfizer common
stock
Wyeth common stock outstanding as of the acquisition date
1,339.6
Multiplied by cash consideration per common share outstanding
$33.00
44,208
Cash
Wyeth stock options canceled for a cash payment
405
Cash
Wyeth restricted stock/restricted stock units and other equity-based awards
canceled for a cash payment
320
Cash
Total fair value of consideration transferred
$68,236
The fair value of Pfizer’s common stock used in the conversion calculation represents the closing market price of Pfizer’s common stock on the acquisition date.
Approximately 1.3 billion shares of Pfizer common stock, previously held as Pfizer treasury stock, were issued to former Wyeth shareholders. The excess of the average cost of Pfizer treasury stock
issued over the fair value of the stock portion of the consideration transferred to acquire Wyeth was recorded as a reduction to
Retained earnings
.
Each Wyeth stock option, whether or not vested and exercisable on the acquisition date, was canceled for a cash payment equal to the excess of the per share value of the merger consideration
(calculated on the basis of the volume-weighted average of the per share price of Pfizer common stock on the New York Stock Exchange Transaction Reporting System for the five consecutive trading
days ending two days prior to the acquisition date) over the per share exercise price of the Wyeth stock option.
2011 Financial Report
63
(a) (a), (b)
(c)
(a)
(b)
(c)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
As of the acquisition date, the fair value of accounts receivable approximated book value acquired. The gross contractual amount receivable was $4.2 billion, of which $140 million was not expected to be
collected.
As part of the acquisition, we acquired liabilities for environmental, legal and tax matters, as well as guarantees and indemnifications that Wyeth incurred in the ordinary course of business. These matters
can include contingencies. Except as specifically excluded by the relevant accounting standard, contingencies are required to be measured at fair value as of the acquisition date, if the acquisition-date
fair value of the asset or liability arising from a contingency can be determined. If the acquisition-date fair value of the asset or liability cannot be determined, the asset or liability would be recognized at
the acquisition date if both of the following criteria were met: (i) it is probable that an asset existed or that a liability had been incurred at the acquisition date, and (ii) the amount of the asset or liability can
be reasonably estimated.
Goodwill is calculated as the excess of the consideration transferred over the net assets recognized and represents the future economic benefits arising from other assets acquired that could not be
individually identified and separately recognized. Specifically, the goodwill recorded as part of the acquisition of Wyeth includes the following:
The assets acquired and liabilities assumed from Wyeth follow:
(MILLIONS OF DOLLARS)
AMOUNTS
RECOGNIZED AS OF
ACQUISITION DATE
(FINAL)
Working capital, excluding inventories
$ 16,366
Inventories
7,971
Property, plant and equipment
9,838
Identifiable intangible assets, excluding in
-
process research and development
36,062
In
-
process research and development
13,822
Other noncurrent assets
2,394
Long
-
term debt
(11,187
)
Benefit obligations
(3,175
)
Net tax accounts
(23,738
)
Other noncurrent liabilities
(1,908
)
Total identifiable net assets
46,445
Goodwill
22,117
Net assets acquired
68,562
Less: Amounts attributable to noncontrolling interests
(326
)
Total consideration transferred
$ 68,236
Includes cash and cash equivalents, short-term investments, accounts receivable, other current assets, assets held for sale, accounts payable and other current liabilities.
As of the acquisition date, included in Taxes and other current assets ($1.2 billion), Taxes and other noncurrent assets ($2.8 billion), Income taxes payable ($500 million), Other current liabilities
($11.1
billion),
Noncurrent deferred tax liabilities
($14.0 billion) and
Other taxes payable
($2.1 billion, including accrued interest of $300 million).
Goodwill recognized as of the acquisition date totaled $19.3 billion for our three biopharmaceutical operating segments and $2.8 billion for our Animal Health and Consumer Healthcare and our Nutrition
operating segments. (Since the acquisition of Wyeth, we have revised our operating segments. See
Note 18A. Segment, Geographic and Other Revenue Information: Segment Information.
)
• Environmental Matters ––In the ordinary course of business, Wyeth incurred liabilities for environmental matters such as remediation work, asset retirement obligations and environmental guarantees
and indemnifications. Virtually all liabilities for environmental matters, including contingencies, were measured at fair value and approximated $570 million as of the acquisition date.
• Legal Matters ––Wyeth was involved in various legal proceedings, including product liability, patent, commercial, environmental, antitrust matters and government investigations, of a nature
considered normal to its business (see Note 17. Commitments and Contingencies ). Due to the uncertainty of the variables and assumptions involved in assessing the possible outcomes of events
related to these items, an estimate of fair value was not determinable. As such, these contingencies were measured under the same “probable and estimable” standard previously used by Wyeth.
Liabilities for legal contingencies approximated $1.3 billion as of the acquisition date, which included the recording of additional adjustments of approximately $260 million for legal matters that we
intended to resolve in a manner different from what Wyeth had planned or intended.
• Tax Matters ––In the ordinary course of business, Wyeth incurred liabilities for income taxes . Income taxes are exceptions to both the recognition and fair value measurement principles associated
with the accounting for business combinations. Reserves for income tax contingencies continue to be measured under the benefit recognition model as previously used by Wyeth (see
Note 1O.
Significant Accounting Policies: Deferred Tax Assets and Liabilities and Income Tax Contingencies ). Net liabilities for income taxes approximated $23.7 billion as of the acquisition date, which
included $1.8 billion for uncertain tax positions (not including $300 million of accrued interest). The net tax liability included the recording of additional adjustments of approximately $14.4 billion for the
tax impact of fair value adjustments and $10.5 billion for income tax matters that we intended to resolve in a manner different from what Wyeth had planned or intended. For example, because we
planned to repatriate certain overseas funds, we provided deferred taxes on Wyeth’s unremitted earnings, as well as on certain book/tax basis differentials related to investments in certain foreign
subsidiaries for which no taxes had been previously provided by Wyeth as it was Wyeth
’
s intention to permanently reinvest those earnings and investments.
64
2011 Financial Report
(a)
(b)
(c)
(a)
(b)
(c)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Goodwill is not amortized and is not deductible for tax purposes (see Note 10A. Goodwill and Other Intangible Assets: Goodwill for additional information).
Actual and Pro Forma Impact of Acquisition
The unaudited pro forma consolidated results were prepared using the acquisition method of accounting and are based on the historical financial information of Pfizer and Wyeth, reflecting Pfizer and
Wyeth results of operations for a 12 month period. The historical financial information has been adjusted to give effect to the pro forma events that are: (i) directly attributable to the acquisition, (ii) factually
supportable and (iii) expected to have a continuing impact on the combined results. The unaudited pro forma consolidated results are not necessarily indicative of what our consolidated results of
operations actually would have been had we completed the acquisition on January 1, 2009. In addition, the unaudited pro forma consolidated results do not purport to project the future results of
operations of the combined company nor do they reflect the expected realization of any cost savings associated with the acquisition. The unaudited pro forma consolidated results reflect primarily the
following pro forma pre-tax adjustments:
In addition, all of the above adjustments were adjusted for the applicable tax impact. The taxes associated with the fair value adjustments for acquired intangible assets, property, plant and equipment and
legacy Wyeth debt, as well as the elimination of the impact of the fair value step-up of acquired inventory reflect the statutory tax rates in the various jurisdictions where the fair value adjustments
occurred. The taxes associated with incremental debt to partially finance the acquisition reflect a 38.3% tax rate since the debt is an obligation of a U.S. entity and is taxed at the combined effective U.S.
federal statutory and state rate. The taxes associated with the elimination of the costs directly attributable to the acquisition reflect a 28.4% effective tax rate since the costs were incurred in the U.S. and
were either taxed at the combined effective U.S. federal statutory and state rate or not deductible for tax purposes depending on the type of expenditure.
•
the expected synergies and other benefits that we believed would result from combining the operations of Wyeth with the operations of Pfizer;
•
any intangible assets that did not qualify for separate recognition, as well as future, as yet unidentified projects and products; and
•
the value of the going
-
concern element of Wyeth
’
s existing businesses (the higher rate of return on the assembled collection of net assets versus if Pfizer had acquired all of the net assets separately).
The revenue and earnings of Wyeth included in Pfizer
’
s consolidated statements of income follow:
(MILLIONS OF DOLLARS)
WYETH’
S OPERATIONS
INCLUDED IN PFIZER’
s 2009
RESULTS
Revenues
$ 3,303
Loss from continuing operations attributable to Pfizer Inc. common shareholders
(2,191
)
The results of Wyeth are included from the acquisition date of October 15, 2009.
Includes purchase accounting adjustments related to the fair value adjustments for acquisition-date inventory that has been sold ($904 million pre-tax), amortization of identifiable intangible assets
acquired from Wyeth ($512 million pre-tax), and restructuring charges and additional depreciation—asset restructuring ($2.1 billion pre-tax).
Supplemental pro forma information follows:
UNAUDITED PRO FORMA
CONSOLIDATED RESULTS
(MILLIONS OF DOLLARS, EXCEPT PER SHARE DATA)
YEAR ENDED DECEMBER 31,2009
Revenues
$67,859
Income from continuing operations attributable to Pfizer Inc. common shareholders
11,436
Diluted earnings per common share attributable to Pfizer Inc. common shareholders
1.41
The pro forma information assumes that the acquisition of Wyeth had occurred on January 1, 2009 for the year ended December 31, 2009.
•
Elimination of Wyeth
’
s historical intangible asset amortization expense (approximately $88 million).
•
Additional amortization expense (approximately $2.4 billion) related to the fair value of identifiable intangible assets acquired.
•
Additional depreciation expense (approximately $200 million) related to the fair value adjustment to property, plant and equipment acquired.
•
Additional interest expense (approximately $316 million) associated with the incremental debt we issued in 2009 to partially finance the acquisition and a reduction of interest income (approximately
$320 million) associated with short-term investments under the assumption that a portion of these investments would have been used to partially fund the acquisition. In addition, a reduction in interest
expense (approximately $129 million) related to the fair value adjustment of Wyeth debt.
•
Elimination of $904 million related to the fair value adjustments to acquisition-date inventory that has been sold, which is considered non-recurring. There is no long-term continuing impact of the fair
value adjustments to acquisition
-
date inventory, and, as such, the impact of those adjustments is not reflected in the unaudited pro forma operating results.
•
Elimination of $834 million of costs which are directly attributable to the acquisition, and which do not have a continuing impact on the combined company’s operating results. Included in these costs
are advisory, legal and regulatory costs incurred by both legacy Pfizer and legacy Wyeth and costs related to a bridge term loan credit agreement with certain financial institutions that has been
terminated.
2011 Financial Report
65
(a)
(b)
(a)
(b)
(a)
(a)
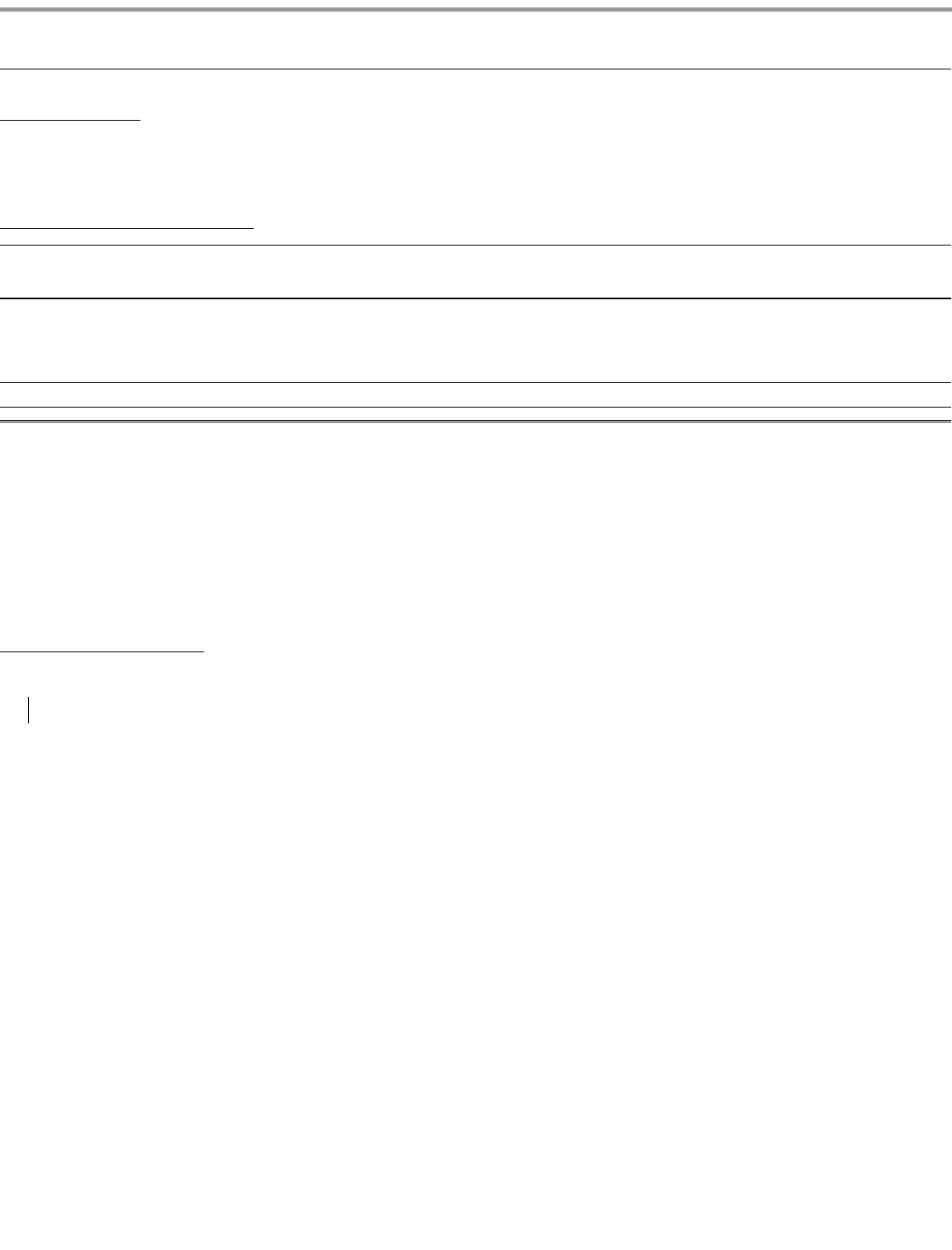
Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
B. Acquisition of King Pharmaceuticals, Inc.
Description of the Transaction
On January 31, 2011 (the acquisition date), we completed a tender offer for the outstanding shares of common stock of King at a purchase price of $14.25 per share in cash and acquired approximately
92.5% of the outstanding shares. On February 28, 2011, we acquired all of the remaining shares of King for $14.25 per share in cash. As a result, the total fair value of consideration transferred for King
was approximately $3.6 billion in cash ($3.2 billion, net of cash acquired).
King’s principal businesses consist of a prescription pharmaceutical business focused on delivering new formulations of pain treatments designed to discourage common methods of misuse and abuse;
the Meridian auto-injector business for emergency drug delivery, which develops and manufactures the EpiPen; an established products portfolio; and an animal health business that offers a variety of
feed-additive products for a wide range of species.
Recording of Assets Acquired and Liabilities Assumed
As of the acquisition date, the fair value of accounts receivable approximated book value acquired. The gross contractual amount receivable was $200 million, virtually all of which was expected to be
collected.
Goodwill is calculated as the excess of the consideration transferred over the net assets recognized and represents the future economic benefits arising from other assets acquired that could not be
individually identified and separately recognized. Specifically, the goodwill recorded as part of the acquisition of King includes the following:
Goodwill is not amortized and is not deductible for tax purposes (see Note 10A. Goodwill and Other Intangible Assets: Goodwill for additional information).
The assets and liabilities arising from contingencies recognized as of the acquisition date are not significant to Pfizer’s consolidated financial statements.
Actual and Pro Forma Impact of Acquisition
Revenues from King are included in Pfizer’s consolidated statements of income from the acquisition date, January 31, 2011, through Pfizer’s domestic and international year-ends and were $1.3 billion in
2011. We are no longer able to provide the results of operations attributable to King as those operations have now been substantially integrated.
The assets acquired and liabilities assumed from King follow:
(MILLIONS OF DOLLARS)
AMOUNTS
RECOGNIZED AS OF
ACQUISITION DATE
(FINAL)
Working capital, excluding inventories
$ 155
Inventories
340
Property, plant and equipment
412
Identifiable intangible assets, excluding in
-
process research and development
1,806
In
-
process research and development
303
Net tax accounts
(328
)
All other long
-
term assets and liabilities, net
102
Total identifiable net assets
2,790
Goodwill
765
Net assets acquired/total consideration transferred
$ 3,555
Measurement period adjustments were not significant and did not have a significant impact on our earnings, balance sheets or cash flows in any interim period in 2011 and, therefore, we did not
retrospectively adjust our interim financial statements.
Goodwill recorded as of the acquisition date totaled $720 million for our three biopharmaceutical operating segments and $45 million for our Animal Health and Consumer Healthcare operating
segment. (Since the acquisition of King, we have revised our operating segments. See
Note 18A. Segment, Geographic and Other Revenue Information: Segment Information.)
•
the expected synergies and other benefits that we believed would result from combining the operations of King with the operations of Pfizer;
•
any intangible assets that did not qualify for separate recognition, as well as future, yet unidentified projects and products; and
•
the value of the going
-
concern element of King
’
s existing businesses (the higher rate of return on the assembled collection of net assets versus if Pfizer had acquired all of the net assets separately).
66
2011 Financial Report
(a)
(b)
(a)
(b)
Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The unaudited pro forma consolidated results do not purport to project the future results of operations of the combined company nor do they reflect the expected realization of any cost savings associated
with the acquisition. The unaudited pro forma consolidated results reflect the historical financial information of Pfizer and King, adjusted for the following pre
-tax amounts:
C. Other Acquisitions
Excaliard
On November 30, 2011, we completed our acquisition of Excaliard Pharmaceuticals, Inc. (Excaliard), a privately-owned biopharmaceutical company focused on developing novel drugs for the treatment
of skin fibrosis, more commonly referred to as skin scarring. Excaliard ‘s lead compound, EXC-001, is an antisense oligonucleotide designed to interrupt the process of fibrosis by inhibiting expression of
connective tissue growth factor (CTGF), and has produced positive clinical results in reducing scar severity in certain Phase 2 trials. The total consideration for the acquisition was approximately $174
million, which consisted of an upfront payment to Excaliard’s shareholders of about $86 million and contingent consideration with an estimated acquisition-date fair value of about $88 million. The
contingent consideration consists of up to $230 million in additional payments that are contingent upon attainment of future regulatory and revenue milestones. In connection with this acquisition, we
recorded approximately $257 million in Identifiable intangible assets — in-process research and development .
The fair value of the contingent consideration at the acquisition date was estimated by utilizing a probability-weighted income approach. We started with an estimate of the timing of the potential cash
payments by year, based on our expectation as to when the future regulatory and commercial milestones might be achieved, adjusted the payments to reflect the likelihood of payment, and then
discounted each of those projected payments to arrive at a present value amount. Subsequent to the acquisition date, we remeasure the contingent consideration liability at current fair value at every
reporting period with changes recorded in Other deductions—net .
Icagen
On September 20, 2011, we completed our cash tender offer for the outstanding shares of Icagen, Inc. (Icagen), resulting in approximately 70% ownership of the outstanding shares of Icagen, a
biopharmaceutical company focused on discovery, development and commercialization of novel orally-administered small molecule drugs that modulate ion channel targets. On October 27, 2011, we
acquired all of the remaining shares of Icagen. In connection with this acquisition, we recorded approximately $19 million in Identifiable intangible assets .
FoldRx Pharmaceuticals, Inc.
On October 6, 2010, we completed our acquisition of FoldRx Pharmaceuticals, Inc. (FoldRx), a privately-held drug discovery and clinical development company, whose portfolio includes clinical and
preclinical programs for investigational compounds to treat diseases caused by protein misfolding. The total consideration for the acquisition was approximately $400 million, which consisted of an upfront
payment to FoldRx’s shareholders of about $200 million and contingent consideration with an estimated acquisition-date fair value of about $200 million. The contingent consideration consists of up to
$455 million in additional payments that are contingent upon the attainment of future regulatory and commercial milestones. In connection with this acquisition, we recorded approximately $500 million in
Identifiable intangible assets — in-process research and development and approximately $60 million in Goodwill.
The fair value of the contingent consideration at the acquisition date was estimated by utilizing a probability-weighted income approach. We started with an estimate of the probability weighted potential
cash payments by year, based on our expectation as to when the future regulatory and commercial milestones might be achieved, and then we discounted each of those projected payments to arrive at a
present value amount. Subsequent to the acquisition date, we remeasure the contingent consideration liability at current fair value at every reporting period with changes recorded in Other deductions—
net .
Supplemental pro forma information follows:
UNAUDITED PRO FORMA
CONSOLIDATED RESULTS
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS, EXCEPT PER SHARE DATA)
2011
2010
Revenues
$67,534
$68,432
Net income attributable to Pfizer Inc. common shareholders
10,228
8,013
Diluted earnings per share attributable to Pfizer Inc. common shareholders
1.30
0.99
The pro forma information for December 31, 2011 and 2010 assumes that the acquisition of King occurred on January 1, 2010.
•
Elimination of King
’
s historical intangible asset amortization expense (approximately $6 million in 2011 and $116 million in 2010).
•
Additional amortization expense (approximately $15 million in 2011 and $190 million in 2010) related to the fair value of identifiable intangible assets acquired.
•
Additional depreciation expense (approximately $3 million in 2011 and $35 million in 2010) related to the fair value adjustment to property, plant and equipment acquired.
•
Adjustment related to the fair value adjustments to acquisition
-
date inventory estimated to have been sold (elimination of $160 million charge in 2011 and addition of $160 million charge in 2010).
•
Adjustment for acquisition-
related costs directly attributable to the acquisition (elimination of $224 million of charges in 2011 and addition of $224 million of charges in 2010, reflecting charges incurred
by both King and Pfizer).
2011 Financial Report
67
(a)
(a)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
FoldRx’s lead product candidate, Vyndaqel (tafamidis meglumine), a first-in-class oral therapy for the treatment of transthyretin familial amyloid polyneuropathy (TTR-FAP), a progressively fatal genetic
neurodegenerative disease, for which liver transplant is the only treatment option currently available, was approved in the EU in November 2011 and our new drug application was accepted for review in
the U.S. in February 2012. As a result of the November EU approval and changes in the commercial forecasts, we increased our contingent consideration liability by approximately $85 million in 2011,
with the changes recorded in
Other deductions – net .
D. Divestitures
On August 1, 2011, we completed the sale of our Capsugel business for approximately $2.4 billion in cash. In connection with the decision to sell, the operating results associated with the Capsugel
business are classified as Discontinued operations—net of tax in the consolidated statements of income for all periods presented, and the assets and liabilities associated with this business are classified
into Assets of discontinued operations and other assets held for sale and Liabilities of discontinued operations , as appropriate, in the consolidated balance sheets for all applicable periods presented.
The components of Discontinued operations—net of tax , substantially all of which relate to our Capsugel business, follow:
The net cash flows of our discontinued operations for each of the categories of operating, investing and financing activities were not significant for any period presented.
E. Collaborative Arrangements
In the normal course of business, we enter into collaborative arrangements with respect to in-line medicines, as well as medicines in development that require completion of research and regulatory
approval. Collaborative arrangements are contractual agreements with third parties that involve a joint operating activity, typically a research and/or commercialization effort, where both we and our
partner are active participants in the activity and are exposed to the significant risks and rewards of the activity. Our rights and obligations under our collaborative arrangements vary. For example, we
have agreements to co-promote pharmaceutical products discovered by us or other companies, and we have agreements where we partner to co-develop and/or participate together in commercializing,
marketing, promoting, manufacturing and/or distributing a drug product.
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Revenues
$ 507
$ 752
$ 740
Pre
-
tax (loss)/income from discontinued operations
$ 31
$ 140
$ 148
Provision for taxes on income
23
52
51
Income from discontinued operations
—
net of tax
8
88
97
Pre
-
tax gain/(loss) on sale of discontinued operations
1,688
(11)
15
Provision/(benefit) for taxes on income
384
—
(
2)
Gain/(loss) on sale of discontinued operations
—
net of tax
1,304
(11)
17
Discontinued operations
—
net of tax
$1,312
$ 77
$ 114
Deferred tax amounts are not significant for 2011.
Includes a deferred tax expense of $190 million for 2011.
Includes deferred tax expense of $16 million and $8 million, respectively for 2010 and 2009.
Deferred tax amounts are not significant for 2010 and 2009.
The components of
Assets of discontinued operations and other assets held for sale
and
Liabilities of discontinued operations
, most of which relate to our Capsugel business, follow:
(MILLIONS OF DOLLARS)
YEAR ENDED
DECEMBER 31,
2010
Accounts receivable
$ 186
Inventories
130
Taxes and other current assets
47
Property, plant and equipment
1,009
Goodwill
19
Identifiable intangible assets
3
Taxes and other noncurrent assets
45
Assets of discontinued operations and other assets held for sale
$1,439
Current liabilities
$ 124
Other liabilities
27
Liabilities of discontinued operations
$ 151
68
2011 Financial Report
(a), (c)
(b), (d)
(a)
(b)
(c)
(d)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The amounts disclosed in the above table do not include transactions with third parties other than our collaboration partners, or other costs associated with the products under the collaborative
arrangements. In addition, during 2011 we paid $61 million in milestones to a collaboration partner. These payments were recorded in Identifiable intangible assets-developed technology rights .
F. Equity-Method Investments
Investment in Laboratório Teuto Brasileiro
On November 8, 2010, we consummated our partnership to develop and commercialize generic medicines with Laboratório Teuto Brasileiro S.A. (Teuto) a leading generics company in Brazil. As part of
the transaction, we acquired a 40 percent equity stake in Teuto, and entered into a series of commercial agreements. The partnership is enhancing our position in Brazil, a key emerging market, by
providing access to Teuto’s portfolio of products. Through this partnership, we have access to significant distribution networks in rural and suburban areas in Brazil and the opportunity to register and
commercialize Teuto’s products in various markets outside of Brazil. Under the terms of our purchase agreement with Teuto, we made an upfront payment at the closing of approximately $230 million. In
addition, Teuto will be eligible to receive a performance-based milestone payment from us in 2012 of up to approximately $200 million. We have an option to acquire the remaining 60 percent of Teuto’s
shares beginning in 2014, and Teuto’s shareholders have an option to sell their 60 percent stake to us beginning in 2015. The portion of the total arrangement consideration that was allocated to the net
call/put option, based on relative fair values of the 40% equity investment and net option respectively, is being accounted for at cost and will be evaluated for impairment on an ongoing basis.
We are accounting for our interest in Teuto as an equity method investment due to the significant influence we have over the operations of Teuto through our board representation, minority veto rights and
40% voting interest. Our investment in Teuto is reported as a private equity investment in Long-term investments and loans . Our share of Teuto’s income and expenses is recorded in Other deductions
— net .
Formation of ViiV
On October 30, 2009, we and GlaxoSmithKline plc (GSK) created a new company, ViiV Healthcare Limited (ViiV), which is focused solely on research, development and commercialization of human
immunodeficiency virus (HIV) medicines. We and GSK have contributed certain existing HIV-related products, pipeline assets and research assets to ViiV and will perform R&D and manufacturing
services. The R&D Services Agreement provides that we will perform R&D services for pipeline and marketed products contributed by us and that such services be billed at our internal cost plus a profit
margin. After two and a half years, either party may terminate this agreement with six months’ notice. The Contract Manufacturing Agreement provides that we will manufacture and supply products to
ViiV for four years at a price that incorporates a profit margin. Prior to the agreed termination date, ViiV may terminate this agreement at any time with approximately one-year’s notice. Further, Pfizer and
GSK have entered into a 3-year Research Alliance Agreement with ViiV under which each party, at its sole discretion, may conduct research programs in order to achieve Proof of Concept for an HIV
Therapy Compound. ViiV will have a right of first negotiation on compounds that reach Proof of Concept.
We recognized a gain of approximately $482 million in connection with the formation, which was recorded in Other deductions — net in the fourth quarter of 2009. Since we held a 15% equity interest in
ViiV, we had an indirect retained interest in the contributed assets; as such, 15% of the gain, or $72 million, is the portion of the gain associated with that indirect retained interest. In valuing our
investment in ViiV (which includes the indirect retained interest in the contributed assets), we used discounted cash flow techniques, utilizing an 11% discount rate and a terminal year growth factor of 3%.
We currently hold a 15% equity interest and GSK holds an 85% equity interest in ViiV. The equity interests will be adjusted in the event that specified sales and regulatory milestones are achieved. Our
equity interest in ViiV could vary from 9% to 30.5%, and GSK’s equity interest could vary from 69.5% to 91%, depending upon the milestones achieved with respect to the original assets contributed to
ViiV by us and by GSK. Each company also may be entitled to preferential dividend payments to the extent that specific sales thresholds are met in respect of the marketed products and pipeline assets
originally contributed.
The amounts and classification of payments (income/(expense)) between us and our collaboration partners follow:
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Revenues
—Revenues
$1,029
$ 710
$ 676
Revenue
s
—
Alliance revenues
3,630
4,084
2,925
Total revenues from collaborative arrangements
4,659
4,794
3,601
Cost of sales
(420)
(124
)
(175)
Selling, informational and administrative expenses
(237)
(131
)
10
Research and development expenses
(299)
(316
)
(361)
Other deductions
—
net
34
37
37
Represents sales to our partners of products manufactured by us.
Substantially all relate to amounts earned from our partners under co-promotion agreements.
Primarily relates to royalties earned by our partners and cost of sales associated with inventory purchased from our partners.
Represents net reimbursements from our partners/(to our partners) for selling, informational and administrative expenses incurred.
Primarily related to net reimbursements, as well as upfront payments and milestone payments earned by our partners. The upfront and milestone payments were as follows: $210 million in 2011, $147
million in 2010 and $150 million in 2009.
2011 Financial Report
69
(a)
(b)
(c)
(d)
(e)
(a)
(b)
(c)
(d)
(e)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
We are accounting for our interest in ViiV as an equity method investment due to the significant influence we have over the operations of ViiV through our board representation and minority veto rights.
Our investment in ViiV is reported as a private equity investment in Long-term investments and loans . Our share of ViiV’s income and expenses is recorded in Other deductions—net .
We incur significant costs in connection with acquiring businesses and restructuring and integrating acquired businesses and in connection with our global cost-reduction and productivity initiatives. For
example:
All of our businesses and functions can be impacted by these actions, including sales and marketing, manufacturing and research and development, as well as functions such as information technology,
shared services and corporate operations. In early February 2011, we announced a new research and productivity initiative to accelerate our strategies to improve innovation and overall productivity in
R&D by prioritizing areas with the greatest scientific and commercial promise, utilizing appropriate risk/return profiles and focusing on areas with the highest potential to deliver value in the near term and
over time.
The components of costs incurred in connection with our acquisitions and our cost-reduction/productivity initiatives follow:
The restructuring charges in 2011 are associated with the following:
3.
Restructuring Charges and Other Costs Associated with Acquisitions and Cost-Reduction/Productivity Initiatives
•
for our cost-reduction and productivity initiatives, we typically incur costs and charges associated with site closings and other facility rationalization actions, workforce reductions and the expansion of
shared services, including the development of global systems; and
•
for our acquisition activity, we typically incur costs that can include transaction costs, integration costs (such as expenditures for consulting and the integration of systems and processes) and
restructuring charges, related to employees, assets and activities that will not continue in the combined company.
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Transaction costs
$
30
$
22
$
768
Integration costs
730
1,004
569
Restructuring charges
Employee termination costs
1,791
1,114
2,564
Asset impairments
256
870
159
Other
127
191
270
Restructuring charges and certain acquisition
-
related costs
2,934
3,201
4,330
Additional depreciation—asset restructuring, recorded in our consolidated
statements of income as follows :
Cost of Sales
557
527
133
Selling, informational and administrative expenses
75
227
53
Research and development expenses
607
34
55
Total additional depreciation—asset restructuring
1,239
788
241
Implementation costs, recorded in our consolidated statements of income
as follows
Cost of sales
250
—
46
Selling, informational and administrative expenses
25
—
159
Research and development expenses
72
—
36
Other deductions
—
net
—
—
9
Total implementation costs
347
—
250
Total costs associated with cost-reduction and productivity initiatives and
acquisition activity
$
4,520
$
3,989
$
4,821
Transaction costs represent external costs directly related to acquired businesses and primarily include expenditures for banking, legal, accounting and other similar services. Substantially all of the
costs incurred in 2009 were fees related to a $22.5 billion bridge term loan credit agreement entered into with certain financial institutions on March 12, 2009 to partially fund our acquisition of Wyeth.
The bridge term loan credit agreement was terminated in June 2009 as a result of our issuance of approximately $24.0 billion of senior unsecured notes in the first half of 2009.
Integration costs represent external, incremental costs directly related to integrating acquired businesses, and primarily include expenditures for consulting and the integration of systems and
processes.
From the beginning of our cost-reduction and productivity initiatives in 2005 through December 31, 2011, Employee termination costs represent the expected reduction of the workforce by
approximately 57,400 employees, mainly in manufacturing and sales and research, of which approximately 42,800 employees have been terminated as of December 31, 2011. Employee termination
costs are generally recorded when the actions are probable and estimable and include accrued severance benefits, pension and postretirement benefits, many of which may be paid out during periods
after termination.
Asset impairments
primarily include charges to write down property, plant and equipment to fair value.
Other
primarily includes costs to exit certain assets and activities.
•
Primary Care operating segment ($593 million), Specialty Care and Oncology operating segment ($220 million), Established Products and Emerging Markets operating segment ($110
million), Animal Health and Consumer Healthcare operating segment ($51 million), Nutrition operating segment ($4 million), research and development operations ($489 million),
manufacturing operations ($280 million) and Corporate ($427 million).
70
2011 Financial Report
(a)
(b)
(c)
(d)
(e)
(a)
(b)
(c)
Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The restructuring charges in 2010 are associated with the following:
The restructuring charges in 2009 are associated with the following:
The components of Other deductions—net follow:
In 2011, the impairment charges of $863 million include (i) approximately $475 million of IPR&D assets, primarily related to two compounds for the treatment of certain autoimmune and inflammatory
diseases; (ii) approximately $195 million related to our biopharmaceutical indefinite-lived brand, Xanax; and (iii) approximately $185 million of Developed Technology Rights comprising the
impairments of five assets. These impairment charges reflect, among other things, the impact of new scientific findings and the increased competitive environment. The impairment charges are
associated with the following: Worldwide Research and Development ($394 million); Established Products ($193 million); Specialty Care ($135 million); Primary Care ($56 million); Oncology ($56
million); Animal Health ($17 million); and other ($12 million).
In 2010, the impairment charges of $2.2 billion include (i) approximately $950 million of IPR&D assets, primarily Prevnar 13/Prevenar 13 Adult, a compound for the prevention of pneumococcal
disease in adults age 50 and older, and Neratinib, a compound for the treatment of breast cancer; (ii) approximately $700 million of indefinite-lived Brands, related to Third Age, infant formulas for the
first 12-36 months of age, and Robitussin, a cough suppressant; and (iii) approximately $550 million of Developed Technology Rights, primarily Thelin, a product that treated pulmonary hypertension,
and Protonix, a product that treats erosive gastroesophageal reflux disease.
•
Primary Care operating segment ($71 million), Specialty Care and Oncology operating segment ($197 million), Established Products and Emerging Markets operating segment ($43 million),
Animal Health and Consumer Healthcare operating segment ($46 million), Nutrition operating segment ($4 million), research and development operations ($292 million), manufacturing
operations ($1.1 billion) and Corporate ($455 million).
•
Our three biopharmaceutical operating segments ($1.3 billion), Animal Health and Consumer Healthcare operating segment ($250 million), Nutrition operating segment ($4 million income),
research and development operations ($339 million), manufacturing operations ($292 million) and Corporate ($781 million).
Additional depreciation—asset restructuring represents the impact of changes in the estimated useful lives of assets involved in restructuring actions.
Implementation costs generally represent external, incremental costs directly related to implementing our non
-
acquisition
-
related cost
-
reduction and productivity initiatives.
The components of restructuring charges follow:
COSTS
INCURRED
ACTIVITY
THROUGH
DECEMBER 31,
ACCRUAL
AS OF
DECEMBER 31,
(MILLIONS OF DOLLARS)
2005-
2011
2011
2011
Employee termination costs
$10,602
$ 8,167
$2,434
Asset impairments
2,564
2,564
—
Other
1,022
931
92
Total
$14,188
$11,662
$2,526
Includes adjustments for foreign currency translation.
Included in Other current liabilities ($1.6 billion) and Other noncurrent liabilities ($928 million).
4.
Other Deductions—Net
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Interest income
$
(458
)
$
(402
)
$
(747
)
Interest expense
1,681
1,797
1,232
Net interest expense
1,223
1,395
485
Royalty-related income
(570
)
(579
)
(243
)
Net gains on asset disposals
(1
)
(262
)
(188
)
Certain legal matters, net
790
1,737
234
Certain asset impairment charges
863
2,175
417
Gain related to ViiV
—
—
(
482
)
Other, net
174
(130
)
62
Other deductions
—
net
$
2,479
$
4,336
$
285
2011 vs. 2010 - Interest income increased due to higher cash balances and higher interest rates earned on investments. Interest expense decreased due to lower long- and short-term debt balances
and the conversion of some fixed-rate liabilities to floating rate liabilities. 2010 vs. 2009 – Interest expense increased due to our issuance of $13.5 billion of senior unsecured notes on March 24, 2009
and approximately $10.5 billion of senior unsecured notes on June 3, 2009, primarily related to the acquisition of Wyeth, as well as the addition of legacy Wyeth debt. Interest income decreased due
to lower interest rates, coupled with lower average cash balances. Capitalized interest expense totaled $50 million in 2011, $36 million in 2010 and $34 million in 2009.
In 2010 and 2009, represents gains on sales of certain investments and businesses. Net gains primarily include realized gains and losses on sales of available-for-sale securities: in 2011, 2010 and
2009, gross realized gains were $79 million, $153 million and $186 million, respectively. Gross realized losses were $73 million in 2011, $12 million in 2010 and $43 million in 2009. Proceeds, primarily
from the sale of available-for-sale securities, were $10.2 billion in 2011, $5.3 billion in 2010 and $27.0 billion in 2009.
In 2011, primarily relates to charges for hormone-replacement therapy litigation. In 2010, includes a $1.3 billion charge for asbestos litigation related to our wholly owned subsidiary, Quigley Company,
Inc.
The majority of the asset impairment charges for 2011 and 2010 are related to intangible assets, including in-process research and development (IPR&D) assets, which were acquired as part of our
acquisition of Wyeth.
2011 Financial Report
71
(d)
(e)
(a) (b)
(a)
(b)
(a)
(b)
(c)
(d)
(e)
( a )
(b)
(c)
(d)
Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
These impairment charges, most of which occurred in the third quarter of 2010, reflect, among other things, the following: for IPR&D assets, the impact of changes to the development programs, the
projected development and regulatory timeframes and the risk associated with these assets; for Brand assets, the current competitive environment and planned investment support; and, for
Developed Technology Rights, in the case of Thelin, we voluntarily withdrew the product in regions where it was approved and discontinued all clinical studies worldwide, and for the others, an
increased competitive environment. The impairment charges are generally associated with the following: Specialty Care ($708 million); Oncology ($396 million); Nutrition ($385 million); Consumer
Healthcare ($292 million); Established Products ($182 million); Primary Care ($145 million); Worldwide Research and Development ($54 million); and other ($13 million).
In 2009, the impairment charge of $417 million primarily relates to certain materials used in our research and development activities that were no longer considered recoverable.
A. Taxes on Income
The components of Income from continuing operations before provision for taxes on income follow:
Represents a gain related to ViiV, an equity method investment, which is focused solely on research, development and commercialization of HIV medicines (see Note 2F. Acquisitions, Divestitures,
Collaborative Arrangements and Equity
-
Method Investments: Equity
-
Method Investments
).
5.
Taxes on Income
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
United States
$
(2,254)
$
(2,513)
$
(3,694)
International
15,016
11,795
14,368
Income from continuing operations before provision for taxes on income
$
12,762
$
9,282
$
10,674
2011 vs. 2010 – The decrease in the domestic loss was primarily due to the non-recurrence of a charge of $1.3 billion (pre-tax) in 2010 for asbestos litigation related to our wholly owned subsidiary,
Quigley Company, Inc., partially offset by a reduction in revenues due to the loss of exclusivity for several biopharmaceutical products and the impact of the U.S. Healthcare Legislation. The increase in
international income was due to the favorable impact of foreign exchange, higher impairment charges in 2010, as well as increased revenues from the biopharmaceutical products such as the
Prevnar/Prevenar franchise, Enbrel and Celebrex.
2010 vs. 2009 – The decrease in the domestic loss was due to revenues from legacy Wyeth products and a reduction in domestic restructuring charges partially offset by increased amortization
charges primarily related to identifiable intangibles in connection with our acquisition of Wyeth and litigation charges primarily related to our wholly owned subsidiary Quigley Company, Inc. The
decrease in international income was due primarily to an increase in international restructuring and amortization charges plus the non-recurrence of the gain in 2009 in connection with the formation of
ViiV, partially offset by revenues from legacy Wyeth products.
The components of
Provision for taxes on income
based on the location of the taxing authorities, follow:
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
United States:
Current income taxes:
Federal
$
1,349
$
(2,763
)
$
10,151
State and local
208
(315
)
68
Deferred income taxes:
Federal
349
2,010
(10,005
)
State and local
(242
)
(6
)
(93
)
Total U.S. tax provision/(benefit)
1,664
(1,074
)
121
International:
Current income taxes
2,202
2,212
1,516
Deferred income taxes
157
(67
)
508
Total international tax provision
2,359
2,145
2,024
Provision for taxes on income
$
4,023
$
1,071
$
2,145
In 2011, the Federal deferred income tax expense includes approximately $2.1 billion as a result of providing U.S. deferred income taxes on certain current-year funds earned outside of the U.S. that
will not be permanently reinvested overseas. (See
Note 5C. Taxes on Income: Deferred Taxes.
)
In 2010, the Federal current income tax benefit is primarily due to the tax benefit recorded in connection with our $1.4 billion settlement with the U.S. Internal Revenue Service and the reversal of $600
million of accruals related to interest on these unrecognized tax benefits. (See below). The Federal deferred income tax expense includes approximately $2.5 billion as a result of providing U.S.
deferred income taxes on certain current
-
year funds earned outside of the U.S. that will not be permanently reinvested overseas. (See
Note 5C. Taxes on Income: Deferred Taxes
).
In 2009, virtually all of the Federal current income tax expense was due to increased tax costs associated with certain business decisions executed to finance the Wyeth acquisition, including the
decision to repatriate certain funds earned outside of the U.S. In addition, virtually all of the Federal deferred income tax benefit was due to a reduction of deferred tax liabilities recorded in connection
with our acquisition of Wyeth. (See
Note 2A. Acquisitions, Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Acquisition of Wyeth
).
In 2011, federal, state and international net tax liabilities assumed or established on the date of the acquisition primarily of King are excluded. In 2010 and 2009, federal, state and international net tax
liabilities assumed or established on the date of the acquisition primarily of Wyeth are excluded. (See Note 2A. Acquisitions, Divestitures, Collaborative Arrangements and Equity-Method Investments:
Acquisition of Wyeth
and
Note 2B. Acquisitions, Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Acquisition of King Pharmaceuticals, Inc.
)
72
2011 Financial Report
(e)
( a), (b)
(a)
(b)
(a), (b), (c)
(d)
(a)
(b)
(c)
(d)
Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Settlements and Other Items Impacting Provision for Taxes on Income
In 2011, the Provision for taxes on income was impacted by the following:
In 2011, the $248 million fee payable to the federal government, recorded in Selling, informational and administrative expenses, as a result of the U.S. Healthcare Legislation, is not deductible for U.S.
income tax purposes.
In 2010, the Provision for taxes on income was impacted by the following:
In 2009, the Provision for taxes on income was impacted by the following:
In 2009, we incurred certain costs associated with the Wyeth acquisition that are not deductible for tax purposes.
See also Note 5D . Taxes on Income: Tax Contingencies.
B. Tax Rate Reconciliation
The reconciliation of the U.S. statutory income tax rate to our effective tax rate for Income from continuing operations follows:
•
Tax benefits of approximately $190 million resulting from the resolution of certain tax positions pertaining to prior years with various foreign tax authorities, and from the expiration of certain statutes of
limitations, as well as the reversal of approximately $77 million of accruals related to interest on these unrecognized tax benefits;
•
A tax benefit of approximately $80 million, inclusive of interest, resulting from the settlement of certain audits with the U.S. Internal Revenue Service; and
•
Tax benefits of approximately $270 million resulting from charges related to the hormone-therapy litigation.
•
A tax benefit of approximately $1.4 billion recorded in the fourth quarter, related to an audit settlement with the U.S. Internal Revenue Service and the reversal of approximately $600 million of accruals
related to interest on these unrecognized tax benefits;
•
The write-off of approximately $270 million of deferred tax assets related to the Medicare Part D subsidy for retiree prescription drug coverage, resulting from the provisions of the U.S. Healthcare
Legislation enacted in March 2010 concerning the tax treatment of that subsidy effective for tax years beginning after December 31, 2012;
•
Tax benefits of approximately $320 million resulting from the resolution of certain tax positions pertaining to prior years with various foreign tax authorities, and the expiration of certain statute of
limitations, as well as the reversal of approximately $140 million of accruals related to interest on these unrecognized tax benefits; and
•
Tax benefits of approximately $506 million resulting from charges for asbestos litigation related to our wholly owned subsidiary, Quigley Company, Inc.
•
A tax benefit of approximately $174 million, recorded in the third quarter, related to the final resolution of an agreement-in-principle with the DOJ to settle investigations of past promotional practices
concerning Bextra and certain other investigations. This resulted in the receipt of information that raised our assessment of the likelihood of prevailing on the technical merits of our tax position; and
•
A tax benefit of approximately $556 million related to the sale of one of our biopharmaceutical companies, Vicuron Pharmaceuticals, Inc. The sale, for nominal consideration, resulted in a loss for tax
purposes. This tax benefit is a result of the significant initial investment in the entity at the time of acquisition, primarily reported as an income statement charge for IPR&D at acquisition date.
YEAR ENDED DECEMBER 31,
2011
2010
2009
U.S. statutory income tax rate
35.0%
35.0%
35.0%
Taxation of non
-
U.S. operations
(3.3)
2.2
(9.4)
Resolution of certain tax positions
(2.7)
(26.4)
—
Sales of biopharmaceutical companies
0.2
—
(
5.1)
U.S. Healthcare Legislation
0.7
2.8
—
U.S. research tax credit and manufacturing deduction
(0.9)
(2.3)
(1.3)
Legal settlements
—
0.4
(1.6)
Acquired IPR&D
—
0.5
0.2
Wyeth acquisition
-
related costs
—
0.5
2.4
All other
—
net
2.5
(1.2)
(0.1)
Effective tax rate for income from continuing operations
31.5%
11.5%
20.1%
For taxation of non-U.S. operations, this rate impact reflects the fact that we operate manufacturing subsidiaries in Puerto Rico, Ireland, and Singapore. We benefit from a Puerto Rican incentive grant
that expires in 2029. Under the grant, we are partially exempt from income, property and municipal taxes. In Ireland, we benefited from an incentive tax rate effective through 2010 on income from
manufacturing operations. In Singapore, we benefit from incentive tax rates effective through 2031 on income from manufacturing operations. The rate impact also reflects the jurisdictional location of
earnings, the costs of certain repatriation decisions and uncertain tax positions.
2011 Financial Report
73
(a)
(b)
(c)
(c)
(c)
(d)
(c)
(a)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
C. Deferred Taxes
Deferred taxes arise as a result of basis differentials between financial statement accounting and tax amounts.
The components of our deferred tax assets and liabilities, shown before jurisdictional netting follow:
We have carryforwards, primarily related to foreign tax credits, net operating and capital losses, and charitable contributions, which are available to reduce future U.S. federal and state, as well as
international, income taxes payable with either an indefinite life or expiring at various times from 2012 to 2031. Certain of our U.S. net operating losses are subject to limitations under Internal Revenue
Code Section 382.
Valuation allowances are provided when we believe that our deferred tax assets are not recoverable based on an assessment of estimated future taxable income that incorporates ongoing, prudent and
feasible tax planning strategies.
As of December 31, 2011, we have not made a U.S. tax provision on approximately $63.0 billion of unremitted earnings of our international subsidiaries. As of December 31, 2011, as these earnings are
intended to be permanently reinvested overseas, the determination of a hypothetical unrecognized deferred tax liability is not practicable.
D. Tax Contingencies
We are subject to income tax in many jurisdictions, and a certain degree of estimation is required in recording the assets and liabilities related to income taxes. All of our tax positions are subject to audit
by the local taxing authorities in each tax jurisdiction. These tax audits can involve complex issues, interpretations and judgments and the resolution of matters may span multiple years, particularly if
subject to negotiation or litigation. For a description of our accounting policies associated with accounting for income tax contingencies, see Note 1O. Significant Accounting Policies: Deferred Tax Assets
and Liabilities and Income Tax Contingencies. For a description of the risks associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions.
Uncertain Tax Positions
As tax law is complex and often subject to varied interpretations, it is uncertain whether some of our tax positions will be sustained upon audit. As of December 31, 2011 and 2010, we had approximately
$6.1 billion and $5.8 billion, respectively, in net liabilities associated with uncertain tax positions, excluding associated interest:
For a discussion about the resolution of certain tax positions, see Note 5D. Taxes on Income: Tax Contingencies .
For a discussion about the sales of the biopharmaceutical companies, the impact of U.S. Healthcare Legislation, legal settlements and Wyeth acquisition related costs, see Note 5A. Taxes on Income:
Taxes on Income
.
The charges for acquired IPR&D are primarily not deductible for tax purposes.
2011 DEFERRED TAX
2010 DEFERRED TAX
(MILLIONS OF DOLLARS)
ASSETS
(LIABILITIES)
ASSETS
(LIABILITIES)
Prepaid/deferred items
$
1,611
$ (211
)
$
1,321
$ (112)
Inventories
324
(52
)
132
(59)
Intangibles
1,713
(16,014
)
1,165
(17,104)
Property, plant and equipment
226
(1,326
)
420
(2,146)
Employee benefits
4,285
(524
)
4,479
(56)
Restructurings and other charges
554
(95
)
1,359
(70)
Legal and product liability reserves
1,812
—
1,411
—
Net operating loss/credit carryforwards
4,414
—
4,575
—
Unremitted earnings
—
(
11,699
)
—
(
9,524)
State and local tax adjustments
476
—
452
—
All other
1,197
(125
)
601
(554)
Subtotal
16,612
(30,046
)
15,915
(29,625)
Valuation allowance
(1,201
)
—
(
894
)
—
Total deferred taxes
$
15,411
$(30,046
)
$
15,021
$(29,625)
Net deferred tax liability
$(14,635
)
$(14,604)
2011 vs. 2010 – The net deferred tax liability position in 2011 was about the same as 2010 and reflects an increase in noncurrent deferred tax liabilities related to intangibles established in connection
with our acquisition of King and an increase in noncurrent deferred tax liabilities on unremitted earnings, partially offset by the reduction in noncurrent deferred tax liabilities related to the amortization of
identifiable intangibles, and an increase in current deferred tax assets established as a result of litigation charges related to hormone therapy.
In 2011, included in Taxes and other current assets ($4.0 billion), Taxes and other noncurrent assets ($1.2 billion), Other current liabilities ($291 million) and Noncurrent deferred tax liabilities ($19.6
billion). In 2010, included in Taxes and other current assets ($3.0 billion), Taxes and other noncurrent assets ($1.2 billion), Other current liabilities ($108 million) and Noncurrent deferred tax liabilities
($18.6 billion).
•
Tax assets associated with uncertain tax positions primarily represent our estimate of the potential tax benefits in one tax jurisdiction that could result from the payment of income taxes in another tax
jurisdiction. These potential benefits generally result from cooperative efforts among taxing authorities, as required by tax treaties to minimize double taxation, commonly referred to as the competent
authority process. The recoverability of these assets, which we believe to be more likely than not, is dependent upon the actual payment of taxes in one tax jurisdiction and, in some cases, the
successful petition for recovery in another tax jurisdiction. As of December 31, 2011 and 2010, we had approximately $1.2 billion and $1.0 billion, respectively, in assets associated with uncertain tax
positions recorded in
Taxes and other noncurrent assets
.
74
2011 Financial Report
(b)
(c)
(d)
(a) ,(b)
(a)
(b)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The reconciliation of the beginning and ending amounts of gross unrecognized tax benefits follows:
Status of Tax Audits and Potential Impact on Accruals for Uncertain Tax Positions
The United States is one of our major tax jurisdictions:
In addition to the open audit years in the U.S., we have open audit years in other major tax jurisdictions, such as Canada (1998-2011), Japan (2006-2011), Europe (2002-2011, primarily reflecting Ireland,
the United Kingdom, France, Italy, Spain and Germany) and Puerto Rico (2007-2011). During 2011, we recognized approximately $190 million in tax benefits resulting from the resolution of certain tax
positions pertaining to prior years with various foreign tax authorities, as well as from the expiration of certain statutes of limitations. The 2011 effective tax rate was also favorably impacted by
approximately $77 million related to the reversal of accruals for interest on these unrecognized tax benefits. During 2010, we also recognized approximately $320 million in tax benefits resulting from the
resolution of certain tax positions pertaining to prior years with various foreign tax authorities, as well as from the expiration of certain statutes of limitations. The 2010 effective tax rate was also favorably
impacted by approximately $140 million related to the reversal of accruals for interest on these unrecognized tax benefits.
•
Tax liabilities associated with uncertain tax positions represent unrecognized tax benefits, which arise when the estimated benefit recorded in our financial statements differs from the amounts taken
or expected to be taken in a tax return because of the uncertainties described above. These unrecognized tax benefits relate primarily to issues common among multinational corporations.
Substantially all of these unrecognized tax benefits, if recognized, would impact our effective income tax rate.
(MILLIONS OF DOLLARS)
2011
2010
2009
Balance, January 1
$
(6,759
)
$(7,657
)
$(5,372)
Acquisitions
(72
)
(49
)
(1,785)
Increases based on tax positions taken during a prior period
(502
)
(513
)
(79)
Decreases based on tax positions taken during a prior period
271
2,384
38
Decreases based on cash payments for a prior period
575
280
—
Increases based on tax positions taken during the current period
(855
)
(1,396
)
(941)
Decreases based on tax positions taken during the current period
—
—
712
Impact of foreign exchange
(89
)
104
(284)
Other, net
122
88
54
Balance, December 31
$
(7,309
)
$(6,759
)
$(7,657)
The amount in 2011 primarily relates to the acquisition of King and the amounts in 2010 and 2009 primarily relate to the acquisition of Wyeth.
Primarily included in
Provision for taxes on income.
In 2011, 2010, and 2009, the decreases are primarily a result of effectively settling certain issues with the U.S. and foreign tax authorities. See discussions below
.
Primarily includes decreases as a result of a lapse of applicable statutes of limitations.
In 2011, included in Income taxes payable ($357 million), Taxes and other current assets ($11 million), Taxes and other noncurrent assets ($225 million), Noncurrent deferred tax liabilities ($677
million) and Other taxes payable ($6.0 billion). In 2010, included in Income taxes payable ($421 million), Taxes and other current assets ($279 million), Taxes and other noncurrent assets ($169
million),
Noncurrent deferred tax liabilities
($369 million) and
Other taxes payable
($5.5 billion).
•
Interest related to our unrecognized tax benefits is recorded in accordance with the laws of each jurisdiction and is recorded in Provision for taxes on income
in our consolidated statements of income.
In 2011, we recorded net interest expense of $203 million. In 2010, we recorded net interest income of $545 million, primarily as a result of settling certain issues with the U.S. and various foreign tax
authorities, which are discussed below. In 2009, we recorded net interest expense of $191 million. Gross accrued interest totaled $951 million as of December 31, 2011 (reflecting a decrease of
approximately $203 million as a result of cash payments) and $952 million as of December 31, 2010. In 2011, these amounts were included in Income taxes payable ($120 million), Taxes and other
current assets ($2 million) and Other taxes payable ($829 million). In 2010, these amounts were included in Income taxes payable ($112 million), Taxes and other current assets ($122 million) and
Other taxes payable
($718 million). Accrued penalties are not significant.
• During the first quarter of 2011, we reached a settlement with the U.S. Internal Revenue Service (IRS) with respect to the audits of the Wyeth tax returns for the years 2002 through 2005. The
settlement resulted in an income tax benefit to Pfizer of approximately $80 million for income tax and interest. Tax years 2006 through the Wyeth acquisition date (October 15, 2009) are currently
under audit.
• During the fourth quarter of 2010, we reached a settlement with the IRS related to issues we had appealed with respect to the audits of the Pfizer Inc. tax returns for the years 2002 through 2005, as
well as the Pharmacia audit for the year 2003 through the date of merger with Pfizer (April 16, 2003). The IRS concluded its examination of the aforementioned tax years and issued a final Revenue
Agent’s Report (RAR). The Company agreed with all of the adjustments and computations contained in the RAR. As a result of settling these audit years, in the fourth quarter of 2010, we reduced our
unrecognized tax benefits by approximately $1.4 billion and recorded a corresponding tax benefit. The fourth quarter and full year 2010 effective tax rates were also favorably impacted by the reversal
of $600 million of accruals related to interest on these unrecognized tax benefits. The tax years 2006-2010 are currently under audit and the tax year 2011 is open but not under audit. All other tax
years in the U.S. for Pfizer Inc. are closed under the statute of limitations.
• King’s tax year 2008 and Alpharma, Inc.’s (a company acquired through the King acquisition) tax years 2005-2007 are currently under audit. Tax years 2009 through the date of acquisition (January
31, 2011) are open but not under audit. King’s tax years prior to 2008 have been settled with the IRS. The open tax years and audits of King and its subsidiaries are not considered significant to
Pfizer.
2011 Financial Report
75
(a)
(b)
(b), (c)
(b)
(d)
(e)
(a)
(b)
(c)
(d)
(e)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Any settlements or statute of limitations expirations could result in a significant decrease in our uncertain tax positions. We estimate that it is reasonably possible that within the next twelve months, our
gross unrecognized tax benefits, exclusive of interest, could decrease by as much as $500 million, as a result of settlements with taxing authorities or the expiration of the statute of limitations. Our
assessments are based on estimates and assumptions that have been deemed reasonable by management, but our estimates of unrecognized tax benefits and potential tax benefits may not be
representative of actual outcomes, and variation from such estimates could materially affect our financial statements in the period of settlement or when the statutes of limitations expire, as we treat these
events as discrete items in the period of resolution. Finalizing audits with the relevant taxing authorities can include formal administrative and legal proceedings, and, as a result, it is difficult to estimate
the timing and range of possible change related to our uncertain tax positions, and such changes could be significant.
The components and changes in Accumulated other comprehensive income/(loss) follow:
Income taxes are not provided for foreign currency translation relating to permanent investments in international subsidiaries.
6.
Other Comprehensive Income/(Loss)
NET UNREALIZED GAINS/(LOSSES)
BENEFIT PLANS
(MILLIONS OF DOLLARS)
CURRENCY
TRANSLATION
ADJUSTMENT
AND OTHER
DERIVATIVE
FINANCIAL
INSTRUMENTS
AVAILABLE
FOR-
SALE
SECURITIES
ACTUARIAL
GAINS/
(LOSSES)
PRIOR
SERVICE
(COSTS)/
CREDITS AND
OTHER
ACCUMULATED
OTHER
COMPREHENSIVE
INCOME/(LOSS)
Balance, January 1, 2009
$(1,389
)
$ 28
$ (86
)
$(3,132
)
$ 10
$(4,569)
Other comprehensive income/(loss)—Pfizer Inc. :
Foreign currency translation adjustments
4,978
—
—
—
—
4,978
Unrealized holding gains
—
291
576
—
—
867
Reclassification adjustments to income
5
(299
)
(143
)
—
—
(
437)
Actuarial gains/(losses) and other benefit plan items
—
—
—
(
701
)
154
(547)
Amortization of actuarial losses and other benefit plan items
—
—
—
291
(6
)
285
Curtailments and settlements
—
net
—
—
—
390
(5
)
385
Other
2
—
—
(
196
)
(3
)
(197)
Income taxes
(46
)
(14
)
(78
)
(19
)
(56
)
(213)
5,121
Balance, December 31, 2009
3,550
6
269
(3,367
)
94
552
Other comprehensive income/(loss)
—
Pfizer Inc.
:
Foreign currency translation adjustments
(3,544
)
—
—
—
—
(
3,544)
Unrealized holding gains/(losses)
—
(
1,043
)
7
—
—
(
1,036)
Reclassification adjustments to income
(7
)
702
(141
)
—
—
554
Actuarial gains/(losses) and other benefit plan items
—
—
—
(
1,426
)
550
(876)
Amortization of actuarial losses and other benefit plan items
—
—
—
262
(42
)
220
Curtailments and settlements
—
net
—
—
—
266
(49
)
217
Other
5
—
—
88
5
98
Income taxes
165
127
22
230
(169
)
375
(3,992)
Balance, December 31, 2010
169
(208
)
157
(3,947
)
389
(3,440)
Other comprehensive income/(loss)
–
–
Pfizer Inc.
:
Foreign currency translation adjustments
837
—
—
—
—
837
Unrealized holding losses
—
(
502
)
(143
)
—
—
(
645)
Reclassification adjustments to income
(127
)
239
15
—
—
127
Actuarial gains/(losses) and other benefit plan items
—
—
—
(
2,459
)
106
(2,353)
Amortization of actuarial losses and other benefit plan items
—
—
—
284
(69
)
215
Curtailments and settlements
—
net
—
—
—
355
(91
)
264
Other
4
—
—
(
100
)
3
(93)
Income taxes
61
110
17
747
24
959
(689)
Balance, December 31, 2011
$ 944
$ (361
)
$ 46
$(5,120)
$ 362
$(4,129)
Amounts do not include adjustments attributable to noncontrolling interests of $45 million loss in 2011, $5 million income in 2010 and $6 million income in 2009.
The currency translation adjustments reclassified to income resulted from the sale of legal entities.
76
2011 Financial Report
(a)
(b)
(a)
(b)
(a)
(b)
(a)
(b)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
As of December 31, 2011, we estimate that we will reclassify into 2012 income the following pre-tax amounts currently held in Accumulated other comprehensive income/(loss) : $21 million of the
unrealized holding gains on derivative financial instruments; $466 million of actuarial losses related to benefit plan obligations and plan assets and other benefit plan items; and $75 million of prior service
credits primarily related to benefit plan amendments.
A. Selected Financial Assets and Liabilities
Information about certain of our financial assets and liabilities follows:
A single estimate of fair value can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For a description of our general
accounting policies associated with developing fair value estimates, see Note 1E. Significant Accounting Policies: Fair Value . For a description of the risks associated with estimates and assumptions,
see Note 1C. Significant Accounting Policies: Estimates and Assumptions .
7.
Financial Instruments
AS OF DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
Selected financial assets measured at fair value on a recurring basis
:
Trading securities
$ 154
$ 173
Available-for-sale debt securities
29,179
32,699
Available-for-sale money market funds
1,370
1,217
Available
-
for
-
sale equity securities, excluding money market funds
317
230
Derivative financial instruments in receivable positions
:
Interest rate swaps
1,033
603
Foreign currency forward
-
exchange contracts
349
494
Foreign currency swaps
17
128
Total
32,419
35,544
Other selected financial assets
:
Held
-
to
-
maturity debt securities, carried at amortized cost
1,155
1,178
Private equity securities, carried as equity method or at cost
1,020
1,134
Short-term loans, carried at cost
51
467
Long
-
term loans, carried at cost
381
299
Total
2,607
3,078
Total selected financial assets
$35,026
$38,622
Financial liabilities measured at fair value on a recurring basis :
Derivative financial instruments in a liability position
:
Foreign currency swaps
$ 1,396
$ 623
Foreign currency forward
-
exchange contracts
355
257
Interest rate swaps
14
4
Total
1,765
884
Other financial liabilities
:
Short-term borrowings, carried at historical proceeds, as adjusted
4,018
5,603
Long
-
term debt, carried at historical proceeds, as adjusted
34,931
38,410
Total
38,949
44,013
Total selected financial liabilities
$40,714
$44,897
Fair values are determined based on valuation inputs categorized as Level 1, 2 or 3 (see Note 1E. Significant Accounting Policies: Fair Value ). All of our financial assets and liabilities measured at
fair value on a recurring basis use Level 2 inputs in the calculation of fair value, except that included in available-for-sale equity securities, excluding money market funds, are $85 million as of
December 31, 2011 and $105 million as of December 31, 2010 of investments that use Level 1 inputs in the calculation of fair value, and $25 million as of December 31, 2011 that use Level 3 inputs.
Trading securities are held in trust for legacy business acquisition severance benefits.
Gross unrealized gains and losses are not significant.
Includes approximately $625 million as of December 31, 2011 and December 31, 2010 of money market funds held in escrow to secure certain of Wyeth’s payment obligations under its 1999
Nationwide Class Action Settlement Agreement, which relates to litigation against Wyeth concerning its former weight
-
loss products, Redux and Pondimin.
Designated as hedging instruments, except for certain foreign currency contracts used as offsets; namely, foreign currency forward-
exchange contracts with fair values of $169 million and interest rate
swaps with fair values of $8 million at December 31, 2011; and foreign currency forward-exchange contracts with fair values of $326 million and foreign currency swaps with fair values of $17 million
at December 31, 2010.
The differences between the estimated fair values and carrying values of these financial assets and liabilities not measured at fair value on a recurring basis were not significant as of December 31,
2011 or December 31, 2010.
Our private equity securities represent investments in the life sciences sector.
Our short
-
term and long
-
term loans are due from companies with highly rated securities (Standard & Poor
’
s (S&P) ratings that are virtually all AA or better).
Designated as hedging instruments, except for certain foreign currency contracts used as offsets; namely, foreign currency forward-exchange contracts with fair values of $141 million and foreign
currency swaps with fair values of $123 million at December 31, 2011; and foreign currency forward-exchange contracts with fair values of $186 million and foreign currency swaps with fair values of
$93 million at December 31, 2010.
Some carrying amounts may include adjustments for discount or premium amortization or for the effect of interest rate swaps designated as hedges.
Includes foreign currency borrowings with fair values of $2 billion at December 31, 2010, which are used as hedging instruments.
Includes foreign currency debt with fair values of $919 million at December 31, 2011 and $880 million at December 31, 2010, which are used as hedging instruments.
The fair value of our long-term debt is $40.1 billion at December 31, 2011 and $42.3 billion at December 31, 2010.
2011 Financial Report
77
(a)
(b)
(c)
(d)
(c)
(e)
(f)
(c)
(g)
(h)
(h)
(a)
(i)
(j)
(f), (k)
(l), (m)
(a)
(b)
(c)
(d)
(e)
(f)
(g)
(h)
(i)
(j)
(k)
(l)
(m)
Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Specifically, the following methods and assumptions were used to estimate the fair value of our financial assets and liabilities:
In addition, we have long-term receivables where the determination of fair value employs discounted future cash flows, using current interest rates at which similar loans would be made to borrowers with
similar credit ratings and for the same remaining maturities.
We periodically review the methodologies, inputs and outputs of third-party pricing services for reasonableness. Our procedures can include, for example, referencing other third-party pricing models,
monitoring key observable inputs (like LIBOR interest rates) and selectively performing test-comparisons of values with actual sales of financial instruments.
The selected financial assets and liabilities are presented in our consolidated balance sheets as follows :
There were no significant impairments of financial assets recognized in any period presented.
•
Trading equity securities
—
quoted market prices.
•
Trading debt securities
—
observable market interest rates.
•
Available-for-sale debt securities—third-party matrix-pricing model that uses significant inputs derived from or corroborated by observable market data and credit-adjusted interest rate yield curves.
•
Available
-
for
-
sale money market funds
—
observable Net Asset Value prices.
•
Available
-
for
-
sale equity securities, excluding money market funds
—
third
-
party pricing services that principally use a composite of observable prices.
•
Derivative financial instruments (assets and liabilities)—third-party matrix-pricing model that uses significant inputs derived from or corroborated by observable market data. Where applicable, these
models discount future cash flow amounts using market-based observable inputs, including interest rate yield curves, and forward and spot prices for currencies. The credit risk impact to our
derivative financial instruments was not significant.
•
Held-to-maturity debt securities—third-party matrix-pricing model that uses significant inputs derived from or corroborated by observable market data and credit-adjusted interest rate yield curves.
•
Private equity securities, excluding equity-method investments—application of the implied volatility associated with an observable biotech index to the carrying amount of our portfolio and, to a lesser
extent, performance multiples of comparable securities adjusted for company
-
specific information.
•
Short-term and long-term loans—third-party model that discounts future cash flows using current interest rates at which similar loans would be made to borrowers with similar credit ratings and for the
same remaining maturities.
•
Short
-
term borrowings and long
-
term debt
—
third
-
party matrix
-
pricing model that uses significant inputs derived from or corroborated by observable market data and our own credit rating.
AS OF DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
Assets
Cash and cash equivalents
$ 900
$ 906
Short
-
term investments
23,219
26,277
Short
-
term loans
51
467
Long
-
term investments and loans
9,457
9,747
Taxes and other current assets
357
515
Taxes and other noncurrent assets
1,042
710
Total
$35,026
$38,622
Liabilities
Short
-
term borrowings, including current portion of long
-
term debt
$4,018
$ 5,603
Other current liabilities
459
339
Long-term debt
34,931
38,410
Other noncurrent liabilities
1,306
545
Total
$40,714
$44,897
As of December 31, 2011, derivative instruments at fair value include foreign currency forward-exchange contracts ($349 million) and interest rate swaps ($8 million) and, as of December 31, 2010,
include foreign currency forward
-
exchange contracts ($494 million) and foreign currency swaps ($21 million).
As of December 31, 2011, derivative instruments at fair value include interest rate swaps ($1.0 billion) and foreign currency swaps ($17 million) and, as of December 31, 2010, include interest rate
swaps ($603 million) and foreign currency swaps ($107 million).
At December 31, 2011, derivative instruments at fair value include foreign currency forward-exchange contracts ($355 million) and foreign currency swaps ($104 million) and, at December 31, 2010,
include foreign currency forward
-
exchange contracts ($257 million), foreign currency swaps ($79 million) and interest rate swaps ($3 million).
At December 31, 2011, derivative instruments at fair value include foreign currency swaps ($1.3 billion) and interest rate swaps ($14 million) and, at December 31, 2010, include foreign currency swaps
($544 million) and interest rate swaps ($1 million).
78
2011 Financial Report
(a)
(b)
(c)
(d)
(a)
(b)
(c)
(d)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
B. Investments in Debt Securities
The contractual maturities of the available-for-sale and held-to-maturity debt securities follow:
C. Short-Term Borrowings
Short-term borrowings include amounts for commercial paper of $2.7 billion as of December 31, 2011, and $1.2 billion as of December 31, 2010. The weighted-average effective interest rate on short-
term borrowings outstanding was 0.2% as of December 31, 2011, and 2.8% as of December 31, 2010.
As of December 31, 2011, we had access to $9.4 billion of lines of credit, of which $2.3 billion expire within one year. Of these lines of credit, $8.6 billion are unused, of which our lenders have committed
to loan us $7.5 billion at our request. Also, $7.0 billion of our unused lines of credit, all of which expire in 2016, may be used to support our commercial paper borrowings.
D. Long-Term Debt
The components of our long-term debt follow:
Long-term debt outstanding as of December 31, 2011 matures in the following years:
YEARS
(MILLIONS OF DOLLARS)
WITHIN 1
OVER 1
TO 5
OVER 5
TO 10
TOTAL AS OF
DECEMBER 31,
2011
Available-for-sale debt securities:
Western European, Scandinavian and other government debt
$ 9,895
$1,177
$
–
–
$
11,072
Corporate debt
3,921
2,321
284
6,526
U.S. Government debt
5,431
–
–
257
5,688
Supranational debt
1,872
433
––
2,305
Federal Home Loan Mortgage Corporation and Federal National Mortgage Association asset
-
backed securities
–
–
2,225
9
2,234
Western European and other government agency debt
1,101
253
–
–
1,354
Held
-
to
-
maturity debt securities:
Certificates of deposit and other
1,150
5
––
1,155
Total debt securities
$23,370
$6,414
$550
$30,334
Primarily issued by above-investment-grade institutions in the financial services sector.
AS OF DECEMBER 31,
(MILLIONS OF DOLLARS)
MATURITY DATE
2011
2010
Senior unsecured notes:
6.20%
March 2019
$ 3,248
$ 3,247
5.35%
March 2015
3,069
3,000
7.20%
March 2039
2,948
2,564
4.75% euro
June 2016
2,583
2,665
5.75% euro
June 2021
2,581
2,662
3.625% euro
June 2013
2,392
2,466
6.50% U.K. pound
June 2038
2,306
2,306
5.95%
April 2037
2,088
2,089
5.50%
February 2014
1,893
1,921
5.50%
March 2013
1,564
1,608
4.55% euro
May 2017
1,325
1,322
4.75% euro
December 2014
1,266
1,302
5.50%
February 2016
1,061
1,074
4.45%
March 2012
–
–
3,543
Notes and other debt with a weighted-average interest rate of 5.28%
2012–2018
2,302
2,342
Notes and other debt with a weighted
-
average interest rate of 6.51%
2021
–
2036
3,440
3,464
Foreign currency notes and other foreign currency debt with a weighted-
average interest rate of 2.48%
2014-2016
865
835
Total long
-
term debt
$34,931
$38,410
Current portion not included above
$ 6
$ 3,502
Instrument is callable by us at any time at the greater of 100% of the principal amount or the sum of the present values of the remaining scheduled payments of principal and interest discounted at
the U.S. Treasury rate plus 0.50% plus, in each case, accrued and unpaid interest.
Instrument is callable by us at any time at the greater of 100% of the principal amount or the sum of the present values of the remaining scheduled payments of principal and interest discounted at a
comparable government bond rate plus 0.20% plus, in each case, accrued and unpaid interest.
At December 31, 2011, the note was called.
Contains debt issuances with a weighted-average maturity of approximately 5 years.
Contains debt issuances with a weighted
-
average maturity of approximately 18 years.
Contains debt issuances with a weighted
-
average maturity of approximately 4 years.
(MILLIONS OF DOLLARS)
2013
2014
2015
2016
AFTER 2016
TOTAL
Maturities
$
3,964
$
3,987
$
3,074
$
4,500
$19,406
$
34,931
2011 Financial Report
79
(a)
(a)
(a)
(a)
(a)
(b)
(b)
(b)
(b)
(c)
(d)
(e)
(f)
(a)
(b)
(c)
(d)
(e)
(f)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
In March 2007, we filed a securities registration statement with the SEC. The registration statement was filed under the automatic shelf registration process available to “well–known seasoned issuers”
and expired in March 2010. On March 24, 2009, in order to partially finance our acquisition of Wyeth, we issued $13.5 billion of senior unsecured notes under this registration statement. On June 3, 2009,
also in order to partially finance our acquisition of Wyeth, we issued approximately $10.5 billion of senior unsecured notes in a private placement pursuant to Regulation S under the Securities Act of
1933, as amended (Securities Act of 1933). The notes issued on June 3, 2009 have not been and will not be registered under the Securities Act of 1933 and, subject to certain exceptions, may not be
sold, offered or delivered within the U.S. to, or for the account or benefit of, U.S. persons.
Foreign Exchange Risk
A significant portion of our revenues, earnings and net investments in foreign affiliates is exposed to changes in foreign exchange rates. We seek to manage our foreign exchange risk, in part, through
operational means, including managing expected same-currency revenues in relation to same-currency costs and same-currency assets in relation to same-currency liabilities. Depending on market
conditions, foreign exchange risk also is managed through the use of derivative financial instruments and foreign currency debt. These financial instruments serve to protect net income and net
investments against the impact of the translation into U.S. dollars of certain foreign exchange-denominated transactions. The aggregate notional amount of foreign exchange derivative financial
instruments hedging or offsetting foreign currency exposures is $48.1 billion. The derivative financial instruments primarily hedge or offset exposures in the euro, Japanese yen and U.K. pound. The
maximum length of time over which we are hedging future foreign exchange cash flows relates to our $2.3 billion U.K. pound debt maturing in 2038.
All derivative contracts used to manage foreign currency risk are measured at fair value and are reported as assets or liabilities on the consolidated balance sheet. Changes in fair value are reported in
earnings or in Other comprehensive income/(loss) , depending on the nature and purpose of the financial instrument (offset or hedge relationship) and the effectiveness of the hedge relationships, as
follows:
Any ineffectiveness is recognized immediately into earnings. There was no significant ineffectiveness for any period presented.
Interest Rate Risk
Our interest-bearing investments, loans and borrowings are subject to interest rate risk. We seek to invest and loan primarily on a short-term or variable-rate basis; however, in light of current market
conditions, we currently borrow primarily on a long-term, fixed-rate basis. From time to time, depending on market conditions, we will change the profile of our outstanding debt by entering into derivative
financial instruments like interest rate swaps.
We entered into derivative financial instruments to hedge or offset the fixed interest rates on the hedged item, matching the amount and timing of the hedged item. The aggregate notional amount of
interest rate derivative financial instruments is $10.6 billion. The derivative financial instruments primarily hedge U.S. dollar and euro fixed-rate debt.
All derivative contracts used to manage interest rate risk are measured at fair value and reported as assets or liabilities on the consolidated balance sheet. Changes in fair value are reported in earnings,
as follows:
Any ineffectiveness is recognized immediately into earnings. There was no significant ineffectiveness for any period presented.
E.
Derivative Financial Instruments and Hedging Activities
•
We record in Other comprehensive income/(loss) the effective portion of the gains or losses on foreign currency forward-exchange contracts and foreign currency swaps that are designated as cash
flow hedges and reclassify those amounts, as appropriate, into earnings in the same period or periods during which the hedged transaction affects earnings.
•
We recognize the gains and losses on forward-exchange contracts and foreign currency swaps that are used to offset the same foreign currency assets or liabilities immediately into earnings along
with the earnings impact of the items they generally offset. These contracts essentially take the opposite currency position of that reflected in the month
-end balance sheet to counterbalance the effect
of any currency movement.
•
We recognize the gain and loss impact on foreign currency swaps designated as hedges of our net investments in earnings in three ways: over time—for the periodic net swap payments;
immediately—to the extent of any change in the difference between the foreign exchange spot rate and forward rate; and upon sale or substantial liquidation of our net investments—to the extent of
change in the foreign exchange spot rates.
•
We record in Other comprehensive income/(loss) the foreign exchange gains and losses related to foreign exchange-denominated debt designated as a hedge of our net investments in foreign
subsidiaries and reclassify those amounts into earnings upon the sale or substantial liquidation of our net investments.
•
We recognize the gains and losses on interest rate swaps that are designated as fair value hedges in earnings upon the recognition of the change in fair value of the hedged risk. We recognize the
offsetting earnings impact of fixed-rate debt attributable to the hedged risk also in earnings.
80
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Information about the gains/(losses) incurred to hedge or offset operational foreign exchange or interest rate risk follows:
For information about the fair value of our derivative financial instruments, and the impact on our consolidated balance sheet, see
Note 7A. Financial Instruments: Selected Financial Assets and Liabilities.
Certain of our derivative instruments are covered by associated credit-support agreements that have credit-risk-related contingent features designed to reduce our counterparties’ exposure to our risk of
defaulting on amounts owed. The aggregate fair value of these derivative instruments that are in a liability position is $502 million, for which we have posted collateral of $555 million in the normal course
of business. These features include the requirement to pay additional collateral in the event of a downgrade in our debt ratings. If there had been a downgrade to below an A rating by S&P or the
equivalent rating by Moody’s Investors Service, on December 31, 2011, we would have been required to post an additional $46 million of collateral to our counterparties. The collateral advanced
receivables are reported in Cash and cash equivalents.
F. Credit Risk
On an ongoing basis, we review the creditworthiness of counterparties to our foreign exchange and interest rate agreements and do not expect to incur a significant loss from failure of any counterparties
to perform under the agreements. There are no significant concentrations of credit risk related to our financial instruments with any individual counterparty. As of December 31, 2011, we had $2.8 billion
due from a well-diversified, highly rated group (S&P ratings of mostly A+ or better) of bank counterparties around the world. See Note 7B. Financial Instruments: Investment in Debt Securities for a
distribution of our investments.
In general, there is no requirement for collateral from customers. However, derivative financial instruments are executed under master netting agreements with financial institutions. These agreements
contain provisions that provide for the ability for collateral payments, depending on levels of exposure, our credit rating and the credit rating of the counterparty. As of December 31, 2011, we received
cash collateral of $491 million against various counterparties. The collateral primarily supports the approximate fair value of our derivative contracts. With respect to the collateral received, the obligations
are reported in Short-term borrowings, including current portion of long-term debt.
AMOUNT OF
GAINS/(LOSSES)
RECOGNIZED IN OID
AMOUNT OF
GAINS/(LOSSES)
RECOGNIZED IN OCI
(EFFECTIVE PORTION)
AMOUNT OF
GAINS/(LOSSES)
RECLASSIFIED FROM
OCI INTO OID
(EFFECTIVE PORTION)
(MILLIONS OF DOLLARS)
Dec. 31,
2011
Dec. 31,
2010
Dec. 31,
2011
Dec. 31,
2010
Dec. 31,
2011
Dec. 31,
2010
Derivative Financial Instruments in Cash Flow
Hedge Relationships
Foreign currency swaps
$
–
–
$
–
–
$ (
496)
$(1,054)
$(243)
$(704)
Derivative Financial Instruments in Net Investment
Hedge Relationships
Foreign currency swaps
7
(1)
(1,059)
(97)
–
–
–
–
Derivative Financial Instruments Not Designated
as Hedges
Foreign currency forward
-
exchange contracts
(260
)
(454)
–
–
–
–
–
–
–
–
Foreign currency swaps
106
20
–
–
–
–
–
–
–
–
Non-Derivative Financial Instruments in Net
Investment Hedge Relationships
Foreign currency short
-
term borrowings
–
–
–
–
940
(241)
–
–
–
–
Foreign currency long
-
term debt
–
–
–
–
(
41)
(91)
–
–
–
–
All other, net
15
1
(4)
(6)
4
2
Total
$(132
)
$(434)
$ (660)
$(1,489)
$(239)
$(702)
OID = Other (income)/deductions––net, included in the income statement account, Other deductions—net . OCI = Other comprehensive income/(loss) , included in the balance sheet account
Accumulated other comprehensive loss
.
Also includes gains and losses attributable to the hedged risk in fair value hedged relationships.
There was no significant ineffectiveness for any of the periods presented.
Amounts presented represent the effective portion of the gain or loss. For derivative financial instruments in cash flow hedge relationships, the effective portion is included in Other comprehensive
income/(loss)–derivative financial instruments . For derivative financial instruments in net investment hedge relationships and for foreign currency debt designated as hedging instruments, the effective
portion is included in
Other comprehensive income/(loss)
–
–
currency translation adjustment and other.
2011 Financial Report
81
(a), (b), (c)
(a), (d)
(a), (d)
(a)
(b)
(c)
(d)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The components of inventories follow:
The components of property, plant and equipment follow:
A. Goodwill
The components and changes in the carrying amount of goodwill follow:
8.
Inventories
AS OF DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
Finished goods
$2,765
$3,665
Work
-
in
-
process
4,119
3,727
Raw materials and supplies
885
883
Total inventories
$7,769
$8,275
The decrease in total inventories is primarily due to the inventory sold during 2011 that was acquired from Wyeth and had been recorded at fair value, partially offset by the acquisition of King (see
Note
2B. Acquisitions, Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Acquisition of King Pharmaceuticals, Inc.
) and the impact of foreign exchange.
Certain amounts of inventories are in excess of one year’s supply. There are no recoverability issues associated with those amounts.
9.
Property, Plant and Equipment
AS OF DECEMBER 31,
USEFUL LIVES
(MILLIONS OF DOLLARS)
(YEARS)
2011
2010
Land
—
$
747
$ 791
Buildings
33
/3
-
50
12,804
13,200
Machinery and equipment
8-
20
11,541
11,744
Furniture, fixtures and other
3
-
12
/2
4,291
4,643
Construction in progress
—
1,139
999
30,522
31,377
Less: Accumulated depreciation
13,584
12,732
Total property, plant and equipment
$16,938
$18,645
The decrease in total property, plant and equipment is primarily due to depreciation, disposals and impairments, partially offset by capital additions, the impact of foreign exchange and the acquisition of
King (see
Note 2B. Acquisitions, Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Acquisition of King Pharmaceuticals, Inc.)
.
10.
Goodwill and Other Intangible Assets
(MILLIONS OF DOLLARS)
P
RIMARY
C
ARE
S
PECIALTY
C
ARE
AND
O
NCOLOGY
E
STABLISHED
P
RODUCTS
AND
E
MERGING
M
ARKETS
A
NIMAL
H EALTH
AND
C
ONSUMER
H
EALTHCARE
N
UTRITION
O
THER
T
OTAL
Balance, January 1, 2010
$3,272
$ 9,010
$ 9,883
$ 154
$
—
$
20,038
$42,357
Additions
11
29
32
19
—
2,163
2,254
Other
(71
)
(195
)
(214
)
(14
)
—
(
189
)
(683)
Allocation of other goodwill
2,838
7,815
8,573
2,290
496
(22,012
)
—
Balance, December 31, 2010
6,050
16,659
18,274
2,449
496
—
43,928
Additions
129
300
321
55
—
—
805
Other
50
138
151
(7
)
2
—
334
Balance, December 31, 2011
$6,229
$17,097
$18,746
$2,497
$498
$
—
$
45,067
The Other goodwill related to our acquisition of Wyeth and was unallocated and subject to change until we completed the recording of the assets acquired and liabilities assumed (see Note 2A.
Acquisitions, Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Acquisition of Wyeth).
Beginning in the first quarter of 2011, our Company is managed through five operating segments, as shown in the table above (see also Note 18. Segment, Product and Geographic Area Information
for further discussion about the change in management approach). As part of the change, we have retrospectively presented goodwill according to the new operating segment structure.
Primarily reflects the impact of measurement period adjustments related to Wyeth (see Note 2A. Acquisitions, Divestitures, Collaborative Arrangements and Equity-Method Investments: Acquisition of
Wyeth)
.
Primarily reflects the impact of foreign exchange.
Primarily reflects the acquisition of King (see Note 2B. Acquisitions, Divestitures, Collaborative Arrangements and Equity-Method Investments: Acquisition of King Pharmaceuticals, Inc.) .
82
2011 Financial Report
(a), (b)
(a)
(b)
1
1
(a)
(a)
(a)
(b)
(c)
(d)
(b)
(e)
(d)
(a)
(b)
(c)
(d)
(e)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
B. Other Intangible Assets
The components of identifiable intangible assets follow:
At December 31, 2011, our identifiable intangible assets are associated with the following, as a percentage of identifiable intangible assets, less accumulated amortization:
There are no percentages for our Emerging Markets business unit as it is a geographic-area unit, not a product-based unit. The carrying value of the assets associated with our Emerging Markets
business unit is included within the assets associated with the other four biopharmaceutical business units.
For information about intangible asset impairments, see Note 4. Other Deductions––Net.
Developed Technology Rights
Developed technology rights represent the amortized cost associated with developed technology, which has been acquired from third parties and which can include the right to develop, use, market, sell
and/or offer for sale the product, compounds and intellectual property that we have acquired with respect to products, compounds and/or processes that have been completed. We possess a well-
diversified portfolio of hundreds of developed technology rights across therapeutic categories, primarily representing the commercialized products included in our five biopharmaceutical business units.
Virtually all of these assets were acquired in connection with our Wyeth acquisition in 2009 and our Pharmacia acquisition in 2003. The more significant components of developed technology rights are
the following (in order of significance): Prevnar 13/Prevenar 13 Infant and Enbrel and, to a lesser extent, Premarin, Prevnar 13/Prevenar 13 Adult, Effexor, Celebrex, Pristiq, Tygacil, BMP-2, BeneFIX,
Refacto AF and Genotropin. Also included in this category are the post-approval milestone payments made under our alliance agreements for certain biopharmaceutical products, such as Rebif and
Spiriva.
Brands
Brands represent the amortized or unamortized cost associated with tradenames and know-how, as the products themselves do not receive patent protection. Most of these assets are associated with
our Consumer Healthcare and Nutrition business units. Virtually all of these assets were acquired in connection with our Wyeth acquisition in 2009 and our Pharmacia acquisition in 2003. The more
significant components of indefinite-lived brands are the following (in order of significance): Advil, Xanax, Centrum, Medrol, 1st Age Nutrition and 2nd Age Nutrition. The more significant components of
finite-lived brands are the following (in order of significance): Depo-Provera, Advil Cold and Sinus, and Dimetapp.
AS OF DECEMBER 31,
2011
2010
(MILLIONS OF DOLLARS)
GROSS
CARRYING
AMOUNT
ACCUMULATED
AMORTIZATION
IDENTIFIABLE
INTANGIBLE
ASSETS, LESS
ACCUMULATED
AMORTIZATION
GROSS
CARRYING
AMOUNT
ACCUMULATED
AMORTIZATION
IDENTIFIABLE
INTANGIBLE
ASSETS, LESS
ACCUMULATED
AMORTIZATION
Finite-lived intangible assets:
Developed technology rights
$73,088
$(32,013)
$41,075
$68,432
$(26,223)
$42,209
Brands
1,678
(687)
991
1,626
(607)
1,019
License agreements
425
(215)
210
637
(248)
389
Other
623
(362)
261
533
(324)
209
Total finite-lived intangible assets
75,814
(33,277)
42,537
71,228
(27,402)
43,826
Indefinite-lived intangible assets:
Brands
10,027
—
10,027
10,219
—
10,219
In
-
process research and development
1,197
—
1,197
3,438
—
3,438
Trademarks
72
—
72
72
—
72
Total indefinite
-
lived intangible assets
11,296
—
11,296
13,729
—
13,729
Total identifiable intangible assets
$87,110
$(33,277)
$53,833
$84,957
$(27,402)
$57,555
In the fourth quarter of 2011, Prevenar 13 Adult and Vyndaqel (tafamidis meglumine) received regulatory approval in a major market, and as a result, we reclassified these assets, with a combined book
value of approximately $2.3 billion, from IPR&D to Developed Technology Rights and began to amortize the assets.
The decrease is primarily related to amortization and impairment charges (see Note 4. Other Deductions—Net ), partially offset by assets acquired as part of the acquisition of King (see Note 2B.
Acquisitions, Divestitures, Collaborative Arrangements and Equity
-
Method Investments: Acquisition of King Pharmaceuticals, Inc.)
and the impact of foreign exchange.
•
Developed technology rights: Specialty Care (64%); Established Products (17%); Primary Care (15%); Animal Health (2%); Oncology (1%); and Nutrition (1%)
•
Brands, finite
-
lived: Consumer Healthcare (57%); Established Products (29%); and Animal Health (14%)
•
Brands, indefinite
-
lived: Consumer Healthcare (51%); Established Products (26%); and Nutrition (23%)
•
IPR&D: Worldwide Research and Development (57%); Specialty Care (14%); Primary Care (14%); Established Products (8%); Oncology (5%); and Animal Health (2%)
2011 Financial Report
83
(a)
(a)
(b)
(a)
(b)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
In-Process Research and Development
IPR&D assets represent research and development assets that have not yet received regulatory approval in a major market. The majority of these IPR&D assets were acquired in connection with our
acquisition of Wyeth. The more significant components of IPR&D are a treatment for skin fibrosis and a program for the treatment of rheumatoid arthritis.
IPR&D assets are required to be classified as indefinite-lived assets until the successful completion or the abandonment of the associated research and development effort. Accordingly, during the
development period after the date of acquisition, these assets will not be amortized until approval is obtained in a major market, typically either the U.S. or the EU, or in a series of other countries, subject
to certain specified conditions and management judgment. At that time, we will determine the useful life of the asset, reclassify the asset out of in-process research and development and begin
amortization. If the associated research and development effort is abandoned, the related IPR&D assets will likely be written-off, and we will record an impairment charge.
As these compounds were approved in a major market, we reclassified the associated assets with a combined book value of approximately $2.3 billion from IPR&D to Developed Technology Rights and
began to amortize those assets.
For information about impairments of IPR&D assets, see Note 4. Other Deductions––Net.
For IPR&D assets, the risk of failure is significant and there can be no certainty that these assets ultimately will yield a successful product. The nature of the biopharmaceutical business is high-risk and,
as such, we expect that many of these IPR&D assets will become impaired and be written off at some time in the future.
Amortization
The weighted-average life of both our total finite-lived intangible assets and the largest component, Developed technology rights, is approximately 11 years. Total amortization expense for finite-lived
intangible assets was $5.8 billion in 2011, $5.5 billion in 2010 and $3.0 billion in 2009.
The annual amortization expense expected for the years 2012 through 2016 follows:
11.
Pension and Postretirement Benefit Plans and Defined Contribution Plans
The majority of our employees worldwide are covered by defined benefit pension plans, defined contribution plans or both. In the U.S., we have both qualified and supplemental (non-qualified) defined
benefit plans. A qualified plan meets the requirements of certain sections of the Internal Revenue Code, and, generally, contributions to qualified plans are tax deductible. A qualified plan typically
provides benefits to a broad group of employees with restrictions on discriminating in favor of highly compensated employees with regard to coverage, benefits and contributions. A supplemental (non-
qualified) plan provides additional benefits to certain employees. In addition, we provide medical and life insurance benefits to certain retirees and their eligible dependents through our postretirement
plans. In 2009, we assumed all of Wyeth’s defined benefit obligations and related plan assets for qualified and non-qualified pension plans and postretirement plans in connection with our acquisition of
Wyeth (see Note 2A. Acquisitions, Divestitures, Collaborative Arrangements and Equity-Method Investments: Acquisition of Wyeth ) .
Beginning on January 1, 2011, for employees hired in the U.S. and Puerto Rico after December 31, 2010, we no longer offer a defined benefit plan and, instead, offer an enhanced benefit under our
defined eligible contribution plan. In addition to the standard matching contribution by the Company, the enhanced benefit provides an automatic Company contribution for such eligible employees based
on age and years of service.
•
On December 30, 2011, the FDA approved the Company’s 13-valent pneumococcal conjugate vaccine, Prevenar 13, for active immunization for the prevention of pneumonia and invasive disease
caused by the 13 Streptococcus pnueumoniae serotypes contained in the vaccine in adults age 50 years and older. On October 25, 2011, the European Commission approved Prevenar 13 for active
immunization for the prevention of vaccine
-
type invasive disease caused by Streptococcus pneumoniae in adults age 50 years and older.
•
In November, 2011, FoldRx’s lead product candidate, Vyndaqel (tafamidis meglumine), was approved in the EU and our new drug application was accepted for review in the U.S. in February 2012.
This product is a first-in-class oral therapy for the treatment of transthyretin familial amyloid polyneuropathy (TTR-FAP), a progressively fatal genetic neurodegenerative disease, for which liver
transplant is the only treatment option currently available.
(MILLIONS OF DOLLARS)
2012
2013
2014
2015
2016
Amortization expense
$
5,350
$
4,856
$
4,150
$
3,741
$
3,494
84
2011 Financial Report
Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The annual cost and other amounts recognized in other comprehensive (income)/loss for our benefit plans follow:
The amounts in Accumulated other comprehensive income/(loss) expected to be amortized into 2012 net periodic benefit costs follow:
A.
Components of Net Periodic Benefit Costs and Other Amounts Recognized in Other Comprehensive (Income)/Loss
YEAR ENDED DECEMBER 31,
PENSION PLANS
U.S. QUALIFIED
U.S.
SUPPLEMENTAL
(NON-QUALIFIED)
INTERNATIONAL
POSTRETIREMENT
PLANS
(MILLIONS OF DOLLARS)
2011
2010
2009
2011
2010
2009
2011
2010
2009
2011
2010
2009
Service cost
$ 351
$347
$ 252
$ 36
$ 28
$ 24
$251
$230
$ 188
$ 68
$ 79
$ 39
Interest cost
734
740
526
72
77
53
453
427
342
195
211
145
Expected return on plan assets
(871
)
(782
)
(527
)
—
—
—
(
448
)
(434
)
(375
)
(35
)
(31
)
(26
)
Amortization of:
Actuarial losses
145
151
212
36
29
31
86
67
30
17
15
18
Prior service (credits)/costs
(8
)
2
2
(3
)
(2
)
(2
)
(5
)
(4
)
(3
)
(53
)
(38
)
(3
)
Curtailments and settlements
—
net
95
(52
)
110
23
1
(2
)
3
(3
)
3
(68
)
(23
)
(3
)
Special termination benefits
23
73
61
26
180
137
4
6
8
3
19
24
Net periodic benefit costs
469
479
636
190
313
241
344
289
193
127
232
194
Other changes recognized in other comprehensive (income)/loss
1,879
260
(783
)
36
117
(23
)
(365
)
152
1,004
421
(183
)
(122
)
Total recognized in net periodic benefit costs and other comprehensive (income)/loss
$2,348
$739
$(147
)
$226
$430
$218
$(21
)
$441
$1,197
$ 548
$ 49
$ 72
The acquisition of Wyeth during fourth quarter 2009 contributed to the increase in certain components of net periodic benefit costs, such as service cost and interest cost, which was largely offset by
higher expected returns on plan assets during 2010 from the inclusion of Wyeth plan assets. Further declines in interest rates during 2011 resulted in service costs continuing to increase on an overall
basis. The decrease in 2011 postretirement plans
’
service and interest costs is largely driven by the harmonization of the Wyeth plans.
For details, see
Note 6. Other Comprehensive Income/(Loss).
2011 vs. 2010 – The decrease in the U.S. qualified pension plans’ net periodic benefit costs was largely driven by lower special termination benefits costs and higher expected returns due to
contributions made to the plans, partially offset by lower curtailment gains and an increase in settlement costs associated with on-going restructuring efforts. 2010 vs. 2009 – The decrease in the U.S.
qualified pension plans
’
net periodic benefit costs was largely driven by curtailment gains and lower settlement charges associated with Wyeth
-
related restructuring initiatives.
2011 vs. 2010 – The decrease in the U.S. supplemental (non-qualified) plans’ net periodic benefit costs was primarily driven by lower special termination benefits costs associated with Wyeth-related
restructuring initiatives. 2010 vs. 2009 – The increase in the U.S. supplemental (non-qualified) plans’ net periodic benefit costs was primarily driven by special termination benefits recognized for
certain executives as part of ongoing Wyeth
-
related restructuring initiatives.
2011 vs. 2010 and 2010 vs. 2009 – The increase in the international plans’ net periodic benefit costs as compared to the prior year was primarily driven by changes in assumptions, including the
decrease in discount rates across most plans.
2011 vs. 2010 – The decrease in the postretirement plans’ net periodic benefit costs was due to the harmonization of the Wyeth postretirement medical program initiated in mid-2010 . 2010 vs. 2009 –
The increase postretirement plans
’
net periodic benefit costs was due to the Wyeth acquisition, offset partially by the postretirement harmonization program.
PENSION PLANS
(MILLIONS OF DOLLARS)
U.S. QUALIFIED
U.S. SUPPLEMENTAL
(NON
-
QUALIFIED)
INTERNATIONAL
POSTRETIREMENT
PLANS
Actuarial losses
$(320)
$(44)
$(69)
$(33)
Prior service credits and other
15
3
7
50
Total
$(305)
$(41)
$(62)
$17
2011 Financial Report
85
(c) (d) (e)
(f)
(a)
(a)
(a)
(b)
(a)
(b)
(c)
(d)
(e)
(f)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The weighted-average actuarial assumptions of our benefit plans follow:
The assumptions above are used to develop the benefit obligations at fiscal year-end and to develop the net periodic benefit cost for the subsequent fiscal year. Therefore, the assumptions used to
determine net periodic benefit cost for each year are established at the end of each previous year, while the assumptions used to determine benefit obligations are established at each year-end.
The net periodic benefit cost and the benefit obligations are based on actuarial assumptions that are reviewed on an annual basis. We revise these assumptions based on an annual evaluation of long-
term trends, as well as market conditions that may have an impact on the cost of providing retirement benefits.
The expected rates of return on plan assets for our U.S. qualified, international and postretirement plans represent our long-
term assessment of return expectations, which we may change based on shifts
in economic and financial market conditions. The 2011 expected rates of return for these plans reflect our long-term outlook for a globally diversified portfolio, which is influenced by a combination of
return expectations for individual asset classes, actual historical experience and our diversified investment strategy. The historical returns are one of the inputs used to provide context for the development
of our expectations for future returns. Using this information, we develop ranges of returns for each asset class and a weighted-average expected return for our targeted portfolio, which includes the
impact of portfolio diversification and active portfolio management.
The healthcare cost trend rate assumptions for our U.S. postretirement benefit plans follow:
A one-percentage-point increase or decrease in the healthcare cost trend rate assumed for postretirement benefits would have the following effects as of December 31, 2011:
Actuarial and other assumptions for pension and postretirement plans can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and
assumptions. For a description of the risks associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions .
B.
Actuarial Assumptions
(PERCENTAGES)
2011
2010
2009
Weighted-average assumptions used to determine benefit obligations:
Discount rate:
U.S. qualified pension plans
5.1
%
5.9
%
6.3
%
U.S. non
-
qualified pension plans
5.0
5.8
6.2
International pension plans
4.7
4.8
5.1
Postretirement plans
4.8
5.6
6.0
Rate of compensation increase:
U.S. qualified pension plans
3.5
4.0
4.0
U.S. non-qualified pension plans
3.5
4.0
4.0
International pension plans
3.3
3.5
3.6
Weighted
-
average assumptions used to determine net periodic benefit cost:
Discount rate:
U.S. qualified pension plans
5.9
6.3
6.4
U.S. non-qualified pension plans
5.8
6.2
6.4
International pension plans
4.8
5.1
5.6
Postretirement plans
5.6
6.0
6.4
Expected return on plan assets:
U.S. qualified pension plans
8.5
8.5
8.5
International pension plans
6.0
6.4
6.7
Postretirement plans
8.5
8.5
8.5
Rate of compensation increase:
U.S. qualified pension plans
4.0
4.0
4.3
U.S. non
-
qualified pension plans
4.0
4.0
4.3
International pension plans
3.5
3.6
3.2
2011
2010
Healthcare cost trend rate assumed for next year
7.8
%
8.0
%
Rate to which the cost trend rate is assumed to decline
4.5
%
4.5
%
Year that the rate reaches the ultimate trend rate
2027
2027
(MILLIONS OF DOLLARS)
INCREASE
DECREASE
Effect on total service and interest cost components
$ 18
$ (17
)
Effect on postretirement benefit obligation
304
(270
)
86
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
An analysis of the changes in our benefit obligations, plan assets and funded status of our benefit plans follow:
C.
Obligations and Funded Status
YEAR ENDED DECEMBER 31,
PENSION PLANS
U.S. QUALIFIED
U.S. SUPPLEMENTAL
(NON
-
QUALIFIED)
INTERNATIONAL
POSTRETIREMENT
PLANS
(MILLIONS OF DOLLARS)
2011
2010
2011
2010
2011
2010
2011
2010
Change in benefit obligation:
Benefit obligation at beginning of year
$13,035
$12,578
$ 1,401
$ 1,368
$ 9,132
$ 9,049
$ 3,582
$ 3,733
Service cost
351
347
36
28
251
230
68
79
Interest cost
734
740
72
77
453
427
195
211
Employee contributions
—
—
—
—
16
18
45
22
Plan amendments
(73)
(46)
(9)
(6)
4
(3)
(28)
(495)
Changes in actuarial assumptions and other
1,808
980
111
180
(536)
361
300
281
Foreign exchange impact
—
—
—
—
311
(504)
—
4
Acquisitions
56
1
—
(
1)
2
10
14
—
Curtailments
(97)
(233)
(10)
(29)
(121)
(33)
17
1
Settlements
(476)
(905)
(128)
(235)
(64)
(53)
—
—
Special termination benefits
23
73
26
180
4
6
3
19
Benefits paid
(526)
(500)
(68)
(161)
(398)
(376)
(296)
(273)
Benefit obligation at end of year
14,835
13,035
1,431
1,401
9,054
9,132
3,900
3,582
Change in plan assets:
Fair value of plan assets at beginning of year
10,596
9,977
—
—
6,699
6,516
414
370
Actual gain on plan assets
398
1,123
—
—
171
454
9
46
Company contributions
1,969
901
196
396
491
455
250
249
Employee contributions
—
—
—
—
16
18
45
22
Foreign exchange impact
—
—
—
—
203
(315)
—
—
Acquisitions
44
—
—
—
—
—
—
—
Settlements
(476)
(905)
(128)
(235)
(64)
(53)
—
—
Benefits paid
(526)
(500)
(68)
(161)
(398)
(376)
(296)
(273)
Fair value of plan assets at end of year
12,005
10,596
—
—
7,118
6,699
422
414
Funded status
—
Plan assets less than the benefit obligation at end of year
$(2,830)
$(2,439)
$(1,431)
$(1,401)
$(1,936)
$(2,433)
$(3,478)
$(3,168)
The unfavorable change in our U.S. qualified plans’ projected benefit obligations funded status was largely driven by changes in interest rates and lower than expected asset returns, partially offset by
plan contributions of $2.0 billion.
The U.S. supplemental (non-qualified) pension plans are not generally funded and these obligations, which are substantially greater than the annual cash outlay for these liabilities, are paid from cash
generated from operations.
The favorable change in our international plans’ projected benefit obligations funded status was largely driven by changes in actuarial assumptions, partially offset by the weakening of the U.S. dollar
against the U.K. pound and euro. Outside the U.S., in general, we fund our defined benefit plans to the extent that tax or other incentives exist and we have accrued liabilities on our consolidated
balance sheet to reflect those plans that are not fully funded.
The unfavorable change in our postretirement plans’ accumulated benefit obligations (ABO) funded status was largely driven by changes in actuarial assumptions.
For the U.S. and international pension plans, the benefit obligation is the projected benefit obligation. For the postretirement plans, the benefit obligation is the accumulated postretirement benefit
obligation. The ABO for all of our U.S. qualified pension plans was $13.8 billion in 2011 and $12.0 billion in 2010. The ABO for our U.S. supplemental (non-qualified) pension plans was $1.2 billion in
both 2011 and 2010. The ABO for our international pension plans was $8.3 billion in 2011 and $8.1 billion in 2010.
The U.S. qualified pension plans loan securities to other companies. Such securities may be onward loaned, sold or pledged by the other companies, but they may be required to be returned in a short
period of time. We also require cash collateral from these companies and a maintenance margin of 103% of the fair value of the collateral relative to the fair value of the loaned securities. As of
December 31, 2011, the fair value of collateral received was $2 million and, as of December 31, 2010, the fair value of collateral received was $581 million. The securities loaned continue to be included
in the table above in
Fair value of plan assets
, and the securities
-
lending program for the pension plans will be discontinued in 2012.
2011 Financial Report
87
(a) ( b)
(c)
(d)
(e)
(f)
(a)
(b)
(c)
(d)
(e)
(f)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The funded status is recognized in our consolidated balance sheets as follows:
The components of amounts recognized in Accumulated other comprehensive income/(loss) follow:
Information related to the funded status of selected benefit plans follows:
All of our U.S. plans were underfunded as of December 31, 2011.
AS OF DECEMBER 31,
PENSION PLANS
U.S. QUALIFIED
U.S. SUPPLEMENTAL
(NON
-
QUALIFIED)
INTERNATIONAL
POSTRETIREMENT
PLANS
(MILLIONS OF DOLLARS)
2011
2010
2011
2010
2011
2010
2011
2010
Noncurrent assets
$
—
$
—
$
—
$
—
$
329
$ 118
$
—
$
—
Current liabilities
—
—
(
130)
(156)
(41)
(41)
(134)
(133)
Noncurrent liabilities
(2,830)
(2,439)
(1,301)
(1,245)
(2,224)
(2,510)
(3,344)
(3,035)
Funded status
$ (2,830)
$ (2,439)
$ (1,431)
$ (1,401)
$ (1,936)
$ (2,433)
$ (3,478)
$ (3,168)
Included primarily in Taxes and other noncurrent assets .
Included in Other current liabilities .
Included in Pension benefit obligations and Postretirement benefit obligations , as appropriate.
AS OF DECEMBER 31,
PENSION PLANS
U.S. QUALIFIED
U.S. SUPPLEMENTAL
(NON-QUALIFIED)
INTERNATIONAL
POSTRETIREMENT
PLANS
(MILLIONS OF DOLLARS)
2011
2010
2011
2010
2011
2010
2011
2010
Actuarial losses
$ (4,638)
$ (2,699)
$ (566)
$ (525)
$ (2,020)
$ (2,388)
$ (759)
$ (451)
Prior service (costs)/credits and other
123
63
26
21
(21)
(18)
468
581
Total
$ (4,515)
$ (2,636)
$ (540)
$ (504)
$ (2,041)
$ (2,406)
$ (291)
$ 130
The actuarial losses primarily represent the cumulative difference between the actuarial assumptions and actual return on plan assets, changes in discount rates and changes in other assumptions
used in measuring the benefit obligations. These actuarial losses are recognized in Accumulated other comprehensive income/(loss) and are amortized into net periodic benefit costs over an average
period of 9.9 years for our U.S. qualified plans, an average period of 9.7 years for our U.S. supplemental (non-qualified) plans, an average period of 14 years for our international plans and an average
period of 11.1 years for our postretirement plans.
AS OF DECEMBER 31,
PENSION PLANS
U.S. SUPPLEMENTAL
U.S. QUALIFIED
(NON-QUALIFIED)
INTERNATIONAL
(MILLIONS OF DOLLARS)
2011
2010
2011
2010
2011
2010
Pension plans with an accumulated benefit obligation in excess of plan assets:
Fair value of plan assets
$ 12,005
$10,596
$
—
$
—
$
2,529
$ 2,228
Accumulated benefit obligation
13,799
11,953
1,225
1,177
4,446
4,069
Pension plans with a projected benefit obligation in excess of plan assets:
Fair value of plan assets
12,005
10,596
—
—
2,686
5,731
Projected benefit obligation
14,835
13,035
1,431
1,401
4,951
8,283
88
2011 Financial Report
(a)
(b)
(c)
(a)
(b)
(c)
(a)
(a)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The components of plan assets follow:
D.
Plan Assets
FAIR VALUE
FAIR VALUE
(MILLIONS OF DOLLARS)
AS OF
DECEMBER 31,
2011
LEVEL 1
LEVEL 2
LEVEL 3
AS OF
DECEMBER 31,
2010
LEVEL 1
LEVEL 2
LEVEL 3
U.S. qualified pension plans:
Cash and cash equivalents
$ 2,111
$
—
$
2,111
$
—
$
1,196
$
—
$
1,196
$
—
Equity securities:
Global equity securities
2,522
2,509
12
1
2,766
2,765
—
1
Equity commingled funds
1,794
—
1,794
—
1,708
—
1,708
–
–
Debt securities:
Fixed income commingled funds
870
—
870
—
817
—
817
—
Government bonds
808
—
805
3
660
—
660
—
Corporate debt securities
1,971
—
1,966
5
2,085
—
2,083
2
Other investments:
Private equity funds
920
—
—
920
899
—
—
899
Insurance contracts
353
353
—
—
—
—
—
Other
656
—
—
656
465
—
—
465
Total
12,005
2,509
7,911
1,585
10,596
2,765
6,464
1,367
International pension plans:
Cash and cash equivalents
311
—
311
—
518
—
518
—
Equity securities:
Global equity securities
1,513
1,432
81
—
1,458
1,166
292
—
Equity commingled funds
2,047
—
2,047
—
1,881
–
–
1,881
—
Debt securities:
Fixed income commingled funds
786
—
786
—
804
—
804
—
Government bonds
1,015
—
1,015
—
932
—
932
—
Corporate debt securities
542
—
542
—
376
—
376
—
Other investments:
Private equity funds
55
—
4
51
21
—
4
17
Insurance contracts
433
—
67
366
435
—
69
366
Other
416
—
67
349
274
—
59
215
Total
7,118
1,432
4,920
766
6,699
1,166
4,935
598
U.S. postretirement plans
:
Cash and cash equivalents
19
—
19
—
12
—
12
—
Equity securities:
Global equity securities
24
24
–
–
—
29
29
—
—
Equity commingled funds
17
—
17
—
18
—
18
—
Debt securities:
Fixed income commingled funds
8
—
8
—
9
—
9
—
Government bonds
8
—
8
—
7
—
7
—
Corporate debt securities
19
—
19
—
21
—
21
—
Other investments
Insurance contracts
312
—
312
—
306
—
306
—
Others
15
—
15
—
12
—
12
—
Total
$ 422
$ 24
$ 398
$
—
$
414
$ 29
$ 385
$
—
Fair values are determined based on valuation inputs categorized as Level 1, 2 or 3 (see Note 1E. Significant Accounting Policies: Fair Value ).
Reflects postretirement plan assets, which support a portion of our U.S. retiree medical plans.
An analysis of changes in our more significant investments valued using significant unobservable inputs follows:
ACTUAL RETURN ON PLAN
ASSETS
(MILLIONS OF DOLLARS)
FAIR VALUE
BEGINNING
OF YEAR
ASSETS
HELD,
END OF YEAR
ASSETS SOLD
DURING THE
PERIOD
PURCHASES,
SALES AND
SETTLEMENTS,
NET
TRANSFER
INTO/(OUT OF)
LEVEL 3
EXCHANGE
RATE
CHANGES
FAIR
VALUE,
END OF
YEAR
2011
U.S. qualified pension plans:
Private equity funds
$ 899
$ (246)
$ 55
$ 212
$
—
$
—
$
920
Other
465
24
(6
)
173
—
—
656
International pension plans:
Insurance contracts
366
8
—
(
12
)
(15
)
19
366
Other
215
(4
)
—
120
12
6
349
2010
U.S. qualified pension plans:
Private equity funds
843
45
42
(31
)
—
—
899
Other
454
21
—
(
10
)
—
—
465
International pension plans:
Insurance contracts
346
12
—
(
10
)
52
(34
)
366
Other
127
(3
)
—
37
58
(4
)
215
2011 Financial Report
89
( a ) ( a )
(b)
(a)
(b)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
A single estimate of fair value can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For a description of our general
accounting policies associated with developing fair value estimates, see Note 1E. Significant Accounting Policies: Fair Value.
For a description of the risks associated with estimates and assumptions, see
Note 1C. Significant Accounting Policies: Estimates and Assumptions.
Specifically, the following methods and assumptions were used to estimate the fair value of our pension and postretirement plans’ assets:
We periodically review the methodologies, inputs and outputs of third-party pricing services for reasonableness.
The long-term target asset allocations ranges and the percentage of the fair value of plan assets for benefit plans follow:
We utilize long-term asset allocation ranges in the management of our plans’ invested assets. Our long-term return expectations are developed based on a diversified, global investment strategy that
takes into account historical experience, as well as the impact of portfolio diversification, active portfolio management, and our view of current and future economic and financial market conditions. As
market conditions and other factors change, we may adjust our targets accordingly and our asset allocations may vary from the target allocations.
Our long-term asset allocation ranges reflect our asset class return expectations and tolerance for investment risk within the context of the respective plans’ long-term benefit obligations. These ranges
are supported by analysis that incorporates historical and expected returns by asset class, as well as volatilities and correlations across asset classes and our liability profile. This analysis, referred to as
an asset-liability analysis, also provides an estimate of expected returns on plan assets, as well as a forecast of potential future asset and liability balances.
The plans’ assets are managed with the objectives of minimizing pension expense and cash contributions over the long term. Asset liability studies are performed periodically in order to support asset
allocations.
The investment managers of each separately managed account are permitted to use derivative securities as described in their investment management agreements.
Investment performance is reviewed on a monthly basis in total, as well as by asset class and individual manager, relative to one or more benchmarks. Investment performance and detailed statistical
analysis of both investment performance and portfolio holdings are conducted, a large portion of which is presented to senior management on a quarterly basis. Periodic formal meetings are held with
each investment manager to review the investments.
It is our practice to fund amounts for our qualified pension plans that are at least sufficient to meet the minimum requirements set forth in applicable employee benefit laws and local tax laws.
•
Cash and cash equivalents, Equity commingled funds, Fixed-income commingled funds––observable prices.
•
Global equity securities
—
quoted market prices.
•
Government bonds, Corporate debt securities
—
observable market prices.
•
Other investments—principally unobservable inputs that are significant to the estimation of fair value. These unobservable inputs could include, for example, the investment managers’ assumptions
about earnings multiples and future cash flows.
AS OF DECEMBER 31,
TARGET
ALLOCATION
PERCENTAGE
PERCENTAGE OF PLAN ASSETS
(PERCENTAGES)
2011
2011
2010
U.S. qualified pension plans:
Cash and cash equivalents
0
-
5
17.6
11.3
Equity securities
25
-
50
36.0
42.2
Debt securities
30
-
55
30.4
33.6
Real estate and other investments
10
-
15
16.0
12.9
Total
100
100.0
100.0
International pension plans:
Cash and cash equivalents
0
-
5
4.4
7.7
Equity securities
25
-
50
50.0
49.8
Debt securities
30
-
55
32.9
31.6
Real estate and other investments
10
-
15
12.7
10.9
Total
100
100.0
100.0
U.S. postretirement plans:
Cash and cash equivalents
0
-
5
4.6
2.9
Equity securities
5
-
20
9.7
11.3
Debt securities
5
-
20
8.1
8.9
Real estate, insurance contracts and other investments
65
-
80
77.6
76.9
Total
100
100.0
100.0
E.
Cash Flows
90
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The expected future cash flow information related to our benefit plans follows:
The table reflects the total U.S. and international plan benefits projected to be paid from the plans or from our general assets under the current actuarial assumptions used for the calculation of the benefit
obligation and, therefore, actual benefit payments may differ from projected benefit payments.
F. Defined Contribution Plans
We have savings and investment plans in several countries, including the U.S., U.K., Japan, Spain and the Netherlands. For the U.S. plans, employees may contribute a portion of their salaries and
bonuses to the plans, and we match, largely in company stock or company stock units, a portion of the employee contributions. In the U.S., the matching contributions in company stock are sourced
through open market purchases. Employees are permitted to subsequently diversify all or any portion of their company matching contribution. The contribution match for certain legacy Pfizer U.S.
participants is held in an employee stock ownership plan. We recorded charges related to our plans of $288 million in 2011, $259 million in 2010 and $191 million in 2009.
A. Common Stock
During 2009, in connection with our acquisition of Wyeth on October 15, 2009, we issued approximately 1.3 billion shares of common stock, which were previously held as Pfizer treasury stock, to former
Wyeth shareholders to partially fund the acquisition. The excess of the average cost of Pfizer treasury stock issued over the fair value of the stock portion of the consideration transferred to acquire Wyeth
was recorded as a reduction to
Retained Earnings . We purchase our common stock via privately negotiated transactions or in open market purchases as circumstances and prices warrant. Purchased
shares under each of the share-purchase plans, which are authorized by our Board of Directors, are available for general corporate purposes.
From June 2005 through year-end 2011, we purchased approximately 1.2 billion shares of our stock for approximately $28 billion. On February 1, 2011, we announced that the Board of Directors
authorized a new $5 billion share-purchase plan. On December 12, 2011, we announced that the Board of Directors authorized an additional $10 billion share-purchase plan. In 2011, we purchased
approximately 459 million shares of our common stock for approximately $9.0 billion. In 2010, we purchased approximately 61 million shares of our common stock for approximately $1.0 billion. We did
not purchase any shares of our common stock in 2009.
After giving effect to share purchases through year-end 2011, our remaining share-purchase authorization is approximately $10 billion at December 31, 2011.
B. Preferred Stock
The Series A convertible perpetual preferred stock is held by an Employee Stock Ownership Plan (Preferred ESOP) Trust and provides dividends at the rate of 6.25%, which are accumulated and paid
quarterly. The per-share stated value is $40,300 and the preferred stock ranks senior to our common stock as to dividends and liquidation rights. Each share is convertible, at the holder’s option, into
2,574.87 shares of our common stock with equal voting rights. The conversion option is indexed to our common stock and requires share settlement, and, therefore, is reported at the fair value at the date
of issuance. We may redeem the preferred stock at any time or upon termination of the Preferred ESOP, at our option, in cash, in shares of common stock or, a combination of both at a price of $40,300
per share.
C. Employee Stock Ownership Plans
We have two employee stock ownership plans (collectively, the ESOPs), the Preferred ESOP and another that holds common stock of the Company (Common ESOP).
Allocated shares held by the Common ESOP are considered outstanding for the earnings per share (EPS) calculations and the eventual conversion of allocated preferred shares held by the Preferred
ESOP is assumed in the diluted EPS calculation. As of December 31, 2011, the Preferred ESOP held preferred shares with a stated value of approximately $45 million, convertible into approximately
3 million shares of our common stock. As of December 31, 2011, the Common ESOP held approximately 4 million shares of our common stock. As of December 31, 2011, all preferred and common
shares held by the ESOPs have been allocated to the Pharmacia U.S. and certain Puerto Rico savings plan participants.
PENSION PLANS
(MILLIONS OF DOLLARS)
U.S.
QUALIFIED
U.S.
SUPPLEMENTAL
(NON
-
QUALIFIED)
INTERNATIONAL
POST
RETIREMENT
PLANS
Expected employer contributions:
2012
$ 19
$ 130
$ 431
$ 394
Expected benefit payments:
2012
$ 874
$ 130
$ 394
$ 295
2013
806
173
403
308
2014
825
174
416
317
2015
819
165
436
326
2016
839
141
455
331
2017
–
2021
4,891
706
2,496
1,780
12.
Equity
2011 Financial Report
91

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
D. Employee Benefit Trust
The Pfizer Inc. Employee Benefit Trust (EBT) was established in 1999 to fund our employee benefit plans through the use of its holdings of Pfizer Inc. stock. Our consolidated balance sheets reflect the
fair value of the shares owned by the EBT as a reduction of Shareholders’ equity. Beginning in May 2009, the Company began using the shares held in the EBT to help fund the Company’s matching
contribution in the Pfizer Savings Plan.
Our compensation programs can include share-based payments, in the form of stock options, Restricted Stock Units (RSUs), Performance Share Awards (PSAs) and Total Shareholder Return Units
(TSRUs).
The Company’s shareholders approved the amendment and restatement of the 2004 Stock Plan at the Annual Meeting of Shareholders held on April 23, 2009. The primary purpose of the amendment
was to increase the number of shares of common stock available for grants by 425 million shares. In addition, the amendment provided other changes, including that the number of stock options, Stock
Appreciation Rights (SARs) (now known as TSRUs) or other performance-based awards that may be granted to any one individual during any 36-month period is limited to eight million shares, and that
RSUs, PSAs and restricted stock grants count as two shares, while stock options and TSRUs count as one share, toward the maximums for the incremental 425 million shares. As of December 31, 2011,
319 million shares were available for award. The 2004 Stock Plan, as amended, is the only Pfizer plan under which equity-based compensation may currently be awarded to executives and other
employees.
The Company’s shareholders originally approved the 2004 Stock Plan at the Annual Meeting of Shareholders held on April 22, 2004, and, effective upon that approval, new stock option and other share-
based awards could be granted only under the originally approved 2004 Stock Plan. As originally approved, the 2004 Stock Plan allowed a maximum of three million shares to be awarded to any
employee per year and 475 million shares in total. RSUs, PSAs and restricted stock grants counted as three shares, while stock options and SARs counted as one share, toward the maximums under the
Plan, as originally approved.
Although not required to do so, we have used authorized and unissued shares and, to a lesser extent, shares held in our Employee Benefit Trust and treasury stock to satisfy our obligations under these
programs.
A. Impact on Net Income
The components of share-based compensation expense and the associated tax benefit follow:
Amounts capitalized as part of inventory cost and the impact of modifications under our cost-reduction and productivity initiatives to share-based awards were not significant for any period presented.
Generally, the modifications resulted in an acceleration of vesting, either in accordance with plan terms or at management’s discretion.
B. Stock Options
Stock options are issued to select employees and, when vested, entitle the holder to purchase a specified number of shares of Pfizer common stock at a price per share equal to the closing market price
of Pfizer common stock on the date of grant.
All eligible employees may receive stock option grants. No stock options were awarded to senior and other key management in any period presented; however, stock options were awarded to certain
other employees. In virtually all instances, stock options granted since 2005 vest after three years of continuous service from the grant date and have a contractual term of 10 years. In most cases, stock
options must be held for at least one year from the grant date before any vesting may occur. In the event of a divestiture or restructuring, options held by employees are immediately vested and are
exercisable for a period from three months to their remaining term, depending on various conditions.
We measure the value of stock option grants as of the grant date using, for virtually all grants, the Black-Scholes-Merton option-pricing model. The values determined through this fair-value-
based method
generally are amortized on a straight-line basis over the vesting term into Cost of sales, Selling, informational and administrative expenses , and Research and development expenses , as appropriate.
13.
Share-Based Payments
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Stock option expense
$166
$150
$165
RSU expense
228
211
183
TSRU expense
17
28
15
Directors
’
compensation and other
5
2
3
PSA expense/(expense reduction)
3
14
(17
)
Share
-
based payment expense
419
405
349
Tax benefit for share-based compensation expense
(139
)
(129
)
(99
)
Share-based payment expense, net of tax
$280
$276
$250
92
2011 Financial Report
Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The weighted-average assumptions used in the valuation of stock options follow:
The following table summarizes all stock option activity during 2011:
The following table provides data related to all stock option activity:
C. Restricted Stock Units (RSUs)
RSUs are issued to select employees and, when vested, entitle the holder to receive a specified number of shares of Pfizer common stock, including shares resulting from dividend equivalents paid on
such RSUs. For RSUs granted during the periods presented, in virtually all instances, the units vest after three years of continuous service from the grant date.
We measure the value of RSU grants as of the grant date using the closing price of Pfizer common stock. The values determined through this fair-value-based method generally are amortized on a
straight-line basis over the vesting term into Cost of sales, Selling, informational and administrative expenses , and Research and development expenses , as appropriate.
The following table summarizes all RSU activity during 2011:
YEAR ENDED DECEMBER 31,
2011
2010
2009
Expected dividend yield
4.14
%
4.00
%
4.90
%
Risk-free interest rate
2.59
%
2.87
%
2.69
%
Expected stock price volatility
25.55
%
26.85
%
41.36
%
Expected term
(years)
6.25
6.25
6.0
Determined using a constant dividend yield during the expected term of the option.
Determined using the interpolated yield on U.S. Treasury zero-coupon issues.
Determined using implied volatility, after consideration of historical volatility.
Determined using historical exercise and post-vesting termination patterns.
SHARES
(THOUSANDS)
WEIGHTED-
AVERAGE
EXERCISE PRICE
PER SHARE
WEIGHTED-
AVERAGE
REMAINING
CONTRACTUAL TERM
(YEARS)
AGGREGATE
INTRINSIC
VALUE
(MILLIONS)
Outstanding, December 31, 2010
458,604
$28.29
Granted
66,850
18.92
Exercised
(9,406
)
16.31
Forfeited
(6,513
)
17.41
Canceled
(79,982
)
38.73
Outstanding, December 31, 2011
429,553
25.31
4.9
$751
Vested and expected to vest
, December 31, 2011
421,754
25.46
4.9
$715
Exercisable, December 31, 2011
273,563
30.09
3.0
$17
Market price of underlying Pfizer common stock less exercise price.
The number of options expected to vest takes into account an estimate of expected forfeitures.
YEAR ENDED/AS OF
DECEMBER 31,
(MILLIONS OF DOLLARS, EXCEPT PER STOCK OPTION AMOUNTS)
2011
2010
2009
Weighted
-
average grant date fair value per stock option
$
3.15
$
3.25
$
3.30
Aggregate intrinsic value on exercise
32
5
2
Cash received upon exercise
153
16
7
Tax benefits realized related to exercise
10
1
1
Total compensation cost related to nonvested stock options not yet recognized,
pre
-
tax
$
177
$
178
$
147
Weighted-average period over which stock option compensation cost is expected to
be recognized (years)
1.3
1.3
1.2
SHARES
(THOUSANDS)
WEIGHTED-
AVERAGE
GRANT DATE
FAIR VALUE
PER SHARE
Nonvested, December 31, 2010
41,177
$17.57
Granted
15,671
18.91
Vested
(13,281)
20.99
Reinvested dividend equivalents
1,740
19.28
Forfeited
(3,367)
17.27
Nonvested, December 31, 2011
41,940
$17.08
2011 Financial Report
93
(a)
(b)
(c)
(d)
(
a
)
( b )
(
c
)
(
d
)
(a)
(b)
(a
)
(b )

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The following table provides data related to all RSU activity:
PSAs are awarded to senior and other key members of management. The target number of shares is determined by reference to the fair value of share-based awards to similar employees in the industry
peer group.
We measure the value of PSA grants as of the grant date using a Monte Carlo simulation model. The values determined through this fair-value-based methodology generally are amortized on a straight-
line basis over the vesting term into Cost of sales, Selling, informational and administrative expenses , and Research and development expenses , as appropriate.
The weighted-average assumptions used in the valuation of PSAs follow:
TSRUs are awarded to senior and other key management. The contractual terms for TSRUs were for 5 years for certain awards and for 7 years for the balance of the awards in 2011, and for 5 years for
all awards in each of 2009 and 2010. The target number of shares is determined by reference to the fair value of share-based awards to similar employees in the industry peer group.
We measure the value of TSRU grants as of the grant date using a Monte Carlo simulation model. The values determined through this fair-value-based methodology generally are amortized on a straight-
line basis over the vesting term into Cost of sales, Selling, informational and administrative expenses , and Research and development expenses , as appropriate.
The weighted-average assumptions used in the valuation of TSRUs follow:
(MILLIONS OF DOLLARS)
YEAR ENDED DECEMBER
31,
2011
2010
2009
Total grant date fair
-
value
-
based amount of shares vested
$279
$311
$131
Total compensation cost related to nonvested RSU awards not yet recognized, pre
-
tax
$264
$230
$198
Weighted-average period over which RSU cost is expected to be recognized (years)
1.3
1.4
1.3
D.
Performance Share Awards (PSAs)
YEAR ENDED DECEMBER
31,
2011
2010
2009
Risk-free interest rate
1.22
%
1.24
%
1.95
%
Expected Pfizer stock price volatility
25.55
%
26.75
%
40.40
%
Average peer stock price volatility
21.63
%
23.64
%
36.30
%
Contractual term (years)
3
3
3
Determined using the interpolated yield on U.S. Treasury zero-coupon issues.
Determined using implied volatility, after consideration of historical volatility.
E.
Total Shareholder Return Units (TSRUs)
YEAR ENDED DECEMBER
31,
2011
2010
2009
Expected dividend yield
4.15%
3.99%
4.55
%
Risk
-
free interest rate
2.51%
2.34%
2.35
%
Expected stock price volatility
25.55%
26.76%
36.92
%
Contractual term (years)
5.95
5.00
5.00
Determined using a constant dividend yield during the expected term of the TSRU.
Determined using the interpolated yield on U.S. Treasury zero-coupon issues.
Determined using implied volatility, after consideration of historical volatility.
94
2011 Financial Report
(a)
(b)
(b)
( a )
(b
)
(a)
(b)
(c)
( a )
(
b
)
(
c
)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
14.
Earnings per Common Share Attributable to Common Shareholders
Basic and diluted EPS were computed using the following common share data:
15.
Lease Commitments
We lease properties and equipment for use in our operations. In addition to rent, the leases may require us to pay directly for taxes, insurance, maintenance and other operating expenses or to pay higher
rent when operating expenses increase. Rental expense, net of sublease income, was $382 million in 2011, $387 million in 2010 and $356 million in 2009.
The future minimum rental commitments under non-cancelable operating leases follow:
16.
Insurance
Our insurance coverage reflects market conditions (including cost and availability) existing at the time it is written, and our decision to obtain insurance coverage or to self-insure varies accordingly.
Depending upon the cost and availability of insurance and the nature of the risk involved, the amount of self-insurance may be significant. The cost and availability of coverage have resulted in self-
insuring certain exposures, including product liability. If we incur substantial liabilities that are not covered by insurance or substantially exceed insurance coverage and that are in excess of existing
accruals, there could be a material adverse effect on our results of operations or cash flows in the period in which the amounts are paid and/or accrued (see Note 17. Commitments and Contingencies ).
17.
Commitments and Contingencies
We and certain of our subsidiaries are subject to numerous contingencies arising in the ordinary course of business. For a discussion of our tax contingencies, see Note 5D. Taxes on Income: Tax
Contingencies .
LEGAL PROCEEDINGS
Our non-tax contingencies include, among others, the following:
YEAR ENDED DECEMBER 31,
(IN MILLIONS)
2011
2010
2009
EPS Numerator
—
Basic:
Income from continuing operations
$ 8,739
$8,211
$8,529
Less: Net income attributable to noncontrolling interests
42
31
8
Income from continuing operations attributable to Pfizer Inc.
8,697
8,180
8,521
Less: Preferred stock dividends—net of tax
2
2
2
Income from continuing operations attributable to Pfizer Inc. common shareholders
8,695
8,178
8,519
Discontinued operations
—
net of tax
1,312
77
114
Net income attributable to Pfizer Inc. common shareholders
$10,007
$8,255
$8,633
EPS Numerator
—
Diluted:
Income from continuing operations attributable to Pfizer Inc. common shareholders and assumed conversions
$ 8,697
$8,180
$8,521
Discontinued operations—net of tax
1,312
77
114
Net income attributable to Pfizer Inc. common shareholders and assumed conversions
$10,009
$8,257
$8,635
EPS Denominator:
Weighted
-
average number of common shares outstanding
—
Basic
7,817
8,036
7,007
Common-share equivalents: stock options, stock issuable under employee compensation plans and convertible preferred stock
53
38
38
Weighted
-
average number of common shares outstanding
—
Diluted
7,870
8,074
7,045
Stock options that had exercise prices greater than the average market price of our common stock issuable under employee compensation plans
272
413
400
These common stock equivalents were outstanding during 2011, 2010 and 2009 but were not included in the computation of diluted EPS for those years because their inclusion would have had an anti-
dilutive effect.
(MILLIONS OF DOLLARS)
2012
2013
2014
2015
2016
AFTER
2016
Lease commitments
$187
$166
$144
$105
$83
$723
•
Patent litigation, which typically involves challenges to the coverage and/or validity of our patents on various products or processes. We are the plaintiff in the vast majority of these actions. An
adverse outcome in actions in which we are the plaintiff could result in a loss of patent protection for the drug at issue, a significant loss of revenues from that drug and impairments of any associated
assets.
•
Product liability and other product-related litigation, which can include personal injury, consumer, off-label promotion, securities-law, antitrust and breach of contract claims, among others, often
involves highly complex issues relating to medical causation, label warnings and reliance on those warnings, scientific evidence and findings, actual provable injury and other matters.
2011 Financial Report
95
(a)
(a)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Certain of these contingencies could result in losses, including damages, fines and/or civil penalties, and/or criminal charges, which could be substantial.
We believe that our claims and defenses in these matters are substantial, but litigation is inherently unpredictable and excessive verdicts do occur. We do not believe that any of these matters will have a
material adverse effect on our financial position. However, we could incur judgments, enter into settlements or revise our expectations regarding the outcome of certain matters, and such developments
could have a material adverse effect on our results of operations or cash flows in the period in which the amounts are paid and/or accrued.
We have accrued for losses that are both probable and reasonably estimable. Substantially all of these contingencies are subject to significant uncertainties and, therefore, determining the likelihood of a
loss and/or the measurement of any loss can be complex. Consequently, we are unable to estimate the range of reasonably possible loss in excess of amounts accrued. Our assessments are based on
estimates and assumptions that have been deemed reasonable by management, but the assessment process relies heavily on estimates and assumptions that may prove to be incomplete or inaccurate,
and unanticipated events and circumstances may occur that might cause us to change those estimates and assumptions.
Amounts recorded for legal and environmental contingencies can result from a complex series of judgments about future events and uncertainties and can rely heavily on estimates and assumptions. For
information about the risks associated with estimates and assumptions, see Note 1C. Significant Accounting Policies: Estimates and Assumptions.
The principal pending matters to which we are a party are discussed below. In determining whether a pending matter is a principal matter, we consider both quantitative and qualitative factors in order to
assess materiality, such as, among other things, the amount of damages and the nature of any other relief sought in the proceeding, if such damages and other relief are specified; our view of the merits
of the claims and of the strength of our defenses; whether the action purports to be a class action and our view of the likelihood that a class will be certified by the court; the jurisdiction in which the
proceeding is pending; any experience that we or, to our knowledge, other companies have had in similar proceedings; whether disclosure of the action would be important to a reader of our financial
statements, including whether disclosure might change a reader’s judgment about our financial statements in light of all of the information about the Company that is available to the reader; the potential
impact of the proceeding on our reputation; and the extent of public interest in the matter. In addition, with respect to patent matters, we consider, among other things, the financial significance of the
product protected by the patent. As a result of considering qualitative factors in our determination of principal matters, there are some matters discussed below with respect to which management believes
that the likelihood of possible loss in excess of amounts accrued is remote.
A. Patent Litigation
Like other pharmaceutical companies, we are involved in numerous suits relating to our patents, including but not limited to those discussed below. Most of the suits involve claims by generic drug
manufacturers that patents covering our products, processes or dosage forms are invalid and/or do not cover the product of the generic manufacturer. Also, counterclaims, as well as various independent
actions, have been filed claiming that our assertions of, or attempts to enforce, our patent rights with respect to certain products constitute unfair competition and/or violations of the antitrust laws. In
addition to the challenges to the U.S. patents on a number of our products that are discussed below, we note that the patent rights to certain of our products are being challenged in various other
countries.
ACTIONS IN WHICH WE ARE THE PLAINTIFF AND CERTAIN RELATED ACTIONS
Lipitor (atorvastatin)
In November 2008, Apotex Inc. notified us that it had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Lipitor. In December 2008, we filed patent-
infringement suits against Apotex Inc. in the U.S. District Court for the District of Delaware and the U.S. District Court for the Northern District of Illinois. In August 2009, our action in the District of
Delaware was transferred to the Northern District of Illinois and consolidated with our pending action there. Apotex Inc. asserts the invalidity of our patent covering the crystalline form of atorvastatin,
which (including the six
-month pediatric exclusivity period) expires in 2017. We assert the infringement of our crystalline patent and are defending against the allegations of invalidity.
In November 2011, our previously reported patent-infringement actions related to Lipitor against KUDCO Ireland, Ltd. and Kremers Urban LLC and against Aurobindo Pharma Ltd. in the U.S. District
Court for the District of Delaware were settled on terms that are not material to Pfizer.
Lipitor began to face generic competition in the U.S. in November 2011.
In the U.K., while the basic patent for Lipitor expired in November 2011, the exclusivity period has been extended by six months to May 2012 by virtue of the supplementary protection certificate and
pediatric extension. In September 2011, Dr. Reddy’s Laboratories (UK) Limited filed an action in the High Court of Justice seeking revocation of the six-month pediatric extension. We are defending this
action, which is based upon the interpretation of the EU Pediatric Medicines Regulation.
•
Commercial and other litigation, which can include merger
-
related and product
-
pricing claims and environmental claims and proceedings, can involve complexities that will vary from matter to matter.
•
Government investigations, which often are related to the extensive regulation of pharmaceutical companies by national, state and local government agencies in the U.S. and in other countries.
96
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Caduet (atorvastatin/amlodipine combination)
In December 2011, our previously reported patent-infringement action related to Caduet against Sandoz, Inc., a division of Novartis AG (Sandoz), in the U.S. District Court for the District of Delaware was
voluntarily dismissed by us.
Caduet began to face generic competition in the U.S. in November 2011.
Viagra (sildenafil)
In March 2010, we brought a patent-infringement action in the U.S. District Court for the Eastern District of Virginia against Teva Pharmaceuticals USA, Inc. (Teva USA) and Teva Pharmaceutical
Industries Ltd. (Teva Pharmaceutical Industries), which had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Viagra. Teva USA and Teva
Pharmaceutical Industries assert the invalidity and non-infringement of the Viagra use patent, which expires in 2019, but have not challenged the basic patent, which expires in 2012. In August 2011, the
court ruled that our Viagra use patent is valid and infringed, thereby preventing Teva USA and Teva Pharmaceutical Industries from receiving approval for a generic version of Viagra before October
2019. In September 2011, Teva USA and Teva Pharmaceutical Industries appealed the decision to the U.S. Court of Appeals for the Federal Circuit.
In October 2010, we filed a patent-infringement action with respect to Viagra in the U.S. District Court for the Southern District of New York against Apotex Inc. and Apotex Corp., Mylan Pharmaceuticals
Inc. and Mylan Inc., Actavis, Inc. and Amneal Pharmaceuticals LLC. These generic manufacturers have filed abbreviated new drug applications with the FDA seeking approval to market their generic
versions of Viagra. They assert the invalidity and non-infringement of the Viagra use patent, but have not challenged the basic patent.
In May and June 2011, respectively, Watson Laboratories Inc. (Watson) and Hetero Labs Limited (Hetero) notified us that they had filed abbreviated new drug applications with the FDA seeking approval
to market their generic versions of Viagra. Each asserts the invalidity and non-infringement of the Viagra use patent. Neither has challenged the basic patent. In June and July 2011, respectively, we filed
actions against Watson and Hetero in the U.S. District Court for the Southern District of New York asserting the validity and infringement of the use patent.
Sutent (sunitinib malate)
In May 2010, Mylan Pharmaceuticals Inc. notified us that it had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Sutent and challenging on various
grounds the Sutent basic patent, which expires in 2021, and two other patents, which expire in 2020 and 2021. In June 2010, we filed suit against Mylan Pharmaceuticals Inc. in the U.S. District Court for
the District of Delaware asserting the infringement of those three patents.
Detrol and Detrol LA (tolterodine)
In January 2008, Impax Laboratories, Inc. (Impax) notified us that it had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Detrol LA. Impax is
challenging on various grounds the basic patent, which (including the six-month pediatric exclusivity period) expires in 2012, and three formulation patents, which (including the six-month pediatric
exclusivity period) expire in 2020. We filed an action against Impax in the U.S. District Court for the Southern District of New York asserting the infringement of the basic patent and two of the formulation
patents. This action subsequently was transferred to the U.S. District Court for the District of New Jersey.
In March 2008 and May 2010, respectively, Sandoz and Mylan Pharmaceuticals Inc. notified us that they had filed abbreviated new drug applications with the FDA seeking approval to market generic
versions of Detrol LA. They assert the invalidity and/or non-infringement of three formulation patents for Detrol LA. They have not challenged the basic patent. In June 2010, we filed actions against
Sandoz and Mylan Pharmaceuticals Inc. in the U.S. District Court for the District of New Jersey asserting the infringement of two of the formulation patents.
In April 2011, Impax notified us that it had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Detrol. Impax asserts the non-infringement of the basic
patent, which (including the six-month pediatric exclusivity period) expires in 2012. In June 2011, we filed an action against Impax in the U.S. District Court for the District of New Jersey asserting
infringement of the basic patent.
In June 2011, Torrent Pharmaceuticals Ltd. (Torrent) notified us that it had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Detrol LA. Torrent
asserted the invalidity and non-infringement of three formulation patents. Torrent did not challenge the basic patent. In July 2011, we filed an action against Torrent in the U.S. District Court for the District
of New Jersey asserting the validity and infringement of the challenged patents. In February 2012, this action was settled on terms that are not material to Pfizer.
Lyrica (pregabalin)
Beginning in March 2009, several generic manufacturers notified us that they had filed abbreviated new drug applications with the FDA seeking approval to market generic versions of Lyrica capsules
and, in the case of one generic manufacturer, Lyrica oral solution. Each of the generic manufacturers is challenging one or more of three patents for Lyrica: the basic patent, which expires in 2018, and
two other patents, which expire in 2013 and 2018. Each of the generic manufacturers asserts the invalidity and/or the non-
infringement of the patents subject to challenge. Beginning in April 2009, we filed
actions against these generic manufacturers in the U.S. District Court for the District of Delaware asserting the infringement and validity of our patents for Lyrica. All of these cases have been
consolidated in the District of Delaware.
In November 2010, Novel Laboratories, Inc. (Novel) notified us that it had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Lyrica oral solution and
asserting the invalidity and/or infringement of our three patents for Lyrica referred to above. In January 2011, we filed an action against Novel in the U.S. District Court for the District of Delaware asserting
the validity and infringement of all three patents.
2011 Financial Report
97

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Apotex Inc. notified us, in May and June 2011, respectively, that it had filed abbreviated new drug applications with the FDA seeking approval to market generic versions of Lyrica oral solution and Lyrica
capsules. Apotex Inc. asserts the invalidity and non-infringement of the basic patent, as well as the seizure patent that expires in 2013. In July 2011, we filed an action against Apotex Inc. in the U.S.
District Court for the District of Delaware asserting the validity and infringement of the challenged patents in connection with both of the abbreviated new drug applications.
In October 2011, Alembic Pharmaceuticals Limited (Alembic) notified us that it had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Lyrica capsules
and asserting the invalidity of the basic patent. In December 2011, we filed an action against Alembic in the U.S. District Court for the District of Delaware asserting the validity and infringement of the
basic patent.
We also have filed patent-infringement actions in Canada against certain generic manufacturers who are seeking approval to market generic versions of Lyrica capsules in that country.
Zyvox (linezolid)
In December 2009, Teva Parenteral Medicines Inc. (Teva Parenteral) notified us that it had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Zyvox.
Teva Parenteral asserts the invalidity and non-infringement of the basic Zyvox patent, which (including the six-month pediatric exclusivity period) expires in 2015, and another patent that expires in 2021.
In January 2010, we filed suit against Teva Parenteral in the U.S. District Court for the District of Delaware asserting the infringement of the basic patent.
Relpax (eletriptan)
In June 2010, we received notices from Apotex Inc. and Apotex Corp. and from Teva USA that they had filed abbreviated new drug applications with the FDA seeking approval to market generic versions
of Relpax. They asserted the non-infringement of our patent covering the crystalline form of eletriptan, which expires in 2017. They did not challenge the basic patent, which expires in 2016. In July 2010,
we filed actions against Apotex Inc. and Apotex Corp. and against Teva USA in the U.S. District Court for the Southern District of New York asserting the infringement of the crystalline patent. In July
2011, the action against Teva USA was settled on terms that are not material to Pfizer. In October 2011, the action against Apotex Inc. and Apotex Corp. was voluntarily dismissed by the parties without
prejudice.
Protonix (pantoprazole sodium)
Wyeth has a license to market Protonix in the U.S. from Nycomed GmbH (Nycomed), which owns the patents relating to Protonix. The basic patent (including the six-
month pediatric exclusivity period) for
Protonix expired in January 2011.
Following their respective filings of abbreviated new drug applications with the FDA, Teva USA and Teva Pharmaceutical Industries, Sun Pharmaceutical Advanced Research Centre Ltd. and Sun
Pharmaceutical Industries Ltd. (collectively, Sun) and KUDCO Ireland, Ltd. (KUDCO Ireland) received final FDA approval to market their generic versions of Protonix 20mg and 40mg delayed-release
tablets. Wyeth and Nycomed filed actions against those generic manufacturers in the U.S. District Court for the District of New Jersey, which subsequently were consolidated into a single proceeding,
alleging infringement of the basic patent and seeking declaratory and injunctive relief. Following the court’s denial of a preliminary injunction sought by Wyeth and Nycomed, Teva USA and Teva
Pharmaceutical Industries and Sun launched their generic versions of Protonix tablets at risk in December 2007 and January 2008, respectively. Wyeth launched its own generic version of Protonix
tablets in January 2008, and Wyeth and Nycomed filed amended complaints in the pending patent-infringement action seeking compensation for damages resulting from Teva USA’s, Teva
Pharmaceutical Industries’ and Sun’s at-risk launches.
In April 2010, the jury in the pending patent-infringement action upheld the validity of the basic patent for Protonix. In July 2010, the court upheld the jury verdict, but it did not issue a judgment against
Teva USA, Teva Pharmaceutical Industries or Sun because of their other claims relating to the patent that still are pending. Wyeth and Nycomed will continue to pursue all available legal remedies
against those generic manufacturers, including compensation for damages resulting from their at-risk launches.
Separately, Wyeth and Nycomed are defendants in purported class actions brought by direct and indirect purchasers of Protonix in the U.S. District Court for the District of New Jersey. Plaintiffs seek
damages, on behalf of the respective putative classes, for the alleged violation of antitrust laws in connection with the procurement and enforcement of the patents for Protonix. These purported class
actions have been stayed pending resolution of the underlying patent litigation in the U.S. District Court for the District of New Jersey.
Rapamune (sirolimus)
In March 2010, Watson and Ranbaxy Laboratories Limited (Ranbaxy) notified us that they had filed abbreviated new drug applications with the FDA seeking approval to market generic versions of
Rapamune. Watson and Ranbaxy assert the invalidity and non-infringement of a method-of-use patent which (including the six-month pediatric exclusivity period) expires in 2014 and a solid-dosage
formulation patent which (including the six-month pediatric exclusivity period) expires in 2018. In April 2010, we filed actions against Watson and Ranbaxy in the U.S. District Court for the District of
Delaware and against Watson in the U.S. District Court for the Southern District of Florida asserting the infringement of the method-of-use patent. In June 2010, our action in the Southern District of
Florida was transferred to the District of Delaware and consolidated with our pending action there.
98
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Tygacil (tigecycline)
In October 2009, Sandoz notified Wyeth that it had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Tygacil. Sandoz asserts the invalidity and non-
infringement of two of Wyeth’s patents relating to Tygacil, including the basic patent, which expires in 2016. In December 2009, Wyeth filed suit against Sandoz in the U.S. District Court for the District of
Delaware asserting infringement of the basic patent.
Avinza (morphine sulfate)
King Pharmaceuticals, Inc. (King) and Elan Pharma International LTD (EPI) brought a patent-
infringement action in the U.S. District Court for the District of New Jersey against Sandoz in July 2009 as the
result of its abbreviated new drug application with the FDA seeking approval to market a generic version of Avinza. Sandoz is challenging a formulation patent for Avinza, which is owned by EPI, that
expires in 2017.
EpiPen
King brought patent-infringement actions against Sandoz in the U.S District Court for the District of New Jersey in July 2010 and against Teva Pharmaceutical Industries and Intelliject, Inc. (Intelliject) in
the U.S. District Court for the District of Delaware in August 2009 and January 2011, respectively, as the result of their abbreviated new drug applications with the FDA seeking approval to market
epinephrine injectable products. The two actions in Delaware subsequently were consolidated. Sandoz and Teva Pharmaceutical Industries are challenging and Intelliject challenged two patents, which
expire in 2025, covering the next generation autoinjector for use with epinephrine that is sold under the EpiPen brand name. In February 2012, the action against Intelliject was settled. Under the
settlement agreement, Intellject may launch its epinephrine injectable product no earlier than November 15, 2012, subject to final approval by the FDA.
Embeda (morphine sulfate/naltrexone hydrochloride extended-release capsules)
In August 2011, Watson Laboratories Inc. –
Florida (Watson Florida) notified us that it had filed an abbreviated new drug application with the FDA seeking approval to market a generic version of Embeda
extended-release capsules. Watson Florida asserts the invalidity and non-infringement of three formulation patents that expire in 2027. In October 2011, we filed an action against Watson Florida in the
U.S. District Court for the District of Delaware asserting the infringement of, and defending against the allegations of the invalidity of, the three formulation patents.
Torisel (temsirolimus)
In November 2011, Sandoz and Accord Healthcare, Inc. USA and certain of its affiliates (collectively, Accord) notified us that they had filed abbreviated new drug applications with the FDA seeking
approval to market generic versions of Torisel. Sandoz and Accord assert the invalidity and non-infringement of two patents for Torisel, including the basic patent, which expires in 2014. In December
2011, we filed suit against Sandoz and Accord in the U.S. District Court for the District of Delaware asserting the infringement of, and defending against the allegation of the invalidity of, the basic patent.
ACTION IN WHICH WE ARE THE DEFENDANT AND A RELATED ACTION
ReFacto AF and Xyntha
In February 2008, Novartis Vaccines and Diagnostics, Inc. (Novartis) filed suit against Wyeth and a subsidiary of Wyeth in the U.S. District Court for the Eastern District of Texas alleging that Wyeth’s
ReFacto AF and Xyntha products infringe two Novartis patents. Novartis’s complaint seeks damages, including treble damages, for alleged willful infringement. Wyeth and its subsidiary assert, among
other things, the invalidity and non-infringement of the Novartis patents. In November 2009, Novartis added a third patent to its infringement claim against Wyeth and its subsidiary. In August 2010,
Novartis granted Wyeth and its subsidiary a covenant not to sue on the third patent and withdrew that patent from its pending action.
In May 2008, a subsidiary of Wyeth filed suit in the U.S. District Court for the District of Delaware against Novartis seeking a declaration that the two Novartis patents initially asserted against Wyeth and
its subsidiary in the action referred to in the preceding paragraph are invalid on the ground that the Wyeth subsidiary was the first to invent the subject matter. In February 2010, the District of Delaware
declined to invalidate those two Novartis patents. In March 2010, the Wyeth subsidiary appealed the decision to the U.S. Court of Appeals for the Federal Circuit. In August 2011, the Federal Circuit
affirmed the District Court’s decision. In November 2011, the Federal Circuit denied the Wyeth subsidiary’s petition for a rehearing. The Federal Circuit’s decision does not address the defenses that
Wyeth and its subsidiary are asserting in the action referred to in the previous paragraph.
B. Product Litigation
Like other pharmaceutical companies, we are defendants in numerous cases, including but not limited to those discussed below, related to our pharmaceutical and other products. Plaintiffs in these cases
seek damages and other relief on various grounds for alleged personal injury and economic loss.
Asbestos
Quigley Company, Inc. (Quigley), a wholly owned subsidiary, was acquired by Pfizer in 1968 and sold products containing small amounts of asbestos until the early 1970s. In September 2004, Pfizer and
Quigley took steps that were intended to resolve all pending and future claims against Pfizer and Quigley in which the claimants allege personal injury from exposure to Quigley products containing
asbestos, silica or mixed dust. We recorded a charge of $369 million pre-tax ($229 million after-tax) in the third quarter of 2004 in connection with these matters.
•
Quigley
2011 Financial Report
99

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
In September 2004, Quigley filed a petition in the U.S. Bankruptcy Court for the Southern District of New York seeking reorganization under Chapter 11 of the U.S. Bankruptcy Code. In March 2005,
Quigley filed a reorganization plan in the Bankruptcy Court that needed the approval of 75% of the voting claimants, as well as the Bankruptcy Court and the U.S. District Court for the Southern District of
New York. In connection with that filing, Pfizer entered into settlement agreements with lawyers representing more than 80% of the individuals with claims related to Quigley products against Quigley and
Pfizer. The agreements provide for a total of $430 million in payments, of which $215 million became due in December 2005 and has been and is being paid to claimants upon receipt by the Company of
certain required documentation from each of the claimants. The reorganization plan provided for the establishment of a Trust (the Trust) for the evaluation and, as appropriate, payment of all unsettled
pending claims, as well as any future claims alleging injury from exposure to Quigley products.
In February 2008, the Bankruptcy Court authorized Quigley to solicit an amended reorganization plan for acceptance by claimants. According to the official report filed with the court by the balloting agent
in July 2008, the requisite votes were cast in favor of the amended plan of reorganization.
The Bankruptcy Court held a confirmation hearing with respect to Quigley’s amended plan of reorganization that concluded in December 2009. In September 2010, the Bankruptcy Court declined to
confirm the amended reorganization plan. As a result of the foregoing, Pfizer recorded additional charges for this matter of approximately $1.3 billion pre-tax (approximately $800 million after-tax) in 2010.
Further, in order to preserve its right to address certain legal issues raised in the court’s opinion, in October 2010, Pfizer filed a notice of appeal and motion for leave to appeal the Bankruptcy Court’s
decision denying confirmation.
In March 2011, Pfizer entered into a settlement agreement with a committee (the Ad Hoc Committee) representing approximately 40,000 claimants in the Quigley bankruptcy proceeding (the Ad Hoc
Committee claimants). Consistent with the additional charges recorded in 2010 referred to above, the principal provisions of the settlement agreement provide for a settlement payment in two installments
and other consideration, as follows:
Following the execution of the settlement agreement with the Ad Hoc Committee, Quigley filed a revised plan of reorganization and accompanying disclosure statement with the Bankruptcy Court in April
2011. Under the revised plan, and consistent with the additional charges recorded in 2010 referred to above, we expect to contribute an additional amount to the Trust, if and when the Bankruptcy Court
confirms the plan, of cash and non-cash assets (including insurance proceeds) with a value in excess of $550 million. The Bankruptcy Court must find that the revised plan meets the requisite standards
of the U.S. Bankruptcy Code before it confirms the plan. We expect that, if approved by claimants, confirmed by the Bankruptcy Court and the District Court and upheld on any subsequent appeal, the
revised reorganization plan will result in the District Court entering a permanent injunction directing pending claims, as well as future claims, alleging personal injury from exposure to Quigley products to
the Trust. There is no assurance that the plan will be confirmed by the courts.
In a separately negotiated transaction with an insurance company in August 2004, we agreed to a settlement related to certain insurance coverage which provides for payments to an insurance proceeds
trust established by Pfizer and Quigley over a ten-year period of amounts totaling $405 million. Most of these insurance proceeds, as well as other payments from insurers that issued policies covering
Pfizer and Quigley, would be paid, following confirmation, to the Trust for the benefit of present unsettled and future claimants with claims arising from exposure to Quigley products.
Between 1967 and 1982, Warner-Lambert owned American Optical Corporation, which manufactured and sold respiratory protective devices and asbestos safety clothing. In connection with the sale of
American Optical in 1982, Warner-Lambert agreed to indemnify the purchaser for certain liabilities, including certain asbestos-related and other claims. As of December 31, 2011, approximately 67,700
claims naming American Optical and numerous other defendants were pending in various federal and state courts seeking damages for alleged personal injury from exposure to asbestos and other
allegedly hazardous materials. Warner-Lambert is actively engaged in the defense of, and will continue to explore various means to resolve, these claims.
Warner-Lambert and American Optical brought suit in state court in New Jersey against the insurance carriers that provided coverage for the asbestos and other allegedly hazardous materials claims
related to American Optical. A majority of the carriers subsequently agreed to pay for a portion of the costs of defending and resolving those claims. The litigation continues against the carriers who have
disputed coverage or how costs should be allocated to their policies, and the court held that Warner-Lambert and American Optical are entitled to payment from each of those carriers of a proportionate
share of the costs associated with those claims. Under New Jersey law, a special allocation master was appointed to implement certain aspects of the court’s rulings.
•
the payment to the Ad Hoc Committee, for the benefit of the Ad Hoc Committee claimants, of a first installment of $500 million upon receipt by Pfizer of releases of asbestos-related claims against
Pfizer Inc. from Ad Hoc Committee claimants holding $500 million in the aggregate of claims (Pfizer began paying this first installment in June 2011);
•
the payment to the Ad Hoc Committee, for the benefit of the Ad Hoc Committee claimants, of a second installment of $300 million upon Pfizer’s receipt of releases of asbestos-related claims against
Pfizer Inc. from Ad Hoc Committee claimants holding an additional $300 million in the aggregate of claims following the earlier of the effective date of a revised plan of reorganization and April 6, 2013;
•
the payment of the Ad Hoc Committee
’
s legal fees and expenses incurred in this matter up to a maximum of $19 million (Pfizer began paying these legal fees and expenses in May 2011); and
•
the procurement by Pfizer of insurance for the benefit of certain Ad Hoc Committee claimants to the extent such claimants with non-malignant diseases have a future disease progression to a
malignant disease (Pfizer procured this insurance in August 2011).
•
Other Matters
100
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Numerous lawsuits are pending against Pfizer in various federal and state courts seeking damages for alleged personal injury from exposure to products containing asbestos and other allegedly
hazardous materials sold by Gibsonburg Lime Products Company (Gibsonburg). Gibsonburg was acquired by Pfizer in the 1960s and sold products containing small amounts of asbestos until the early
1970s.
There also is a small number of lawsuits pending in various federal and state courts seeking damages for alleged exposure to asbestos in facilities owned or formerly owned by Pfizer or its subsidiaries.
Celebrex and Bextra
Beginning in late 2004, actions, including purported class actions, were filed in various federal and state courts against Pfizer, Pharmacia Corporation (Pharmacia) and certain current and former officers,
directors and employees of Pfizer and Pharmacia. These actions include (i) purported class actions alleging that Pfizer and certain current and former officers of Pfizer violated federal securities laws by
misrepresenting the safety of Celebrex and Bextra, and (ii) purported class actions filed by persons who claim to be participants in the Pfizer or Pharmacia Savings Plan alleging that Pfizer and certain
current and former officers, directors and employees of Pfizer or, where applicable, Pharmacia and certain former officers, directors and employees of Pharmacia, violated certain provisions of the
Employee Retirement Income Security Act of 1974 (ERISA) by selecting and maintaining Pfizer stock or Pharmacia stock as an investment alternative when it allegedly no longer was a suitable or
prudent investment option. In June 2005, the federal securities and ERISA actions were transferred for consolidated pre-trial proceedings to a Multi-
District Litigation (In re Pfizer Inc. Securities, Derivative
and “ERISA” Litigation MDL-1688) in the U.S. District Court for the Southern District of New York.
In 2003, several purported class action complaints were filed in the U.S. District Court for the District of New Jersey against Pharmacia, Pfizer and certain former officers of Pharmacia. The plaintiffs seek
damages, alleging that the defendants violated federal securities laws by misrepresenting the data from a study concerning the gastrointestinal effects of Celebrex. These cases were consolidated for pre-
trial proceedings in the District of New Jersey (Alaska Electrical Pension Fund et al. v. Pharmacia Corporation et al.). In January 2007, the court certified a class consisting of all persons who purchased
Pharmacia securities from April 17, 2000 through February 6, 2001 and were damaged as a result of the decline in the price of Pharmacia’s securities allegedly attributable to the misrepresentations.
In October 2007, the court granted defendants’ motion for summary judgment and dismissed the plaintiffs’ claims. In November 2007, the plaintiffs appealed the decision to the U.S. Court of Appeals for
the Third Circuit. In January 2009, the Third Circuit vacated the District Court’s grant of summary judgment in favor of defendants and remanded the case to the District Court for further proceedings. The
Third Circuit also held that the District Court erred in determining that the class period ended on February 6, 2001, and directed that the class period end on August 5, 2001. In June 2009, the District
Court stayed proceedings in the case pending a determination by the U.S. Supreme Court with regard to defendants’ petition for certiorari seeking reversal of the Third Circuit’s decision. In May 2010, the
U.S. Supreme Court denied defendants’ petition for certiorari, and the case was remanded to the District Court for further proceedings.
Pfizer and several predecessor and affiliated companies, including Monsanto Company (Monsanto), are defendants in an action brought by Brigham Young University (BYU) and a BYU professor in the
U.S. District Court for the District of Utah alleging, among other things, breach by Monsanto of a 1991 research agreement with BYU. Plaintiffs claim that research under that agreement led to the
discovery of Celebrex and that, as a result, they are entitled to a share of the profits from Celebrex sales. Plaintiffs seek, among other things, compensatory and punitive damages.
Various Drugs: Off-Label Promotion Actions
In May 2010, a purported class action was filed in the U.S. District Court for the Southern District of New York against Pfizer and several of our current and former officers. The complaint alleges that the
defendants violated federal securities laws by failing to disclose that Pfizer was engaged in off-label marketing of certain drugs. Plaintiffs seek damages in an unspecified amount.
In June 2010, Health Care Service Corporation (HCSC), for itself and its affiliates, Blue Cross and Blue Shield plans in Illinois, New Mexico, Oklahoma and Texas, filed an action against us in the U.S.
District Court for the Eastern District of Texas. In July 2010, HCSC amended its complaint. The complaint, as amended, alleges that we engaged in deceptive marketing activities, including off-label
promotion, and the payment of improper remuneration to healthcare professionals with respect to Bextra and Celebrex in violation of, among other things, the federal Racketeer Influenced and Corrupt
Organizations (RICO) Act and the Illinois Consumer Fraud Act. In December 2010, this action was transferred to a Multi-District Litigation (In re Celebrex and Bextra Marketing, Sales Practices and
Product Liability Litigation MDL-1699) in the U.S. District Court for the Northern District of California. In July 2010, HCSC also filed a separate lawsuit against us in the U.S. District Court for the Eastern
District of Texas including substantially similar allegations regarding Geodon, Lyrica and Zyvox.
•
Securities and ERISA Actions
•
Securities Action in New Jersey
•
Other
•
Securities Action
•
Actions by Health Care Service Corporation
2011 Financial Report
101

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
In this latter action, in October 2011, HCSC filed an amended complaint that is substantially similar to the original complaint except that it no longer includes allegations regarding Lyrica or claims under
the Illinois Consumer Fraud Act. In both actions, HCSC seeks to recover the amounts that it paid for the specified drugs on behalf of its members in Illinois, New Mexico, Oklahoma, and Texas, as well as
treble damages and punitive damages.
Hormone-Replacement Therapy
Pfizer and certain wholly owned subsidiaries and limited liability companies, including Wyeth and King, along with several other pharmaceutical manufacturers, have been named as defendants in
approximately 10,000 actions in various federal and state courts alleging personal injury or economic loss related to the use or purchase of certain estrogen and progestin medications prescribed for
women to treat the symptoms of menopause. Although new actions are occasionally filed, the number of new actions was not significant in 2011, and we do not expect a substantial change in the rate of
new actions being filed. Plaintiffs in these suits allege a variety of personal injuries, including breast cancer, ovarian cancer, stroke and heart disease. Certain co-
defendants in some of these actions have
asserted indemnification rights against Pfizer and its affiliated companies. The cases against Pfizer and its affiliated companies involve one or more of the following products, all of which remain approved
by the FDA: femhrt (which Pfizer divested in 2003); Activella and Vagifem (which are Novo Nordisk products that were marketed by a Pfizer affiliate from 2000 to 2004); Premarin, Prempro, Aygestin,
Cycrin and Premphase (which are legacy Wyeth products); and Provera, Ogen, Depo-Estradiol, Estring and generic MPA (which are legacy Pharmacia & Upjohn products). The federal cases have been
transferred for consolidated pre-trial proceedings to a Multi-District Litigation (In re Prempro Products Liability Litigation MDL-1507) in the U.S. District Court for the Eastern District of Arkansas. Certain of
the federal cases have been remanded to their respective District Courts for further proceedings including, if necessary, trial.
This litigation consists of individual actions, a few purported statewide class actions and a purported provincewide class action in Quebec, Canada, a statewide class action in California and a nationwide
class action in Canada. In March 2011, in an action against Wyeth seeking the refund of the purchase price paid for Wyeth’s hormone-replacement therapy products by individuals in the State of
California during the period from January 1995 to January 2003, the U.S. District Court for the Southern District of California certified a class consisting of all individual purchasers of such products in
California who actually heard or read Wyeth’s alleged misrepresentations regarding such products. This is the only hormone-replacement therapy action to date against Pfizer and its affiliated companies
in the U.S. in which a class has been certified. In addition, in August 2011, in an action against Wyeth seeking damages for personal injury, the Supreme Court of British Columbia certified a class
consisting of all women who were prescribed Premplus and/or Premarin in combination with progestin in Canada between January 1, 1997 and December 1, 2003 and who thereafter were diagnosed
with breast cancer.
Pfizer and its affiliated companies have prevailed in many of the hormone-replacement therapy actions that have been resolved to date, whether by voluntary dismissal by the plaintiffs, summary
judgment, defense verdict or judgment notwithstanding the verdict; a number of these cases have been appealed by the plaintiffs. Certain other hormone
-replacement therapy actions have resulted in
verdicts for the plaintiffs and have included the award of compensatory and, in some instances, punitive damages; each of these cases has been appealed by Pfizer and/or its affiliated companies. The
decisions in a few of the cases that had been appealed by Pfizer and/or its affiliated companies or by the plaintiffs have been upheld by the appellate courts, while several other cases that had been
appealed by Pfizer and/or its affiliated companies or by the plaintiffs have been remanded by the appellate courts to their respective trial courts for further proceedings. Trials of additional hormone-
replacement therapy actions are underway or scheduled in 2012.
As of December 31, 2011, Pfizer and its affiliated companies had settled, or entered into definitive agreements or agreements-in-principle to settle, approximately 52% of the hormone-replacement
therapy actions pending against us and our affiliated companies. We have recorded aggregate charges with respect to those actions, as well as with respect to the actions that have resulted in verdicts
against us or our affiliated companies, of $336 million in 2011 and $300 million in prior years. In addition, we have recorded a charge of $359 million in 2011 that provides for the minimum expected costs
to resolve all remaining hormone-replacement therapy actions against Pfizer and its affiliated companies, consistent with our current ability to quantify such future costs. The $359 million charge is an
estimate and, while we cannot reasonably estimate the range of reasonably possible loss in excess of the amount accrued for these contingencies given the uncertainties inherent in this product liability
litigation, as described below, additional charges may be required in the future.
Most of the unresolved actions against Pfizer and/or its affiliated companies have been outstanding for more than five years and could take many more years to resolve. However, opportunistic
settlements could occur at any time. The litigation process is time-consuming, as every hormone-replacement action being litigated involves contested issues of medical causation and knowledge of risk.
Even though the vast majority of hormone-replacement therapy actions concern breast cancer, the underlying facts (e.g., medical causation, family history, reliance on warnings, physician/patient
interaction, analysis of labels, actual provable injury and other critical factors) can differ significantly from action to action, and the process of discovery has not yet begun for a majority of the unresolved
actions. Our ability to estimate the range of possible loss in excess of amounts accrued is complicated by these factors. In addition, the hormone-replacement therapy litigation involves fundamental
issues of science and medicine that often are uncertain and continue to evolve. Key scientific court rulings may have a significant impact on the litigation as a whole. An integral part of the litigation
process involves understanding the evolving science, as well as seeking key scientific rulings. Equally important, the discovery process is lengthy and complex and has not yet begun for a majority of the
unresolved actions. Therefore, we may not have sufficient information to determine the percentage of unresolved actions that could be impacted by scientific developments and/or key scientific rulings.
Our ability to estimate the range of possible loss in excess of amounts accrued is complicated by these fundamental issues of science and medicine, because we do not know how the science may
evolve, how the courts will rule on key motions or which unresolved actions will be impacted by these scientific matters.
•
Personal Injury and Economic Loss Actions
102
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Accordingly, we cannot reasonably estimate the range of possible loss in excess of amounts accrued for these contingencies.
Pfizer and/or its affiliated companies also have received inquiries from various federal and state agencies and officials relating to the marketing of their hormone-
replacement products. In November 2008,
the State of Nevada filed an action against Pfizer, Pharmacia & Upjohn Company and Wyeth in state court in Nevada alleging that they had engaged in deceptive marketing of their respective hormone-
replacement therapy medications in Nevada in violation of the Nevada Deceptive Trade Practices Act. The action seeks monetary relief, including civil penalties and treble damages. In February 2010, the
action was dismissed by the court on the grounds that the statute of limitations had expired. In July 2011, the Nevada Supreme Court reversed the dismissal and remanded the case to the district court for
further proceedings.
Zoloft and Effexor
A number of individual lawsuits and multi-plaintiff lawsuits have been filed against us and/or our subsidiaries in various federal and state courts alleging personal injury as a result of the purported
ingesting of Zoloft or Effexor.
Beginning in May 2011, purported class actions were filed in certain federal courts against Wyeth and, in certain of the actions, affiliates of Wyeth and certain other defendants relating to Effexor XR,
which is the extended
-release formulation of Effexor. The plaintiffs in each of these actions seek to represent a class consisting of all persons in the U.S. and its territories who purchased Effexor XR or
generic Effexor XR directly (in certain of the actions) or indirectly (in the other actions) from any of the defendants from June 14, 2008 until the time the defendants’ allegedly unlawful conduct ceased (the
Class Period). The plaintiffs allege delay in the launch of generic Effexor XR in the U.S. and its territories, in violation of federal antitrust laws and, in the indirect-purchaser actions, the antitrust, consumer
protection and various other laws of certain states, as the result of Wyeth fraudulently obtaining and improperly listing certain patents for Effexor XR, enforcing certain patents for Effexor XR, and entering
into litigation settlement agreements with various generic manufacturers with respect to Effexor XR. Each of the actions seeks treble damages on behalf of the putative class for alleged price overcharges
for Effexor XR or generic Effexor XR in the U.S. and its territories during the Class Period. All of the purported class actions brought by direct purchasers have been consolidated in the U.S. District Court
for the District of New Jersey, and all of the purported class actions brought by indirect purchasers have been separately consolidated in the same court. In addition, a few individual actions are pending in
the same court that assert claims and seek relief for the plaintiffs that are substantially similar to the claims asserted and the relief sought in the purported class actions.
Neurontin
A number of lawsuits, including purported class actions, have been filed against us in various federal and state courts alleging claims arising from the promotion and sale of Neurontin. The plaintiffs in the
purported class actions seek to represent nationwide and certain statewide classes consisting of persons, including individuals, health insurers, employee benefit plans and other third-party payers, who
purchased or reimbursed patients for the purchase of Neurontin that allegedly was used for indications other than those included in the product labeling approved by the FDA. In 2004, many of the suits
pending in federal courts, including individual actions as well as purported class actions, were transferred for consolidated pre-trial proceedings to a Multi-District Litigation (In re Neurontin Marketing,
Sales Practices and Product Liability Litigation MDL-1629) in the U.S. District Court for the District of Massachusetts.
In the Multi-District Litigation, in 2009, the court denied the plaintiffs’ renewed motion for certification of a nationwide class of all consumers and third-party payers who allegedly purchased or reimbursed
patients for the purchase of Neurontin for off-label uses from 1994 through 2004. In May 2011, the court denied a motion to reconsider its class certification ruling.
In 2010, the Multi-District Litigation court partially granted the Company’s motion for summary judgment, dismissing the claims of all of the proposed class representatives for third-
party payers and four of
the six proposed class representatives for individual consumers. In June 2011, the plaintiffs whose claims were dismissed appealed both the dismissal and the denial of class certification to the U.S. Court
of Appeals for the First Circuit.
Also in the Multi-District Litigation, in February 2011, a third-party payer who was not included in the proposed class action appealed a dismissal order to the U.S. Court of Appeals for the First Circuit.
Plaintiffs are seeking certification of statewide classes of Neurontin purchasers in actions pending in California, Illinois and Oklahoma. State courts in New York, Pennsylvania, Missouri and New Mexico
have declined to certify statewide classes of Neurontin purchasers. In November 2011, the plaintiff in the Missouri action and a proposed intervenor appealed the denial of class certification.
In January 2011, the U.S. District Court for the District of Massachusetts entered an order trebling a jury verdict against us in an action by a third-party payer seeking damages for the alleged off-label
promotion of Neurontin in violation of the federal Racketeer Influenced and Corrupt Organizations (RICO) Act. The verdict was for $47.4 million, which was subject to automatic trebling to $142.1 million
under the RICO Act. In November 2010, the court had entered a separate verdict against us in the amount of $65.4 million, together with prejudgment interest, under California’s Unfair Trade Practices
law relating to the same alleged conduct, which amount is included within and is not additional to the $142.1 million trebled amount of the jury verdict. In August 2011, we appealed the District Court’s
judgment to the U.S. Court of Appeals for the First Circuit.
•
Government Inquiries; Action by State of Nevada
•
Personal Injury Actions
•
Antitrust Actions
•
Off
-
Label Promotion Actions in the U.S.
2011 Financial Report
103

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
A number of individual lawsuits have been filed against us in various U.S. federal and state courts and in certain other countries alleging suicide, attempted suicide and other personal injuries as a result
of the purported ingesting of Neurontin. Certain of the U.S. federal actions have been transferred for consolidated pre-trial proceedings to the same Multi-District Litigation referred to in the first paragraph
of this section.
In addition, purported class actions have been filed against us in various Canadian provincial courts alleging claims arising from the promotion, sale and labeling of Neurontin and generic gabapentin. In a
proceeding pending in Ontario, Canada, the court certified a class consisting of all persons in Canada who purchased and ingested Neurontin prior to August 2004. The plaintiffs claim that Pfizer failed to
provide adequate warning of the alleged risks of personal injury associated with Neurontin.
In January 2011, in a Multi-District Litigation (In re Neurontin Antitrust Litigation MDL-1479) that consolidates four actions, the U.S. District Court for the District of New Jersey certified a nationwide class
consisting of wholesalers and other entities who purchased Neurontin directly from Pfizer and Warner-Lambert during the period from December 11, 2002 to August 31, 2008 and who also purchased
generic gabapentin after it became available. The complaints allege that Pfizer and Warner-Lambert engaged in anticompetitive conduct in violation of the Sherman Act that included, among other things,
submitting patents for listing in the Orange Book and prosecuting and enforcing certain patents relating to Neurontin, as well as engaging in off-label marketing of Neurontin. Plaintiffs seek compensatory
damages, which may be subject to trebling.
Lipitor
In 2004, a former employee filed a “whistleblower” action against us in the U.S. District Court for the Eastern District of New York. The complaint remained under seal until September 2007, at which time
the U.S. Attorney for the Eastern District of New York declined to intervene in the case. We were served with the complaint in December 2007. Plaintiff alleges off-label promotion of Lipitor in violation of
the Federal Civil False Claims Act and the false claims acts of certain states, and he seeks treble damages and civil penalties on behalf of the federal government and the specified states as the result of
their purchase, or reimbursement of patients for the purchase, of Lipitor allegedly for such off-label uses. Plaintiff also seeks compensation as a whistleblower under those federal and state statutes. In
addition, plaintiff alleges that he was wrongfully terminated, in violation of the anti-retaliation provisions of applicable federal and New York law, and he seeks damages and the reinstatement of his
employment. In 2009, the court dismissed without prejudice the off-label promotion claims and, in 2010, plaintiff filed an amended complaint containing off-
label promotion allegations that are substantially
similar to the allegations in the original complaint.
Beginning in November 2011, purported class actions relating to Lipitor were filed in various federal and state courts against Pfizer, certain affiliates of Pfizer, and, in most of the actions, Ranbaxy, among
others. The plaintiffs seek to represent nationwide or statewide classes consisting of persons or entities who directly purchased, indirectly purchased or reimbursed patients for the purchase of Lipitor (or,
in certain of the actions, generic Lipitor) from any of the defendants from March 2010 until the cessation of the defendants’ allegedly unlawful conduct (the Class Period). The plaintiffs allege delay in the
launch of generic Lipitor, in violation of federal antitrust laws and/or state antitrust, consumer protection and various other laws resulting from (i) the 2008 agreement pursuant to which Pfizer and Ranbaxy
settled certain patent litigation involving Lipitor, and Pfizer granted Ranbaxy a license to sell a generic version of Lipitor in various markets beginning on varying dates, and (ii) in certain of the federal
actions, the procurement and/or enforcement of certain patents for Lipitor. Each of the actions seeks, among other things, treble damages on behalf of the putative class for alleged price overcharges for
Lipitor (or, in certain of the actions, generic Lipitor) during the Class Period. In addition, an individual action by several California pharmacies was filed in January 2012 in state court in California against
Pfizer, Ranbaxy and certain of their affiliates, among others, that asserts claims and seeks relief for the plaintiff pharmacies that are substantially similar to the claims asserted and the relief sought in the
purported class actions described above.
Chantix/Champix
A number of individual lawsuits have been filed against us in various federal and state courts alleging suicide, attempted suicide and other personal injuries as a result of the purported ingesting of
Chantix, as well as economic loss. Plaintiffs in these actions seek compensatory and punitive damages and the disgorgement of profits resulting from the sale of Chantix. In October 2009, the federal
cases were transferred for consolidated pre-trial proceedings to a Multi-District Litigation (In re Chantix (Varenicline) Products Liability Litigation MDL-2092) in the U.S. District Court for the Northern
District of Alabama.
Beginning in December 2008, purported class actions were filed against us in the Ontario Superior Court of Justice (Toronto Region), the Superior Court of Quebec (District of Montreal), the Court of
Queen’s Bench of Alberta, Judicial District of Calgary, and the Superior Court of British Columbia (Vancouver Registry) on behalf of all individuals and third-party payers in Canada who have purchased
and ingested Champix or reimbursed patients for the purchase of Champix. Each of these actions asserts claims under Canadian product liability law, including with respect to the safety and efficacy of
Champix, and, on behalf of the putative class, seeks monetary relief, including punitive damages. The actions in Quebec, Alberta and British Columbia have been stayed pending the decision regarding
class certification in the Ontario action.
•
Personal
Injury Actions in the U.S. and Certain Other Countries
•
Antitrust
Action in the U.S.
•
Whistleblower
Action
•
Antitrust
Actions
104
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Bapineuzumab
In June 2010, a purported class action was filed in the U.S. District Court for the District of New Jersey against Pfizer, as successor to Wyeth, and several former officers of Wyeth. The complaint alleges
that Wyeth and the individual defendants violated federal securities laws by making or causing Wyeth to make false and misleading statements, and by failing to disclose or causing Wyeth to fail to
disclose material information, concerning the results of a clinical trial involving bapineuzumab, a product in development for the treatment of Alzheimer’s disease. The plaintiff seeks to represent a class
consisting of all persons who purchased Wyeth securities from May 21, 2007 through July 2008 and seeks damages in an unspecified amount on behalf of the putative class. In February 2012, the court
granted the defendants’ motion to dismiss the complaint. The court’s decision is subject to possible appeal by the plaintiff.
In July 2010, a related action was filed in the U.S. District Court for the Southern District of New York against Elan Corporation (Elan), certain directors and officers of Elan, and Pfizer, as successor to
Wyeth. Elan participated in the development of bapineuzumab until September 2009. The complaint alleges that Elan, Wyeth and the individual defendants violated federal securities laws by making or
causing Elan to make false and misleading statements, and by failing to disclose or causing Elan to fail to disclose material information, concerning the results of a clinical trial involving bapineuzumab.
The plaintiff seeks to represent a class consisting of all persons who purchased Elan call options from June 17, 2008 through July 29, 2008 and seeks damages in an unspecified amount on behalf of the
putative class. In June 2011, the court granted Pfizer’s and Elan’s motions to dismiss the complaint. In July 2011, the plaintiff filed a supplemental memorandum setting forth the bases that the plaintiff
believed supported amendment of the complaint. In August 2011, the court dismissed the complaint with prejudice. In September 2011, the plaintiff appealed the District Court’s decision to the U.S. Court
of Appeals for the Second Circuit.
Thimerosal
Wyeth is a defendant in a number of suits by or on behalf of vaccine recipients alleging that exposure through vaccines to cumulative doses of thimerosal, a preservative used in certain childhood
vaccines formerly manufactured and distributed by Wyeth and other vaccine manufacturers, caused severe neurological damage and/or autism in children. While several suits were filed as purported
nationwide or statewide class actions, all of the purported class actions have been dismissed, either by the courts or voluntarily by the plaintiffs. In addition to the suits alleging injury from exposure to
thimerosal, certain of the cases were brought by parents in their individual capacities for, among other things, loss of services and loss of consortium of the injured child.
The National Childhood Vaccine Injury Act (the Vaccine Act) requires that persons alleging injury from childhood vaccines first file a petition in the U.S. Court of Federal Claims asserting a vaccine-
related
injury. At the conclusion of that proceeding, petitioners may bring a lawsuit against the manufacturer in federal or state court, provided that they have satisfied certain procedural requirements. Also under
the terms of the Vaccine Act, if a petition has not been adjudicated by the U.S. Court of Federal Claims within a specified time period after filing, the petitioner may opt out of the proceeding and pursue a
lawsuit against the manufacturer by following certain procedures. Some of the vaccine recipients who have sued Wyeth to date may not have satisfied the conditions to filing a lawsuit that are mandated
by the Vaccine Act. The claims brought by parents for, among other things, loss of services and loss of consortium of the injured child are not covered by the Vaccine Act.
In 2002, the Office of Special Masters of the U.S. Court of Federal Claims established an Omnibus Autism Proceeding with jurisdiction over petitions in which vaccine recipients claim to suffer from autism
or autism spectrum disorder as a result of receiving thimerosal-containing childhood vaccines and/or the measles, mumps and rubella (MMR) vaccine. There currently are several thousand petitions
pending in the Omnibus Autism Proceeding. Special masters of the court have heard six test cases on petitioners’ theories that either thimerosal-
containing vaccines in combination with the MMR vaccine
or thimerosal-containing vaccines alone can cause autism or autism spectrum disorder.
Pristiq
In late 2007 and early 2008, the following actions were filed in various federal courts: (i) a purported class action alleging that Wyeth and certain former officers of Wyeth violated federal securities laws by
misrepresenting the safety of Pristiq during the period before the FDA’s issuance in July 2007 of an “approvable letter” for Pristiq for the treatment of vasomotor symptoms, which allegedly caused a
decline in the price of Wyeth stock; and (ii) a purported class action against Wyeth, the Wyeth Savings Plan Committee, the Wyeth Savings Plan-Puerto Rico Committee, the Wyeth Retirement
Committee and certain former Wyeth officers and committee members alleging that they violated certain provisions of ERISA by maintaining Wyeth stock as an investment alternative under certain Wyeth
plans notwithstanding their alleged knowledge of the aforementioned alleged misrepresentation.
The U.S. District Court for the Southern District of New York dismissed the ERISA action and denied the plaintiff’s motion to amend the complaint in March and August 2010, respectively. In September
2010, the plaintiff appealed both of those rulings to the U.S. Court of Appeals for the Second Circuit. In November 2010, the plaintiff withdrew the appeal, but has reserved the right to reinstate the appeal
by March 2012. The purported securities class action remains pending.
•
In February 2009, special masters of the U.S. Court of Federal Claims rejected the three cases brought on the theory that a combination of MMR and thimerosal-containing vaccines caused petitioners’
conditions. After these rulings were affirmed by the U.S. Court of Federal Claims, two of them were appealed by petitioners to the U.S. Court of Appeals for the Federal Circuit. In 2010, the Federal
Circuit affirmed the decisions of the special masters in both of these cases.
•
In March 2010, special masters of the U.S. Court of Federal Claims rejected the three additional test cases brought on the theory that thimerosal-containing vaccines alone caused petitioners’
conditions. Petitioners did not seek review by the U.S. Court of Federal Claims of the decisions of the special masters in these latter three test cases, and judgments were entered dismissing the cases
in April 2010.
•
Petitioners in each of the six test cases have filed an election to bring a civil action.
2011 Financial Report
105

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Rebif
We have an exclusive collaboration agreement with EMD Serono, Inc. (Serono) to co-promote Rebif, a treatment for multiple sclerosis, in the U.S. In August 2011, Serono filed a complaint in the
Philadelphia Court of Common Pleas seeking a declaratory judgment that we are not entitled to a 24-month extension of the Rebif co-promotion agreement, which otherwise would terminate at the end of
2013. We disagree with Serono’s interpretation of the agreement and believe that we have the right to extend the agreement to the end of 2015. In October 2011, the court sustained our preliminary
objections and dismissed Serono’s complaint, and Serono has appealed the decision to the Superior Court of Pennsylvania.
Acquisition of Wyeth
In 2009, a number of retail pharmacies in California brought an action against Pfizer and Wyeth in the U.S. District Court for the Northern District of California. The plaintiffs alleged, among other things,
that our acquisition of Wyeth violated various federal antitrust laws by creating a monopoly in the manufacture, distribution and sale of prescription drugs in the U.S. In April 2010, the District Court
granted our motion to dismiss the second amended complaint. In May 2011, the U.S. Court of Appeals for the Ninth Circuit affirmed the dismissal by the District Court and, in June 2011, it denied
plaintiffs’ petition for a rehearing. In December 2011, the U.S. Supreme Court denied the plaintiffs’ petition for certiorari seeking reversal of the Ninth Circuit’s decision.
Acquisition of King Pharmaceuticals, Inc.
In October 2010, several purported class action complaints were filed in state court in Tennessee by shareholders of King challenging Pfizer’s acquisition of King. King and the individuals who served as
the members of King’s Board of Directors at the time of the execution of the merger agreement are named as defendants in all of these actions. Pfizer and Parker Tennessee Corp., a subsidiary of Pfizer,
also are named as defendants in most of these actions.
In November 2010, all of these actions were consolidated in the Chancery Court for Sullivan County, Tennessee Second Judicial District, at Bristol. The parties to the consolidated action have reached an
agreement-in-principle to resolve that action as a result of certain disclosures regarding the transaction made by King in its amended Schedule 14D-
9 recommendation statement for the tender offer dated
January 21, 2011. The proposed settlement is subject to, among other things, court approval.
Average Wholesale Price Litigation
A number of states, as well as most counties in New York, have sued Pharmacia, Pfizer and other pharmaceutical manufacturers alleging that they provided average wholesale price (AWP) information
for certain of their products that was higher than the actual prices at which those products were sold. The AWP is used to determine reimbursement levels under Medicare Part B and Medicaid and in
many private-sector insurance policies and medical plans. The plaintiffs claim that the alleged spread between the AWPs at which purchasers were reimbursed and the actual sale prices was promoted
by the defendants as an incentive to purchase certain of their products. In addition to suing on their own behalf, many of the plaintiff states seek to recover on behalf of individual Medicare Part B co-
payers and private-sector insurance companies and medical plans in their states. These various actions generally assert fraud claims, as well as claims under state deceptive trade practice laws, and
seek monetary and other relief, including civil penalties and treble damages. Several of the suits also allege that Pharmacia and/or Pfizer did not report to the states their best price for certain products
under the Medicaid program.
In addition, Pharmacia, Pfizer and other pharmaceutical manufacturers are defendants in a number of purported class action suits in various federal and state courts brought by employee benefit plans
and other third-party payers that assert claims similar to those in the state and county actions. These suits allege, among other things, fraud, unfair competition and unfair trade practices and seek
monetary and other relief, including civil penalties and treble damages.
All of these state, county and purported class action suits were transferred for consolidated pre-trial proceedings to a Multi-District Litigation (In re Pharmaceutical Industry Average Wholesale Price
Litigation MDL-1456) in the U.S. District Court for the District of Massachusetts. Certain of the state and private suits have been remanded to their respective state courts. In 2006, the claims against
Pfizer in the Multi-District Litigation were dismissed with prejudice.
In 2008, the court in the Multi-District Litigation granted preliminary approval with respect to the fairness of a proposed settlement of the claims against 11 defendants, including Pharmacia, for a total of
$125 million. In December 2011, the court granted final approval of the settlement. Pharmacia’s contribution to the settlement was not material to Pfizer.
In addition, Wyeth is a defendant in AWP actions brought by certain states, which are not included in the Multi-
District Litigation. Wyeth also is a defendant in a purported class action in state court in New
Jersey brought by a union health and welfare plan on behalf of a putative class consisting of third-party payers in New Jersey. In addition, King and/or certain of its subsidiaries are defendants in AWP
actions brought by certain states, which are not included in the Multi-District Litigation.
Monsanto-Related Matters
In 1997, Monsanto Company (Former Monsanto) contributed certain chemical manufacturing operations and facilities to a newly formed corporation, Solutia Inc. (Solutia), and spun off the shares of
Solutia. In 2000, Former Monsanto merged with Pharmacia & Upjohn Company to form Pharmacia Corporation (Pharmacia). Pharmacia then transferred its agricultural operations to a newly created
subsidiary, named Monsanto Company (New Monsanto), which it spun off in a two-stage process that was completed in 2002. Pharmacia was acquired by Pfizer in 2003 and is now a wholly owned
subsidiary of Pfizer.
C.
Commercial and Other Matters
106
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
In connection with its spin-off that was completed in 2002, New Monsanto assumed, and agreed to indemnify Pharmacia for, any liabilities related to Pharmacia’s former agricultural business. New
Monsanto is defending and indemnifying Pharmacia in connection with various claims and litigation arising out of, or related to, the agricultural business.
In connection with its spin-off in 1997, Solutia assumed, and agreed to indemnify Pharmacia for, liabilities related to Former Monsanto’s chemical businesses. As the result of its reorganization under
Chapter 11 of the U.S. Bankruptcy Code, Solutia’s indemnification obligations related to Former Monsanto’s chemical businesses are limited to sites that Solutia has owned or operated. In addition, in
connection with its spinoff that was completed in 2002, New Monsanto assumed, and agreed to indemnify Pharmacia for, any liabilities primarily related to Former Monsanto’s chemical businesses,
including, but not limited to, any such liabilities that Solutia assumed. Solutia’s and New Monsanto’
s assumption of and agreement to indemnify Pharmacia for these liabilities apply to pending actions and
any future actions related to Former Monsanto’s chemical businesses in which Pharmacia is named as a defendant, including, without limitation, actions asserting environmental claims, including alleged
exposure to polychlorinated biphenyls. Solutia and New Monsanto are defending and indemnifying Pharmacia in connection with various claims and litigation arising out of, or related to, Former
Monsanto’s chemical businesses.
Trade Secrets Action in California
In 2004, Ischemia Research and Education Foundation (IREF) and its chief executive officer brought an action in California Superior Court, Santa Clara County, against a former IREF employee and
Pfizer. Plaintiffs allege that defendants conspired to misappropriate certain information from IREF’s allegedly proprietary database in order to assist Pfizer in designing and executing a clinical study of a
Pfizer drug. In 2008, the jury returned a verdict for compensatory damages of approximately $38.7 million. In March 2009, the court awarded prejudgment interest, but declined to award punitive
damages. In July 2009, the court granted our motion for a new trial and vacated the jury verdict.
Trimegestone
Aventis filed a breach of contract action against Wyeth in the Commercial Court of Nanterre in France arising out of the December 2003 termination by Wyeth of an October 2000 agreement between
Wyeth and Aventis relating to the development of hormone-therapy drugs utilizing Aventis’s trimegestone (TMG) progestin. Aventis alleges that the termination was improper and seeks
monetary damages. In 2009, a three-judge tribunal rendered its decision in favor of Wyeth. In May 2010, the Versailles Court of Appeals reversed the Commercial Court’s decision and appointed experts
to hear evidence and make a recommendation to the Court of Appeals concerning damages. In November 2011, the Supreme Court of France affirmed the decision of the Court of Appeals. The damage
proceeding by the experts appointed by the Court of Appeals is continuing.
Environmental Matters
In 2009, we submitted to the U.S. Environmental Protection Agency (EPA) a corrective measures study report with regard to Pharmacia Corporation’s discontinued industrial chemical facility in North
Haven, Connecticut and a revised site-wide feasibility study with regard to Wyeth’s discontinued industrial chemical facility in Bound Brook, New Jersey. In September 2010, our corrective measures
study report with regard to the North Haven facility was approved by the EPA, and we commenced construction of the site remedy in late 2011 under an Updated Administrative Order on Consent with the
EPA. In July 2011, we finalized an Administrative Settlement Agreement and Order on Consent for Removal Action with the EPA with regard to the Bound Brook facility and commenced construction of
an interim remedy to address the discharge of impacted groundwater from that facility to the Raritan River. In February 2012, the EPA issued a proposed remediation plan for the Bound Brook facility. The
proposed plan, which is subject to public comment, is generally in accordance with one of the remedies evaluated in the Company’s revised site-wide feasibility study. The estimated costs of the site
remedy for the North Haven facility and the proposed remediation plan for the Bound Brook facility are covered by accruals previously taken by the Company.
We are a party to a number of other proceedings brought under the Comprehensive Environmental Response, Compensation, and Liability Act of 1980, as amended (CERCLA or Superfund), and other
state, local or foreign laws in which the primary relief sought is the cost of past and/or future remediation.
In February 2011, King received notice from the U.S. Department of Justice (DOJ) advising that the EPA has requested that DOJ initiate enforcement action seeking injunctive relief and penalties against
King for alleged non-
compliance with certain provisions of the federal Clean Air Act at its Bristol, Tennessee manufacturing facility. King has executed a tolling agreement with the DOJ in order to facilitate
the possible resolution of this matter.
In October 2011, we voluntarily disclosed to the EPA potential non-compliance with certain provisions of the federal Clean Air Act at our Barceloneta, Puerto Rico manufacturing facility. We do not expect
that any penalties that may result from this matter will be material to the Company.
D. Government Investigations
Like other pharmaceutical companies, we are subject to extensive regulation by national, state and local government agencies in the U.S. and in the other countries in which we operate. As a result, we
have interactions with government agencies on an ongoing basis. Among the investigations by government agencies are those discussed below. It is possible that criminal charges and substantial fines
and/or civil penalties could result from government investigations, including but not limited to those discussed below.
The Company has voluntarily provided the DOJ and the U.S. Securities and Exchange Commission (SEC) with information concerning potentially improper payments made by certain Pfizer and Wyeth
subsidiaries in connection with certain sales activities outside the U.S. In recent discussions, we have reached agreements-in-principle with the SEC staff and with the DOJ, and we are in the process of
finalizing a resolution of these matters. In addition, certain potentially improper payments and other matters are the subject of investigations by government authorities in certain foreign countries. The
previously reported investigation in Germany with respect to certain tax matters relating to a wholly owned subsidiary of Pfizer was resolved in December 2011 with no criminal charges and with the
payment of an amount, primarily for interest, that was not material to Pfizer.
2011 Financial Report
107

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
The DOJ is conducting civil and criminal investigations regarding Wyeth’s promotional practices with respect to Protonix and its practices relating to the pricing for Protonix for Medicaid rebate purposes.
In connection with the pricing investigation, in 2009, the DOJ filed a civil complaint in intervention in two qui tam actions that had been filed under seal in the U.S. District Court for the District of
Massachusetts. The complaint alleges that Wyeth’s practices relating to the pricing for Protonix for Medicaid rebate purposes between 2001 and 2006 violated the Federal Civil False Claims Act and
federal common law. The two qui tam actions have been unsealed and the complaints include substantially similar allegations. In addition, in 2009, several states and the District of Columbia filed a
complaint under the same docket number asserting violations of various state laws based on allegations substantially similar to those set forth in the civil complaint filed by the DOJ. We are exploring with
the DOJ various ways to resolve its civil and criminal investigations relating to Protonix.
The DOJ, including the U.S. Attorney’s Office for the Western District of Oklahoma, is conducting a civil and criminal investigation with respect to Wyeth’s promotional practices relating to Rapamune. In
addition, in October 2010, the DOJ was permitted to intervene in a qui tam action, which alleges off-label promotion of Rapamune, that was pending in the U.S. District Court for the Eastern District of
Pennsylvania. In December 2010, the qui tam action was transferred to the Western District of Oklahoma, where it was consolidated with the proceedings underway there. We are exploring with the DOJ
various ways to resolve this matter.
We have received civil investigative demands and informal inquiries from the consumer protection divisions of several states seeking information and documents concerning the promotion of Lyrica and
Zyvox. We are in discussions with those states regarding a resolution of this matter. These requests appear to relate to the same past promotional practices concerning these products that were the
subject of previously reported settlements in September 2009 with the DOJ and the Medicaid fraud control units of various states.
GUARANTEES AND INDEMNIFICATIONS
In the ordinary course of business and in connection with the sale of assets and businesses, we often indemnify our counterparties against certain liabilities that may arise in connection with the
transaction or related to activities prior to the transaction. These indemnifications typically pertain to environmental, tax, employee and/or product-related matters and patent-infringement claims. If the
indemnified party were to make a successful claim pursuant to the terms of the indemnification, we would be required to reimburse the loss. These indemnifications are generally subject to threshold
amounts, specified claim periods and other restrictions and limitations. Historically, we have not paid significant amounts under these provisions and, as of December 31, 2011, recorded amounts for the
estimated fair value of these indemnifications were not significant.
PURCHASE COMMITMENTS
As of December 31, 2011, we have agreements totaling $3.8 billion to purchase goods and services that are enforceable and legally binding and include amounts relating to advertising, information
technology services, employee benefit administration services, and potential milestone payments deemed reasonably likely to occur.
18.
Segment, Geographic and Other Revenue Information
A. Segment Information
We manage our operations through five operating segments––Primary Care, Specialty Care and Oncology, Established Products and Emerging Markets, Animal Health and Consumer Healthcare and
Nutrition. Each operating segment has responsibility for its commercial activities and for certain research and development activities related to in-line products and IPR&D projects that generally have
achieved proof-of-concept. Previously, we managed our operations through two operating segments––Biopharmaceutical and Diversified. We have restated our prior period segment information to
conform with the current period presentation.
We regularly review our segments and the approach used by management to evaluate performance and allocate resources.
Operating Segments
A description of each of our five operating segments follows:
• Primary Care operating segment––includes revenues and earnings, as defined by management, from human pharmaceutical products primarily prescribed by primary-care physicians, and may
include products in the following therapeutic and disease areas: Alzheimer’s disease, cardiovascular (excluding pulmonary arterial hypertension), erectile dysfunction, genitourinary, major
depressive disorder, pain, respiratory and smoking cessation. Examples of products in this unit include Celebrex, Chantix/Champix, Lipitor, Lyrica, Premarin, Pristiq and Viagra. All revenues and
earnings for such products are allocated to the Primary Care unit, except those generated in Emerging Markets and those that are managed by the Established Products unit.
108
2011 Financial Report

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Our chief operating decision maker uses the revenues and earnings of the five operating segments, among other factors, for performance evaluation and resource allocation. For the operating segments
that comprise more than one business unit, a single segment manager has responsibility for those business units.
Other Costs and Business Activities
Certain costs are not allocated to our operating segment results, such as costs associated with the following:
•
Specialty Care and Oncology operating segment
–
–
comprises the Specialty Care business unit and the Oncology business unit.
-
Specialty Care––includes revenues and earnings, as defined by management, from most human pharmaceutical products primarily prescribed by physicians who are specialists, and may
include products in the following therapeutic and disease areas: anti-infectives, endocrine disorders, hemophilia, inflammation, multiple sclerosis, ophthalmology, pulmonary arterial
hypertension, specialty neuroscience and vaccines. Examples of products in this unit include BeneFIX, Enbrel, Genotropin, Geodon, the Prevnar/Prevenar franchise, Rebif, ReFacto AF,
Revatio, Xalatan , Xyntha and Zyvox. All revenues and earnings for such products are allocated to the Specialty Care unit, except those generated in Emerging Markets and those that are
managed by the Established Products unit.
-
Oncology––includes revenues and earnings, as defined by management, from human prescription pharmaceutical products addressing oncology and oncology-related illnesses. Examples of
products in this unit include Aromasin, Sutent, Torisel and Xalkori. All revenues and earnings for such products are allocated to the Oncology unit, except those generated in Emerging Markets
and those that are managed by the Established Products unit.
•
Established Products and Emerging Markets operating segment––comprises the Established Products business unit and the Emerging Markets business unit.
-
Established Products––generally includes revenues and earnings, as defined by management, from human prescription pharmaceutical products that have lost patent protection or marketing
exclusivity in certain countries and/or regions. Typically, products are transferred to this unit in the beginning of the fiscal year following loss of patent protection or marketing exclusivity. In
certain situations, products may be transferred to this unit at a different point than the beginning of the fiscal year following loss of patent protection or marketing exclusivity in order to maximize
their value. This unit also excludes revenues and earnings generated in Emerging Markets. Examples of products in this unit include Arthrotec, Effexor, Medrol, Norvasc, Protonix, Relpax and
Zosyn/Tazocin.
-
Emerging Markets––includes revenues and earnings, as defined by management, from all human prescription pharmaceutical products sold in Emerging Markets, including Asia (excluding
Japan and South Korea), Latin America, Middle East, Africa, Central and Eastern Europe and Turkey.
•
Animal Health and Consumer Healthcare operating segment—comprises the Animal Health business unit and the Consumer Healthcare business unit.
-
Animal Health––includes worldwide revenues and earnings, as defined by management, from products and services to prevent and treat disease in livestock and companion animals, including
vaccines, parasiticides and anti
-
infectives.
-
Consumer Healthcare––generally includes worldwide revenues and earnings, as defined by management, from non-prescription products in the following therapeutic categories: dietary
supplements, pain management, respiratory and personal care. Products marketed by Consumer Healthcare include Advil, Caltrate, Centrum, ChapStick, Preparation H and Robitussin.
•
Nutrition operating segment––generally includes revenues and earnings, as defined by management, from a full line of infant and toddler nutritional products sold outside of the U.S. and Canada.
•
Worldwide Research and Development (WRD), which is generally responsible for human health research projects until proof-of-concept is achieved and then for transitioning those projects to the
appropriate business unit for possible clinical and commercial development. R&D spending may include upfront and milestone payments for intellectual property rights. This organization also has
responsibility for certain science-based platform services, which provide technical expertise and other services to the various research and development projects.
• Pfizer Medical, which is responsible for all human-health-related regulatory submissions and interactions with regulatory agencies. This organization is also responsible for the collection, evaluation
and reporting of all safety event information related to our human health products and for conducting clinical trial audits and readiness reviews and for providing Pfizer-related medical information to
healthcare providers.
•
Corporate, which is responsible for platform functions such as finance, global real estate operations, human resources, legal, compliance, science and technology, worldwide procurement,
worldwide public affairs and policy and worldwide technology. These costs also include compensation costs and other miscellaneous operating expenses not charged to our operating segments, as
well as interest income and expense.
•
Certain transactions and events such as (i) purchase accounting adjustments, where we incur expenses associated with the amortization of fair value adjustments to inventory, intangible assets and
property, plant and equipment; (ii) acquisition-related activities, where we incur costs for restructuring, integration, implementation and executing the transaction; and (iii) certain significant items,
which include non
-acquisition-related restructuring costs, as well as costs incurred for legal settlements, asset impairments and sales of assets or businesses.
2011 Financial Report
109
Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Segment Assets
We manage our assets on a total company basis, not by operating segment, as many of our operating assets are shared (such as our plant network assets) or commingled (such as accounts receivable,
as many of our customers are served by multiple operating segments). Therefore, our chief operating decision maker does not regularly review any asset information by operating segment and,
accordingly, we do not report asset information by operating segment. Total assets were approximately $188 billion at December 31, 2011 and approximately $195 billion at December 31, 2010.
Selected Income Statement Information
Selected income statement information follows:
(MILLIONS OF DOLLARS)
REVENUES
R&D EXPENSES
EARNINGS
DEPRECIATION &
AMORTIZATION
YEAR ENDED DECEMBER 31, 2011
Primary Care
$22,670
$1,307
$15,001
$ 247
Specialty Care and Oncology
16,568
1,561
10,789
419
Established Products and Emerging Markets
18,509
441
9,417
422
Animal Health and Consumer Healthcare
7,241
425
2,020
232
Total reportable segments
64,988
3,734
37,227
1,320
Nutrition and other business activities
2,437
3,378
(2,793
)
230
Reconciling Items:
Corporate
—
1,309
(7,430
)
541
Purchase accounting adjustments
—
(
2
)
(6,801
)
5,565
Acquisition
-
related costs
—
23
(1,983
)
624
Certain significant items
—
656
(4,354
)
615
Other unallocated
—
14
(1,104
)
131
$67,425
$9,112
$12,762
$9,026
YEAR ENDED DECEMBER 31, 2010
Primary Care
$23,328
$1,473
$15,773
$ 201
Specialty Care and Oncology
16,435
1,624
10,571
432
Established Products and Emerging Markets
18,760
452
10,100
418
Animal Health and Consumer Healthcare
6,347
428
1,569
197
Total reportable segments
64,870
3,977
38,013
1,248
Nutrition and other business activities
2,187
3,743
(3,263
)
242
Reconciling Items:
Corporate
—
1,567
(7,990
)
619
Purchase accounting adjustments
—
26
(8,257
)
5,477
Acquisition-related costs
—
34
(3,989
)
788
Certain significant items
—
18
(3,964
)
—
Other unallocated
—
27
(1,268
)
113
$67,057
$9,392
$9,282
$8,487
YEAR ENDED DECEMBER 31, 2009
Primary Care
$22,576
$1,407
$15,100
$ 130
Specialty Care and Oncology
8,925
1,060
4,661
269
Established Products and Emerging Markets
13,947
392
6,955
360
Animal Health and Consumer Healthcare
3,258
297
812
142
Total reportable segments
48,706
3,156
27,528
901
Nutrition and other business activities
563
2,706
(2,751
)
181
Reconciling Items:
Corporate
—
1,296
(4,657
)
526
Purchase accounting adjustments
—
37
(3,787
)
2,799
Acquisition-related costs
—
13
(4,025
)
241
Certain significant items
—
56
(1,511
)
—
Other unallocated
—
560
(123
)
109
$49,269
$7,824
$10,674
$4,757
Income from continuing operations before provision for taxes on income.
Certain production facilities are shared. Depreciation is allocated based on estimates of physical production.
For 2011, includes King commencing on the acquisition date of January 31, 2011. For 2009, includes Wyeth commencing on the acquisition date of October 15, 2009.
Other business activities includes the revenues and operating results of Pfizer CentreSource, our contract manufacturing and bulk pharmaceutical chemical sales operation, and the research and
development costs managed by our Worldwide Research and Development organization and our Pfizer Medical organization.
Corporate for R&D expenses includes, among other things, administration expenses and compensation expenses associated with our research and development activities and for Earnings includes,
among other things, administration expenses, interest income/(expense), certain compensation and other costs not charged to our operating segments.
Purchase accounting adjustments include certain charges related to the fair value adjustments to inventory, intangible assets and property, plant and equipment.
Acquisition-related costs can include costs associated with acquiring, integrating and restructuring newly acquired businesses, such as transaction costs, integration costs, restructuring charges and
additional depreciation associated with asset restructuring (see Note 3. Restructuring Charges and Other Costs Associated with Acquisitions and Cost-Reduction/Productivity Initiatives for additional
information).
Certain significant items are substantive, unusual items that, either as a result of their nature or size, would not be expected to occur as part of our normal business on a regular basis. Such items
primarily include restructuring charges and implementation costs associated with our cost-reduction and productivity initiatives that are not associated with an acquisition, the impact of certain tax
and/or legal settlements and certain asset impairments.
Includes overhead expenses associated with our manufacturing and commercial operations not directly attributable to an operating segment. In 2009, R&D expenses include approximately $550
million of Wyeth R&D expenses and Earnings include approximately $900 million of Wyeth earnings and $290 million of operating expenses incurred in Japan associated with our three
biopharmaceutical operating segments, where allocation among the segments is not practicable.
110
2011 Financial Report
(a) (b)
(c)
(d)
(e)
(f)
(g)
(h)
(i)
(d)
(e)
(f)
(g)
(h)
(i)
(c)
(d)
(e)
(f)
(g)
(h)
(i)
(a)
(b)
(c)
(d)
(e)
(f)
(g)
(h)
(i)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Revenues exceeded $500 million in each of 18 countries outside the U.S. in 2011 and 2010, and in each of 13 countries outside the U.S. in 2009. The U.S. was the only country to contribute more than
10% of total revenues in each year.
Revenues by geographic region follow:
Long-lived assets by geographic region follow:
Significant Customers
We sell our products primarily to customers in the wholesale sector. In 2011, sales to our three largest U.S. wholesaler customers represented approximately 13%, 10% and 9% of total revenues and,
collectively, represented approximately 13% of total accounts receivable as of December 31, 2011. These sales and related accounts receivable were concentrated in our three biopharmaceutical
operating segments. In 2010, sales to our three largest U.S. wholesaler customers represented approximately 14%, 10% and 9% of total revenues and, collectively, represented approximately 18% of
total accounts receivable as of December 31, 2010.
B.
Geographic Information
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Revenues
United States
$
26,933
$
28,855
$
21,540
Developed Europe
16,297
16,345
12,586
Developed Rest of World
11,091
10,008
8,097
Emerging Markets
13,104
11,849
7,046
Consolidated
$
67,425
$
67,057
$
49,269
For 2011, includes King commencing on the acquisition date of January 31, 2011. For 2009, includes Wyeth commencing on the acquisition date of October 15, 2009.
Developed Europe region includes the following markets: Western Europe, Finland and the Scandinavian countries. Euro revenues were approximately $12 billion for each of 2011 and 2010 and $10
billion for 2009.
Developed Rest of World region includes the following markets: Australia, Canada, Japan, New Zealand, and South Korea.
Emerging Markets region includes, but is not limited to, the following markets: Asia (excluding Japan and South Korea), Latin America, Middle East, Africa, Central and Eastern Europe and Turkey.
AS OF DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
Property, plant and equipment, net
United States
$ 7,893
$ 8,537
Developed Europe
6,023
7,159
Developed Rest of World
904
854
Emerging Markets
2,118
2,095
Consolidated
$16,938
$18,645
Developed Europe region includes the following markets: Western Europe, Finland and the Scandinavian countries.
Developed Rest of World region includes the following markets: Australia, Canada, Japan, New Zealand, and South Korea.
Emerging Markets region includes, but is not limited to, the following markets: Asia (excluding Japan and South Korea), Latin America, Middle East, Africa, Central and Eastern Europe and Turkey.
C.
Other Revenue Information
2011 Financial Report
111
(a)
(b)
(c)
(d)
(a)
(b)
(c )
(d)
(a)
(b)
(c)
(a)
(b
)
(c)

Notes to Consolidated Financial Statements
Pfizer Inc. and Subsidiary Companies
Significant Product Revenues
Significant product revenues follow:
YEAR ENDED DECEMBER 31,
(MILLIONS OF DOLLARS)
2011
2010
2009
Revenues from biopharmaceutical products
:
Lipitor
$9,577
$10,733
$11,434
Lyrica
3,693
3,063
2,840
Prevnar 13/Prevenar 13
3,657
2,416
—
Enbrel (Outside the U.S. and Canada)
3,666
3,274
378
Celebrex
2,523
2,374
2,383
Viagra
1,981
1,928
1,892
Norvasc
1,445
1,506
1,973
Zyvox
1,283
1,176
1,141
Xalatan/Xalacom
1,250
1,749
1,737
Sutent
1,187
1,066
964
Geodon/Zeldox
1,022
1,027
1,002
Premarin family
1,013
1,040
213
Genotropin
889
885
887
Detrol/Detrol LA
883
1,013
1,154
Vfend
747
825
798
Chantix/Champix
720
755
700
BeneFIX
693
643
98
Effexor
678
1,718
520
Zosyn/Tazocin
636
952
184
Pristiq
577
466
82
Zoloft
573
532
516
Caduet
538
527
548
Revatio
535
481
450
Medrol
510
455
457
ReFacto AF/Xyntha
506
404
47
Prevnar/Prevenar (7
-
valent)
488
1,253
287
Zithromax/Zmax
453
415
430
Aricept
450
454
435
Fragmin
382
341
359
Cardura
380
413
457
Rapamune
372
388
57
Aromasin
361
483
483
BMP2
340
400
81
Relpax
341
323
326
Xanax XR
306
307
318
Tygacil
298
324
54
Neurontin
289
322
327
Diflucan
265
278
281
Arthrotec
242
250
270
Unasyn
231
244
245
Sulperazon
218
213
204
Skelaxin
203
—
—
Inspra
195
157
130
Dalacin/Cleocin
192
214
241
Methotrexate
191
164
21
Toviaz
187
137
59
Somavert
183
157
147
Alliance revenues
3,630
4,084
2,925
All other biopharmaceutical products
6,768
6,194
4,913
Total revenues from biopharmaceutical products
57,747
58,523
45,448
Revenues from other products
:
Animal Health
4,184
3,575
2,764
Consumer Healthcare
3,057
2,772
494
Nutrition
2,138
1,867
191
Pfizer CentreSource
299
320
372
Total revenues
$67,425
$67,057
$49,269
For 2011, includes King commencing on the acquisition date of January 31, 2011. For 2009, includes Wyeth commencing on the acquisition date of October 15, 2009.
On November 30, 2011, Lipitor lost exclusivity in the U.S. This loss of exclusivity reduced revenues by $326 million in 2011, in comparison with 2010.
Acquired from Wyeth.
Represents direct sales under license agreement with Eisai Co., Ltd.
Acquired from King.
Enbrel (in the U.S. and Canada), Aricept, Exforge, Rebif and Spiriva.
Includes products from legacy Pfizer, legacy Wyeth and legacy King.
112
2011 Financial Report
(a)
(b)
(c)
(c)
(c)
(c)
(c)
(c)
(c)
(c)
(c)
(d)
(c)
(c)
(c)
(e)
(f)
(g)
(a)
(g)
(c)
(c)
(a)
(a)
(b)
(c)
(d)
(
e
)
(f)
(
g
)

Quarterly Consolidated Financial Data (Unaudited)
Pfizer Inc. and Subsidiary Companies
Basic and diluted EPS are computed independently for each of the periods presented. Accordingly, the sum of the quarterly EPS amounts may not agree to the total for the year.
As of January 31, 2012, there were 223,038 holders of record of our common stock (New York Stock Exchange symbol PFE).
QUARTER
(MILLIONS OF DOLLARS, EXCEPT PER COMMON SHARE DATA)
FIRST
SECOND
THIRD
FOURTH
2011
Revenues
$
16,502
$
16,984
$
17,193
$
16,746
Costs and expenses
12,490
12,823
12,423
13,993
Acquisition
-
related in
-
process research and development charges
—
—
—
—
Restructuring charges and certain acquisition-related costs
894
479
1,101
460
Income from continuing operations before provision for taxes on income
3,118
3,682
3,669
2,293
Provision for taxes on income
894
1,094
1,235
800
Income from continuing operations
2,224
2,588
2,434
1,493
Discontinued operations
—
net of tax
10
30
1,315
(43)
Net income before allocation to noncontrolling interests
2,234
2,618
3,749
1,450
Less: Net income attributable to noncontrolling interests
12
8
11
11
Net income attributable to Pfizer Inc.
$
2,222
$
2,610
$
3,738
$
1,439
Earnings per common share
—
basic:
Income from continuing operations attributable to Pfizer Inc. common shareholders
$
0.28
$
0.33
$
0.31
$
0.19
Discontinued operations
—
net of tax
—
—
0.17
(0.01)
Net income attributable to Pfizer Inc. common shareholders
$
0.28
$
0.33
$
0.48
$
0.19
Earnings per common share—diluted:
Income from continuing operations attributable to Pfizer Inc. common shareholders
$
0.28
$
0.33
$
0.31
$
0.19
Discontinued operations
—
net of tax
—
—
0.17
(0.01)
Net income attributable to Pfizer Inc. common shareholders
$
0.28
$
0.33
$
0.48
$
0.19
Cash dividends paid per common share
$
0.20
$
0.20
$
0.20
$
0.20
Stock prices
High
$
20.57
$
21.45
$
20.95
$
21.90
Low
$
17.62
$
19.10
$
16.63
$
17.05
The fourth quarter of 2011 reflects historically higher Q4 costs in Cost of sales, Selling, informational and administrative expenses, Research and development expenses and Other deductions—net.
The third quarter of 2011 reflects higher employee termination costs.
The third quarter of 2011 reflects the gain on the sale of Capsugel.
2011 Financial Report
113
(a)
(b)
(c)
(a)
(b)
(c)

Quarterly Consolidated Financial Data (Unaudited)
Pfizer Inc. and Subsidiary Companies
Basic and diluted EPS are computed independently for each of the periods presented. Accordingly, the sum of the quarterly EPS amounts may not agree to the total for the year.
QUARTER
(MILLIONS OF DOLLARS, EXCEPT PER COMMON SHARE DATA)
FIRST
SECOND
THIRD
FOURTH
2010
Revenues
$
16,576
$
17,132
$
15,995
$
17,354
Costs and expenses
12,647
12,321
14,082
15,399
Acquisition
-
related in
-
process research and development charges
74
—
—
51
Restructuring charges and certain acquisition-related costs
706
885
499
1,111
Income from continuing operations before provision/(benefit) for taxes on income
3,149
3,926
1,414
793
Provision/(benefit) for taxes on income
1,135
1,472
558
(2,094)
Income from continuing operations
2,014
2,454
856
2,887
Discontinued operations
—
net of tax
21
31
15
10
Net income before allocation to noncontrolling interests
2,035
2,485
871
2,897
Less: Net income attributable to noncontrolling interests
9
10
5
7
Net income attributable to Pfizer Inc.
$
2,026
$
2,475
$
866
$
2,890
Earnings per common share
—
basic:
Income from continuing operations attributable to Pfizer Inc. common shareholders
$
0.25
$
0.30
$
0.11
$
0.36
Discontinued operations
—
net of tax
—
—
—
—
Net income attributable to Pfizer Inc. common shareholders
$
0.25
$
0.31
$
0.11
$
0.36
Earnings per common share—diluted:
Income from continuing operations attributable to Pfizer Inc. common shareholders
$
0.25
$
0.30
$
0.11
$
0.36
Discontinued operations
—
net of tax
—
—
—
—
Net income attributable to Pfizer Inc. common shareholders
$
0.25
$
0.31
$
0.11
$
0.36
Cash dividends paid per common share
$
0.18
$
0.18
$
0.18
$
0.18
Stock prices
High
$
20.36
$
17.39
$
17.50
$
17.90
Low
$
16.80
$
14.00
$
14.14
$
16.25
The fourth quarter of 2010 reflects historically higher Q4 costs in Cost of sales and Selling, informational and administrative expenses, partially offset by lower charges recorded in Other deductions—
net
.
The fourth quarter of 2010 reflects higher integration charges and restructuring costs, primarily related to our acquisition of Wyeth.
The fourth quarter of 2010 includes a $2.0 billion tax benefit recorded as a result of a settlement of certain tax audits covering the years 2002-2005.
114
2011 Financial Report
(a)
(b)
(c)
(a)
(b)
(c)

Financial Summary
Pfizer Inc. and Subsidiary Companies
2011—Restructuring charges of $2.2 billion related to our cost-reduction and productivity initiatives.
2010—Restructuring charges of $2.2 billion related to our acquisition of Wyeth and other cost-reduction initiatives.
2009—Restructuring charges of $3.0 billion related to our acquisition of Wyeth and other cost-reduction initiatives.
2008—Restructuring charges of $2.6 billion related to our cost-reduction initiatives.
2007—Restructuring charges of $2.5 billion related to our cost-reduction initiatives.
operations—net of tax for the year ended December 31, 2011. Capsugel is presented as a discontinued operation and we have made
certain reclassification adjustments to conform the prior year amounts to current-year presentation.
tax liabilities associated with the acquisition of Wyeth.
those held by our employee benefit trusts). The increase in 2009 was due to the issuance of equity to partially finance the Wyeth acquisition.
YEAR ENDED/AS OF DECEMBER 31,
(MILLIONS, EXCEPT PER COMMON SHARE DATA)
2011
2010
2009
2008
2007
Revenues
$ 67,425
$ 67,057
$ 49,269
$ 47,529
$ 47,733
Research and development expenses
9,112
9,392
7,824
7,924
8,071
Other costs and expenses
42,617
45,057
26,373
26,790
27,728
Acquisition
-
related in
-
process research and development charges
—
125
68
633
283
Restructuring charges and certain acquisition-related costs
2,934
3,201
4,330
2,662
2,524
Income from continuing operations before provision for taxes on income
12,762
9,282
10,674
9,520
9,127
Provision for taxes on income
4,023
1,071
2,145
1,582
977
Income from continuing operations
8,739
8,211
8,529
7,938
8,150
Discontinued operations
—
net of tax
1,312
77
114
188
34
Less: Net income attributable to noncontrolling interests
42
31
8
22
40
Net income attributable to Pfizer Inc.
$ 10,009
$ 8,257
$ 8,635
$ 8,104
$ 8,144
Effective tax rate
—
continuing operations
31.5
%
11.5
%
20.1
%
16.6
%
10.7%
Depreciation and amortization
$ 9,026
$ 8,487
$ 4,757
$ 5,090
$ 5,200
Property, plant and equipment additions
1,660
1,513
1,205
1,701
1,880
Cash dividends paid
6,234
6,088
5,548
8,541
7,975
Working capital
29,659
32,377
24,929
16,748
25,415
Property, plant and equipment, less accumulated depreciation
16,938
18,645
22,291
12,864
15,315
Total assets
188,002
195,014
212,949
111,148
115,268
Long
-
term debt
34,931
38,410
43,192
7,955
7,299
Long
-
term capital
137,149
145,303
151,454
68,637
80,103
Total Pfizer Inc. shareholders
’
equity
82,190
87,813
90,014
57,556
65,010
Earnings per common share
—
basic:
Income from continuing operations attributable to Pfizer Inc. common shareholders
$1.11
$1.02
$1.22
$1.18
$1.17
Discontinued operations
—
net of tax
0.17
0.01
0.02
0.03
—
Net income attributable to Pfizer Inc. common Shareholders
$1.28
$1.03
$1.23
$1.20
$1.18
Earnings per common share
—
diluted:
Income from continuing operations attributable to Pfizer Inc. common shareholders
$1.11
$1.01
$1.21
$1.17
$1.17
Discontinued operations—net of tax
0.17
0.01
0.02
0.03
—
Net income attributable to Pfizer Inc. common shareholders
$1.27
$1.02
$1.23
$1.20
$1.17
Market value per share (December 31)
$21.64
$17.51
$18.19
$17.71
$22.73
Return on Pfizer Inc. shareholders’ equity
11.78
%
10.39
%
13.42
%
13.22
%
11.94%
Cash dividends paid per common share
$0.80
$0.72
$0.80
$1.28
$1.16
Shareholders
’
equity per common share
$10.85
$10.96
$11.19
$8.56
$9.65
Current ratio
2.06:1
2.13:1
1.67:1
1.61:1
2.16:1
Weighted
-
average shares used to calculate:
Basic earnings per common share amounts
7,817
8,036
7,007
6,727
6,917
Diluted earnings per common share amounts
7,870
8,074
7,045
6,750
6,939
For 2011, includes King commencing on the acquisition date of January 31, 2011. For 2009, includes Wyeth commencing on the acquisition date of October 15, 2009.
Research and development expenses includes upfront and milestone payments for intellectual property rights of $306 million in 2011, $393 million in 2010; $489 million in 2009; $377 million in 2008;
and $603 million in 2007.
2010 and 2009 amounts relate to the resolution of a contingency related to our 2008 acquisition of CovX. In 2008 and 2007, we recorded charges for the estimated portion of the purchase price of
acquisitions allocated to in-process research and development.
Restructuring charges and certain acquisition-related costs primarily includes the following:
The sale of the Capsugel business closed on August 1, 2011, and we have recognized a gain related to the sale of Capsugel in Discontinued
Includes discontinued operations.
Defined as long-term debt, noncurrent deferred tax liabilities and total shareholders’ equity. In 2009, increase reflects the long-term debt and deferred
EPS amounts may not add due to rounding.
Represents total Pfizer Inc. shareholders’ equity divided by the actual number of common shares outstanding (which excludes treasury shares and
2011 Financial Report
115
(a)
(b)
(c)
(d)
(e)
(f)
(f)
(g)
(h)
(h)
(i)
(a)
(b)
(c)
(d)
(e)
(
f
)
(g)
(h)
(i)
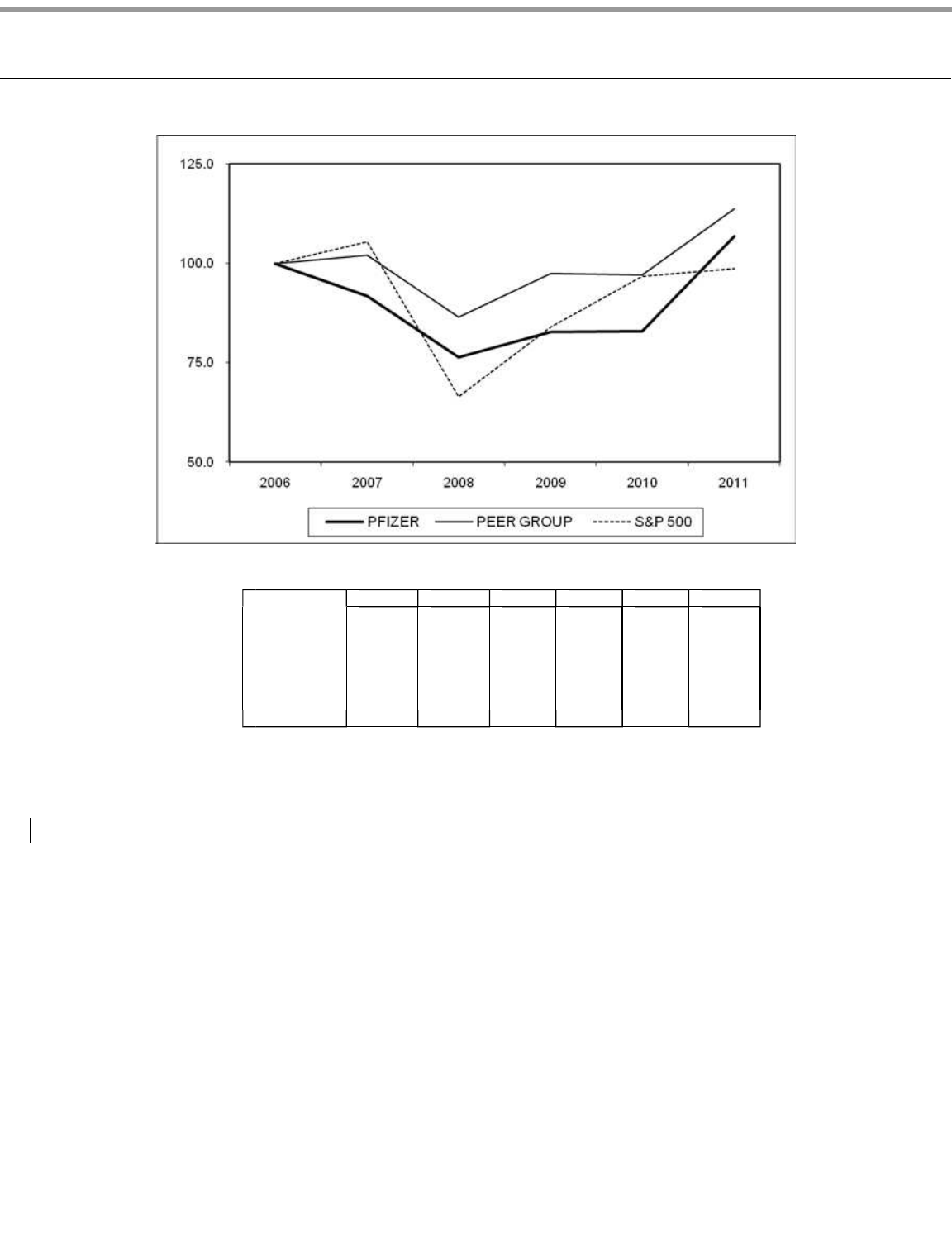
Financial Summary
Pfizer Inc. and Subsidiary Companies
Peer Group Performance Graph
Five Year Performance
2006
2007
2008
2009
2010
2011
PFIZER
100.0
91.9
76.4
82.7
82.9
106.8
PEER
GROUP
100.0
102.0
86.4
97.5
97.1
113.7
S&P 500
100.0
105.5
66.5
84.1
96.7
98.8
Notes:
Pfizer’s pharmaceutical peer group consists of the following
companies: Abbott Laboratories, Amgen, AstraZeneca, Bristol-
Myers Squibb Company, Eli Lilly and Company, GlaxoSmithKline,
Johnson & Johnson and Merck and Co.
116
2011 Financial Report

Exhibit 21
SUBSIDIARIES OF THE COMPANY
The following is a list of subsidiaries of the Company as of December 31, 2011, omitting some subsidiaries which, considered in the aggregate, would not constitute a
significant subsidiary.
NAME
WHERE INCORPORATED
685 TA LLC
Delaware
A. H. Robins (Philippines) Company, Inc.
Philippines
A. H. Robins International Company
Nevada
A/O Pfizer
Russia
AC Acquisition Holding Company
Delaware
Agouron Pharmaceuticals, Inc.
California
AHP Holdings B.V.
Netherlands
AHP Holdings Pty. Limited
Australia
AHP Manufacturing B.V.
Netherlands
Allabinc de Mexico, S.A. de C.V.
Mexico
Alpha
-
Lux Investments S.àr.l.
Luxembourg
Alpharma (Belgium) BVBA
Belgium
Alpharma (Bermuda) Investments Ltd.
Bermuda
Alpharma (Bermuda) Ltd
Bermuda
Alpharma (Bermuda), LLC
Delaware
Alpharma (Luxembourg) S.A.R.L. y Compania Limitada
Chile
Alpharma (Luxembourg) S.àr.l.
Luxembourg
Alpharma Animal Health (Beijing) Trading Co. Ltd.
People
’
s Republic of China
Alpharma Animal Health (Hong Kong) Co. Limited
Hong Kong
Alpharma Animal Health (Shenzhou) Co., Ltd.
People
’
s Republic of China
Alpharma Animal Health (Yantai) Co., Ltd.
People
’
s Republic of China
Alpharma Animal Health Company
Texas
Alpharma Animal Health Italia S.r.l.
Italy
Alpharma Animal Health Pty Ltd.
Australia
Alpharma Bermuda G.P.
Bermuda
Alpharma Canada Corporation
Canada
Alpharma de Argentina S.R.L.
Argentina
Alpharma do Brasil Ltda
Brazil
Alpharma Euro Holdings, LLC
Delaware
Alpharma Holdings (Barbados) SRL
Barbados
Alpharma Holdings Inc.
Delaware
Alpharma International (Luxembourg) Sarl
Luxembourg
Alpharma Ireland Limited
Ireland
Alpharma Operating, LLC
Delaware
Alpharma Pharmaceuticals (Thailand) Limited
Thailand
Alpharma Pharmaceuticals LLC
Delaware
Alpharma Specialty Pharma Inc.
Delaware
Alpharma U.S. Inc.
Delaware
Alpharma USHP Inc.
Delaware
Alpharma, LLC
Delaware
American Food Industries, Inc.
Delaware

NAME
WHERE INCORPORATED
American Home Products Holdings (U.K.) Limited
United Kingdom
Andean Services S.A.
Colombia
Argatroban Royalty Sub LLC
Delaware
Ayerst
-
Wyeth Pharmaceuticals LLC
Delaware
Barre Parent Corporation
Delaware
Berdan Insurance Company
Vermont
BINESA 2002, S.L.
Spain
Biocor Animal Health Inc.
Delaware
Bioren, Inc.
Delaware
BioRexis Pharmaceutical Corporation
Delaware
Blue Whale Re Ltd.
Vermont
C.E. Commercial Holdings C.V.
Netherlands
C.E. Commercial Investments C.V.
Netherlands
C.E. Holdings Europe C.V.
Netherlands
C.P. Pharma Gyógyszerkereskedelmi Korlátolt Felelõsségû Társaság
Hungary
C.P. Pharmaceuticals International C.V.
Netherlands
Carlerba
-
Produtos Químicos e Farmacêuticos, Lda.
Portugal
Charlie Papa Operations, LLC
New Jersey
CICL Corporation
Delaware
COC I Corporation
Delaware
Coley Pharmaceutical GmbH
Germany
Coley Pharmaceutical Group, Inc.
Delaware
Compania Farmaceutica Upjohn, S.A.
Guatemala
Continental Farmaceutica, S.L.
Spain
Continental Pharma, Inc.
Belgium
CovX Research LLC
Delaware
Covx Technologies Ireland Limited
Ireland
Cyanamid de Argentina S.A.
Delaware
Cyanamid de Colombia, S.A.
Delaware
Cyanamid Inter
-
American Corporation
Delaware
Cyanamid of Great Britain Limited
United Kingdom
Davis Medica, S.L.
Spain
Design Group AS
Norway
Design Group Sverige AB
Sweden
Distribuidora Mercantil Centro Americana, S.A
Delaware
Durgon Holdings Limited
British Virgin Islands
Ejendomsselskabet Sydmarken 5 A/S
Denmark
Embrex Bio
-
Tech Trade (Shanghai) Co., Ltd.
People
’
s Republic of China
Embrex Europe Limited
United Kingdom
Embrex Poultry Health, LLC
North Carolina
Embrex, Inc.
North Carolina
Empresa Laboratories de Mexico S.A. de C.V.
Mexico
Encysive (UK) Limited
United Kingdom
Encysive Pharmaceuticals Inc.
Delaware
Encysive, L.P.
Delaware
EP
-
ET, LLC
Delaware

NAME
WHERE INCORPORATED
Esperion LUV Development, Inc.
Delaware
Eurovita A/S
Denmark
Eurovita Holding A/S
Denmark
Eurovita International A/S
Denmark
Eurovita Trading Limited
Ireland
Excaliard Pharmaceuticals, Inc.
Delaware
Farminova Produtos Farmaceuticos de Inovacao, Lda.
Portugal
Farmitalia Carlo Erba Limited
United Kingdom
Farmogene Productos Farmaceuticos Lda
Portugal
FeNSG AS
Norway
Ferrosan A/S
Denmark
Ferrosan AB
Sweden
Ferrosan Do Brasil Ltda.
Brazil
Ferrosan Finance S.A.
Panama
Ferrosan Holding A/S
Denmark
Ferrosan Inc.
Texas
Ferrosan International A/S
Denmark
Ferrosan Limited
United Kingdom
Ferrosan Norge AS
Norway
Ferrosan Poland Sp. z o.o
Poland
Ferrosan S.R.L.
Romania
FoldRx Pharmaceuticals, Inc.
Delaware
Fort Dodge (Hong Kong) Limited
Hong Kong
Fort Dodge Animal Health Limited
United Kingdom
Fort Dodge Animal Health Philippines, Inc.
Philippines
Fort Dodge Animal Health, S. de R.L. de C.V.
Mexico
Fort Dodge Asia Exports, Inc.
Delaware
Fort Dodge Australia Pty. Limited
Australia
Fort Dodge de Venezuela, C.A.
Venezuela
Fort Dodge Laboratories Inc.
Iowa
Fort Dodge Manufatura Ltda.
Brazil
Fort Dodge Sanidad Animal S.A.
Argentina
Fort Dodge Saude Animal Ltda.
Brazil
G. D. Searle & Co. Limited
United Kingdom
G. D. Searle International Capital LLC
Delaware
G. D. Searle LLC
Delaware
Genetics Institute, LLC
Delaware
GenTrac, Inc.
Wisconsin
GI Europe, Inc.
Delaware
GI Japan, Inc.
Delaware
Gödecke GmbH
Germany
Grangematic Limited
Ireland
Greenstone LLC
Delaware
Hälseprodukter Forserum AB
Sweden
Haptogen Limited
United Kingdom
Icagen, Inc.
Delaware

NAME
WHERE INCORPORATED
ImmunoPharmaceutics, Inc.
California
Industrial Santa Agape, S.A.
Guatemala
Instituto Pasteur de Lisboa Virginio Leitao Vieira dos Santos & Filhos S.A.
Portugal
Interfarma
-
Produtos Quimicos e Farmaceuticos, Lda.
Portugal
International Affiliated Corporation LLC
Delaware
Invicta Farma, S.A.
Spain
JMI
-
Daniels Pharmaceuticals, Inc.
Florida
John Wyeth & Brother Limited
United Kingdom
Kenfarma, S.A.
Spain
Kiinteistö oy Espoon Pellavaniementie 14
Finland
King Pharmaceuticals Canada, Inc.
Canada
King Pharmaceuticals Holdings, Inc.
Delaware
King Pharmaceuticals Research and Development, Inc.
Delaware
King Pharmaceuticals, Inc.
Tennessee
Kommanditbolaget Hus Gron
Sweden
Korea Pharma Holding Company Limited
Hong Kong
Laboratoires Pfizer SA
Morocco
Laboratorios Parke Davis, S.L.
Spain
Laboratorios Pfizer Ltda.
Brazil
Laboratórios Pfizer, Lda.
Portugal
Laboratorios Wyeth Inc.
Delaware
Laboratorios Wyeth S.A.
Peru
Laboratorios Wyeth S.A.
Venezuela
Lederle S.L.
Spain
LLC Ferrosan Consumer Health
Russia
Lothian Developments V SPRL
Belgium
MDP Holdings, Inc.
Delaware
MED Urological, Inc.
Minnesota
Meridian Medical Technologies Limited
Ireland
Meridian Medical Technologies, Inc.
Delaware
Meridica Limited
United Kingdom
Mikjan Corporation
Arkansas
Monarch Pharmaceuticals Ireland Limited
Ireland
Monarch Pharmaceuticals, Inc.
Tennessee
MPP Trustee Limited
United Kingdom
MTG Divestitures LLC
Delaware
Nefox Farma, S.A.
Spain
Neusentis Limited
United Kingdom
Nordic Sales Group AS
Norway
Nostrum Farma, S.A.
Spain
Nutrifarma Ferrosan Sa
ğ
lik Ürün ve Hizmetieri A.
Ş
.
Turkey
O.C.T. (Thailand) Ltd.
Thailand
Orfi Farma S.L.
Spain
Oy Ferrosan AB
Finland
PAH Colombia S.A.S.
Colombia
PAH Holdco SARL
Luxembourg

NAME
WHERE INCORPORATED
PAH Holdings LLC
Delaware
PAH Luxembourg 1 SARL
Luxembourg
PAH Luxembourg 2 SARL
Luxembourg
PAH Luxembourg 3 SARL
Luxembourg
PAH Luxembourg 4 SARL
Luxembourg
PAH Luxembourg 5 SARL
Luxembourg
PAH Mexico Holdco SARL
Luxembourg
PAH Panama B.V.
Netherlands
PAH West Europe 2 SARL
Luxembourg
PAH West Europe SARL
Luxembourg
Parke Davis Limited
Hong Kong
Parke Davis Productos Farmaceuticos Lda
Portugal
Parke, Davis & Company LLC
Michigan
Parkedale Pharmaceuticals, Inc.
Michigan
Parke
-
Davis Manufacturing Corp.
Delaware
P
-
D Co., Inc.
Delaware
Peak Enterprises LLC
Delaware
PF Americas Holding C.V.
Netherlands
PF Asia Manufacturing Coöperatief U.A.
Netherlands
PF PR Holdings C.V.
Netherlands
PF PRISM C.V.
Netherlands
PF PRISM Holdings S.a.r.l.
Luxembourg
PF Prism S.á.r.l.
Luxembourg
PF Prism US LLC
Delaware
Pfizer (China) Research and Development Co. Ltd.
People
’
s Republic of China
Pfizer (Far East) Limited
Hong Kong
Pfizer (H.K.) Holding Limited
Hong Kong
Pfizer (Malaysia) Sdn Bhd
Malaysia
Pfizer (Perth) Pty Limited
Australia
Pfizer (S.A.S.)
France
Pfizer (Thailand) Limited
Thailand
Pfizer (Wuhan) Research and Development Co. Ltd.
People
’
s Republic of China
Pfizer AB
Sweden
Pfizer Africa & Middle East for Pharmaceuticals, Veterinary Products & Chemicals S.A.E.
Egypt
Pfizer Afrique de L
’
Ouest
Senegal
Pfizer AG
Switzerland
Pfizer Animal Health B.V.
Netherlands
Pfizer Animal Health India Limited
India
Pfizer Animal Health Japan G.K.
Japan
Pfizer Animal Health Korea Ltd.
Republic of Korea
Pfizer Animal Health MA EEIG
United Kingdom
Pfizer Animal Health S.A.
Belgium
Pfizer ApS
Denmark
Pfizer AS
Norway
Pfizer Asia Manufacturing Pte. Ltd.
Singapore

NAME
WHERE INCORPORATED
Pfizer Asia Pacific Pte Ltd.
Singapore
Pfizer AsiaPac Holdings SARL
Luxembourg
Pfizer Asset Management Luxembourg SARL
Luxembourg
Pfizer Atlantic Holdings S.a.r.l.
Luxembourg
Pfizer Australia Holdings Pty Limited
Australia
Pfizer Australia Investments B.V.
Netherlands
Pfizer Australia Investments Pty. Ltd.
Australia
Pfizer Australia Pty Limited
Australia
Pfizer B.V.
Netherlands
Pfizer BH D.o.o.
Bosnia and Herzegovina
Pfizer Biologics Ireland Holdings Limited
Ireland
Pfizer Biotech Corporation
Taiwan
Pfizer Biotechnology Ireland
Ireland
Pfizer Bolivia S.A.
Bolivia
Pfizer Canada Inc.
Canada
Pfizer CentreSource Asia Pacific Pte. Ltd.
Singapore
Pfizer Chile S.A.
Chile
Pfizer Cia. Ltda.
Ecuador
Pfizer Commercial Holdings Coöperatief U.A.
Netherlands
Pfizer Consumer Healthcare GmbH
Germany
Pfizer Consumer Healthcare Ltd.
United Kingdom
Pfizer Continental Holdings SARL
Luxembourg
Pfizer Continental Services LLC
Delaware
Pfizer Convention III LLC
Delaware
Pfizer Convention IV LLC
Delaware
Pfizer Cork Limited
Ireland
Pfizer Corporation
Panama
Pfizer Corporation Austria Gesellschaft m.b.H.
Austria
Pfizer Corporation Hong Kong Limited
Hong Kong
Pfizer Croatia d.o.o.
Croatia
Pfizer Deutschland GmbH
Germany
Pfizer Development LP
United Kingdom
Pfizer Development Services (UK) Limited
United Kingdom
Pfizer Distribution Company
Ireland
Pfizer Domestic Ventures Limited
Isle of Jersey
Pfizer Dominicana, S.A.
Dominican Republic
Pfizer Eastern Investments B.V.
Netherlands
Pfizer Egypt S.A.E.
Egypt
Pfizer Enterprises Inc.
Delaware
Pfizer Enterprises SARL
Luxembourg
Pfizer ESP Pty Ltd
Australia
Pfizer Europe Holdings SARL
Luxembourg
Pfizer Europe MA EEIG
United Kingdom
Pfizer Europe Services LLC
Delaware
Pfizer European Service Center BVBA
Belgium
Pfizer Export AB
Sweden

NAME
WHERE INCORPORATED
Pfizer Export Company
Ireland
Pfizer Finance GmbH & Co. KG
Germany
Pfizer Finance Holding S.r.l.
Italy
Pfizer Finance Italy S.r.l.
Italy
Pfizer Finance Share Service (Dalian) Co., Ltd.
People
’
s Republic of China
Pfizer Finance Verwaltungs GmbH
Germany
Pfizer Financial Services N.V./S.A.
Belgium
Pfizer France Investment Holdings
France
Pfizer Global Holdings B.V.
Netherlands
Pfizer Global Investments SARL
Luxembourg
Pfizer Global Supply
Ireland
Pfizer Global Supply Japan Inc.
Japan
Pfizer Global Trading
Ireland
Pfizer GmbH
Germany
Pfizer Gulf FZ
-
LLC
United Arab Emirates
Pfizer H.C.P. Corporation
New York
Pfizer Health AB
Sweden
Pfizer Health Solutions Inc.
Delaware
Pfizer Healthcare Consultant (Shanghai) Co., Ltd
People
’
s Republic of China
Pfizer Healthcare Ireland
Ireland
Pfizer Hellas, A.E.
Greece
Pfizer HK Service Company Limited
Hong Kong
Pfizer Holding France (S.C.A.)
France
Pfizer Holding Italy S.p.A.
Italy
Pfizer Holding Ventures
Ireland
Pfizer Holdings Europe
Ireland
Pfizer Holdings International Luxembourg (PHIL) Sarl
Luxembourg
Pfizer Holdings K.K.
Japan
Pfizer Holdings Luxembourg SARL
Luxembourg
Pfizer Holdings Netherlands B.V.
Netherlands
Pfizer Holdings North America SARL
Luxembourg
Pfizer Holdings Turkey Limited
Isle of Jersey
Pfizer Hungary Asset Management LLC
Hungary
Pfizer Ilaclari Limited Sirketi
Turkey
Pfizer International Business Europe
Ireland
Pfizer International Corporation
Panama
Pfizer International Holdings
Ireland
Pfizer International Investments Ltd.
Bermuda
Pfizer International LLC
New York
Pfizer International Luxembourg SA
Luxembourg
Pfizer International Operations (S.A.S.)
France
Pfizer International Sweden
Sweden
Pfizer International Trading (Shanghai) Limited
People
’
s Republic of China
Pfizer Investment Capital
Ireland
Pfizer Investment Co. Ltd.
People
’
s Republic of China
Pfizer Investment Holdings S.a.r.l.
Luxembourg

NAME
WHERE INCORPORATED
Pfizer Investments Netherlands B.V.
Netherlands
Pfizer Ireland Investments Limited
Ireland
Pfizer Ireland Pharmaceuticals
Ireland
Pfizer Ireland Ventures
Ireland
Pfizer Italia S.r.l.
Italy
Pfizer Japan Inc.
Japan
Pfizer Jersey Capital Limited
Isle of Jersey
Pfizer Jersey Company Limited
Isle of Jersey
Pfizer Jersey Finance Limited
Isle of Jersey
Pfizer Laboratories (Pty) Limited
South Africa
Pfizer Laboratories Limited
Kenya
Pfizer Limitada
Angola
Pfizer Limited
India
Pfizer Limited
Taiwan
Pfizer Limited
Tanzania
Pfizer Limited
United Kingdom
Pfizer LLC
Russia
Pfizer Luxco Holdings Sarl
Luxembourg
Pfizer Luxembourg SARL
Luxembourg
Pfizer Manufacturing Belgium N.V.
Belgium
Pfizer Manufacturing Deutschland GmbH
Germany
Pfizer Manufacturing Holdings Coöperatief U.A.
Netherlands
Pfizer Manufacturing Holdings LLC
Delaware
Pfizer Manufacturing Ireland
Ireland
Pfizer Manufacturing LLC
Delaware
Pfizer Manufacturing Services
Ireland
Pfizer Medical Technology Group (Belgium) N.V.
Belgium
Pfizer Medicamentos Genericos e Participacoes Ltda.
Brazil
Pfizer Mexico Luxco SARL
Luxembourg
Pfizer Mexico, S.A. de C.V.
Mexico
Pfizer Middle East for Pharmaceuticals, Animal Health and Chemicals S.A.E.
Egypt
Pfizer New Zealand Investments Limited
New Zealand
Pfizer New Zealand Limited
New Zealand
Pfizer North American Holdings Inc.
Delaware
Pfizer Nutritionals Ireland Limited
Ireland
Pfizer Olot, S.L.
Spain
Pfizer OTC B.V.
Netherlands
Pfizer Overseas LLC
Delaware
Pfizer Overseas Services Inc.
Delaware
Pfizer Oy
Finland
Pfizer Pacific Coöperatief U.A.
Netherlands
Pfizer Pacific Holdings B.V.
Netherlands
Pfizer Pacific Investments B.V.
Netherlands
Pfizer Pakistan Limited
Pakistan
Pfizer Parke Davis
Philippines

NAME
WHERE INCORPORATED
Pfizer Parke Davis (Thailand) Ltd.
Thailand
Pfizer Parke Davis Pte. Ltd.
Singapore
Pfizer PGM (S.A.S.)
France
Pfizer PGRD (S.A.S.)
France
Pfizer Pharm Algerie
Algeria
Pfizer Pharma GmbH
Germany
Pfizer Pharma Trade LLC
Egypt
Pfizer Pharmaceutical (Wuxi) Co., Ltd.
People
’
s Republic of China
Pfizer Pharmaceutical India Pvt. Ltd.
India
Pfizer Pharmaceutical Trading Limited Liability Company (a/k/a Pfizer Kft. or Pfizer LLC)
Hungary
Pfizer Pharmaceuticals B.V.
Netherlands
Pfizer Pharmaceuticals Global Coöperatief U.A.
Netherlands
Pfizer Pharmaceuticals Israel Ltd.
Israel
Pfizer Pharmaceuticals Korea Limited
Republic of Korea
Pfizer Pharmaceuticals Limited
Cayman Islands
Pfizer Pharmaceuticals LLC
Delaware
Pfizer Pharmaceuticals Ltd.
People
’
s Republic of China
Pfizer Pharmaceuticals Tunisie Sarl
Tunisia
Pfizer PHF
Ireland
Pfizer Philippines Holdings B.V.
Netherlands
Pfizer Pigments Inc.
Delaware
Pfizer Polska Sp. z.o.o.
Poland
Pfizer Precision Holdings SARL
Luxembourg
Pfizer Private Limited
Malaysia
Pfizer Private Ltd.
Singapore
Pfizer Production LLC
Delaware
Pfizer Products Inc.
Connecticut
Pfizer Products India Private Limited
India
Pfizer Romania SRL
Romania
Pfizer S.A.
Peru
Pfizer S.A. (Belgium)
Belgium
Pfizer S.A.S.
Colombia
Pfizer S.G.P.S. Lda.
Portugal
Pfizer S.R.L.
Argentina
Pfizer Saidal Manufacturing
Algeria
Pfizer Santé Familiale SAS
France
Pfizer Saudi Limited
Saudi Arabia
Pfizer Science and Technology Ireland Limited
Ireland
Pfizer Searle Investment Limited
Isle of Jersey
Pfizer Service Company BVBA
Belgium
Pfizer Service Company Ireland
Ireland
Pfizer Services 1 (S.N.C.)
France
Pfizer Services 3 (S.N.C.)
France
Pfizer Services 4 (S.N.C.)
France
Pfizer Services LLC
Delaware

NAME
WHERE INCORPORATED
Pfizer Shared Services
Ireland
Pfizer Shareholdings
Intermediate SARL
Luxembourg
Pfizer Singapore Trading Pte. Ltd.
Singapore
Pfizer Spain Holdings Coöperatief U.A.
Netherlands
Pfizer Specialities Ghana
Ghana
Pfizer Specialties Limited
Nigeria
Pfizer Specialty UK Limited
United Kingdom
Pfizer Sterling Investments Limited
Isle of Jersey
Pfizer Strategic Investment Company Limited
Isle of Jersey
Pfizer Suzhou Animal Health Products Co., Ltd.
People
’
s Republic of China
Pfizer Suzhou Pharmaceutical Co., Ltd.
People
’
s Republic of China
Pfizer Trading Polska sp.z.o.o.
Poland
Pfizer Transactions Ireland
Ireland
Pfizer Transactions LLC
Delaware
Pfizer Transactions Luxembourg SARL
Luxembourg
Pfizer Tunisie SA
Tunisia
Pfizer UK Group Limited
United Kingdom
Pfizer Ukraine
Ukraine
Pfizer Vaccines LLC
Delaware
Pfizer Venezuela, S.A.
Venezuela
Pfizer Warner Lambert Luxembourg SARL
Luxembourg
Pfizer Zona Franca, S.A.
Costa Rica
Pfizer, Inc.
Philippines
Pfizer, S.A.
Costa Rica
Pfizer, S.A. de C.V.
Mexico
Pfizer, S.L.
Spain
Pfizer, spol. s r.o.
Czech Republic
Pharmacia & Upjohn Company LLC
Delaware
Pharmacia & Upjohn Company, Inc.
Delaware
Pharmacia & Upjohn LLC
Delaware
Pharmacia & Upjohn Trading Corporation
Michigan
Pharmacia & Upjohn, S.A. de C.V.
Mexico
Pharmacia (South Africa) (Pty) Ltd
South Africa
Pharmacia Brasil Ltda.
Brazil
Pharmacia Corporation
Delaware
Pharmacia de Centroamerica S.A.
Panama
Pharmacia GmbH
Germany
Pharmacia Grupo Pfizer, S.L.
Spain
Pharmacia Hepar Inc.
Delaware
Pharmacia Holding AB
Sweden
Pharmacia Inter
-
American LLC
Michigan
Pharmacia International B.V.
Netherlands
Pharmacia International Inc.
South Dakota
Pharmacia Ireland
Ireland
Pharmacia Korea Ltd.
Republic of Korea
Pharmacia Laboratories Limited
United Kingdom

NAME
WHERE INCORPORATED
Pharmacia Limited
United Kingdom
Pharmacia Limited Company
Michigan
Pharmacia Malaysia Sdn Bhd
Malaysia
Pharmacia Searle Limited
United Kingdom
PHIVCO Corp.
Delaware
PHIVCO Holdco S.à r.l.
Luxembourg
PHIVCO Luxembourg SARL
Luxembourg
PN Mexico LLC
Delaware
PN North America, S. de R.L. de C.V.
Mexico
PowderJect Research Limited
United Kingdom
PowderJect Vaccines, Inc.
Delaware
PowderMed Limited
United Kingdom
PowderMed, Inc.
Delaware
Preve Oy
Finland
Prosec (Ireland) Limited
Ireland
Prosec Forsakrings AB (Prosec Insurance Co. Ltd.)
Sweden
PT. Fort Dodge Indonesia
Indonesia
PT. Pfizer Indonesia
Indonesia
PT. Wyeth Indonesia
Indonesia
Purepac Pharmaceutical Holdings, Inc.
Delaware
PZR Ltd.
United Kingdom
PZR Property Limited
United Kingdom
Quigley Company, Inc.
New York
Renrall LLC
Wyoming
Rinat Neuroscience Corp.
Delaware
Rivepar (S.A.S.)
France
RMV Produtos Veterinarios Ltda.
Brazil
Roerig Produtos Farmaceuticos, Lda.
Portugal
Roerig S.A.
Chile
Roerig, S.A.
Venezuela
Route 24 Holdings, Inc.
Delaware
Sao Cristovao Participacoes Ltda.
Brazil
Searle Laboratorios, Lda.
Portugal
Searle Ltd.
Bermuda
Servicios P&U, S.de R.L. de C.V.
Mexico
Shiley International
California
Shiley LLC
California
Sinergis Farma
-
Produtos Farmaceuticos, Lda.
Portugal
Site Realty, Inc.
Delaware
Solinor LLC
Delaware
STI International Limited
United Kingdom
Sugen, Inc.
Delaware
Synbiotics Corporation
California
Synbiotics Europe S.A.S.
France
Tabor Corporation
Delaware
The Pfizer Incubator LLC
Delaware

NAME
WHERE INCORPORATED
Thiakis Limited
United Kingdom
Trans
-
Europe Assurance Limited
Ireland
Trans
-
Europe Holdings Inc.
Delaware
Upjohn International Holding Company
Delaware
Upjohn Laboratorios Lda.
Portugal
Upjohn Pharmaceuticals Limited
Delaware
US Oral Pharmaceuticals Pty Ltd
Australia
Vermont Whey Company
Vermont
Vesterålens Naturprodukter A/S
Denmark
Vesterålens Naturprodukter AB
Sweden
Vesterålens Naturprodukter AS
Norway
Vesterålens Naturprodukter OY
Finland
Vicuron Holdings LLC
Delaware
Vicuron Pharmaceuticals Italy S.r.l.
Italy
Vinci Farma, S.A.
Spain
Warner Lambert (UK) Limited
United Kingdom
Warner Lambert Company (M) Sdn Bhd
Malaysia
Warner Lambert del Uruguay S.A.
Uruguay
Warner Lambert Ilac Sanayi ve Ticaret Limited Sirketi
Turkey
Warner Lambert Poland Sp.z.o.o.
Poland
Warner
-
Lambert (Thailand) Limited
Thailand
Warner
-
Lambert Company AG
Switzerland
Warner
-
Lambert Company LLC
Delaware
Warner
-
Lambert de El Salvador, S.A. de C.V.
El Salvador
Warner
-
Lambert de Honduras, Sociedad Anonima
Honduras
Warner
-
Lambert de Puerto Rico, Inc.
Puerto Rico
Warner
-
Lambert Guatemala, Sociedad Anonima
Guatemala
Warner
-
Lambert Ireland
Ireland
Warner
-
Lambert Pottery Road Limited
Ireland
Warner
-
Lambert, S.A.
Delaware
WCH Netherlands East LLC
Delaware
WCH Netherlands West LLC
Delaware
Whitehall International Inc.
New York
Whitehall Laboratories Inc.
Delaware
Whitehall Laboratorios S.A.
Uruguay
WL de Guatemala, Sociedad Anonima
Guatemala
W
-
L LLC
Delaware
Wyeth (Asia) Limited
Delaware
Wyeth (Far East) Limited
Hong Kong
Wyeth (H.K.) Limited
Hong Kong
Wyeth (Hong Kong) Holding Company Limited
Hong Kong
Wyeth (Malaysia) SDN. BHD.
Malaysia
Wyeth (Shanghai) Trading Company Limited
People
’
s Republic of China
Wyeth (Singapore) Pte. Ltd.
Singapore
Wyeth (Thailand) Ltd.
Thailand
Wyeth AB
Sweden

NAME
WHERE INCORPORATED
Wyeth Advertising Inc.
New York
Wyeth Australia Pty. Limited
Australia
Wyeth Ayerst Inc.
Delaware
Wyeth Ayerst SARL
Luxembourg
Wyeth Consumer Healthcare Ltd.
Delaware
Wyeth Consumer Healthcare Pty. Limited
Australia
Wyeth Egypt Ltd.
Egypt
Wyeth Egypt Trading Ltd.
Egypt
Wyeth Europa Limited
United Kingdom
Wyeth Farma, S.A.
Spain
Wyeth Holdings Corporation
Maine
Wyeth Ilaclari A.S.
Delaware
Wyeth Industria Farmaceutica Ltda.
Brazil
Wyeth KFT.
Hungary
Wyeth Korea, Inc.
Republic of Korea
Wyeth Lederle S.p.A.
Italy
Wyeth Lederle Vaccines S.A.
Belgium
Wyeth Limited
India
Wyeth LLC
Delaware
Wyeth Nutritional (China) Co., Ltd.
People
’
s Republic of China
Wyeth Nutritionals (Singapore) PTE. LTD.
Singapore
Wyeth Nutritionals Inc.
Delaware
Wyeth OOO
Russia
Wyeth Pakistan Limited
Pakistan
Wyeth Pharmaceutical Co., Ltd.
People
’
s Republic of China
Wyeth Pharmaceuticals Central America Services, S.A.
Panama
Wyeth Pharmaceuticals Company
Puerto Rico
Wyeth Pharmaceuticals FZ
-
LLC
United Arab Emirates
Wyeth Pharmaceuticals Inc.
Delaware
Wyeth Pharmaceuticals India Private Limited
India
Wyeth Pharmaceuticals Limited
Ireland
Wyeth Pharmaceuticals S. de R.L. de C.V.
Mexico
Wyeth Philippines, Inc.
Philippines
Wyeth Puerto Rico, Inc.
Puerto Rico
Wyeth Regional Manufacturing (Singapore) PTE. LTD.
Singapore
Wyeth Research Ireland Limited
Ireland
Wyeth S.A.
Argentina
Wyeth S.A. de C.V.
Mexico
Wyeth Subsidiary Illinois Corporation
Illinois
Wyeth Whitehall Export GmbH
Austria
Wyeth Whitehall SARL
Luxembourg
Wyeth
-
Ayerst (Asia) Limited
Delaware
Wyeth
-
Ayerst International Inc.
New York
Wyeth
-
Ayerst Promotions Limited
Delaware
Yusafarm D.O.O.
Serbia

Exhibit 23
Consent of Independent Registered Public Accounting Firm
To the Shareholders and the Board of Directors of Pfizer Inc.:
We consent to the incorporation by reference in this Form 10-
K of Pfizer Inc. of our reports dated February 28, 2012, with respect to the consolidated balance sheets of
Pfizer Inc. and Subsidiary Companies as of December 31, 2011 and 2010, and the related consolidated statements of income, shareholders’
equity and cash flows for
each of the years in the three-
year period ended December 31, 2011, and the effectiveness of internal control over financial reporting as of December 31, 2011, which
reports appear in the December 31, 2011 annual report on Form 10-K of Pfizer Inc. and Subsidiary Companies.
We also consent to the incorporation by reference of our reports in the following Registration Statements:
-Form S-8 dated October 27, 1983 (File No. 2-87473),
-Form S-8 dated March 22, 1990 (File No. 33-34139),
-Form S-8 dated January 24, 1991 (File No. 33-38708),
-Form S-8 dated November 18, 1991 (File No. 33-44053),
-Form S-8 dated May 27, 1993 (File No. 33-49631),
-Form S-8 dated May 19, 1994 (File No. 33-53713),
-Form S-8 dated October 5, 1994 (File No. 33-55771),
-Form S-8 dated December 20, 1994 (File No. 33-56979),
-Form S-8 dated March 29, 1996 (File No. 33-02061),
-Form S-8 dated September 25, 1997 (File No. 333-36371),
-Form S-8 dated April 24, 1998 (File No. 333-50899),
-Form S-8 dated April 22, 1999 (File No. 333-76839),
-Form S-8 dated June 19, 2000 (File No. 333-39610),
-Form S-8 dated April 27, 2001 (File No. 333-59660),
-Form S-8 dated April 27, 2001 (File No. 333-59654),
-Form S-8 dated April 16, 2003 (File No. 333-104581),
-Form S-8 dated April 16, 2003 (File No. 333-104582),
-Form S-8 dated November 18, 2003 (File No. 333-110571),
-Form S-8 dated December 18, 2003 (File No. 333-111333),
-Form S-8 dated April 26, 2004 (File No. 333-114852),
-Form S-8 dated March 1, 2007 (File No. 333-140987),
-Form S-4 dated March 27, 2009 (File No. 333-158237),
-Form S-3 dated October 15, 2009 (File No. 333-162487),
-Form S-8 dated October 16, 2009 (File No. 333-162519),
-Form S-8 dated October 16, 2009 (File No. 333-162520),
-Form S-8 dated October 16, 2009 (File No. 333-162521) and
-Form S-8 dated March 1, 2010 (File No. 333-165121).
/s/ KPMG LLP
New York, New York
February 28, 2012

Exhibit 31.1
Certification by the Chief Executive Officer Pursuant to
Section 302 of the Sarbanes-Oxley Act of 2002
I, Ian C. Read, certify that:
1. I have reviewed this report on Form 10-K of Pfizer Inc.;
2. Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements
made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3. Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial
condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4. The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act
Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
a) Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure
that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during
the period in which this report is being prepared;
b) Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to
provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with
generally accepted accounting principles;
c) Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of
the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d) Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal
quarter that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
5. The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s
auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions):
a) All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to
adversely affect the registrant’s ability to record, process, summarize and report financial information; and
b) Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over
financial reporting.
Date: February 28, 2012
/s/ I
AN
C. R
EAD
Ian C. Read
Chairman and Chief Executive Officer

Exhibit 31.2
Certification by the Chief Financial Officer Pursuant to
Section 302 of the Sarbanes-Oxley Act of 2002
I, Frank A. D’Amelio, certify that:
1. I have reviewed this report on Form 10-K of Pfizer Inc.;
2. Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements
made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3. Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial
condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4. The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act
Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
a) Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure
that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during
the period in which this report is being prepared;
b) Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to
provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with
generally accepted accounting principles;
c) Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of
the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d) Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal
quarter that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
5. The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s
auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions):
a) All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to
adversely affect the registrant’s ability to record, process, summarize and report financial information; and
b) Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over
financial reporting.
Date: February 28, 2012
/s/ F
RANK
A. D’A
MELIO
Frank A. D’Amelio
Executive Vice President,
Business Operations and
Chief Financial Officer

Exhibit 32.1
Certification by the Chief Executive Officer Pursuant to 18 U. S. C. Section 1350, as Adopted Pursuant to
Section 906 of the Sarbanes-Oxley Act of 2002
Pursuant to 18 U. S. C. Section 1350, I, Ian C. Read, hereby certify that, to the best of my knowledge, the Annual Report on Form 10-K of Pfizer Inc. for the year
ended December 31, 2011 (the “Report”) fully complies with the requirements of Section 13(a) or Section 15(d) of the Securities Exchange Act of 1934, and that the
information contained in that Report fairly presents, in all material respects, the financial condition and results of operations of Pfizer Inc.
/s/ I
AN
C. R
EAD
Ian C. Read
Chairman and Chief Executive Officer
February 28, 2012
This certification accompanies this Report on Form 10-K pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 and shall not, except to the extent required
by such Act, be deemed filed by the Company for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Such certification
will not be deemed to be incorporated by reference into any filing under the Securities Act of 1933, as amended, or the Exchange Act, except to the extent that the
Company specifically incorporates it by reference.

Exhibit 32.2
Certification by the Chief Financial Officer Pursuant to 18 U. S. C. Section 1350, as Adopted Pursuant to
Section 906 of the Sarbanes-Oxley Act of 2002
Pursuant to 18 U. S. C. Section 1350, I, Frank A. D’Amelio, hereby certify that, to the best of my knowledge, the Annual Report on Form 10-K of Pfizer Inc. for
the year ended December 31, 2011 (the “Report”) fully complies with the requirements of Section 13(a) or Section 15(d) of the Securities Exchange Act of 1934, and
that the information contained in that Report fairly presents, in all material respects, the financial condition and results of operations of Pfizer Inc.
/s/ F
RANK
A. D’A
MELIO
Frank A. D’Amelio
Executive Vice President, Business Operations and
Chief Financial Officer
February 28, 2012
This certification accompanies this Report on Form 10-K pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 and shall not, except to the extent required
by such Act, be deemed filed by the Company for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Such certification
will not be deemed to be incorporated by reference into any filing under the Securities Act of 1933, as amended, or the Exchange Act, except to the extent that the
Company specifically incorporates it by reference.
