
IDSA GUIDELINES
International Clinical Practice Guidelines for the
Treatment of Acute Uncomplicated Cystitis and
Pyelonephritis in Women: A 2010 Update by the
Infectious Diseases Society of America and the
European Society for Microbiology and
Infectious Diseases
Kalpana Gupta,
1
Thomas M. Hooton,
2
Kurt G. Naber,
9
Bjo
¨
rn Wullt,
10
Richard Colgan,
3
Loren G. Miller,
4
Gregory J. Moran,
5
Lindsay E. Nicolle,
8
Raul Raz,
11
Anthony J. Schaeffer,
6
and David E. Soper
7
1
Department of Medicine, Veterans Affairs Boston Health Care System and Boston University School of Medicine, Boston, Massachusetts;
2
Department of
Medicine, University of Miami Miller School of Medicine, University of Miami, Miami Florida;
3
Department of Family and Community Medicine, University
of Maryland, Baltimore, Maryland,
4
Division of Infectious Diseases, Harbor -UCLA Medical Center, Torrance, and
5
Department of Emergency Medicine and
Division of Infectious Diseases Olive View-UCLA Medical Center, Slymar , California;
6
Deptartment of urology, Northwestern University , Chicago, Illinois; and
7
Departments of Obstetrics and Gynecology and Medicine, Medical University of South Carolina, Charleston, South Carolina;
8
Department of Internal
Medicine and Department of Medical Mirobiology University of Manitoba, Winnipeg, Canada;
9
Technical University of Munich, Munich, Germany;
10
Lund
University Hospital, Lund, Sweden; and
11
Infectious Diseases Unit, Ha'Emek Medical Center, Afula, and Rappaport Faculty of Medicine, Technion, Haifa, Israel
A Panel of International Experts was convened by the Infectious Diseases Society of America (IDSA) in
collaboration with the European Society for Microbiology and Infectious Diseases (ESCMID) to update the
1999 Uncomplicated Urinary Tract Infection Guidelines by the IDSA. Co-sponsoring organizations include the
American Congress of Obstetricians and Gynecologists, American Urological Association, Association of
Medical Microbiology and Infectious Diseases–Canada, and the Society for Academic Emergency Medicine.
The focus of this work is treatment of women with acute uncomplicated cystitis and pyelonephritis, diagnoses
limited in these guidelines to premenopausal, non-pregnant women with no known urological abnormalities
or co-morbidities. The issues of in vitro resistance prevalence and the ecological adverse effects of
antimicrobial therapy (collateral damage) were considered as important factors in making optimal treatment
choices and thus are reflected in the rankings of recommendations.
EXECUTIVE SUMMARY
BACKGROUND
Acute uncomplicated cystitis remains one of the most
common indications for prescribing of antimicrobials to
otherwise healthy community-dwelling women. Despite
published guidelines for the optimal selection of an
antimicrobial agent and duration of therapy, studies
demonstrate a wide variation in prescribing practices
[1–6]. The Infectious Diseases Society of America (ID-
SA) published a clinical practice guideline on the
treatment of women with acute uncomplicated cystitis
and pyelonephritis in 1999 [1]. Since then, antimicrobial
resistance among uropathogens causing uncomplicated
cystitis has increased, appreciation of the importance of
Received 10 December 2010; accepted 17 December 2010.
The process for evaluating the evidence was based on the IDSA Handbook on
Clinical Practice Guideline Development and involved a systematic weighting of
the quality of the evidence and the grade of recommendation (Table 1) [31]
It is important to realize that guidelines cannot always account for individual
variation among patients. They are not intended to supplant physician judgment
with respect to particular patients or special clinical situations. The IDSA considers
adherence to these guidelines to be voluntary, with the ultimate determination
regarding their application to be made by the physician in the light of each
patient's individual circumstances.
Correspondence: Kalpana Gupta, MD, VA Boston HCS, 1400 VFW Pkwy, 111
Med, West Roxbury, MA 02132 (kalpana.gupta@v a.gov).
Clinical Infectious Diseases 2011;52(5):e103–e120
Ó The Author 2011. Published by Oxford University Press on behalf of the
Infectious Diseases Society of America. All rights reserved. For Permissions,
1058-4838/2011/525-0001$37.00
DOI: 10.1093/cid/ciq257
Clinical Practice Guidelines
d
CID 2011:52 (1 March)
d
e103
the ecological adverse effects of antimicrobial therapy (collateral
damage) has increased, newer agents and different durations of
therapy have been studied, and clinical outcomes have in-
creasingly been reported. In addition, women with uropath-
ogens resistant to the treatment drug have been included in
some studies, allowing for estimations of expected response rates
in a ‘‘real-life’’ clinical setting in which empirical therapy is
prescribed either without a urine culture and susceptibility
testing or before such results are known. In light of these de-
velopments, an update of the guidelines was warranted.
The focus of this guideline is treatment of women with acute
uncomplicated cystitis and pyelonephritis, diagnoses limited
in these guidelines to premenopausal, nonpregnant women
with no known urological abnormalities or comorbidities. It
should be noted that women who are postmenopausal or have
well-controlled diabetes without urological sequelae may be
considered by some experts to have uncomplicated urinary
tract infection (UTI), but a discussion of specific management
of these groups is outside the scope of this guideline. In ad-
dition, management of recurrent cystitis and of UTI in
pregnant women, prevention of UTI, and diagnosis of UTI are
all important issues that are not addressed in this guideline.
The issues of in vitro resistance prevalence and the potential
for collateral damage were considered as important factors in
making optimal treatment choices and thus are reflected in
the rankings of recommendations.
Summarized below are the recommendations made in the
2010 guideline update. The Panel followed a process used in the
development of other IDSA guidelines which included a sys-
tematic weighting of the quality of the evidence and the grade of
recommendation [32] (Table 1). A detailed description of the
methods, background, and evidence summaries that support
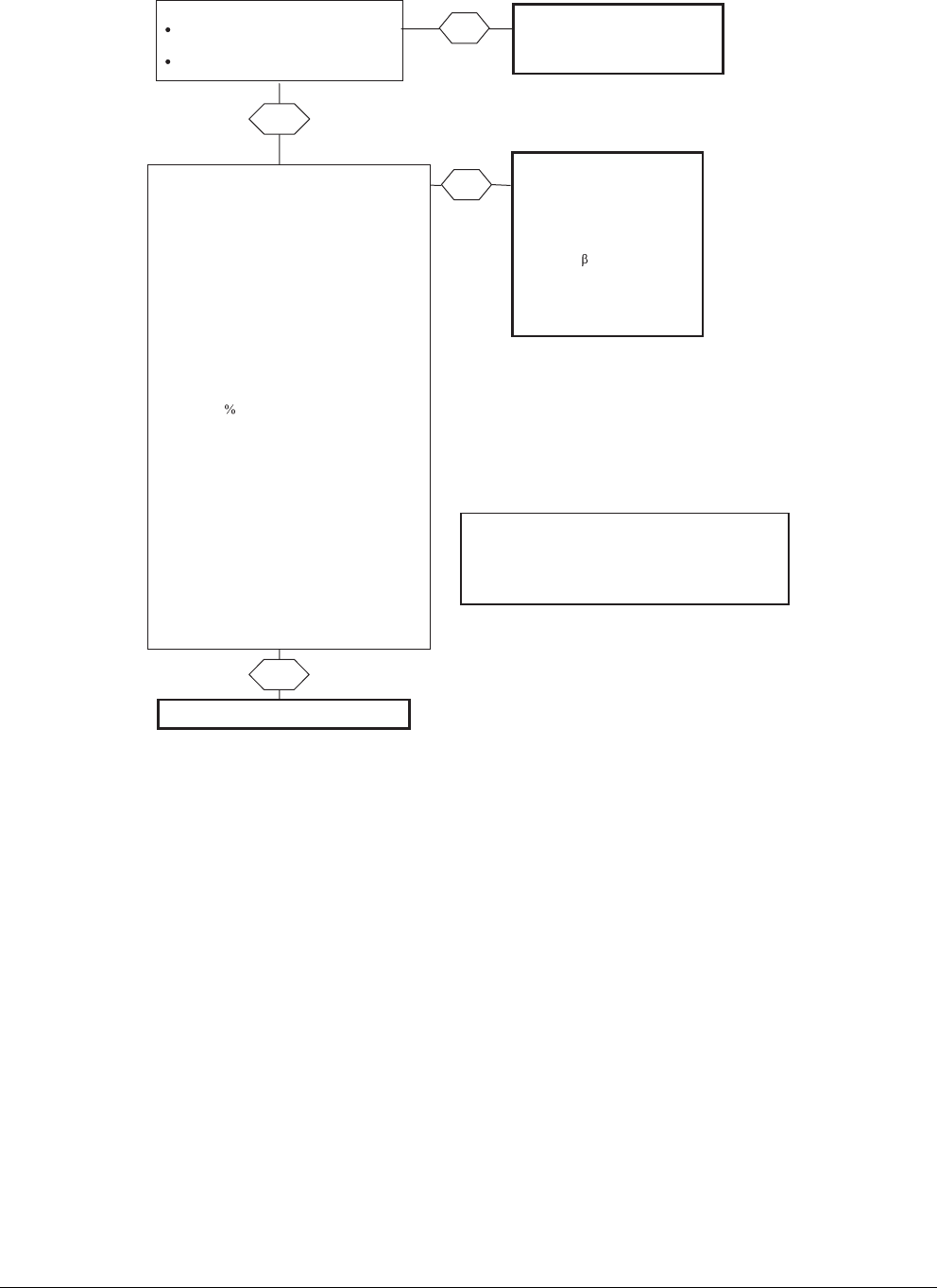
Prescribe a recommended antimicrobial
Consider alternate diagnosis (such
as pyelonephritis or complicated
UTI) & treat accordingly
(see text)
Yes
Woman with acute uncomplicated cystitis
Absence of fever, flank pain, or other
suspicion for pyelonephritis
Able to take oral medication
No
Fluoroquinolones
(resistance prevalence high in
some areas)
OR
-lactams
(avoid ampicillin or amoxicillin
alone; lower efficacy than other
available agents; requires close
follow-up)
No
Can one of the recommended antimicrobials*
below be used considering:
Availability
Allergy history
Tolerance
Nitrofurantoin monohydrate/macrocrystals 100
mg bid X 5 days
(avoid if early pyelonephritis suspected)
OR
Trimethoprim-sulfamethoxazole 160/800 mg
(one DS tablet) bid X 3 days
(avoid if resistance prevalence is known to
exceed 20 or if used for UTI in previous 3
months)
OR
Fosfomycin trometamol 3 gm single dose
(lower efficacy than some other recommended
agents; avoid if early pyelonephritis suspected)
OR
Pivmecillinam 400 mg bid x 5 days
(lower efficacy than some other recommended
agents; avoid if early pyelonephritis suspected)
Yes
*The choice between these agents should be
individualized and based on patient allergy and
compliance history, local practice patterns, local
community resistance prevalence, availability, cost, and
patient and provider threshold for failure (see Table 4)
Figure 1. Approach to choosing an optimal antimicrobial agent for empirical treatment of acute uncomplicated cystitis. DS, double-strength; UTI,
urinary tract infection.
e104
d
CID 2011:52 (1 March)
d
Gupta et al

each of the recommendations can be found in the full text of the
guideline.
I.What Is the Optimal Treatment for Acute Uncomplicated
Cystitis?
Recommendations (Figure 1).
1. Nitrofurantoin monohydrate/macrocrystals (100 mg twice
daily for 5 days) is an appropriate choice for therapy due to
minimal resistance and propensity for collateral damage
(defined above) and efficacy comparable to 3 days of
trimethoprim-sulfamethoxazole (A-I).
2. Trimethoprim-sulfamethoxazole (160/800 mg [1 double-
strength tablet] twice-daily for 3 days) is an appropriate choice
for therapy, given its efficacy as assessed in numerous clinical
trials, if local resistance rates of uropathogens causing acute
uncomplicated cystitis do not exceed 20% or if the infecting
strain is known to be susceptible (A-I).
i. The threshold of 20% as the resistance prevalence at which
the agent is no longer recommended for empirical treatment of
acute cystitis is based on expert opinion derived from clinical,
in vitro, and mathematical modeling studies (B-III).
ii. In some countries and regions, trimethoprim (100 mg twice
daily for 3 days) is the preferred agent and is considered
equivalent to trimethoprim-sulfamethoxazole on the basis of
data presented in the original guideline (A-III) [1].
iii. Data are insufficient to make a recommendation for
othercystitisantimicrobialsastowhatresistanceprevalence
should be used to preclude their use for empirical treatment of
acute cystitis.
3. Fosfomycin trometamol (3 g in a single dose) is an
appropriate choice for therapy where it is available due to
minimal resistance and propensity for collateral damage, but it
appears to have inferior efficacy compared with standard short-
course regimens according to data submitted to the US Food
and Drug Administration (FDA) and summarized in the
Medical Letter (A-I) [7].
4. Pivmecillinam (400 mg bid for 3–7 days) is an
appropriate choice for therapy in regions where it is available
(availability limited to some European countries; not licensed
and/or available for use in North America), because of
minimal resistance and propensity for collateral damage, but
it may have inferior efficacy compared with other available
therapies (A-I).
5. The fluoroquinolones, ofloxacin, ciprofloxacin, and
levofloxacin, are highly efficacious in 3-day regimens (A-I)
but have a propensity for collateral damage and should be
reserved for important uses other than acute cystitis and thus
should be considered alternative antimicrobials for acute
cystitis (A-III).
6. b-Lactam agents, including amoxicillin-clavulanate,
cefdinir, cefaclor, and cefpodoxime-proxetil, in 3–7-day
regimens are appropriate choices for therapy when other
recommended agents cannot be used (B-I). Other b-lactams,
such as cephalexin, are less well studied but may also be
appropriate in certain settings (B-III). The b-lactams generally
have inferior efficacy and more adverse effects, compared with
other UTI antimicrobials (B-I). For these reasons, b-lactams
other than pivmecillinam should be used with caution for
uncomplicated cystitis.
7. Amoxicillin or ampicillin should
not be used for
empirical treatment given the relatively poor efficacy, as
discussed in the 1999 guidelines [1] and the very high
prevalence of antimicrobial resistance to these agents
worldwide [8–11] (A-III).
II.What Is the Treatment for Acute Pyelonephritis?
Recommendations
8. In patients suspected of having pyelonephritis, a urine
culture and susceptibility test should always be performed, and
initial empirical therapy should be tailored appropriately on
the basis of the infecting uropathogen (A-III).
9. Oral ciprofloxacin (500 mg twice daily) for 7 days, with or
without an initial 400-mg dose of intravenous ciprofloxacin, is
an appropriate choice for therapy in patients not requiring
hospitalization where the prevalence of resistance of community
uropathogens to fluoroquinolones is not known to exceed 10%
(A-I). If an initial one-time intravenous agent is used, a long-
acting antimicrobial, such as 1 g of ceftriaxone or a consolidated
24-h dose of an aminoglycoside, could be used in lieu of an
intravenous fluoroquinolone (B-III). If the prevalence of
fluoroquinolone resistance is thought to exceed 10%, an
initial 1-time intravenous dose of a long-acting parenteral
antimicrobial, such as 1 g of ceftriaxone (B-III) or
a consolidated 24-h dose of an aminoglycoside, is
recommended (B-III).
i. Data are insufficient to make a recommendation about
what fluoroquinolone resistance level requires an alternative
agent in conjunction with or to replace a fluoroquinolone
for treatment of pyelonephritis.
10. A once-daily oral fluoroquinolone, including
ciprofloxacin (1000 mg extended release for 7 days)or
levofloxacin (750 mg for 5 days), is an appropriate choice
for therapy in patients not requiring hospitalization where
the prevalence of resistance of community uropathogens is
not known to exceed 10% (B-II). If the prevalence of
fluoroquinolone resistance is thought to exceed 10%, an initial
intravenous dose of a long-acting parenteral antimicrobial, such
as 1 g of ceftriaxone (B-III) or a consolidated 24-h dose of an
aminoglycoside, is recommended (B-III).
Clinical Practice Guidelines
d
CID 2011:52 (1 March)
d
e105

11. Oral trimethoprim-sulfamethoxazole (160/800 mg [1
double-strength tablet] twice-daily for 14 days) is an
appropriate choice for therapy if the uropathogen is known
to be susceptible (A-I). If trimethoprim-sulfamethoxazole is
used when the susceptibility is not known, an initial
intravenous dose of a long-acting parenteral antimicrobial,
such as 1 g of ceftriaxone (B-II) or a consolidated 24-h dose of
an aminoglycoside, is recommended (B-III).
12. Oral b-lactam agents are less effective than other
available agents for treatment of pyelonephritis (B-III). If an
oral b-lactam agent is used, an initial intravenous dose of a
long-acting parenteral antimicrobial, such as 1 g of ceftriaxone
(B-II) or a consolidated 24-h dose of an aminoglycoside, is
recommended (B-III).
i. Data are insufficient to modify the previous guideline
recommendation for a duration of therapy of 10–14 days for
treatment of pyelonephritis with a b-lactam agent.
13. Women with pyelonephritis requiring hospitalization
should be initially treated with an intravenous antimicrobial
regimen, such as a fluoroquinolone; an aminoglycoside, with
or without ampicillin; an extended-spectrum cephalosporin
or extended-spectrum penicillin, with or without an
aminoglycoside; or a carbapenem. The choice between these
agents should be based on local resistance data, and the
regimen should be tailored on the basis of susceptibility results
(B-III).
INTRODUCTION
The focus of this guideline is management of women with acute
uncomplicated cystitis and pyelonephritis who are not pregnant
and have no known urological abnormalities or co-morbidities.
An optimal approach to therapy includes consideration of an-
timicrobial resistance and collateral damage.
Consideration of Antimicrobial Resistance
The microbial spectrum of uncomplicated cystitis and pyelo-
nephritis consists mainly of Escherichia coli (75%–95%), with
occasional other species of Enterobacteriaceae, such as Proteus
mirabilis and Klebsiella pneumoniae, and Staphylococcus sapro-
phyticus. Other gram-negative and gram-positive species are
rarely isolated in uncomplicated UTIs. Therefore, local antimi-
crobial susceptibility patterns of E. coli in particular should be
considered in empirical antimicrobial selection for un-
complicated UTIs. Since the resistance patterns of E. coli strains
causing uncomplicated UTI varies considerably between regions
and countries, a specific treatment recommendation may not be
universally suitable for all regions or countries.
Active surveillance studies of in vitro susceptibility of ur-
opathogens in women with uncomplicated cystitis are helpful
in making decisions about empirical therapy. Four large studies
reporting in vitro susceptibility of E. coli causing un-
complicated UTI in North America and Europe were reviewed
[8–11]. All of these demonstrate considerable geographic var-
iability in susceptibility. For example, resistance rates for all
antimicrobials were higher in US medical centers than in
Canadian medical centers and were usually higher in Portugal
and Spain than other European countries. In general, resistance
rates .20% were reported in all regions for ampicillin, and in
many countries and regions for trimethoprim with or without
sulfamethoxazole. Fluoroquinolone resistance rates were still
,10% in most parts of North America and Europe, but there
was a clear trend for increasing resistance compared with
previous years. Moreover, the resistance data for nalidixic acid
in these studies suggest that .10% (in some countries, .20%)
of the E. coli strains have acquired resistance genes for quino-
lones [10, 11]. First- and second-generation oral cepha-
losporins and amoxicillin-clavulanic acid also show regional
variability, but the resistance rates were generally ,10%. De-
spite wide variability in antimicrobial susceptibility among the
different countries studied, nitrofurantoin, fosfomycin, and
mecillinam (the latter 2 not tested in the Canadian study) had
good in vitro activity in all the countries investigated. Thus,
these 3 antimicrobials could be considered appropriate anti-
microbials for empirical therapy in most regions [8–11]. Given
a trend toward increasing resistance, compared with previous
years, for most antimicrobials, continued monitoring of this
data to evaluate rates over time is necessary for sustained op-
timization of empirical therapy [12].
Because local in vitro resistance rates are not always known,
and change over time is anticipated, identification of individual
predictors of resistance can also be useful to informing empirical
antimicrobial choice. In 2 studies evaluating epidemiological
predictors of resistance, the use of trimethoprim-sulfamethox-
azole in the preceding 3–6 months was an independent risk
factor for trimethoprim-sulfamethoxazole resistance in women
with acute uncomplicated cystitis [13, 14]. In addition, 2 US-
based studies demonstrated that travel outside the United States
in the preceding 3–6 months was independently associated with
trimethoprim-sulfamethoxazole resistance [15, 16]. Predictors
of resistance to other cystitis antimicrobials are not as well
studied but in general support the findings that exposure to the
drug or to an area with endemic resistance are important factors
to consider [17, 18]. Local resistance rates reported in hospital
antibiograms are often skewed by cultures of samples obtained
from inpatients or those with complicated infection and may
not predict susceptibilities in women with uncomplicated
community-acquired infection, in whom resistance rates tend to
be lower [18, 19]. Prospective and unbiased resistance sur-
veillance of uncomplicated uropathogens at the local practice
and/or health care system levels is critical for informing
empirical antimicrobial decisions. In the absence of such
e106
d
CID 2011:52 (1 March)
d
Gupta et al

data, use of individual-level predictors of resistance can be
helpful.
Because treatment of acute uncomplicated cystitis is usually
empirical, it is likely that some women will be treated with a drug
that does not have in vitro activity against the uropathogen. As
the population resistance prevalence of a specific agent increases,
the likelihood of failure outweighs the benefits of using the drug
empirically. For most agents, clinical and bacterial outcomes are
not well studied for varying levels of resistance; thus, recom-
mended thresholds for using alternative agents are based on
expert opinion or secondary analyses of studies that include
patients with isolates resistant to the study drugs. The most ev-
idence in this regard is available for trimethoprim-sulfame-
thoxazole, for which clinical, in vitro, and mathematical
modeling studies consistently suggest a 20% resistance preva-
lence for the threshold at which the agent is no longer recom-
mended for treatment of acute cystitis [20, 21]. There are
insufficient data for other cystitis antimicrobials to recommend
resistance levels at which the likelihood of failure outweighs the
potential benefits, and the decision will vary by individual
practitioner discretion. For pyelonephritis, timely use of an agent
with in vitro activity is essential to treat the infection and min-
imize progression. Thus, thresholds at which a broad-spectrum
agent would be selected empirically followed by directed therapy
or for avoiding selected agents because of anticipated in vitro
resistance are set at a relatively low resistance prevalence. The
recommendation of a 10% fluoroquinolone resistance preva-
lence as the threshold for using an alternative agent in con-
junction with or in place of a fluoroquinolone for pyelonephritis
is primarily based on expert opinion, because there are limited
data to provide evidence-based guidance.
Consideration of Collateral Damage
Collateral damage, a term describing ecological adverse effects of
antimicrobial therapy, such as the selection of drug-resistant
organisms and colonization or infection with multidrug-
resistant organisms, has been associated with use of broad-
spectrum cephalosporins and fluoroquinolones [22, 23]. Use of
broad spectrum cephalosporins has been linked to subsequent
infection with vancomycin-resistant enterococci, extended-
spectrum b-lactamase–producing Klebsiella pneumoniae, b-
lactam-resistant Acinetobacter species, and Clostridium difficile
[22]. Use of fluoroquinolones has been linked to infection with
methicillin-resistant S. aureus and with increasing fluo-
roquinolone resistance in gram-negative bacilli, such as Pseu-
domonas aeruginosa [22]. The preserved in vitro susceptibility of
E. coli to nitrofurantoin, fosfomycin, and mecillinam over many
years of use suggests these antimicrobials cause only minor
collateral damage [8, 10], perhaps because of minimal effects on
normal fecal flora [24–26]. In contrast, increased rates of anti-
microbial resistance have been demonstrated for antimicrobials
that affect the normal fecal flora more significantly, such as
trimethoprim, trimethoprim-sulfamethoxazole, quinolones,
and ampicillin [26, 27].
For uncomplicated cystitis, there are 2 reasons why collateral
damage merits consideration. First, there is minimal risk of
progression to tissue invasion or sepsis. Moreover, studies of
placebo for treatment of uncomplicated cystitis demonstrate
that clinical cure can be achieved in 25%–42% of women who
are not treated or are treated with a drug without in vitro activity
against the uropathogen [28, 29]. Thus, spontaneous resolution
may attenuate differences in clinical outcomes when a drug with
80% efficacy is compared with one with 95% efficacy. Of note,
placebo therapy is associated with prolongation of symptoms as
well as a small risk of progression to pyelonephritis, as dem-
onstrated by the 1 woman out of 38 women treated with placebo
in the study by Christiaens et al [28]. Thus, these data do not
justify withholding antimicrobial therapy for treatment of acute
cystitis. Secondly, uncomplicated UTI is one of the most com-
mon indications for antimicrobial exposure in an otherwise
healthy population; very small increments in collateral damage
repeated many times may in aggregate magnify the impact of
collateral damage when it occurs. Although reducing in-
appropriate use of fluoroquinolones for respiratory infections
could have a greater impact on fluoroquinolone resistance,
limiting use for UTIs may also mitigate increasing fluo-
roquinolone resistance [30].
Clinical Questions Addressed for the 2010 Update
The Expert Panel addressed the following clinical questions in
the 2010 update:
I. What is the optimal treatment for acute uncomplicated
cystitis in adult nonpregnant, premenopausal women?
II. What is the optimal treatment for acute uncomplicated
pyelonephritis in adult nonpregnant, premenopausal women?
PRACTICE GUIDELINES
‘‘Practice guidelines are systematically developed statements to
assist practitioners and patients in making decisions about ap-
propriate health care for specific clinical circumstances’’ [31].
High quality guidelines are clear, reliable and reproducible,
flexible, and based on a multidisciplinary review of evidence
[31]. They should improve quality of care and serve as educa-
tional tools.
METHODOLOGY
Panel Composition
The IDSA Standards and Practice Guidelines Committee
(SPGC) in collaboration with European Society for Microbiol-
ogy and Infectious Diseases (ESCMID) convened experts in the
Clinical Practice Guidelines
d
CID 2011:52 (1 March)
d
e107
management of patients with cystitis and pyelonephritis. A
specific effort was made to include representatives from diverse
geographic areas and a wide breadth of specialties, including
urology, obstetrics and gynecology, emergency medicine, family
medicine, internal medicine, and infectious diseases, with a goal
of improving the generalizability and acceptance of the recom-
mendations and subsequent incorporation into clinical practice.
Process Overview
The evaluation of evidence for each antimicrobial class used in
treatment of cystitis and pyelonephritis was performed by 2
members of the panel. Each member was assigned at least one
antimicrobial class to review. The process for evaluating the
evidence was based on the IDSA Handbook on Clinical Practice
Guideline Development and involved a systematic weighting of
the quality of the evidence and the grade of recommendation
(Table 1) [32]. This scale had been modified from the one used
in the 1999 guideline.
The level of evidence rating (I, II, or III) for recommendations
in this guideline refers to evidence of the antimicrobial’s efficacy
in randomized clinical trials. The strength of the recommen-
dation (A, B, or C) refers to the panel’s level of comfort in
recommending the antimicrobial for the treatment of un-
complicated UTI and is based on the drug’s efficacy in clinical
trials, rates of in vitro resistance among urinary pathogens, and
the drug’s propensity to cause collateral damage and adverse
effects. For example, the panel felt that fosfomycin and piv-
mecillinam should be listed as agents recommended for treat-
ment of uncomplicated cystitis, along with nitrofurantoin and
trimethoprim-sulfamethoxazole, even though they appear to be
less efficacious clinically, because they do not appear to cause
collateral damage. On the other hand, the panel was less en-
thusiastic about strongly recommending fluoroquinolones for
acute cystitis, even though they have high clinical efficacy, be-
cause of concerns about collateral damage and the subsequent
threat to the usefulness of fluoroquinolones for the treatment of
other more serious infections, including pyelonephritis.
It should be emphasized that, as is true with any treatment
guideline, an assessment of the literature for a given agent’s
clinical efficacy is limited by the comparators studied. For ex-
ample, amoxicillin-clavulanate has been shown to be statistically
significantly inferior to ciprofloxacin in a randomized trial re-
cently published. On the other hand, in the only published
randomized study of cefpodoxime, its clinical efficacy appears to
be comparable to that of trimethoprim-sulfamethoxazole. It is
not clear how amoxicillin-clavulanate would compare with
cefpodoxime or to trimethoprim-sulfamethoxazole.
Literature Review and Analysis
For the update, the Expert Panel completed a review and analysis
of data published since 1998. Computerized literature searches
of the Pubmed database were performed. The searches of the
English-language literature from 1998 thru 2008, using the
terms, cystitis or pyelonephritis with MESH terms of ‘‘acute
uncomplicated UTI,’’ ‘‘women,’’ and specific antimicrobials and
or classes of antimicrobials. To be included, the study had to be
an open-label or randomized, clinical trial of treatment of
women with symptoms of acute uncomplicated cystitis or py-
elonephritis. At least 1 follow-up visit assessing microbiological
or clinical response was required. Studies including .10% men
or patients with complicated UTI were excluded. Non–English-
language studies were excluded because they could not be re-
liably reviewed by panel members.
Outcomes of interest included early (first visit after treatment,
typically occurring at 0–7 days after the last dose of the anti-
microbial) clinical and microbiological cure, late (last visit after
treatment, typically occurring 30–45 days after the last dose of
the antimicrobial) clinical cure, and adverse effects.
Guidelines and Conflict of Interest
All members of the Expert Panel complied with the IDSA policy
on conflicts of interest, which requires disclosure of any financial
or other interest that might be construed as constituting an actual,
potential, or apparent conflict. Members of the Expert Panel were
Table 1. Strength of Recommendations and Quality of Evidence
Category/grade Definition
Strength of recommendation
A Good evidence to support a recommendation for or against use
B Moderate evidence to support a recommendation for or against use
C Poor evidence to support a recommendation
Quality of evidence
I Evidence from >1 properly randomized, controlled trial
II Evidence from >1 well-designed clinical trial, without randomization; from cohort or case-
controlled analytic studies (preferably from .1 center); from multiple time-series; or from
dramatic results from uncontrolled experiments
III Evidence from opinions of respected authorities, based on clinical experience, descriptive
studies, or reports of expert committees
NOTE. Data are from the periodic health examination. Canadian Task Force on the Periodic Health Examination. Health Canada, 1979. Adapted and Reproduced
with the permission of the Minister of Public Works and Government Services Canada, 2009 [32].
e108
d
CID 2011:52 (1 March)
d
Gupta et al

provided IDSA’s conflict of interest disclosure statement and were
asked to identify ties to companies developing products that
might be affected by promulgation of the guideline. Information
was requested regarding employment, consultancies, stock own-
ership, honoraria, research funding, expert testimony, and
membership on company advisory committees. The panel made
decisions on a case-by-case basis as to whether an individual’s
role should be limited as a result of a conflict. Potential conflicts
are listed in the Acknowledgements section.
Consensus Development Based on Evidence
The Panel met on 7 occasions via teleconference and once in
person to complete the work of the guideline. The purpose of the
teleconferences was to discuss the questions to be addressed, make
writing assignments and discuss recommendations. Most of the
work was done with e-mail correspondence. All members of the
panel participated in the preparation and review of the draft
guideline. Feedback from external peer reviews was obtained. All
collaborating organizations were also asked to provide feedback
and endorse the guidelines. The following organizations endorsed
the guidelines: American Congress of Obstetricians and Gyne-
cologists, American Urological Association, Association of Med-
ical Microbiology and Infectious Diseases–Canada), and the
Society for Academic Emergency Medicine. The guideline was
reviewed and approved by the IDSA SPGC, the IDSA Board of
Directors, and the ESCMID Board prior to dissemination.
Revision Dates
At annual intervals, the Panel Chair, the SPGC liaison advisor,
and the Chair of the SPGC will determine the need for revisions
to the guideline based on an examination of current literature. If
necessary, the entire Panel will be reconvened to discuss po-
tential changes. When appropriate, the panel will recommend
revision of the guideline to the IDSA SPGC and Board and other
collaborating organizations for review and approval.
RESULTS
Literature Search
The literature search identified 295 potential articles for review,
of which 28 met criteria for inclusion in the analyses. The types
of studies included randomized clinical trials and open label
clinical trials. Expert reviews were also incorporated into the
final grade recommendation. Two panel members were assigned
each antimicrobial class included in the guideline and in-
dependently reviewed the relevant literature. These 2 reviewers
compared their results and reached consensus on their findings
for the antimicrobial class and then presented them to the panel.
Discrepancies were discussed by the panel and final adjudication
was based on review by the chairperson and majority vote.
Limitations in the Literature
There were a limited number of publications directly comparing
the same drug given for different durations of therapy [29,
33]. Thus, there was insufficient new literature to support fur-
ther analyses of single-dose or 3-day therapy versus longer
therapy included in the previous guideline.
The criteria used to define clinical and microbiological cure
and the duration of follow-up and timing of follow-up visits
were not uniform across studies. Many studies did not perform
or report intent to treat analyses; this may inflate the late clinical
and microbiological success rates. Major differences in defi-
nitions of study outcomes are highlighted in the text.
GUIDELINE RECOMMENDATIONS FOR THE
TREATMENT OF ACUTE UNCOMPLICATED
CYSTITIS AND PYELONEPHRITIS
I. What Is the Optimal Treatment for Acute Uncomplicated
Cystitis?
Recommendations (Figure 1).
1. Nitrofurantoin monohydrate/macrocrystals (100 mg twice
daily for 5 days) is an appropriate choice for therapy due to
minimal resistance and propensity for collateral damage
(defined above) and efficacy comparable to 3 days of
trimethoprim-sulfamethoxazole (A-I).
2. Trimethoprim-sulfamethoxazole (160/800 mg [1 double-
strength tablet] twice-daily for 3 days) is an appropriate choice
for therapy, given its efficacy as assessed in numerous clinical
trials, if local resistance rates of uropathogens causing acute
uncomplicated cystitis do not exceed 20% or if the infecting
strain is known to be susceptible (A-I).
i. The threshold of 20% as the resistance prevalence at which
the agent is no longer recommended for empirical treatment of
acute cystitis is based on expert opinion derived from clinical,
in vitro, and mathematical modeling studies (B-III).
ii. In some countries and regions, trimethoprim (100 mg twice
daily for 3 days) is the preferred agent and is considered
equivalent to trimethoprim-sulfamethoxazole on the basis of
data presented in the original guideline (A-III) [1].
iii. Data are insufficient to make a recommendation for other
cystitis antimicrobials as to what resistance prevalence should be
used to preclude their use for empirical treatment of acute cystitis.
3. Fosfomycin trometamol (3 g in a single dose) is an
appropriate choice for therapy where it is available due to
minimal resistance and propensity for collateral damage, but it
appears to have inferior efficacy compared with standard short-
course regimens according to data submitted to the US Food
and Drug Administration (FDA) and summarized in the
Medical Letter (A-I) [7].
4. Pivmecillinam (400 mg bid for 3–7 days) is an
appropriate choice for therapy in regions where it is available
(availability limited to some European countries; not licensed
and/or available for use in North America), because of minimal
Clinical Practice Guidelines
d
CID 2011:52 (1 March)
d
e109
resistance and propensity for collateral damage, but it may have
inferior efficacy compared with other available therapies (A-I).
5. The fluoroquinolones, ofloxacin, ciprofloxacin, and
levofloxacin, are highly efficacious in 3-day regimens (A-I) but
have a propensity for collateral damage and should be reserved for
important uses other than acute cystitis and thus should be
considered alternative antimicrobials for acute cystitis (A-III).
6. b-Lactam agents, including amoxicillin-clavulanate,
cefdinir, cefaclor, and cefpodoxime-proxetil, in 3–7-day
regimens are appropriate choices for therapy when other
recommended agents cannot be used (B-I). Other b-lactams,
such as cephalexin, are less well studied but may also be
appropriate in certain settings (B-III). The b-lactams generally
have inferior efficacy and more adverse effects, compared with
other UTI antimicrobials (B-I). For these reasons, b-lactams
other than pivmecillinam should be used with caution for
uncomplicated cystitis.
7. Amoxicillin or ampicillin should
not be used for empirical
treatment given the relatively poor efficacy, as discussed in the
1999 guidelines [1] and the very high prevalence of antimicrobial
resistance to these agents worldwide [8–11] (A-III).
Evidence Summary
The optimal agent for therapy of a patient with acute un-
complicated cystitis depends on a number of factors (Figure 2).
Each agent has pros and cons related to its use and the choice of
therapy is made on an individual basis.
Trimethoprim-sulfamethoxazole. The traditional first-line
agent in the United States and recommended in the original
IDSA guidelines was trimethoprim-sulfamethoxazole (tri-
methoprim was considered comparable) [1]. However, rising
rates of trimethoprim-sulfamethoxazole resistance among ur-
opathogens, especially outside of the United States, and con-
sistent evidence that in vitro resistance correlates with bacterial
and clinical failures, necessitates revising this recommendation.
Indeed, the guidelines of the European Association of Urology
do not recommend this agent as first choice treatment of un-
complicated cystitis [34].
Four randomized clinical trials compared trimethoprim-
sulfamethoxazole with another agent, including ciprofloxacin,
norfloxacin, nitrofurantoin, and cefpodoxime proxetil, and
evaluated microbiological and clinical outcomes among
women with acute cystitis (Table 2) [35–38]. The 2 studies
including a fluoroquinolone had findings consistent with the
1999 guideline, reporting that trimethoprim-sulfamethoxazole
was noninferior (95% confidence interval of difference at
610%) to ciprofloxacin for early clinical and bacterial
cure rates [35, 37]. Both studies used a longer than standard
(7 days rather than 3 days) course of trimethoprim-
sulfamethoxazole versus a 3-day course of ciprofloxacin. In the
study by Iravani et al [37], 7 days of 160/800 mg twice-daily
trimethoprim-sulfamethoxazole in 174 women had similar
rates of early and late clinical cure as 3 days of 100 mg cipro-
floxacin given twice daily to 168 women (95% early and
90% late for each drug). The late bacterial cure rate (4-6 weeks
after therapy) was lower with trimethoprim-sulfamethoxazole
than for ciprofloxacin (79% vs 91%, respectively), whereas
the early bacterial cure rate was higher with trimethoprim-
sulfamethoxazole (93% vs 88%, respectively). Arredondo-Garcia
et al [35] reported that 7 days of trimethoprim-sulfamethoxazole
(160/800 mg twice daily) in 81 women resulted in early clinical
and bacterial cure rates of 86% and 85%, respectively, non-
inferior to the 89% and 92% cure rates, respectively, achieved in
97 women treated with 3 days of ciprofloxacin (250 mg twice
daily). Of note, these similar outcomes were demonstrated
despite 15% of women in the trimethoprim-sulfamethoxazole
arm having a pretherapy isolate resistant to the treatment drug,
compared with only 1% of women in the ciprofloxacin arm.
Results stratified by susceptibility of the infecting organism to
the treatment regimen were not reported. Each study included
a third treatment arm; results of these comparisons are discussed
below for the relevant antimicrobial class.
A small study compared a 3-day course of trimethoprim-
sulfamethoxazole (160/800 mg twice daily) with a 3-day
course of cefpodoxime-proxetil (100 mg twice daily) [38].
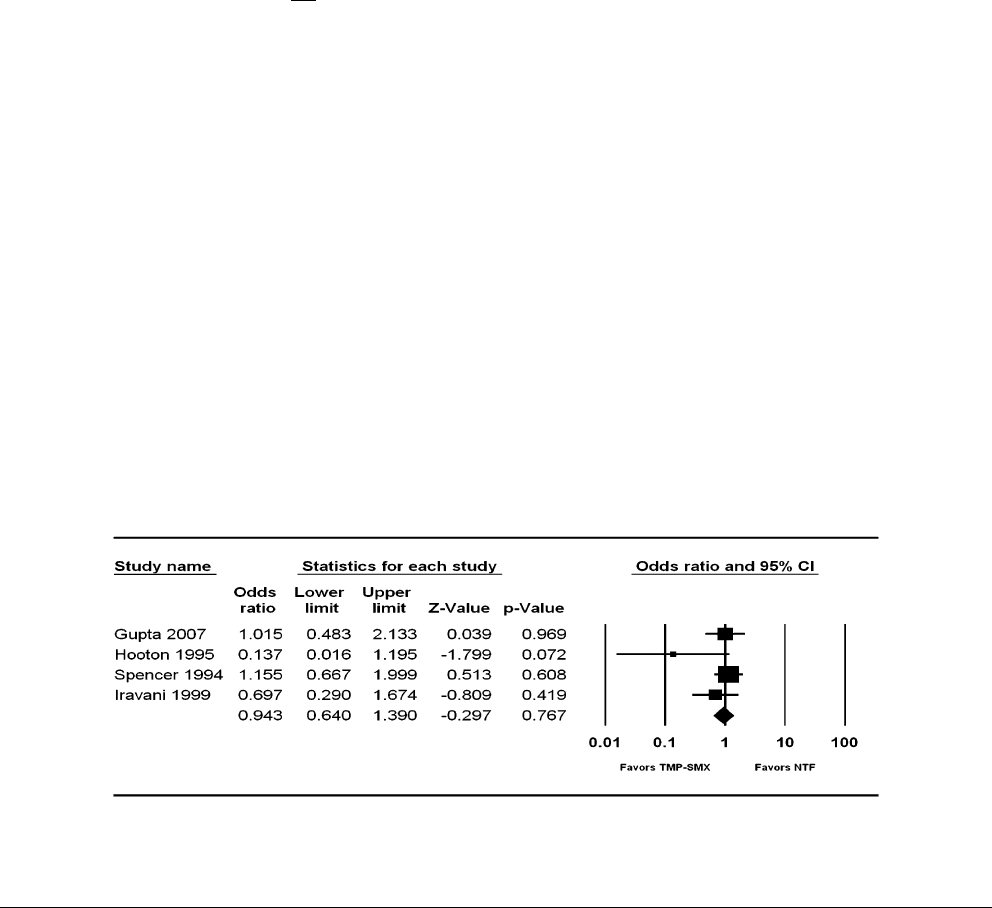
Figure 2. Meta-analysis of studies comparing trimethoprim-sulfamethoxazole (TMP-SMX) with nitrofurantoin (NTF) for acute uncomplicated cystitis. CI,
confidence interval.
e110
d
CID 2011:52 (1 March)
d
Gupta et al

Women with an uropathogen resistant to either study drug
(4 of 82 women in the trimethoprim-sulfamethoxazole arm
and 0 of 81 women in the cefpodoxime arm) were excluded.
Clinical cure was achieved in 100% of the 70 women in the
trimethoprim-sulfamethoxazole arm, compared with 62
(98%) of 63 women in the cefpodoxime arm. The microbio-
logical cure rates were the same as the clinical cure rates in
each arm. Adverse effects were reported in 1 patient in the
trimethoprim-sulfamethoxazole arm and 2 patients in the
cefpodoxime arm.
The fourth study compared a 3-day course of trimethoprim-
sulfamethoxazole (160/800 mg twice daily) with a 5-day
course of nitrofurantoin monohydrate–macrocrystals (100 mg
twice daily) and included women with uropathogens resistant
to the study drugs [36]. The primary end point, overall clinical
cure rate at 30 days, was 79% among the 148 women in the
trimethoprim-sulfamethoxazole arm and 84% among the 160
women in the nitrofurantoin arm, with a nonsignificant dif-
ference of -5%. Rates were also equivalent (predefined as
a 610% difference between agents) at 5-9 days after therapy,
with clinical cure of 90% in each arm and bacterial cure of 91%
in the trimethoprim-sulfamethoxazole arm and 92% in the ni-
trofurantoin arm. There was a significantly higher clinical cure
rate among women in the trimethoprim-sulfamethoxazole
arm who had a trimethoprim-sulfamethoxazole–susceptible
uropathogen, compared with those who had a trimethoprim-
sulfamethoxazole–resistant uropathogen (84% vs 41%,
respectively;1 P , .001).
The fifth study used a prospective observational trial design
to compare clinical and bacterial outcomes among women
with acute cystitis with a trimethoprim-sulfamethoxazole–
susceptible or –resistant uropathogen [21]. All women were
treated with a 5-day course of trimethoprim-sulfamethox-
azole (160/800 mg twice daily). The microbiological cure rates
Table 2. Results from Included Studies of Trimethoprim-Sulfamethoxazole for Treatment of Acute Uncomplicated Cystitis
Study (year) [reference] Treatment regimen
Iravani et al (1999) [37] TMP-SMX,
160/800 mg
twice daily
for 7 days
Nitrofurantoin
monohydrate/
macrocrystals,
100 mg twice
daily for 7 days
Ciprofloxacin, 100 mg twice
daily for 3 days
Early clinical cure 165/174 (95) 166/179 (93) 160/168 (95)
Early bacterial cure 161/174 (93) 153/177 (86) 148/168 (88)
Late clinical cure 137/153 (90) 135/151 (89) 132/147 (90)
Adverse events, % 38 34 28
Arredondo-Garcia et al
(2004) [35]
TMP-SMX,
160/800 mg
twice daily x 7 days
Norfloxacin,
400 mgtwice
daily for 7 days
Ciprofloxacin, 250 mg
twice daily for 3 days
Early clinical cure 70/81 (86) 90/107 (84) 86/97 (89)
Early bacterial cure 69/81 (85) 93/107 (87) 89/97 (92)
Late clinical cure 66/81 (82) 88/107 (82) 81/97 (84)
Adverse events, % 8.7 3.9 4.0
Kavatha et al
(2003) [38]
TMP-SMX,
160/800 mgtwice
daily for 3 days
Cefpodoxime
proxetil, 100
mg twice daily
for 3 days
Early clinical cure 70/70 (100) 62/63 (98.4)
Early bacterial cure 70/70 (100) 62/63 (98.4)
Late clinical cure 51/60 (85) 42/50 (84)
Adverse events, % 1.4 1.6
Gupta et al
(2007) [36]
TMP-SMX, 160/800
mgtwice daily
for 3 days
Nitrofurantoin
monohydrate/
macrocrystals,
100 mg twice
daily for 5 days
Early clinical cure 133/148 (90) 144/160 (90)
Early bacterial cure 131/144 (91) 141/154 (92)
Late clinical cure 117/148 (79) 134/160 (84)
Adverse events, % 31 28%
NOTE. Data are proportion of subjects (%), unless otherwise indicated. Efficacy rates refer to cure rates on the visit closest to a 5–9-day period following
treatment. NA, not available; TMP-SMX, trimethoprim-sulfamethoxazole.
Clinical Practice Guidelines
d
CID 2011:52 (1 March)
d
e111

were significantly higher among women with a trimethoprim-
sulfamethoxazole–susceptible uropathogen than for women
with a –resistant uropathogen (86% vs 42%, respectively; P ,
.001). The clinical cure rate at 5-9 days after completion of
therapy was also higher in the trimethoprim-sulfamethox-
azole–susceptible group (88% of 333 women) than in the
trimethoprim-sulfamethoxazole–resistant group (54% of 151
women; P , .001). The clinical and microbiological differ-
ences remained significant at the 28–42-day follow-up visit.
Because this was not a randomized treatment trial, the data
were not included in the efficacy analyses but are reported as
they provide insight into expected outcomes in patients with
resistant uropathogens.
Overall findings from these studies demonstrate that
trimethoprim-sulfamethoxazole remains a highly effective
treatment for acute uncomplicated cystitis in women when the
rate of resistance is known or expected to be , 20%, supporting
a strong recommendation for use in such settings. Early clinical
and microbiological cure rates are in the 90% - 100% range
(Table 2). Late outcomes are harder to compare across studies,
but when calculated using intent to treat criteria, are 80% - 90%.
Resistance impacts both clinical and bacterial outcomes, so
known or expected resistance should be considered in antimi-
crobial choice. In this regard, resistance to trimethoprim-
sulfamethoxazole is high in many regions of the world.
However, in settings with a 10% - 15% prevalence of resistance
to trimethoprim-sulfamethoxazole, cure rates with trimethoprim-
sulfamethoxazole were equivalent to those with comparator
drugs (ie, ciprofloxacin and nitrofurantoin) to which almost
all isolates were probably susceptible (data on susceptibility
to comparators were not uniformly provided in the studies) [35–
37]. Trimethoprim-sulfamethoxazole use is associated with
increased resistance, but, even though it has a significant impact
on intestinal flora, it is generally not thought to have a propensity
for ‘‘collateral damage’’ as observed with broad-spectrum
cephalosporins or fluoroquinolones.
Nitrofurantoin. There is additional evidence in support
of nitrofurantoin monohydrate/macrocrystals, for which data
were previously limited. There were 4 randomized trials of
nitrofurantoin versus a comparator published since the pre-
vious guideline (Table 3) [28, 36, 37, 39]. These studies
demonstrate that (1) nitrofurantoin monohydrate/
Table 3. Results from Included Studies of Nitrofurantoin for Treatment of Acute Uncomplicated Cystitis
Study Regimen
Iravani et al (1999) [37] Nitrofurantoin monohydrate/
macrocrystals, 100 mg twice
daily for 7 days
TMP-SMX, 160/800
mg twice daily for 7 days
Ciprofloxacin, 100 mg
twice daily for 3 days
Early clinical cure 166/179 (93) 165/174 (95) 160/168 (95)
Early bacterial cure 153/177 (86) 161/174 (93) 148/168 (88)
Late clinical cure 135/151 (89) 137/153 (90) 132/147 (90)
Adverse events, % 34 38 28
Stein et al (1999) [39] Nitrofurantoin monohydrate/
macrocrystals, 100 mg twice
daily for 7 days
Fosfomycin trometamol,
single 3-gdose
Early clinical cure 232/245 (95) 240/263 (90)
Early bacterial cure 189/219 (86) 192/246 (78)
Late clinical cure 168/180 (93) 189/202 (94)
Adverse events, % 5.6 5.3
Christiaens et al (2002) [28] Nitrofurantoin macrocrystals,
100 mg 4 times daily for 3 days
Placebo, 4 times
daily for 3 days
Early clinical cure 21/24 (88) 13/23 (54)
Early bacterial cure 17/23 (74) 9/22 (41)
Late clinical cure NA NA
Adverse events, % 23 26
Gupta et al (2007) [36] Nitrofurantoin monohydrate/
macrocrystals, 100 mg twice
daily for 5 days
TMP-SMX, 160/800
mg twice daily for 3 days
Early clinical cure 144/160 (90) 133/148 (90)
Early bacterial cure 141/154 (92) 131/144 (91)
Late clinical cure 134/160 (84) 117/148 (79)
Adverse events, % 28 31
NOTE. Data are proportion of subjects (%), unless otherwise indicated. Efficacy rates refer to cure rates on the visit closest to a 5–9-day period following
treatment. NA, not available; TMP-SMX, trimethoprim-sulfamethoxazole.
e112
d
CID 2011:52 (1 March)
d
Gupta et al
macrocrystals (100 mg twice daily for 7 days) has similar
clinical cure rates (based on the small differences in early
clinical cure and confidence intervals that are small enough to
suggest no difference in efficacy) to ciprofloxacin (100 mg
twice daily for 3 days; 93% vs 95%), trimethoprim-sulfame-
thoxazole (160/800 mg twice daily for 7 days; 93% vs 95%),
and 3-g single-dose fosfomycin trometamol (89% vs 90%);
(2) nitrofurantoin monohydrate/macrocrystals (100 mg twice
daily in a 5-day regimen) is equivalent in clinical and mi-
crobiological cure rates to trimethoprim-sulfamethoxazole
(160/800 mg twice daily in a 3-day regimen); and (3) nitro-
furantoin macrocrystals (100 mg 4 times daily for 3 days) is
superior to placebo treatment of women with acute cystitis.
Taken together, the studies demonstrate a clinical cure rate
with nitrofurantoin of 88% - 93% and a bacterial cure rate of
81% - 92%. A meta-analysis of studies comparing early clin-
ical cure rates with nitrofurantoin and trimethoprim-sulfa-
methoxazole is shown in Figure 2 and demonstrates
equivalence between the 2 agents. Of note, resistance to ni-
trofurantoin remains low and it is well tolerated and effica-
cious in a 5-day regimen (Table 4).
Thus, current randomized clinical trial data provide strong
support for consideration of nitrofurantoin as an effective agent
for treatment of acute cystitis. Demonstration of efficacy, with
minimal drug resistance or propensity for collateral damage,
makes nitrofurantoin an attractive agent for cystitis. A 5-day
regimen, rather than the traditional 7-day course, can be con-
sidered as an effective duration of treatment based on a recent
randomized clinical trial [36].
Fosfomycin trometamol. There are also new data in support
of fosfomycin trometamol, a phosphonic acid derivative available
in the United States and some European countries for treatment
of UTI. A 3-g single-dose of fosfomycin trometamol was com-
pared with a 7–day course of nitrofurantoin monohydrate/
macrocrystals 100 mg twice daily in one study and with a 5-day
course of trimethoprim 100 mg twice daily in another [39, 40].
The latter study only evaluated the microbiologic outcome and
reported that single-dose fosfomycin trometamol and 5 days of
twice-daily trimethoprim each had an 83% bacterial cure rate
(147 of 177 fosfomycin and 70 of 84 trimethoprim-treated
women, respectively) at the early follow-up visit [34]. The study
by Stein [39] demonstrated that the early clinical response (cure
or improvement at 5-11 days after starting therapy) rates were
not significantly different, at 91% (240 of 263 women) for 3-g
single-dose fosfomycin trometamol treatment and 95% (232 of
245 women) for 100 mg of nitrofurantoin monohydrate/mac-
rocrystals given twice daily. The late clinical response rates re-
mained high for both drugs (93%–94%). However, the
microbiologiccureratewassignificantly higher with nitro-
furantoin (86%), compared with fosfomycin (78%), at the first
follow-up visit (P 5 .02). Microbiologic cure rates 4-6 weeks after
therapy were 96% for fosfomycin and 91% for nitrofurantoin but
Table 4. Treatment Regimens and Expected Early Efficacy Rates for Acute Uncomplicated Cystitis
Mean percentage (range)
Drug (dosage)
Estimated clinical
efficacy
ab
Estimated
microbiological
efficacy
b
Common side effects References
Nitrofurantoin monohydrate/
macrocrystals (100 mg twice
daily for 5–7 days)
93 (84–95) 88 (86–92) Nausea,headache [36, 37, 39]
Trimethoprim-sulfamethoxazole
(160/800 mg twice daily for 3 days)
93 (90–100) 94 (91–100) Rash, urticaria,nausea,
vomiting, hematologic
[36, 37]
Fosfomycin trometamol (3 g
single-dose sachet)
91 80 (78–83) Diarrhea, nausea,headache [39, 40]
Pivmecillinam (400 mg twice
daily for 3–7 days)
73 (55–82) 79 (74–84) Nausea, vomiting, diarrhea [29, 43]
Fluoroquinolones (dose varies
by agent; 3–day regimen)
c
90 (85–98) 91 (81–98) Nausea/vomiting,
diarrhea, headache,
drowsiness, insomnia
[35, 43, 44, 46–52]
b-lactams (dose varies by
agent; 3–5 day regimen)
d
89 (79–98) 82 (74–98) Diarrhea, nausea,
vomiting, rash, urticaria
[38, 52, 54]
a
Efficacy rates refer to cure rates on the visit closest to a 5–9-day period following treatment, and are averages or ranges calculated from clinical trials discussed
in the text.
b
Estimated clinical efficacy and microbiological efficacy rates should not necessarily be compared across agents, because study design, efficacy definition,
therapy duration, and other factors are heterogeneous. Studies represent clinical trials published since publication of the 1999 Infectious Disease Society of America
guidelines so as to represent efficacy rates that account for contemporary prevalence of antibiotic-resistant uropathogens. Note that efficacy rates may vary
geographically depending on local patterns of antimicrobial resistance among uropathogens. See text for details.
c
Data on fluoroquinolones are compiled from regimens of ofloxacin, norfloxacin, and ciprofloxacin from the referenced clinical trials and not other
fluoroquinolones that are no longer commercially available. See text for details.
d
Data on blactams data cited are derived from clinical trials examining second and third generation cephalosporins and amoxicillin-clavulanate. See text for details.
Clinical Practice Guidelines
d
CID 2011:52 (1 March)
d
e113

included only 50% of the original study population. Intent–to-
treat analyses were not reported [39]. Overall, the bacterial effi-
cacy of fosfomycin is lower than that of other first-line agents,
but clinical efficacy (based on a single study) was comparable
(Table 4). Additional information considered by the com-
mittee was the reference in the 1999 IDSA UTI guideline to
unpublished data demonstrating lower bacterial eradication
rates with fosfomycin than with 10 days of trimethoprim-
sulfamethoxazole and with 7 days of ciprofloxacin. These
studies are still not available in the published literature except
as previously referenced in a Medical Letter report [7].
Several in vitro studies examined the activity of fosfomycin
against multidrug-resistant pathogens. These demonstrate that
fosfomycin is active against vancomycin-resistant enterococci
(VRE), methicillin-resistant S. aureus (MRSA), and extended-
spectrum b-lactamase (ESBL)–producing gram-negative rods
[41]. As resistance among uropathogens causing community-
acquired uncomplicated cystitis increases, fosfomycin may be-
come more useful, particularly if no other oral agents with in
vitro activity are available [42]. Clinical outcomes are not yet
reported from randomized, controlled studies; thus, specific
recommendations for the role of fosfomycin in the treatment of
multidrug-resistant uropathogens cannot be included in the
current guideline. However, observational studies are supportive
of clinical efficacy [41, 42].
The convenience of a single-dose regimen, in vitro activity
against resistant gram-negative rods, and minimal propensity
for collateral damage make fosfomycin a useful choice in some
areas. It is recommended as a first-line agent in the guidelines of
the European Association of Urology, although it is not uni-
formly available [34]. Susceptibility data are also not uniformly
available, because testing is not routinely performed in many
clinical laboratories. Furthermore, the effect of fosfomycin on
the intestinal flora after intake of a single 3-g dose (the standard
dosage for uncomplicated UTI) has not been well studied, but
the effect is probably minor [25]. This assumption is supported
by the high rate of E. coli susceptibility in regions with frequent
use of fosfomycin for uncomplicated cystitis in women [10].
Pivmecillinam. Pivmecillinam, the orally bioavailable form
of mecillinam, is distinguished from other b-lactams because of
its specificity for the urinary tract, minimal resistance or pro-
pensity for collateral damage, and reasonable treatment efficacy.
It is an extended gram-negative spectrum penicillin used only
for treatment of UTI. Two studies met our inclusion criteria
[43]. One study compared different doses of pivmecillinam with
placebo. Pivmecillinam at 200 mg 3 times daily for 7 days,
200 mg twice daily for 7 days, and 400 mg twice daily for 3 days
resulted in early clinical cure rates of 62% (132 of 217 women),
64% (136 of 220 women), and 55% (119 of 220 women), re-
spectively, and bacteriologic cure rates of 93%, 94%, and 84%,
respectively. Placebo therapy resulted in a clinical cure rate of
25% (54 of 227 women) and a bacteriologic cure rate of 34%,
both inferior to active drug. In another randomized trial com-
paring 3 days of pivmecillinam (400 mg bid) with 3 days of
norfloxacin (400 mg bid), pivmecillinam treatment resulted in
lower bacterial cure rates (222 (75%) of 298 vs 276 (91%) of 302,
respectively; P , .001) and lower clinical cure rates (360 (82%)
of 437 vs 381 (88%) of 433, respectively; P 5 .02) [43]. In vitro
resistance to pivmecillinam was not associated with a high rate
of failure; 30 (88%) of 34 pivmecillinam-treated patients who
had a pivmecillinam-resistant uropathogen achieved bacterial
cure.
Although not available in the United States or Canada, piv-
mecillinam is one of the agents of choice in many Nordic
countries due to low resistance rates and low propagation of
resistance [24]. Different doses and durations have been asso-
ciated with varying efficacy rates, and a 5-day or 7-day regimen
is probably superior to a 3-day regimen. Similarly, a 400-mg
dose fared better than a 200-mg dose for both bacterial and
clinical efficacy. The efficacy rates are notably lower than other
recommended agents (Table 4). Of note, the rate of resistance
among E. coli to pivmecillinam remains low despite its frequent
use in some European countries [24].
Fluoroquinolones. There were 12 randomized trials of
fluoroquinolones for treatment of acute cystitis. The majority of
these compared one fluoroquinolone with another, often in
varying doses or durations. Sparfloxacin and gatifloxacin are no
longer widely available because of their adverse effects, and thus,
results related to these 2 agents are not included in the analyses
[44–47]. Two large studies compared 500 mg of extended-
release once-daily ciprofloxacin to the 250-mg twice-daily for-
mulation of ciprofloxacin and demonstrated equivalent cure
rates [48, 49]. Another study compared ciprofloxacin (250 mg
twice daily) in a 3-day versus a 7-day regimen and demonstrated
equivalent cure rates but significantly higher adverse event rates
with the longer regimen [50]. A small study compared nor-
floxacin 400 mg twice daily with norfloxacin 800 mg once daily
and demonstrated similar bacterial and clinical outcomes, albeit
with limited power to detect true differences [33]. One study
compared single-dose ciprofloxacin with 3 days of norfloxacin
and found the agents to be equivalent, with microbiological and
clinical cure rates in the 91% - 94% range [51].
Three studies compared a fluoroquinolone with a drug from
another class. Two demonstrated better clinical and microbio-
logical cure rates with the fluoroquinolone regimen (norfloxacin
vs pivmecillinam and ciprofloxacin vs amoxicillin-clavulanate)
[43, 52]. The third demonstrated early clinical and bacterial
cure rates to be similar with 3 days of low-dose ciprofloxacin
or a standard dose but longer duration (7 days each) of
trimethoprim-sulfamethoxazole and nitrofurantoin [37]. The
details for each of these studies are discussed under the
respective comparator agent. Overall clinical and bacterial
e114
d
CID 2011:52 (1 March)
d
Gupta et al

efficacy rates in the studies are consistently high, although they
were occasionally ,90% (Table 4).
Thus, fluoroquinolones remain very effective for the treat-
ment of acute cystitis, although increased fluoroquinolone re-
sistance among community uropathogens is mitigating the
usefulness of this antimicrobial class. Once-daily dosing of ci-
profloxacin is now available and of equal efficacy as the twice-
daily formulation, albeit more expensive, since the latter is now
generic. Single-dose fluoroquinolone therapy remains an option
but with possibly lower efficacy rates than with longer regimens
[1]. Fluoroquinolones with longer half-lives, such as pefloxacin
and fleroxacin, may be useful for single-dose therapy, but no
studies met our eligibility criteria and neither agent is available
in all locales, including North America and many parts of Eu-
rope. The main concern regarding fluoroquinolone use for acute
cystitis is the promotion of fluoroquinolone resistance, not only
among uropathogens but also other organisms, causing more
serious and difficult–to-treat infections at other sites. There is
also concern about the association between fluoroquinolone use
and increased rates of MRSA [22]. Many experts now call for
restricting use of fluoroquinolones to those episodes of un-
complicated cystitis when other UTI antimicrobials are not
suitable [53]. The panel agrees and recommends that fluo-
roquinolones be reserved as an alternative only when other UTI
agents cannot be used (Figure 1).
b-Lactams. Five randomized trials evaluating b-lactam
antibiotics were identified and included in the analyses.
Only 1 study included a 3-day regimen of trimethoprim-
sulfamethoxazole as the standard comparator [38]. This study
demonstrated that 100 mg of cefpodoxime proxetil twice
daily for 3 days was equivalent to trimethoprim-sulfame-
thoxazole (160/800 mg twice daily for 3 days), with 100% of
70 women treated with trimethoprim-sulfamethoxazole and
98% of 63 women treated with cefpodoxime experiencing
clinical and microbiological cured at day 4–7 after completion
of therapy. Clinical cure at 28 days was somewhat lower but
not different between the treatment arms (Table 2). However,
the statistical power of the study to find differences between
the drugs was limited by its small sample size. Side effects
were not different between the 2 groups. In another study,
amoxicillin-clavulanate (500/125 mg twice daily) was com-
pared with ciprofloxacin (250 mg twice daily), both for 3 days,
with 4 months of follow-up [52]. Clinical cure at the last
follow-up visit was observed in 58% of 160 women treated with
amoxicillin-clavulanate, compared with 77% of 162 women
treated with ciprofloxacin (P , .001). The differences were
significant even among the subgroups of women infected with
strains susceptible to the treatment drug (60% vs 77%, re-
spectively; P 5 .004). Microbiological cure at 2 weeks was
observed in 76% of 156 women treated with amoxicillin-
clavulanate, compared with 95% of 161 women treated with
ciprofloxacin (P , .001) [52]. Vaginal colonization with ur-
opathogens before and after therapy was also measured, and the
higher clinical failure rate observed with amoxicillin-clavulanate
was associated with a lower rate of eradication of vaginal ur-
opathogens in the amoxicillin-clavulanate group. These findings
are consistent with the postulated mechanism for b-lactam
inferiority in the treatment of UTI being, in part, related to
persistence of the vaginal reservoir for infection. Another study
compared 2 b-lactam antibiotics, cefdinir (100 mg twice daily)
versus ceflacor (250 mg 3 times daily), each for 5 days, and
demonstrated equivalent clinical (91% vs 93%, respectively) and
microbiological (85% vs 80%, respectively) cure rates [54].
Thus, the overall evidence of the efficacy of b-lactams for
treatment of acute cystitis has not changed since the previous
guideline [1]. Most studies demonstrate that b-lactams are
generally inferior in cure rates to the fluoroquinolones [42, 52].
The study by Kavatha et al [38] demonstrating that an advanced
generation oral cephalosporin (cefpodoxime proxetil) resulted
in cure rates equivalent to those of trimethoprim-sulfame-
thoxazole is intriguing and needs to be confirmed in a larger
clinical trial [38]. However, even if these observations are con-
firmed, concern about emergence of gram-negative ESBL re-
sistance to these agents limits enthusiasm for any widespread
use. Broad-spectrum cephalosporins, in particular, have been
associated with collateral damage, the most concerning of which
is ESBL resistance among gram-negative bacteria [22]. Nar-
rower-spectrum cephalosporins are often used for treatment of
UTI and may result in less collateral damage, compared with
broad-spectrum cephalosporins; however, there is a lack of ad-
equately powered studies to make specific recommendations for
these agents. Thus, the panel feels that currently available data
supports avoidance of b-lactams other than pivmecillinam for
empirical therapy of uncomplicated cystitis unless none of the
recommended agents are appropriate.
The choice of agent should be individualized on the basis of
patient allergy and compliance history, local practice patterns,
local community resistance prevalence, availability, cost, and
patient and provider threshold for failure. In the event of di-
agnostic uncertainty regarding cystitis versus early pyelone-
phritis, use of agents such as nitrofurantoin, fosfomycin, and
pivmecillinam should be avoided, because they do not achieve
adequate renal tissue levels. Such uncertainly may exist in the
setting of cystitis symptoms accompanied by subjective fever that
is not verified at the time of examination, a prolonged duration
of cystitis symptoms (typically greater than 5–7 days), or vague
flank pain or tenderness which is not otherwise explained.
II. What Is the Treatment for Acute Pyelonephritis?
Recommendations.
8. In patients suspected of having pyelonephritis, a urine
culture and susceptibility test should always be performed, and
Clinical Practice Guidelines
d
CID 2011:52 (1 March)
d
e115

initial empirical therapy should be tailored appropriately on
the basis of the infecting uropathogen (A-III).
9. Oral ciprofloxacin (500 mg twice daily) for 7 days, with or
without an initial 400-mg dose of intravenous ciprofloxacin, is
an appropriate choice for therapy in patients not requiring
hospitalization where the prevalence of resistance of
community uropathogens to fluoroquinolones is not known
to exceed 10% (A-I). If an initial one-time intravenous agent is
used, a long-acting antimicrobial, such as 1 gof ceftriaxone or
a consolidated 24-h dose of an aminoglycoside, could be used
in lieu of an intravenous fluoroquinolone (B-III). If the
prevalence of fluoroquinolone resistance is thought to exceed
10%, an initial 1-time intravenous dose of a long-acting
parenteral antimicrobial, such as 1 g of ceftriaxone (B-III) or
a consolidated 24-h dose of an aminoglycoside, is
recommended (B-III).
i. Data are insufficient to make a recommendation about what
fluoroquinolone resistance level requires an alternative agent in
conjunction with or to replace a fluoroquinolone for treatment
of pyelonephritis.
10. A once-daily oral fluoroquinolone, including
ciprofloxacin (1000 mg extended release for 7 days)or
levofloxacin (750 mg for 5 days), is an appropriate choice for
therapy in patients not requiring hospitalization where the
prevalence of resistance of community uropathogens is not
known to exceed 10% (B-II). If the prevalence of fluoroquinolone
resistance is thought to exceed 10%, an initial intravenous dose of
a long-acting parenteral antimicrobial, such as 1 g of ceftriaxone
(B-III) or a consolidated 24-h dose of an aminoglycoside, is
recommended (B-III).
11. Oral trimethoprim-sulfamethoxazole (160/800 mg [1
double-strength tablet] twice-daily for 14 days) is an
appropriate choice for therapy if the uropathogen is known
to be susceptible (A-I). If trimethoprim-sulfamethoxazole is
used when the susceptibility is not known, an initial
intravenous dose of a long-acting parenteral antimicrobial,
such as 1 g of ceftriaxone (B-II) or a consolidated 24-h dose of
an aminoglycoside, is recommended (B-III).
12. Oral b-lactam agents are less effective than other
available agents for treatment of pyelonephritis (B-III). If an
oral b-lactam agent is used, an initial intravenous dose of
a long-acting parenteral antimicrobial, such as 1 g of
ceftriaxone (B-II) or a consolidated 24-h dose of an
aminoglycoside, is recommended (B-III).
i. Data are insufficient to modify the previous guideline
recommendation for a duration of therapy of 10–14 days for
treatment of pyelonephritis with a b-lactam agent.
13. Women with pyelonephritis requiring hospitalization
should be initially treated with an intravenous antimicrobial
regimen, such as a fluoroquinolone; an aminoglycoside, with or
without ampicillin; an extended-spectrum cephalosporin or
extended-spectrum penicillin, with or without an
aminoglycoside; or a carbapenem. The choice between these
agents should be based on local resistance data, and the
regimen should be tailored on the basis of susceptibility
results (B-III).
Evidence Summary
Optimal therapy for acute uncomplicated pyelonephritis de-
pends on the severity of illness at presentation and local re-
sistance patterns as well as specific host factors (such as
allergies). In addition, urine culture and susceptibility testing
should be performed, and initial empirical therapy should be
tailored appropriately on the basis of the infecting uropathogen.
Strategies for optimizing empirical therapy when local resistance
patterns are not known include using an initial intravenous dose
of a long-acting parenteral antimicrobial and starting with
a broader-spectrum agent and narrowing therapy when labo-
ratory results are available.
There were 6 treatment studies of acute uncomplicated py-
elonephritis identified, but only 1 study met our inclusion cri-
teria. This study compared a 7-day regimen of oral ciprofloxacin
(500 mg twice daily) with a 14-day regimen of trimethoprim-
sulfamethoxazole (160/800 mg twice daily) for treatment of
women presenting to emergency departments or outpatient
clinics with mild to moderate pyelonephritis [55]. An initial
intravenous 400-mg dose of ciprofloxacin in the ciprofloxacin
group or a 1-g dose of ceftriaxone in the trimethoprim-
sulfamethoxazole group was allowed in the protocol at the
discretion of the clinician. Women with an uropathogen re-
sistant to the study drug to which they were randomized con-
tinued to receive the drug unless they experienced clinical failure
(14 women in the trimethoprim-sulfamethoxazole group and 1
woman in the ciprofloxacin group). Ciprofloxacin had signifi-
cantly higher microbiological (99% vs 89%, respectively) and
clinical (96% vs 83%, respectively) cure rates at the early post-
therapy visit. Cure rates were similar regardless of whether
an initial intravenous dose of ciprofloxacin was given. Among
trimethoprim-sulfamethoxazole–treated women, those with a
trimethoprim-sulfamethoxazole–resistant uropathogen had
significantly lower microbiological eradication and clinical cure
rates, compared with those with a susceptible uropathogen.
An initial intravenous dose of ceftriaxone significantly
improved the microbiological eradication rate and moderately
improved the clinical cure rate in women with a trimethoprim-
sulfamethoxazole–resistant uropathogen.
Two additional studies also demonstrated that a 5–7-day
regimen of a once-daily fluoroquinolone (ciprofloxacin, 1000
mg extended release, and levofloxacin, 750 mg, respectively)
were effective for acute pyelonephritis [56, 57]. These studies did
not meet our inclusion criteria because they included
e116
d
CID 2011:52 (1 March)
d
Gupta et al

a mixed population of men and women with acute pyelone-
phritis and/or complicated UTIs, but they do lend additional
support for a 5–7-day fluoroquinolone regimen versus the tra-
ditional 14-day regimen for mild to moderate pyelonephritis. A
study of once-daily gatifloxacin (200 mg or 400 mg) was similar
in design and outcomes but is not discussed further because the
oral formulation is no longer available in most parts of the world
[58]. Another pyelonephritis study demonstrated that 10 days of
an oral fluoroquinolone (norfloxacin, 400 mg twice daily) re-
sulted in lower bacterial relapse rates than did 10 days of cefti-
buten (200 mg twice daily), with each group receiving
intravenous cefuroxime for 2–4 days prior to randomization to
the study drugs [59]. This study also included a mixed pop-
ulation of men and women and thus did not meet our inclusion
criteria. Another study demonstrated that initial therapy with
intravenous ceftriaxone (1 g for 3 days) was comparable to 1
dose of intravenous ceftriaxone (1 g) followed by oral cefixime
(400 mg once daily for 2 days). Both groups received a 10-day
course of an oral antibiotic on the basis of susceptibility test
results after the initial regimen was completed [60].
The findings from the 1 study that met our specific inclusion
and exclusion criteria as well as the additional studies described
support the superior efficacy of fluoroquinolone regimens for
treatment of acute pyelonephritis [55]. These studies also dem-
onstrate efficacy of a 5–7-day regimen and once-daily dosing for
mild to moderate pyelonephritis. In regions with low levels of
fluoroquinolone resistance among outpatient uncomplicated
pyelonephritis isolates, as demonstrated in 2 recent US studies,
the fluoroquinolones are the preferred antimicrobial class for oral
therapy [18, 61]. For some areas of the world, including certain
areas of the United States, the prevalence of fluoroquinolone
resistance is .10%. In such areas, it is recommended that a dose
of a long-acting parenteral antimicrobial, such as a 1-g dose of
ceftriaxone or a consolidated 24-h dose of an aminoglycoside (eg,
one 5–7-mg/kg dose of gentamicin), be given once at the initia-
tion of therapy. Some experts prefer to continue the parenteral
agent until susceptibility data are available; this strategy is not well
studied, but it can be considered depending on feasibility and
clinical judgment. The parenteral agent may be administered via
the intramuscular route if the intravenous route is not available,
but there are limited data supporting this approach.
High rates of resistance to trimethoprim-sulfamethoxazole
with corresponding failure rates for resistant isolates make this
agent an inferior choice for empirical therapy, but it is highly
efficacious in pyelonephritis if the causative organism is suscep-
tible. The current efficacy rates observed for trimethoprim-sulfa-
methoxazole in the treatment of pyelonephritis are based on a 14-
day regimen, which is the FDA-approved duration of treatment
[55]. However, there are no data to suggest a shorter course of
trimethoprim-sulfamethoxazole would not be effective when the
uropathogen is susceptible, and additional studies of short-course
regimens are warranted. If trimethoprim-sulfamethoxazole is
used empirically, an initial intravenous dose of ceftriaxone is
recommended as this combination resulted in improved clinical
and bacterial cure rates in the study by Talan et al [55]. Although
not formally studied, a consolidated 24-h dose of an amino-
glycoside could also be considered in place of ceftriaxone.
Oral b-lactam agents should be used with caution for treat-
ment of pyelonephritis, on the basis of studies outlined in the
previous guideline demonstrating inferior efficacy and higher
relapse rates compared with trimethoprim-sulfamethoxazole
[1]. Those studies primarily evaluated aminopenicillins. Current
data on oral cephalosporins are limited but are suggestive of
inferior efficacy compared with the fluoroquinolones [59]. If an
oral b-lactam is used, an initial intravenous dose of ceftriaxone
or a consolidated 24-h dose of an aminoglycoside is recom-
mended. Continued use of the oral b-lactam is reasonable only if
the uropathogen is susceptible. Initial coadministration of
a parenteral agent is supported in part by the findings of Sanchez
et al [60], who reported similar outcomes with ‘ one dose of
ceftriaxone versus a 3-day course of ceftriaxone followed by oral
b-lactam therapy in women with uncomplicated pyelonephritis.
As outlined in the previous guideline, a total course of 10–14
days of therapy is likely sufficient when using an oral b-lactam
for treatment of uncomplicated pyelonephritis.
Uncomplicated cystitis or pyelonephritis due to MRSA is
uncommon, and at this time, there are insufficient data to rec-
ommend use of an MRSA-active agent for empirical therapy of
uncomplicated UTI. Recommendations for treatment of acute
uncomplicated pyelonephritis that is accompanied by nausea or
vomiting, which precludes oral intake or otherwise requires
hospitalization, are the same as previously outlined in the 1999
IDSA guideline, because no new data are available to warrant
revisions [1]. Given rising rates of ampicillin resistance among
gram-negative organisms, the use of ampicillin should be lim-
ited to patients in whom Enterococcus infection is suspected as
the pathogen (based on previous history) and should be ac-
companied by an aminoglycoside. Broad-spectrum antimicro-
bial coverage should be tailored, as appropriate, on the basis of
urine culture and susceptibility results.
FUTURE DIRECTIONS AND RESEARCH GAPS IN
MANAGEMENT OF ACUTE UNCOMPLICATED
CYSTITIS AND PYELONEPHRITIS
As listed below, the panel identified several areas warranting
further investigation.
d Better understanding of collateral damage in the treatment
of uncomplicated cystitis.
d Better understanding of the public health impact of
antimicrobial use and resistance in women with sporadic
uncomplicated UTI.
Clinical Practice Guidelines
d
CID 2011:52 (1 March)
d
e117

d Role of MRSA in uncomplicated cystitis and pyelonephritis.
d Efficacy of narrow-spectrum cephalosporins in treatment of
uncomplicated cystitis.
d Role of oral broad-spectrum cephalosporins for outpatient
treatment of pyelonephritis in regions with high prevalence of
resistance to fluoroquinolones.
d Efficacy of short course (7–10 day) regimens with
trimethoprim-sulfamethoxazole for pyelonephritis caused by
a trimethoprim-sulfamethoxazole susceptible pathogen.
d Optimal therapy of acute cystitis in women who are
postmenopausal or have well-controlled diabetes without
urological sequelae.
d Better understanding of optimal treatment regimens for
ESBL- producing uropathogens causing uncomplicated UTI.
d Additional studies of clinical efficacy rates achieved in the
setting of an acute cystitis uropathogen that is resistant to the
antimicrobial agent used for treatment.
d Prospective and unbiased resistance surveillance of
uropathogens at the local practice and/or health care system
level in order to best inform antimicrobial decisions.
PERFORMANCE MEASURES
Performance measures are indicators to help guideline users
gauge potential effects and benefits of implementation of the
guidelines. Such tools can be indicators of the actual process,
short-term and long-term outcomes, or both. Deviations from
the recommendations are expected in a proportion of cases, and
compliance in 80%–95% of cases is generally appropriate, de-
pending on the measure. The following measures were identified
as appropriate indicators for management of acute un-
complicated UTI in women.
d Use of a recommended antimicrobial for treatment of
uncomplicated cystitis in cases in which it is not prohibited
because of allergy history or availability
d Use of fluoroquinolones for treatment of acute uncomplicated
cystitis only when a recommended antimicrobial cannot be used
d Use of a recommended antimicrobial for treatment of
uncomplicated pyelonephritis in cases in which it is not
prohibited because of allergy history or availability
d Initiation of empirical therapy for acute uncomplicated
pyelonephritis with performance of a pretherapy urine culture and
modification of empirical therapy as indicated by culture results
Acknowledgments
The Expert Panel dedicates this guideline to the memory of Dr. Walter E.
Stamm, whose work and commitment over several decades enhanced our
understanding ofthe pathogenesis, epidemiology, and management of urinary
tract infections in women. We honor him as a colleague, mentor, and leader.
The Expert Panel wishes to express its gratitude to the IDSA staff for
administrative assistance and external reviewers Drs Patricia Brown, Jack
Sobel, and John Warren for thoughtful advice.
Financial support. The Infectious Diseases Society of America.
Potential conflicts of interest. K.G. (Chair) has served as a consultant
to Pfizer and Pinnacle Pharmaceutical. A.J.S. has served as a consultant to
Novabay Pharmaceuticals, Pfizer, Propagate Pharmaceuticals, Hagen/Sin-
clair Research Recruiting, Swiss Precision Diagnostics Development
Company, and FlashPointMedica; has received honoraria from BMJ Group
(British Medical Journal) and Advanstar Communications; received
a royalty payment from UpToDate; and received remuneration from the
American Urological Association. G.J.M. has served as a consultant to
Cerexa, Cubist, Eisai, Forest, Merck, Ortho-McNeil, Pfizer, and Schering-
Plough and has received honoraria from Cubist and Merck. K.G.N. has
received remuneration as consultant or speaker from Bionorica, Daiichi
Sankyo, Janssen Cilag, Johnson & Johnson, OM Pharma, Pierre Fabre,
Sanofi Aventis, and Zambon and has received research grants from Mer-
Lion Pharmaceuticals, Rosen Pharma, and OM Pharma. L.E.N. has served
as a consultant to Pfizer, Leo pharmaceuticals, Cerexa, and Johnson &
Johnson and served on the advisory board for Leo Pharmaceuticals and
Cerexa. L.G.M. has served as a consultant to Forest and Theravance Lab-
oratories and received research grants from Cubist and Pfizer Pharma-
ceuticals. T.M.H. has served as a consultant to Pfizer, Alita Pharmaceuticals,
and Pinnacle Pharmaceuticals All other authors: no conflicts.
References
1. Warren JW, Abrutyn E, Hebel JR, Johnson JR, Schaeffer AJ, Stamm
WE. Guidelines. for antimicrobial treatment of uncomplicated acute
bacterial cystitis and acute pyelonephritis in women. nfectious Diseases
Society of America (IDSA). Clin Infect Dis 1999; 29:745–58.
2. Huang ES, Stafford RS. National patterns in the treatment of urinary
tract infections in women by ambulatory care physicians. Archives of
internal medicine 2002; 162:41–7.
3. Kahan NR, Chinitz DP, Kahan E. Longer than recommended empiric
antibiotic treatment of urinary tract infection in women: an avoidable
waste of money. Journal of clinical pharmacy and therapeutics 2004;
29:59–63.
4. Kallen AJ, Welch HG, Sirovich BE. Current antibiotic therapy for
isolated urinary tract infections in women. Archives of internal med-
icine 2006; 166:635–9.
5. O’Connor PJ, Solberg LI, Christianson J, Amundson G, Mosser G.
Mechanism of action and impact of a cystitis clinical practice guideline
on outcomes and costs of care in an HMO. The Joint Commission
journal on quality improvement 1996; 22:673–82.
6. Stamm WE. Evaluating guidelines. Clin Infect Dis 2007; 44:775–6.
7. Fosfomycin for urinary tract infections. Med Lett Drugs Ther 1997;
39:66–8. PMID: 9255237.
8. Kahlmeter G. An international survey of the antimicrobial susceptibility of
pathogens from uncomplicated urinary tract infections: the ECO.SENS
Project. J Antimicrob Chemother 2003; 51:69–76.
9. Kahlmeter G. Prevalence and antimicrobial susceptibility of pathogens
in uncomplicated cystitis in Europe. The ECO.SENS study. Int J An-
timicrob Agents 2003; 22(Suppl. 2):49–52.
10. Naber KG, Schito G, Botto H, Palou J, Mazzei T. Surveillance study in
Europe and Brazil on clinical aspects and Antimicrobial Resistance
Epidemiology in Females with Cystitis (ARESC): implications for
empiric therapy. Eur Urol 2008; 54:1164–75.
11. Zhanel GG, Hisanaga TL, Laing NM, et al. Antibiotic resistance in
Escherichia coli outpatient urinary isolates: final results from the North
American Urinary Tract Infection Collaborative Alliance (NAUTICA).
Int J Antimicrob Agents 2006; 27:468–75.
12. Sundqvist M, Geli P, Andersson DI, et al. Little evidence for re-
versibility of trimethoprim resistance after a drastic reduction in tri-
methoprim use. J Antimicrob Chemother 2010; 65:350–60.
e118
d
CID 2011:52 (1 March)
d
Gupta et al

13. Brown PD, Freeman A, Foxman B. Prevalence and predictors of tri-
methoprim-sulfamethoxazole resistance among uropathogenic Escher-
ichia coli isolates in Michigan. Clin Infect Dis 2002; 34:1061–6.
14. Metlay JP, Strom BL, Asch DA. Prior antimicrobial drug exposure:
a risk factor for trimethoprim-sulfamethoxazole-resistant urinary tract
infections. J Antimicrob Chemother 2003; 51:963–70.
15. Burman WJ, Breese PE, Murray BE, et al. Conventional and molecular
epidemiology of trimethoprim-sulfamethoxazole resistance among
urinary Escherichia coli isolates. Am J Med 2003; 115:358–64.
16. Colgan R, Johnson JR, Kuskowski M, Gupta K. Risk factors for
trimethoprim-sulfamethoxazole resistance in patients with acute
uncomplicated cystitis. Antimicrob Agents Chemother 2008; 52:
846–51.
17. Johnson L, Sabel A, Burman WJ, et al. Emergence of fluoroquinolone
resistance in outpatient urinary Escherichia coli isolates. Am J Med
2008; 121:876–84.
18. Talan DA, Krishnadasan A, Abrahamian FM, Stamm WE, Moran GJ.
Prevalence and risk factor analysis of trimethoprim-sulfamethoxazole-
and fluoroquinolone-resistant Escherichia coli infection among emer-
gency department patients with pyelonephritis. Clin Infect Dis 2008;
47:1150–8.
19. Miller LG, Tang AW. Treatment of uncomplicated urinary tract in-
fections in an era of increasing antimicrobial resistance. Mayo Clin
Proc 2004; 79:1048–53; quiz 1053–4.
20. Gupta K. Emerging antibiotic resistance in urinary tract pathogens.
Infect Dis Clin North Am 2003; 17:243–59.
21. Raz R, Chazan B, Kennes Y, et al. Empiric use of trimethoprim-sul-
famethoxazole (TMP-SMX) in the treatment of women with un-
complicated urinary tract infections, in a geographical area with a high
prevalence of TMP-SMX-resistant uropathogens. Clin Infect Dis 2002;
34:1165–9.
22. Paterson DL. "Collateral damage" from cephalosporin or quinolone
antibiotic therapy. Clin Infect Dis 2004; 38(Suppl. 4):S341–5.
23. Ramphal R, Ambrose PG. Extended-spectrum b-lactamases and clin-
ical outcomes: current data. Clin Infect Dis 2006; 42(Suppl. 4):
S164–72.
24. Graninger W. Pivmecillinam–therapy of choice for lower urinary tract
infection. Int J Antimicrob Agents 2003; 22(Suppl. 2):73–8.
25. Knothe H, Schafer V, Sammann A, Shah PM. Influence of fosfomycin
on the intestinal and pharyngeal flora of man. Infection 1991;
19:18–20.
26. Mavromanolakis E, Maraki S, Samonis G, Tselentis Y. Effect of nor-
floxacin, trimethoprim-sulfamethoxazole and nitroffurantoin on fecal
flora of women with recurrent urinary tract infections. J Chemother
1997; 9:203–7.
27. Sullivan A, Edlund C, Nord C. Effect of antimicrobial agents on the
ecological balance of human microflora. Lancet Infect Dis 2001;
1:101–14.
28. Christiaens TC, De Meyere M, Verschraegen G, Peersman W, Heytens
S, De Maeseneer JM. Randomised controlled trial of nitrofurantoin
versus placebo in the treatment of uncomplicated urinary tract in-
fection in adult women. Br J Gen Pract 2002; 52:729–34.
29. Ferry SA, Holm SE, Stenlund H, Lundholm R, Monsen TJ. Clinical and
bacteriological outcome of different doses and duration of piv-
mecillinam compared with placebo therapy of uncomplicated lower
urinary tract infection in women: the LUTIW project. Scand J Prim
Health Care 2007; 25:49–57.
30. Linder JA, Huang ES, Steinman MA, Gonzales R, Stafford RS. Fluo-
roquinolone prescribing in the United States: 1995 to 2002. Am J Med
2005; 118:259–68.
31. Field MJ, Lohr KN, eds. Institute of Medicine Committee to Advise the
Public Health Service on Clinical Practice Guidelines. Clinical practice
guidelines: directions for a new program. Washington, DC: National
Academy Press, 1990; 55–77.
32. Canadian Task Force on the Periodic Health Examination. The peri-
odic health examination. Can Med Assoc J 1979; 121(9);1193–254.
33. Pimentel FL, Dolgner A, Guimaraes J, Quintas M, Mario-Reis J. Effi-
cacy and safety of norfloxacin 800 mg once-daily versus norfloxacin
400 mg twice-daily in the treatment of uncomplicated urinary tract
infections in women: a double-blind, randomized clinical trial. J
Chemother 1998; 10:122–7.
34. Grabe M, Bishop MC, Bjerklund-Johansen TE, et al. Guidelines on
urological infections. In EAU Guidelines edition presented at the 25th
EAU Annual Congress, Barcelona 2010. ISBN 978-90-79754-70-0.
35. Arredondo-Garcia JL, Figueroa-Damian R, Rosas A, Jauregui A, Corral
M, Costa A, et al. Comparison of short-term treatment regimen of
ciprofloxacin versus long-term treatment regimens of trimethoprim/
sulfamethoxazole or norfloxacin for uncomplicated lower urinary tract
infections: a randomized, multicentre, open-label, prospective study. J
Antimicrob Chemother 2004; 54:840–3.
36. Gupta K, Hooton TM, Roberts PL, Stamm WE. Short-course nitro-
furantoin for the treatment of acute uncomplicated cystitis in women.
Arch Intern Med 2007; 167:2207–12.
37. Iravani A, Klimberg I, Briefer C, Munera C, Kowalsky SF, Echols RM. A
trial comparing low-dose, short-course ciprofloxacin and standard 7
day therapy with co-trimoxazole or nitrofurantoin in the treatment of
uncomplicated urinary tract infection. J Antimicrob Chemother 1999;
43(Suppl A):67–75.
38. Kavatha D, Giamarellou H, Alexiou Z, et al. Cefpodoxime-proxetil
versus trimethoprim-sulfamethoxazole for short-term therapy of un-
complicated acute cystitis in women. Antimicrob Agents Chemother
2003; 47:897–900.
39. Stein GE. Comparison of single-dose fosfomycin and a 7-day course of
nitrofurantoin in female patients with uncomplicated urinary tract
infection. Clin Ther 1999; 21:1864–72.
40. Minassian MA, Lewis DA, Chattopadhyay D, Bovill B, Duckworth GJ,
Williams JD. A comparison between single-dose fosfomycin trometa-
mol (Monuril) and a 5-day course of trimethoprim in the treatment of
uncomplicated lower urinary tract infection in women. Int J Anti-
microb Agents 1998; 10:39–47.
41. Popovic M, Steinort D, Pillai S, Joukhadar C. Fosfomycin: an old, new
friend? Eur J Clin Microbiol Infect Dis 2010; 29:127–42.
42. Rodriguez-Bano J, Alcala JC, Cisneros JM, et al. Community infections
caused by extended-spectrum beta-lactamase-producing Escherichia
coli. Arch Intern Med 2008; 168:1897–02.
43. Nicolle LE, Madsen KS, Debeeck GO, et al. Three days of pivmecillinam
or norfloxacin for treatment of acute uncomplicated urinary infection
in women. Scand J Infect Dis 2002; 34:487–92.
44. Henry D, Ellison W, Sullivan J, et al. Treatment of community-ac-
quired acute uncomplicated urinary tract infection with sparfloxacin
versus ofloxacin. The Sparfloxacin Multi Center UUTI Study Group.
Antimicrob Agents Chemother 1998; 42:2262–6.
45. Henry DC, Nenad RC, Iravani A, et al. Comparison of sparfloxacin and
ciprofloxacin in the treatment of community-acquired acute uncomplicated
urinary tract infection in women. parfloxacin Multicenter Uncomplicated
Urinary Tract Infection Study Group. Clin Ther 1999; 21:966–81.
46. Naber KG, Allin DM, Clarysse L, et al. Gatifloxacin 400 mg as a single
shot or 200 mg once daily for 3 days is as effective as ciprofloxacin 250
mg twice daily for the treatment of patients with uncomplicated uri-
nary tract infections. Int J Antimicrob Agents 2004; 23:596–605.
47. Richard GA, Mathew CP, Kirstein JM, Orchard D, Yang JY. Single-dose
fluoroquinolone therapy of acute uncomplicated urinary tract in-
fection in women: results from a randomized, double-blind, multi-
center trial comparing single-dose to 3-day fluoroquinolone regimens.
Urology 2002; 59:334–9.
48. Fourcroy JL, Berner B, Chiang YK, Cramer M, Rowe L, Shore N. Ef-
ficacy and safety of a novel once-daily extended-release ciprofloxacin
tablet formulation for treatment of uncomplicated urinary tract in-
fection in women. Antimicrob Agents Chemother 2005; 49:4137–43.
49. Henry DC Jr, Bettis RB, Riffer E, et al. Comparison of once-
daily extended-release ciprofloxacin and conventional twice-daily
Clinical Practice Guidelines
d
CID 2011:52 (1 March)
d
e119

ciprofloxacin for the treatment of uncomplicated urinary tract in-
fection in women. Clin Ther 2002; 24:2088–104.
50. Vogel T, Verreault R, Gourdeau M, Morin M, Grenier-Gosselinl L,
Rochette L. Optimal duration of antibiotic therapy for uncomplicated
urinary tract infection in older women: a double-blind randomized
controlled trial. CMAJ 2004; 170:469–73.
51. Auquer F, Cordon F, Gorina E, Caballero JC, Adalid C, Batlle J. Single-
dose ciprofloxacin versus 3 days of norfloxacin in uncomplicated
urinary tract infections in women. Clin Microbiol Infect 2002; 8:50–4.
52. Hooton TM, Scholes D, Gupta K, Stapleton AE, Roberts PL, Stamm
WE. Amoxicillin-clavulanate vs ciprofloxacin for the treatment of
uncomplicated cystitis in women: a randomized trial. JAMA 2005;
293:949–55.
53. Hooton TM, Besser R, Foxman B, Fritsche TR, Nicolle LE. Acute un-
complicated cystitis in an era of increasing antibiotic resistance:
a proposed approach to empirical therapy. Clin Infect Dis 2004;
39:75–80.
54. Leigh AP, Nemeth MA, Keyserling CH, Hotary LH, Tack KJ. Cefdinir
versus cefaclor in the treatment of uncomplicated urinary tract in-
fection. Clin Ther 2000; 22:818–25.
55. Talan DA, Stamm WE, Hooton TM, et al. Comparison of ciprofloxacin
(7 days) and trimethoprim-sulfamethoxazole (14 days) for acute un-
complicated pyelonephritis pyelonephritis in women: a randomized
trial. JAMA 2000; 283:1583–90.
56. Peterson J, Kaul S, Khashab M, Fisher AC, Kahn JB. A double-blind,
randomized comparison of levofloxacin 750 mg once-daily for five
days with ciprofloxacin 400/500 mg twice-daily for 10 days for the
treatment of complicated urinary tract infections and acute pyelone-
phritis. Urology 2008; 71:17–22.
57. Talan DA, Klimberg IW, Nicolle LE, Song J, Kowalsky SF, Church DA.
Once daily, extended release ciprofloxacin for complicated urinary
tract infections and acute uncomplicated pyelonephritis. J Urol 2004;
171:734–9.
58. Naber KG, Bartnicki A, Bischoff W, et al. Gatifloxacin 200 mg or 400
mg once daily is as effective as ciprofloxacin 500 mg twice daily for the
treatment of patients with acute pyelonephritis or complicated urinary
tract infections. Int J Antimicrob Agents 2004; 23(Suppl. 1):S41–53.
59. Cronberg S, Banke S, Bergman B, et al. Fewer bacterial relapses after
oral treatment with norfloxacin than with ceftibuten in acute pyelo-
nephritis initially treated with intravenous cefuroxime. Scand J Infect
Dis 2001; 33:339–43.
60. Sanchez M, Collvinent B, Miro O, et al. Short-term effectiveness of
ceftriaxone single dose in the initial treatment of acute uncomplicated
pyelonephritis in women: a randomised controlled trial. Emerg Med J
2002; 19:19–22.
61. Czaja CA, Scholes D, Hooton TM, Stamm WE. Population-based ep-
idemiologic analysis of acute pyelonephritis. Clin Infect Dis 2007;
45:273–80.
e120
d
CID 2011:52 (1 March)
d
Gupta et al
