
Middle School Chemistry 1 ©2023 American Chemical Society
Chapter 2 — Student Reading
Atoms and molecules are in motion
We warm things up and cool things down all the time, but we usually don’t think much
about what’s really happening. If you put a room-temperature metal spoon into a hot
liquid like soup or hot chocolate, the metal gets hotter. But what actually has to happen
for the hot liquid to make the metal hotter?
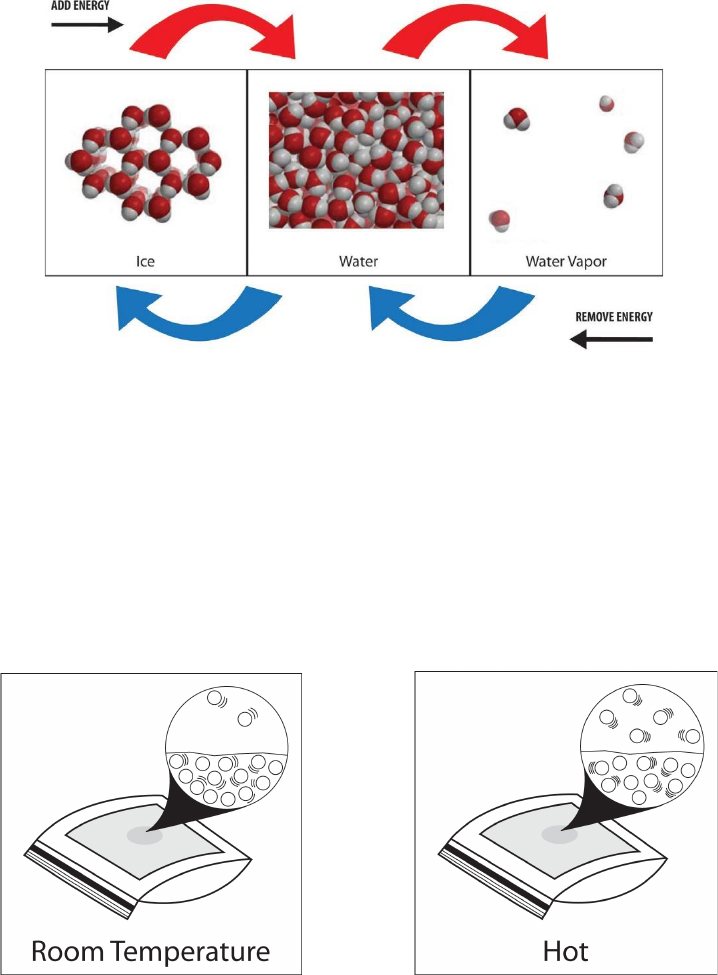
By now you know that substances are made of atoms and molecules. These atoms and
molecules are always in motion. You also know that when atoms and molecules are heated,
they move faster and when they are cooled, they move slower. But how do the atoms and
molecules actually become heated and cooled? In our example of heating a metal spoon in
a hot liquid, what is the process that transfers energy from the water to the spoon?
Moving atoms and molecules have energy
To answer this question, you really have to think about the moving atoms and
molecules as having energy. Anything that has mass and is moving, like a train, a
moving ball, or an atom has a certain amount of energy. The energy of a moving object
is called
kinetic energy
. If the speed of the object increases, its kinetic energy
increases. If the speed of the object decreases, its kinetic energy decreases. So if the
atoms or molecules of a substance are moving fast, they have more kinetic energy than
when they are moving more slowly.
PHOTOS.COM
PHOTOS.COM
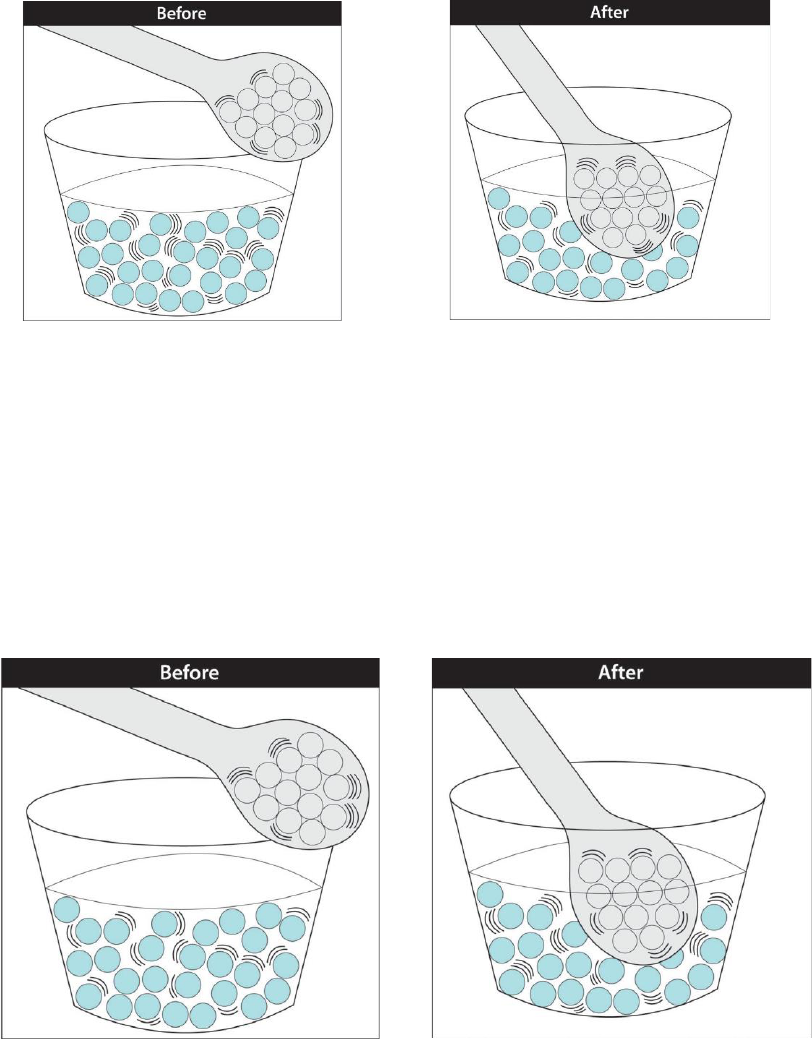
Energy can be transferred to make things warmer
In our example of a spoon in hot liquid, the molecules of hot liquid are moving quickly so
they have a lot of kinetic energy. When the room-temperature spoon is placed in the
liquid, the fast-moving molecules in the liquid contact the slower-moving atoms in the
spoon. The fast-moving molecules hit the slower-moving atoms and speed them up. In
this way, the fast-moving molecules transfer some of their kinetic energy to the slower
atoms so that these slower atoms now have more kinetic energy. This process of
transferring energy by direct contact is called
conduction
.
Middle School Chemistry 2 ©2023 American Chemical Society
Energy can be transferred to make things cooler
Cooling things by conduction works the same way as warming but you just look at the
substance losing energy instead of the substance gaining energy. This time let’s say that
you take a hot metal spoon and put it in room-temperature water. The faster-moving
atoms in the spoon contact the slower-moving molecules in the water. The atoms in the
spoon transfer some of their energy to the molecules in the water. The spoon will get
cooler, and the water will get a little warmer.
Another example is cans of room-temperature soda pop placed in a cooler filled with
ice. Kinet ic energy is transferred from the warmer metal can to the cooler ice. This
makes the can colder.
Energy is then transferred from the warmer soda to the colder can. This transfer of
energy from the soda results in slower motion of the molecules of the soda, which
can be measured as a lower temperature and colder soda.
So, the way to cool something is for its energy to be transferred to something colder.
Middle School Chemistry 3 ©2023 American Chemical Society
This is a rule about conduction: Energy can only be transferred from something at a
higher temperature to something at a lower temperature. You can’t cool something by
adding “coldness” to it. You can only make something colder by allowing its energy to
be transferred to something even colder.
This brings up the question of what exactly is “temperature”. Temperature is related to
the kinetic energy of the moving atoms or molecules of a substance. Temperature is a
measure of the average kinetic energy of the atoms or molecules of a substance. By
taking the temperature of something, you are actually getting information about the
kinetic energy of its atoms and molecules, but not the kinetic energy of any particular
one.
There are more than a billion trillion atoms or molecules in even a small sample of a
substance. They are constantly moving and bumping into each other and transferring
little amounts of energy between each other. So at any time, the atoms and molecules
don’t all have the same kinetic energy. Some are moving faster and some are moving
slower than others but most are about the same. So when you measure the
temperature of something, you are actually measuring the average kinetic energy of its
atoms or molecules.
If temperature is the average kinetic energy of atoms and molecules, then what is “heat”?
The word “heat” has a specific meaning in science even though we use the word all the
time to mean different things in our daily life. The scientific meaning of heat has to do
with energy that is being transferred. Heat is the energy that is transferred from a
substance at a higher temperature to a substance at a lower temperature. During
conduction, the energy transferred from faster-moving atoms to slower-moving atoms is
heat.
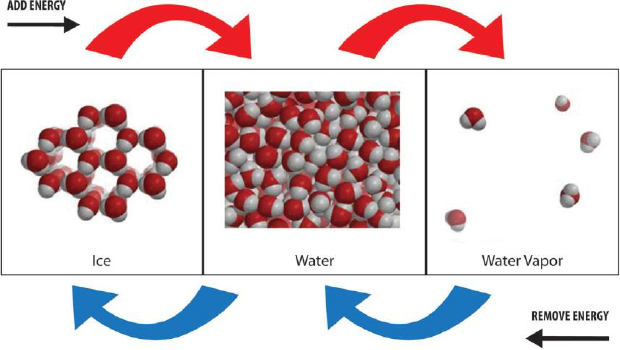
Changing State
Changing from a solid to a liquid—Melting
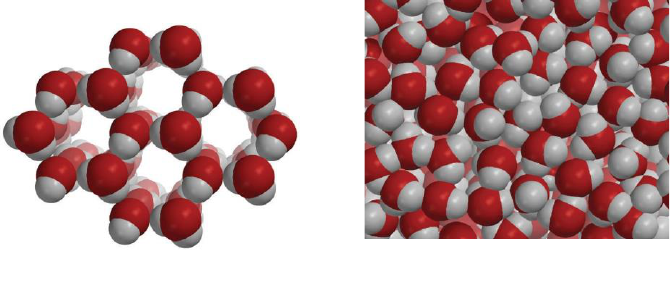
In solids, the atoms or molecules that make up the substance have strong attractions to
each other and stay in fixed positions. These properties give solids their definite shape
and volume.
Ice
Liquid Water

Middle School Chemistry 4 ©2023 American Chemical Society
When a solid is heated, the motion of the particles (atoms or molecules) increases. The
atoms or molecules are still attracted to each other, but their extra movement begins to
compete with their attractions. If enough energy is added, the motion of the particles
begins to overcome the attractions and the particles move more freely. They begin to
slide past each other as the substance begins to change state from a solid to a liquid.
This process is called melting.
The particles of the liquid are only slightly further apart than in the solid. (Water is the
exception because the molecules in liquid water are actually closer together than they are in
ice). The particles of the liquid have more kinetic energy than they did as a solid but their
attractions are still able to hold them together enough so that they retain their liquid state
and do not become a gas.
Different solids melt at different temperatures
The temperature at which a substance begins to melt is called the
melting point. It makes sense that different substances have
different melting points. Since the atoms or molecules that
substances are made of have a different amount of attraction for
each other, a different amount of energy is required to make them
change from a solid to a liquid. A good example is the melting
point of salt and sugar. The melting point of sugar is 186 °C. The
melting point for regular table salt is 801 °C. Metals like iron and
lead also have different melting points. Lead melts at 327 °C and
iron melts at 1,538 °C.
Some solids, like glass do not have a precise melting point but begin to melt over a
range of temperatures. This is because the molecules that make up glass are not
arranged in as orderly a way as those in crystals like salt or sugar or metals like iron.
Depending on the type of glass, the melting point is usually between 1,200–1,600 °C.
Changing from a solid to a gas—Sublimation
Some substances can change directly from a solid to a gas. This process is called
sublimation
. One of the more popular examples of sublimation is dry ice which is frozen
carbon dioxide (CO
2
). To make dry ice, carbon dioxide gas is placed under high pressure
and made very cold (about
−
78.5
°
C). When a piece of dry ice is at room temperature
and normal pressure, the molecules of CO
2
move faster and break away from each
other and go directly into the air as a gas. Regular ice cubes in the freezer will also
sublimate but much more slowly than dry ice.
Middle School Chemistry 5 ©2023 American Chemical Society
Changing from a liquid to a gas—Evaporation
You see evidence of evaporation all the time. Evaporation causes wet clothes to dry and the
water in puddles to “disappear”. But the water doesn’t actually disappear. It changes state
from a liquid to a gas.
The molecules in a liquid evaporate when they have enough energy to overcome the
attractions of the molecules around them. The molecules of a liquid are moving and bumping
into each other all the time, transferring energy between one another. Some molecules will
have more energy than others. If their motion is energetic enough, these molecules can
completely overcome the attractions of the molecules around them. When this happens, the
molecules go into the air as a gas. This process is called evaporation.
Heating increases the rate of evaporation
You’ve probably noticed that higher temperatures seem to make evaporation happen faster.
Wet clothes and puddles seem to dry more quickly when they are heated by the sun or in
some other way.
You can test whether heat affects the rate of evaporation by placing a drop of water on two
paper towels. If one paper is heated and the other remains at room temperature, the water
that is heated will evaporate faster.
Middle School Chemistry 6 ©2023 American Chemical Society
When a liquid is heated, the motion of its molecules increases. The number of molecules
that are moving fast enough to overcome the attractions of other molecules increases.
Therefore, when water is heated, more molecules can break away from the liquid and
the rate of evaporation increases.
Different liquids have different rates of evaporation
It makes sense that different liquids have different rates of evaporation. Different
liquids are made of different molecules. These molecules have their own characteristic
strength of attraction to one another. These molecules require a different amount of
energy to increase their motion enough to overcome the attractions to change from a
liquid to a gas.
Liquids will evaporate over a wide range of temperatures
Even at room temperature, or lower, liquids will evaporate. You can test this by
wetting a paper towel and hanging it up indoors at room temperature. Evaporation at
room temperature might seem strange since the molecules of a liquid need to have a
certain amount of energy to evaporate. Where do they get the energy if the liquid is
not warmed? But remember that the temperature of a substance is the average kinetic
energy of its atoms or molecules. Even cold water, for instance, has a small percentage
of molecules with much more kinetic energy than the others. With all the random
bumping of a billion trillion molecules, there are always a few molecules which gain
enough energy to evaporate. The rate of evaporation will be slow, but it will happen.
Changing from a gas to a liquid—Condensation
If you have seen water form on the outside of a cold cup, you
have seen an example of condensation. Water molecules from
the air contact the cold cup and transfer some of their energy
to the cup. These molecules slow down enough that their
attractions can overcome their motion and hold them together
as a liquid. This process is called condensation.
Cooling increases the rate of condensation
You can test to see if cooling water vapor increases the rate
of condensation by making two similar samples of water
vapor and cooling one more than the other. In the
illustration, two samples of water vapor are trapped inside
the cups. Ice is placed on one top cup, but not the other.
In a few minutes, there are water drops on the inside top of
both cups but more water can be seen on the inside of the
top cup with the ice. This shows that cooling water vapor
Middle School Chemistry 7 ©2023 American Chemical Society
increases the rate of condensation. When a gas is cooled, the motion of its molecules
decreases. The number of molecules moving slow enough for their attractions to hold
them together increases. More molecules join together to form a liquid making the rate
of condensation greater.
The amount of water vapor in the air affects the rate of condensation
Temperature isn’t the only factor that affects the rate of condensation. At a given
temperature, the more water molecules in the air, the greater the rate of
condensation. If there are more molecules, a greater number of molecules will be
moving at different speeds and more will be moving slowly enough to condense.
Different gases condense at different temperatures
Each gas is made up of its own molecules which are attracted to each other a certain amount.
Each gas needs to be cooled to a certain temperature for the molecules to slow down enough
so that the attractions can hold them together as a liquid.
Changing from liquid to solid—Freezing
If a liquid is cooled enough, the molecules slow down to such an extent that their attractions
begin to overcome their motion. The attractions between the molecules cause them to
arrange themselves in more fixed and orderly positions to become a solid. This process is
called freezing.
Water molecules move further apart as water freezes
The freezing of water is very unusual because water molecules move farther apart as they
arrange themselves into the structure of ice as water freezes. The molecules of just about
every other liquid move closer together when they freeze.

Middle School Chemistry 8 ©2023 American Chemical Society
Different liquids have different freezing points
It makes sense that different liquids have different freezing points. Each liquid is made
up of different molecules. The molecules of different liquids are attracted to each other
by different amounts. These molecules must slow down to different extents before their
attractions can take hold and organize themselves into fixed positions as a solid.
Changing from a gas to a solid—Deposition
With the right concentration of gas molecules and
temperature, a gas can change directly to a solid
without going through the liquid phase. This process is
called
deposition
. It is the opposite of sublimation. One
of the most common examples of deposition is the
formation of frost. When there is the right combination
of water vapor in the air and temperature, the water
can change directly to frost without first condensing to
liquid water.
Evaporation, condensation, and the weather
Clouds
Clouds form when liquid water evaporates to become
water vapor and moves up into the sky in upward-
moving air. Air at higher altitudes is usually cooler than
air near the ground. So as the water vapor rises, it cools
and condenses, forming tiny drops of water. These
droplets are suspended in the air as clouds. Clouds at
higher altitudes where the air is even colder also
contain ice crystals. Clouds at very high levels are
composed mostly of ice crystals.
Rain
Rain begins as tiny droplets of water suspended in the
air as clouds. These droplets are so small that they
don’t fall yet to the ground as rain. They are similar to
the tiny droplets in fog or mist. But when these
droplets collect and join together, they become bigger
and heavier drops. Eventually the drops become so
heavy that they fall to the ground as rain.

Middle School Chemistry 9 ©2023 American Chemical Society
Snow
Like rain, snow begins with condensing water vapor that
forms clouds. However, when it’s cold enough, water
vapor not only condenses but also freezes, forming tiny
ice crystals. More and more water vapor condenses and
freezes on these seed crystals, forming the beautiful ice
crystals with six-sided symmetry that we know as
snowflakes.
Hail
Hail forms when a small drop of water freezes, falls, and then gets pushed back up into a
cloud. More water droplets collect and freeze on this ice crystal, which makes it heavier, and
it begins to fall again. The violent air in a thundercloud (cumulonimbus cloud) repeatedly
bounces the ice crystal upward. Each time it gets another coating of freezing water. Finally,
the ice crystal is so heavy that it falls all the way down to the ground as hail.
Dew
Dew is produced when moist air close to the ground
cools enough to condense to form liquid water. Dew
is different than rain because dew doesn’t fall onto
the ground in drops. It slowly accumulates to form
drops on objects near the ground. Dew often forms
on blades of grass and leaves and can make
beautiful designs on spider webs.
Frost
If the temperature of surfaces on the ground is low enough,
water vapor in the air can change directly to solid frost
without first condensing to a liquid. This process of changing
directly from a gas to a solid is called deposition.

Middle School Chemistry 10 ©2023 American Chemical Society
Fog
Usually, the air near the ground is warmer than the air
above it, but the conditions that cause fog are just the
reverse. Fog forms when warm, moist air passes over
cold ground or snow. As the water vapor in the air
cools, it condenses, forming very tiny drops of water
suspended in the air which we call “fog”. Fog is very
much like a cloud, but closer to the ground.
Mist on a Pond
Water evaporates even when the air is cold.
To form mist, the water in a pond, pool, or hot tub
must be warmer than the air above it and the air must
be cold enough to cause the water vapor to condense
as it rises. The mist seems to disappear as the water
droplets evaporate to become water vapor again.
